Abstract
Introduction
To evaluate the rewarming effectiveness of a similar amount of heat (from a charcoal heater) applied to either the head or torso in a human model for severe hypothermia in which shivering is pharmacologically inhibited in mildly hypothermic subjects.
Methods
Six male subjects were cooled on 3 different occasions, each in 8°C water for 60 min, or to a lowest core temperature of 35°C. Shivering was inhibited by intravenous meperidine (1.5 mg·kg−1), administered during the last 10 min of the cold-water immersion. Subjects then exited from the cold water, were dried, and were placed in a 3-season sleeping bag for 120 min in one of the following conditions: spontaneous rewarming only, charcoal heater on the head, or charcoal heater on the torso. Supplemental meperidine (to a maximum cumulative dose of 3.3 mg·kg−1) was administered as required during rewarming to suppress shivering.
Results
No significant differences were found in the postcooling afterdrop amount or core rewarming rates among the 3 conditions (0.8°C·h−1). During the last 30 min of rewarming the net heat gain was significantly higher in the head (85.8±25.3 W) and torso (81.5±6.3 W) conditions compared with the spontaneous condition (56.9±12 W) (P<0.05).
Conclusions
In our study, head and torso warming had the same core rewarming rates when shivering was pharmacologically inhibited in mildly hypothermic subjects. Therefore, in nonshivering cold subjects, head warming is a viable alternative if torso warming is contraindicated (eg, when performing cardiopulmonary resuscitation or working on open chest wounds).
Keywords
Introduction
Accidental hypothermia results from prolonged exposure to cold air or water. Such exposure is a common risk factor for many adventure, commercial, military, and other professional activities. For example, in Canada the number of deaths attributable to cold between 2007 and 2011 averaged 94 per year. 1
Patients exposed to a cold environment for an extended period present a challenge for rescue, medical care, and transport under austere conditions. Effective management of the prolonged cold-exposed patient includes handling as gently as possible, maintaining a horizontal position, preventing continued heat loss, addressing other injuries, and, if possible, using external warming to prevent development or worsening of hypothermia.2,3
Rewarming techniques can be broadly classified as invasive (eg, extracorporeal membrane oxygenation, cardiopulmonary bypass, body cavity lavage) 4 and noninvasive (eg, surface warming techniques). The problem with invasive rewarming is that it normally can only be practiced in a hospital setting. Thus, in the field, noninvasive rewarming techniques are required. 5 Various noninvasive rewarming techniques have been studied in the past, including a charcoal heater,6,7 body-to-body contact, 8 electric 9 or chemical (dry and gel) 10 heating pads or blankets, and arteriovenous anastomoses warming (immersion of distal limbs in 42–45°C water). 11
Traditionally application of external heat to the groin, axillae, and/or neck has been advised because these are areas of high heat transfer. Heat transfer to the core via the groin is limited, however. First, this requires heat to be applied directly against the skin that is immediately overlying the femoral vein; this is a small surface area, and this contact is unlikely to be achievable in field conditions. Second, this location is a relatively long distance for donated heat to be transported convectively via blood flow to the core. Thus, more heat is likely to be lost to other noncore tissues rather than applied to warm the heart. The neck also has areas of high heat transfer, but practically, much of the heat applied to the neck can be lost through the neck/face opening in the insulating bag or enclosure. Because of these limitations, recent recommendations have shifted to focus heat application to the areas immediately surrounding the heart (eg, the upper torso including the axillae, upper chest, and upper back). 3 ,12,13
In individuals who are cold stressed but not hypothermic (core temperature [Tco] >35°C) and mildly hypothermic (Tco 35–32°C), vigorous shivering can increase heat production by up to 5 to 6 times the resting metabolic rate, 12 ,14,15 which can produce an efficient core rewarming rate of 3 to 4°C·h−1.12,16 Related studies have shown that in shivering subjects, external heating generally increases the skin temperature and thereby inhibits shivering heat production by an amount approximately equal to the external heat donated, thus making the rewarming rates similar to rewarming by shivering only. 7 –9,17,18 Nonetheless, application of external heating is not contraindicated because it increases comfort, preserves energy, and reduces cardiac workload.
However, shivering starts to decline and is eventually eliminated in moderate (Tco 32–28°C) or severe (Tco <28°C) hypothermia. Thus, a patient with severe hypothermia who is at high risk for ventricular fibrillation or cardiac arrest 3 ,5,12 does not have significant endogenous heat production to facilitate core rewarming and requires external heat donation. 3 In some cases, the application of heat on the anterior chest may be contraindicated because of the presence of chest wounds or if cardiopulmonary resuscitation is required. In these scenarios, it would be important to find alternate areas for effectively applying heat to the patient.
Aside from the groin and neck, the head is one such option because of its high vascularity and blood flow, which could favor heat transfer to the core. A previous study from our laboratory evaluated the effects of warming the head in mildly hypothermic subjects whose shivering response was intact. 6 A similar amount of exogenous heat was applied to the head or torso using a charcoal heater (a combustion chamber that blows heated air through a set of flexible, noncompressible tubes). In this study, head and torso warming (TW) elicited a similar increase in total mean skin temperatures and rewarming rates. However, one potential limitation of this study was that shivering heat production potentially masked differences between the head and TW conditions. The relative effectiveness of head and TW has not been determined in nonshivering subjects.
