Abstract
Objective
To examine the effects of nicotine on the metabolic and hormonal responses during acute cold exposure.
Methods
Participants in this study included 6 men and 5 women between the ages of 19 and 25 years. Each subject performed 2 cold-air trials (CATs) consisting of a 30-minute baseline (BASE) period and a 120-minute exposure to 10°C air. One CAT was performed after a nicotine (NIC) dosing using a 21-mg transdermal patch, whereas the other CAT was performed after a placebo (PL) treatment. Blood samples for metabolic and hormonal measurements were obtained at the end of BASE and immediately after the cold exposure.
Results
When examining the sexes separately, there was no difference in norepinephrine between PL and NIC (P = .066). There was also no difference in epinephrine between PL and NIC in either sex (P = .634). From BASE to 120 minutes of the CAT, there was a significant decrease in cortisol (P = .036), but this response was similar between the 2 treatments (P = .077). Glucose and glycerol concentrations were not different between the PL and NIC treatments. At BASE, nonesterified fatty acid (NEFA) concentration was lower during PL compared with NIC (P = .021); however, at 120 minutes of the CAT, NEFA was greater during PL compared with NIC (P = .035).
Conclusions
During 120 minutes of cold exposure, NIC resulted in alterations in the responses in NEFA, whereas the other blood measurements were not significantly different between the 2 groups.
Keywords
Introduction
The diversity of humans has long been characterized by their ability to adapt to various surroundings and environmental conditions. Many of these conditions offer physiological challenges to the body's ability to maintain homeostasis. One of the specific environmental conditions that poses a challenge to humans is cold exposure. Cold exposure, both acute and long-term, requires physiological adjustments in order to maintain a relatively stable body temperature. The 2 main mechanisms by which humans respond to cold exposure are via an increase in peripheral vasoconstriction and an increase in metabolic heat production (M) mainly caused by shivering. 1 The increases in peripheral vasoconstriction and M observed during cold exposure result in alterations in the metabolic and hormonal responses during acute cold exposure. Cold exposure results in an increase in sympathetic nervous system and sympathoadrenal activity, thereby causing changes in the levels of circulating catecholamines and possibly glucocorticoids. In addition, the cold-induced increase in M necessitates an increase in the absolute amount of metabolic substrates oxidized for energy and may also result in a preferential shift toward carbohydrate oxidation. Therefore, the combination of these processes may elicit substantial changes in the metabolic and hormonal responses during cold exposure.
In examining the hormonal responses to cold exposure, most studies have shown plasma norepinephrine concentration to increase 2 –9 and little or no change in plasma epinephrine concentration.4,6–9 Although some studies have observed increases in plasma cortisol concentration in response to cold exposure,5,10 most research has reported no change or a decrease.9,11–14
Studies have also examined both the changes in circulating substrate concentrations as well as the rate of utilization of various substrates during cold exposure compared with neutral conditions. Plasma glucose concentration has been shown to increase,15,16 decrease, 17 and stay the same 18 during exposure to the cold compared with thermoneutral conditions. In addition, carbohydrate oxidation has been shown to increase during cold exposure compared with thermoneutral conditions. 19 Much less information is available on the effect of cold exposure on lipid metabolism during rest. However, plasma free-fatty acid concentration has been shown to increase7,15,18 and remain the same 17 during cold exposure.
Any exogenous compound with the potential to alter the 2 main avenues of cold-induced thermoregulation (peripheral vasoconstriction and increase in M) may, in turn, alter the metabolic and hormonal responses to cold exposure. One such compound that may affect the physiological responses during prolonged cold exposure is nicotine (NIC). Previously, the investigators have examined the influence of NIC on the thermoregulatory responses during 120 minutes of exposure to 10°C air. 20 There was no difference in rectal or skin temperature between the placebo (PL) and NIC conditions in men or women. However, in the men, M was significantly greater and tissue insulation was significantly lower during the PL treatment compared with the NIC treatment. Lastly, skin vascular conductance was significantly lower during the PL treatment compared with the NIC treatment.
At NIC dosages comparable with those observed during smoking, NIC has been shown to increase sympathetic nervous system activity and increase the circulating concentrations of norepinephrine and epinephrine. 21 Research has also examined the effects of NIC on plasma cortisol concentrations during rest. Some studies have reported no effects of smoking on plasma cortisol concentrations 22 ; however, other studies have observed increases in plasma cortisol concentration after NIC administration. 23 –26
Plasma glucose concentrations have been shown to be similar 23 and higher 27 as a result of NIC administration. Plasma free-fatty acid concentration as well as plasma glycerol concentration, a better indicator of lipolysis, have also been shown to increase as a result of NIC administration.23,28
No previous studies have examined the effects of NIC on the metabolic and hormonal responses during cold exposure in humans. It is unclear as to whether the effects of NIC on the metabolic and hormonal responses during thermoneutral conditions would be similar during cold conditions. Also, because about 22.5% of adults aged 18 years and older 29 and 30% of military personnel are classified as current smokers, 30 and because of the potential for these populations to be exposed to the cold, there is a need to examine the effects of NIC on the various physiological systems involved in temperature regulation. Therefore, the purpose of this study is to examine the effects of NIC on the metabolic and hormonal responses during 120 minutes of exposure to 10°C air.
Methods
Subjects and Design
Eleven participants (6 men and 5 women) served as subjects in this study. Physical characteristics for the men and women, respectively, included the following: age, 22.6 ± 0.4 years and 21.1 ± 0.7 years (SE); height, 178.1 ± 2.0 cm and 165.2 ± 3.