The purpose of this study was to evaluate the rewarming effectiveness of a similar amount of heat, from a charcoal heater, donated to the head or torso in a human model for severe hypothermia where shivering is pharmacologically inhibited with meperidine in mildly hypothermic subjects. This method allows observations in nonshivering subjects without making them moderately to severely hypothermic and has been successfully used previously without complication.10,19–21 We hypothesized that TW would be most effective in decreasing the afterdrop, providing a greater net heat gain and core rewarming rate, followed by head warming (HW), which would be greater than spontaneous warming (SW) (eg, no external heat application). Even though a similar amount of heat was applied to the head and torso, greater heat donation was expected during TW because 1) the torso has a higher surface area compared with the head; 2) there is greater insulation in TW because the entire heat source is contained within the sleeping bag, whereas during HW some heat is lost through the face opening of the sleeping bag; and 3) the head is smaller than the torso and thus some of the flexible tubes will overlap during HW and heat transfer will be less efficient.
Methods
SUBJECTS
The study was open to both males and females, but only males volunteered. They were asked to complete a physical activity readiness questionnaire and medical screening questionnaire for a hypothermia study. Before participation, a signed informed consent was obtained. The protocol was approved by Health Canada and the biomedical research ethics board, University of Manitoba.
Sample size was calculated according to rewarming rates from a similar study. 10 To achieve 90% power (a=0.05, 1-tailed test; β=0.10; power index of 2.92), a sample size of 6 was required to detect a statistically significant difference (mean±SD) for rate of rewarming of 0.5±0.4°C·h−1.
INSTRUMENTATION
Subjects wore shorts and were instrumented at an ambient temperature of ∼22°C. Core temperature was estimated by esophageal temperature (Tes) measured with a disposable thermocouple (Mallinckrodt Medical Inc, St Louis, MO) inserted into the esophagus to the level of the heart (43 cm past the nares). This site provides the best noninvasive measure for intracardiac temperature22,23 and is a standard procedure in our laboratory.
Skin temperature and cutaneous heat flux were measured using 12 heat flux disks (2 cm in diameter; Concept Engineering, Old Saybrook, CT), which were taped to the skin according to standard lab procedures at the following sites: forehead, left cheek, top of the head, dorsum of the head, right axilla, anterior chest, anterior abdomen, upper back, left anterior upper arm, right anterior forearm, right anterior thigh, and left posterior thigh. Flux was defined as positive when heat flowed from the skin toward the environment. Oxygen consumption (V̇O2) was measured continuously with an open circuit metabolic cart (Vmax 229, Sensormedics, Yorba Linda, CA). Data were recorded at 30-s intervals.
For the purpose of administering meperidine (Demerol) to inhibit shivering, an IV line was inserted in the forearm by a physician before the start of each trial. This arm was kept out of water so that the broken skin site was not exposed to the water.
REWARMING METHODS
For all 3 conditions, subjects were removed from the water, towel dried, and placed in a hypothermia wrap: The subject was wrapped in a plastic vapor barrier and zipped into a 3-season hooded sleeping bag (rated to 0°C). Rewarming was conducted at an ambient air temperature of ∼22°C. One of the following warming conditions were then applied.
Spontaneous warming
No exogenous heat source was used in these trials.
Head warming
A charcoal heater (HEATPAC personal heater; Emergco Tech Solutions, Vancouver, BC) was applied to the head (Figure 1, top). It consists of a combustion chamber, charcoal fuel briquette, and branched, flexible but noncompressible heating ducts. It is small and light (23 × 12 × 6 cm, 1.1 kg) and produces 250 W of heat (according to manufacturer specifications) for 8 to 10 h. Once the charcoal fuel is ignited, it is placed in the chamber and the heated air is blown through the branched ducts by a fan within the charcoal heater. The heater was set to “high” mode and was ignited at least 15 min before being applied to the participant. The combustion chamber is insulated to prevent skin burning. It was placed on the right side of the face and the ducts were wrapped around the head.

(Top) Head warming; charcoal heater on the head. (Bottom) Torso warming; charcoal heater on the torso.
Torso warming
The charcoal heater was applied to the torso (Figure 1, bottom). The combustion chamber was placed on the anterior chest with the flexible ducts travelling over the shoulders posteriorly and then anteriorly under the arms to cross over the lower anterior chest.
PROTOCOL
Each trial was separated by at least 5 d to allow for an adequate washout of meperidine and its metabolite normeperidine. The subjects were cooled at the same time of the day to control for circadian effects. According to a previous study from our laboratory,10,21 the order of trials followed a modified balanced design, with SW being the first condition and the subsequent order of HW and TW conditions, to achieve a balanced design. This modified balanced design was used because external heat donation increases the skin temperature and reduces the thermal stimulus for shivering. The shivering stimulus is expected to be maximum in the SW condition; therefore, a higher dose of meperidine would be required to inhibit shivering in the SW condition compared with the active warming conditions. Because we wanted to use the same dosing amount for all conditions, the dosing schedule from SW was used for the subsequent active warming conditions.
Instrumentation took about 45 min. The participants then sat quietly and baseline measurements were recorded for 10 min at an ambient temperature of ∼22°C. The subjects were then immersed to the level of the sternal notch in a 21°C stirred water bath. The water temperature was lowered to 8°C over a period of 5 to 10 min by addition of about 60 kg of ice. Participants then remained in the water until one of the following criteria was met: core temperature decreased to 35°C, a researcher advised exit for any reason, a time period of 60 min elapsed, or the participant wished to exit.
During the last 10 min of immersion, 1.5 mg·kg−1 of IV meperidine (diluted in 10 mL of saline) was infused slowly in five 2 mL aliquots (ie, 0.3 mg·kg−1 meperidine per injection) over successive 2-min intervals. The participants then exited the water, were dried off, and were placed in the hypothermia enclosure for 120 min. During warming, additional meperidine was administered in doses of 0.3 mg·kg−1 as necessary (to a maximum cumulative dose of 3.3 mg·kg−1) to continuously inhibit shivering.
Treatment continued either for a period of 120 min or until the core temperature returned to baseline values (36.5–37.5°C). If the core temperature did not return to the baseline values, participants were placed in a warm water bath (40–42°C) until they were comfortable and core temperature returned to baseline values.
DATA ANALYSIS
The rate of core cooling (°C·h−1) was calculated by linear regression for Tes data during the final 10 min of immersion. Afterdrop (°C) was calculated as the difference between Tes on exit from cold water and its nadir. Rate of core rewarming (°C·h−1) was calculated by linear regression for Tes data from 60 to 120 min of warming. Head skin temperature (TskHead in°C) and heat flux (HFHead in W) were calculated from the area-weighted average of the temperatures at forehead, left cheek, top of the head, and dorsum of the head sites. Upper torso skin temperature (TskUTorso) and heat flux (HFUTorso) were calculated from the area-weighted average of the anterior chest, right axilla, and upper back sites. Total skin temperature (TskTotal) and heat flux (HFTotal) were calculated from the area-weighted average of all sites.
Metabolic heat production (M) was determined from oxygen consumption by using the following equation 11 :
M (W) = V̇O2 (L O2·min−1) × 69.7 (W·kcal−1·min−1) × [4.686 (kcal·L O2−1) + (0.83 – 0.707) × 1.232 (kcal·L O2−1)]
Respiratory heat loss (RHL) was calculated using the following equation 24 :
RHL (W) = 0.09 × M (W)
Net heat gain (W) was calculated by subtracting the respiratory heat loss and total cutaneous heat flux from the metabolic heat production. Positive values of total heat flux indicate heat loss.
Net heat gain (W) = M (W) – RHL (W) – HFTotal (W)
Data from the 3 conditions were compared using repeated measures analysis of variance for all variables. Post hoc analyses for significant differences between treatments were accomplished using Tukey's test. P<0.05 was used to identify statistically significant differences.
Results
Six male subjects were studied. They were 29±3 y old, 181±6 cm tall, weighed 84±18 kg, and had 2.0±0.2 m2 body surface area, 16±7% body fat, and 26±5 kg·m−2 body mass index.
CORE TEMPERATURE
No significant differences were found among the 3 conditions during the baseline and cold-water immersion periods. Baseline Tes was 37.4±0.2°C, and the core cooling rate was 2.4±1.7°C·h−1 (Figure 2). Likewise, there were no significant differences for either postcooling afterdrop (SW, 1.5±0.4°C; HW, 1.3±0.4°C; and TW, 1.2±0.3°C) or core rewarming rates during 60 to 120 min of warming (SW, 0.7±0.2°C·h−1; HW, 0.8±0.3°C·h−1; and TW, 0.8±0.3°C·h−1) (Figure 2). However, after 90 min of warming, Tes was significantly lower in the SW condition than in the TW condition at 90 min (P<0.05) and 120 min (P<0.01).
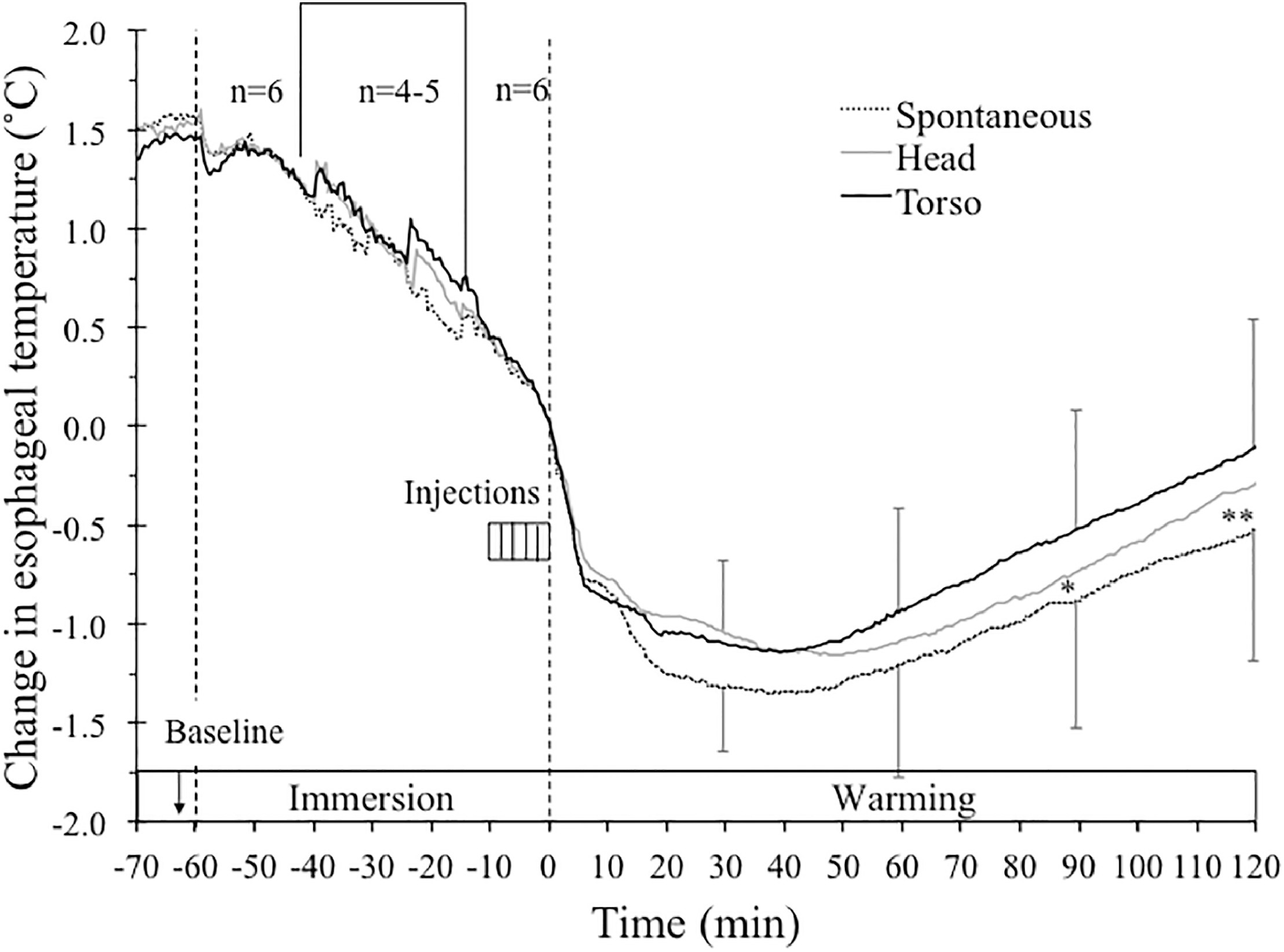
Mean change in esophageal temperature (°C) during baseline, up to 60 min of immersion in 8°C water, and 120 min of warming during spontaneous, head, and torso warming. Time 0 min and temperature 0°C indicates exit from the cold water (bars, SD). Only 3 subjects were immersed for the entire 60-min period in all conditions, with immersion times for the other 3 subjects ranging from 32.5 to 51 min. To show how the whole group responded at the beginning and the end of immersion, data for the immersions less than 60 min are presented for the first 20 min, with the remainder adjusted so that the exit time aligns for everyone at time 0 min. As a result, n=6 for data from −60 to −40 min and from −12.5 to 0 min. In the period between −40 and −12.5 min, n ranges from 4 to 5. For clarity, SD bars are only included for top and bottom lines. * Significantly lower in spontaneous warming than torso warming (90 min, P<0.05). ** Significantly lower in spontaneous warming than torso warming (120 min, P<0.01).
HEART RATE
No significant differences were found in the conditions during baseline, cooling, and warming periods. With all conditions combined, heart rate significantly increased during cooling from baseline values of 77±10 to 84±12 beats·min−1 just before the meperidine administration (P<0.05). During the warming periods heart rate significantly declined to 64±7 beats·min−1 (P<0.05).
SKIN TEMPERATURE AND HEAT FLUX
No significant differences were found among the 3 conditions for either skin temperature or heat flux during baseline and cooling periods. Total heat loss increased from the baseline values of 104±18 W to a peak value of 558±35 W during cold-water immersion. During rewarming, skin temperature responded in an inverse relation to heat flux for each condition; only heat flux data are presented in Figure 3.
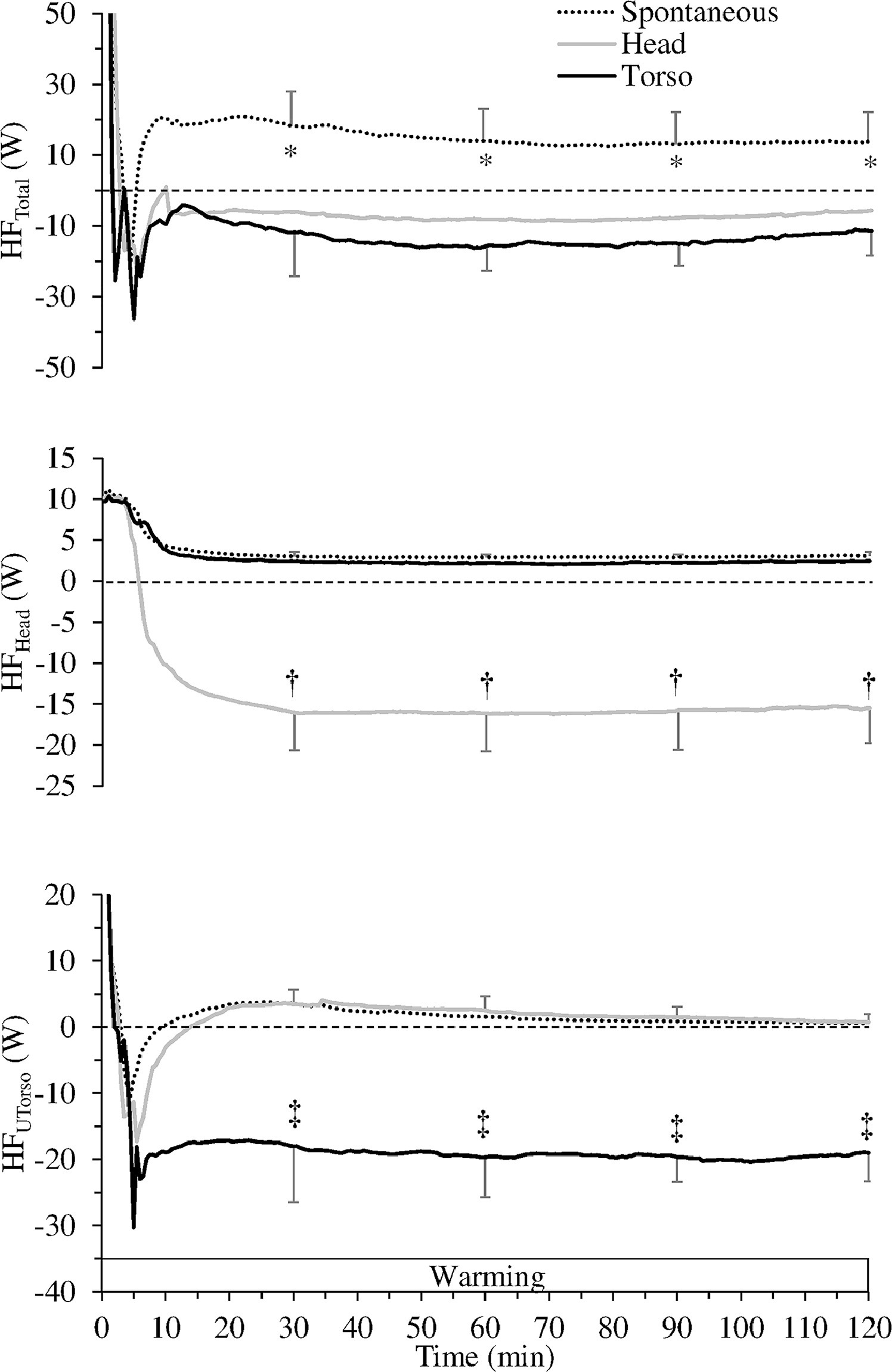
(Top) Total heat flux (HFTotal, W), (middle) head heat flux (HFHead), and (bottom) upper torso heat flux (HFUTorso) during 120 min of warming during spontaneous, head, and torso warming. *Spontaneous warming significantly different from head (P<0.05) and torso (P<0.001) warming. †Significantly greater heat gain during head warming (P<0.001). ‡Significantly greater heat gain during torso warming (P<0.001).
By the end of rewarming, TskTotal was significantly lower (P<0.01) for SW (32.8±0.8°C) than for TW (33.7±0.9°C), with neither condition being different from HW (33.4±0.6°C). This corresponded with HFTotal during SW (positive values indicating heat loss), which was significantly different from values in both HW (P<0.01) and TW (P<0.001) (negative values indicating heat gain) (Figure 3, top).
As expected, TskHead at 120 min of warming was significantly higher (P<0.001) in the HW condition (39±1.4°C) than both the SW (33.5±1.1°C) and TW (33.8±1.4°C; P<0.01) conditions. Consequently, head heat gain during HW was significantly different (P<0.001) from heat loss values for both TW and SW conditions (Figure 3, middle).
Conversely, TskUTorso at 120 min of warming was significantly higher (P<0.001) in the TW condition (37.1±0.5°C) than in both the SW (34.6±0.7°C) and HW (34.8±0.4°C) conditions. Upper torso heat gain during TW was significantly different (P<0.001) from heat loss values for both HW and SW conditions (Figure 3, bottom).
METABOLIC HEAT PRODUCTION
No significant differences were found for metabolic heat production among the conditions during baseline, cooling, and warming periods (Figure 4). With all conditions combined, metabolic heat production significantly increased from 98±13 W during baseline to 269±11 W (P<0.001) during cooling just before meperidine infusion. Meperidine suppressed shivering, and heat production returned to 94±32 W during the warming period.
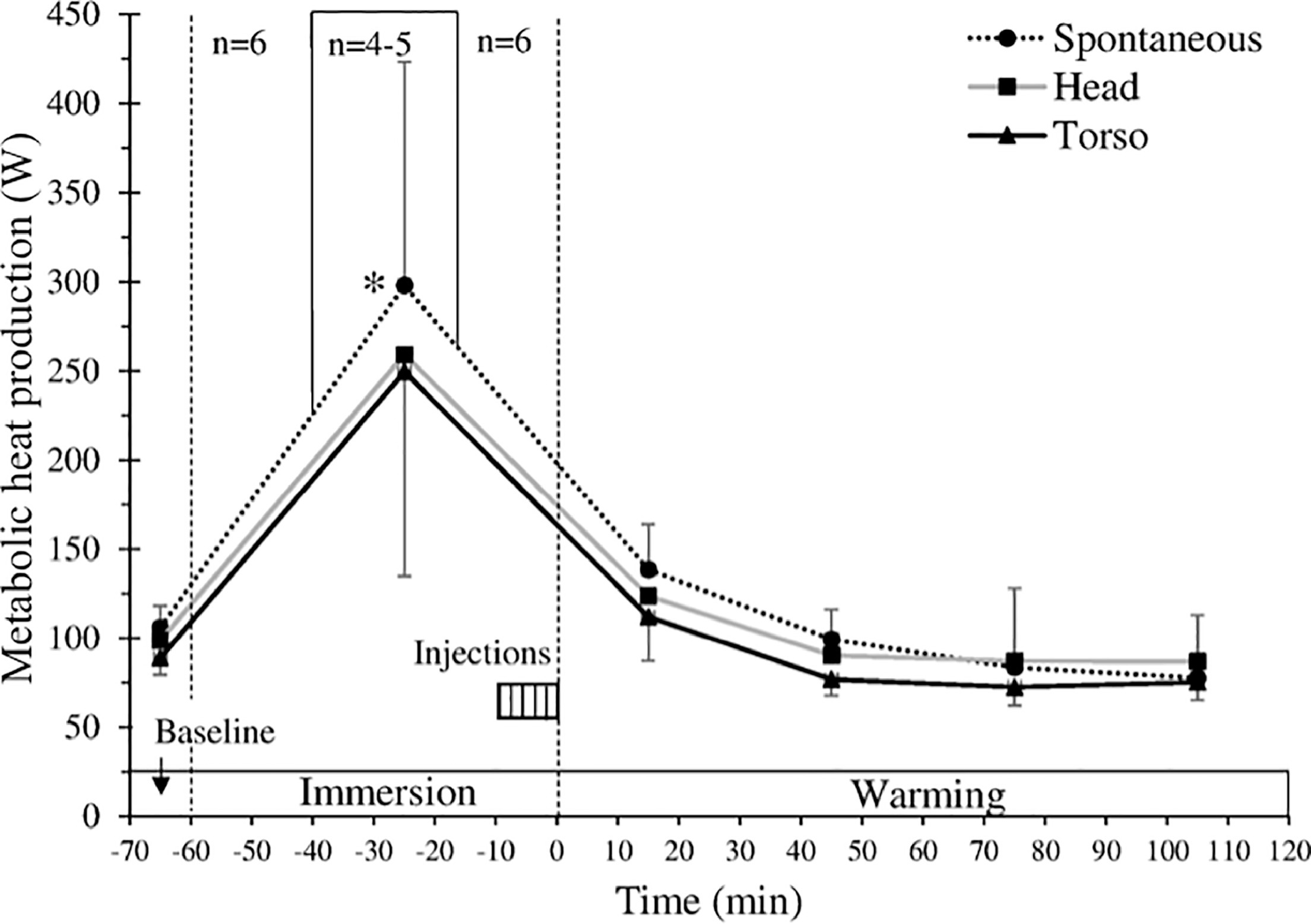
Metabolic heat production (W) during baseline, up to 60 min of immersion in 8°C water, and 120 min of warming during spontaneous, head, and torso warming. Time 0 min and temperature 0°C indicates exit from the cold water (bars, SD). Only 3 subjects were immersed for the entire 60-min period in all conditions, with immersion times for the other 3 subjects ranging from 32.5 to 51 min. To show how the whole group responded at the beginning and the end of immersion, data for the immersions less than 60 min are presented for the first 20 min, with the remainder adjusted so that the exit time aligns for everyone at time 0 min. As a result, n=6 for data from −60 to −40 min and from −12.5 to 0 min. In the period between −40 and −12.5 min, n ranges from 4 to 5. Each data point represents an average of 30 min except for the data point at −65 min, which represents the average for 10 min of baseline. For clarity, SD bars are only included for top and bottom lines. *Significantly greater during cooling than baseline and the entire warming period for all 3 conditions (P< 0.05).
NET HEAT GAIN
There were no significant differences among the 3 conditions during the baseline and cooling periods. During baseline, there was a net heat loss (−15±8 W), which increased rapidly during cold-water immersion to a peak value of −369±195 W. During the last 30 min of rewarming, net heat gain was significantly higher in HW (85.8±25.3 W) and TW (81.5±6.3 W) than for SW (56.9±12.0 W; P<0.05) (Figure 5).
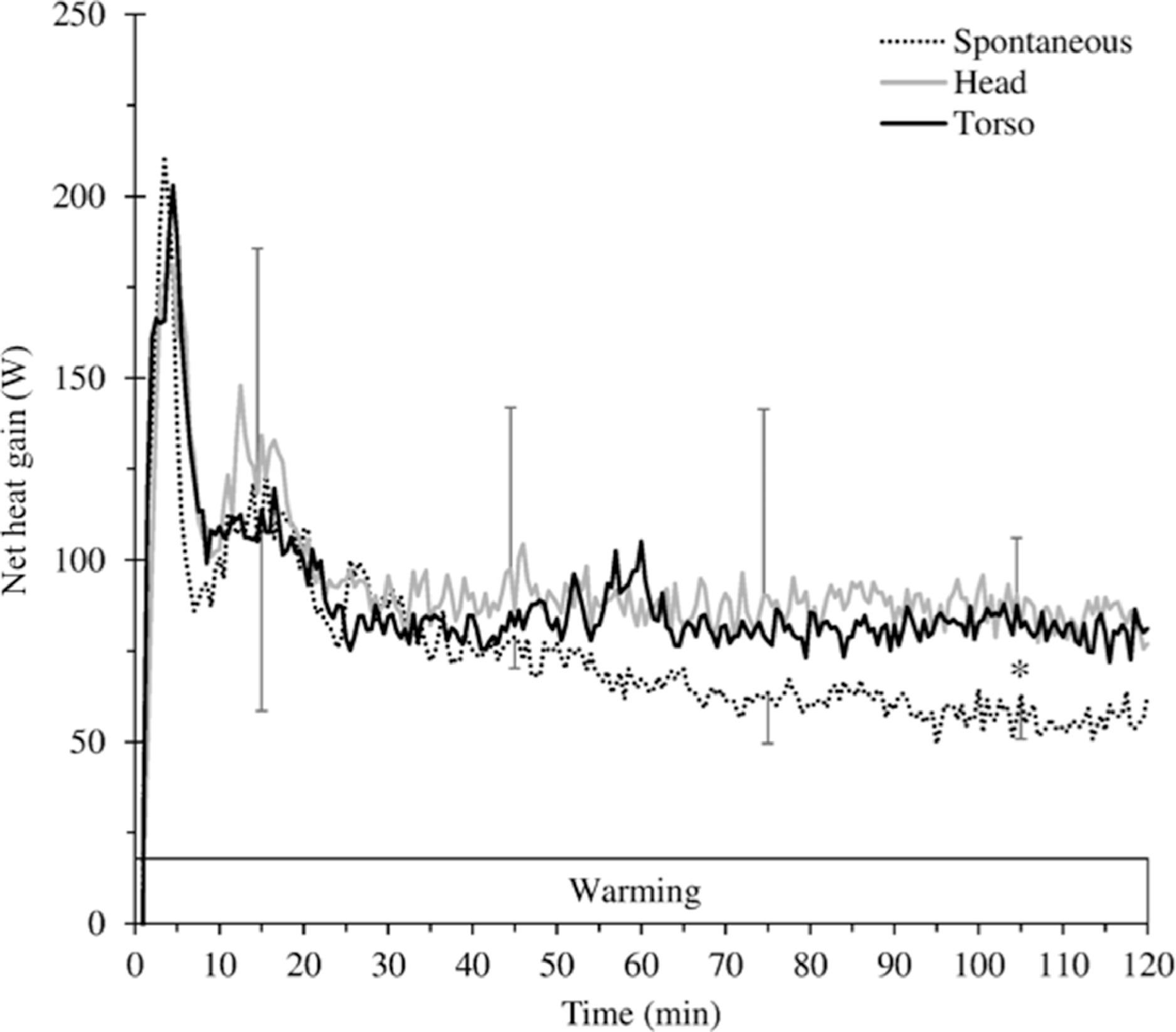
Net heat gain (W) during 120 min of warming during spontaneous, head, and torso warming. For clarity, SD bars are only included for top and bottom lines. *Significantly lower net heat gain during spontaneous warming than during head and torso warming (P<0.05) from 90 to 120 min.
Discussion
This study is the first known to compare the rewarming effectiveness of a similar amount of heat donated to the head or torso in mildly hypothermic subjects in whom shivering was pharmacologically inhibited. No significant differences were found between the 2 active warming or spontaneous (no heat) conditions for afterdrop and core rewarming rate, although TW and HW resulted in a significantly higher net heat gain compared with the SW condition. In the absence of shivering heat production, HW and TW provided a safe and steady rewarming rate (0.8°C·h−1). During the last 30 min of rewarming, there was a tendency for higher Tes with active heating, although this was only statistically significant for the TW condition. The positive aspect of these results is that the head provides a viable alternative for heat donation when TW is contraindicated, not only when shivering is intact 6 but also when shivering is absent.
POSSIBLE MECHANISMS FOR THE RESULTS
We expected that TW would result in lower afterdrop and a higher rate of core rewarming; this was not the case. In this study, the afterdrop values for the SW (1.5°C) and active warming (HW, 1.3°C; TW, 1.2°C) conditions were within the range previously reported for nonshivering subjects (1.4–2.2°C and 1.0–1.8°C, respectively).10,19 A previous study by Goheen et al 19 reported that a prototype forced-air warming system reduced the afterdrop amount by 30 to 40%. This may be due to the high source of heat used in their study (a high-powered forced-air warming system that provided up to 270 W) vs a net gain of 20 W in the present study provided by the charcoal heater on the torso.
In the SW condition, our subjects rewarmed at a higher rate (0.7°C·h−1) than reported in several of our previous studies (0.1–0.4°C·h−1). 10 ,19,21 The higher rate in this current study may partially relate to our cooling/meperidine-dosing protocol. In our initial shivering-inhibition study, 20 the core rewarming rate was 1.2°C·h−1 during the meperidine condition when metabolic heat production increased up to 42% from baseline values; this was because immersion in 8°C water for an average of 52 min resulted in enough core cooling (1.1°C) to provide too high a shivering stimulus to be overcome by our initial cumulative meperidine dose of 1.5 mg·kg−1. This was addressed in the subsequent study, in which shorter immersion (average of 26 min) resulted in less core cooling (∼0.4°C) and the cumulative meperidine dose was increased to 2.5 mg·kg−1. 19 In this case, the average Tes remained at or near nadir levels and did not return to the exit Tes even after 2.5 h of rewarming. In the current study, the immersion period (32–60 min) and core cooling (1.5°C) may have temporarily provided too great a shivering stimulus even though an even higher cumulative meperidine dose (3.3 mg·kg−1) was used. There was slightly higher, though nonsignificant, heat production during the first 30 min postimmersion (138 W) than during baseline (106 W). Although shivering heat production was completely eliminated during the last 90 min of SW, the initial values of heat production during the first 30 min may partially explain why rewarming was 0.7°C·h−1 during this condition.
It is noteworthy that when shivering heat production was eliminated, HW had similar core rewarming rates compared with TW even though TW provided more heat to a larger surface area (20 W to the torso vs 15 W to the head with HW). The similarity in rewarming rates could be due to the similar net heat gain throughout rewarming (Figure 5). Further work is needed to quantify tissue temperatures and blood flow during rewarming to confirm the mechanisms for these results.
PRACTICAL IMPLICATIONS
The Wilderness Medical Society practice guidelines for out-of-hospital evaluation and treatment of accidental hypothermia 3 recommends active external warming for severely hypothermic patients in whom shivering is absent. In the absence of active warming, core temperature can continue to drop and remain low for hours or longer because there is minimal spontaneous heat production to warm the core. 19 Because of the proximity to the heart, the torso is an effective area for applying external heat. In this study, we found that practically, it was easier to apply the charcoal heater to the torso than to the head. Also, once the torso is wrapped in a vapor barrier and a sleeping bag, heat loss from the heater to the environment would be less compared with HW.
Although TW was the preferred and a more comfortable method of providing active warming in a previous study, 6 HW does seem to provide a viable alternative to donate external heat to hypothermic subjects in whom TW may be contraindicated. This may occur when there is an open wound to the chest or emergency personnel are performing cardiopulmonary resuscitation on the patient. In some cases, the hypothermic patient is already insulated and wrapped in a sleeping bag, and thus gaining access to the torso to apply heat may be difficult or impractical. In such cases, application of heat to the head is recommended. This should apply to most sources of heat, including gel and dry chemical heat pads, warm water bladders, and electric heating pads. Future studies could address these other sources of heat.
LIMITATIONS
The study was open to both sexes, but no females volunteered. The lack of female participation was unlikely to affect our conclusions, however, because sex is not expected to influence the effect of skin heat donation on skin temperature, heat flux, or the relative rates of core rewarming between conditions. For example, we have recently evaluated data from a single female subject, who was shorter and lighter than her male counterparts, and found that this subject's results for metabolic heat production and net heat gain during cold exposure were within 0.05 to 1.4 standard deviations from the group mean (Dutta et al, unpublished manuscript currently in peer review, September 2018). Therefore, we believe our conclusion that HW is a viable alternative to TW is likely valid for both sexes, although a future study could address this specific question.
This study used a modified balanced design in which SW was done first for each subject to establish a standard meperidine dosing schedule. Thus, meperidine was given in response to shivering onset during SW but generally before shivering occurred during subsequent active warming trials. Therefore, heat production during the first 30 min of warming was slightly (though nonsignificantly) elevated during SW. Although this may have contributed to an SW rewarming rate that was higher than in our previous studies, we still observed a difference in core temperature between SW and TW from 90 to 120 min. This design is the standard protocol used in our previous studies, and we do not believe this affected our general conclusions that HW is a viable alternative to TW when applying heat to the torso is contraindicated.
A recent comparison of body surface area (BSA) formulae has revealed discrepancies in calculated results by as much as 0.5 m2. 25 First, this variability is greater for people with abnormal physiques or for children; therefore, potential errors with our estimates would likely be much less in our young healthy subjects. Second, the significance of errors in BSA calculation are primarily important in clinical situations such as cancer chemotherapy, transplantology, and burn treatment, where BSA may affect choice of treatment or drug dosage.
Finally, it is noteworthy that this human model for severe hyopthermia does not mimic all of the signs and symptoms of severe hypothermia (eg, decreased cardiorespiratory function and brain activity, multiple organ dysfunction). The purpose of meperidine injection is to abolish shivering such that a mildly hypothermic subject approximates moderate to severe hypothermia from a thermal perspective (eg, effects of heat donation are determined in a cold body in the absence of shivering heat production). Although it is impossible, or extremely difficult, to compare thermal results from our model to clinically moderate to severe hypothermia, we believe comparative results found in this study would be valid for actual nonshivering cold patients.
Conclusions
This study demonstrated that the external heat donation to the head and torso resulted in similar core rewarming rates. Although it is preferable to apply heat to the torso, heat donation to the head can be a beneficial alternative in severely hypothermic subjects in whom the application of heat to the torso is contraindicated; these results are consistent whether shivering is intact 6 or absent. A charcoal heater, which is light, portable, and easy to use, can be an effective modality to provide a safe and steady rate of core rewarming in severely hypothermic patients. The charcoal heater does not need any external power supply and can be used safely by laypersons or search and rescue personnel during rescue operations to provide prehospital warming to cold patients.
Footnotes
Acknowledgments
The authors thank the Natural Sciences and Engineering Research Council of Canada (NSERC) and the University of Manitoba Graduate Fellowship for providing financial support. They thank all the research volunteers for their participation in the study.
Author Contributions
Conception (KK, GG); design (KK, GG, SW, SP); data collection (EH, KK, RD, GG); data analysis (KK); data interpretation (KK, RD, GG, SW, GM, SP); manuscript draft and revision (GM, GG, SW).
Financial/Material Support
Natural Sciences and Engineering Research Council of Canada (NSERC), and The University of Manitoba Graduate Fellowship.
Disclosures
None.
Supplementary materials
Supplementary material associated with this article can be found in the online version at doi:
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
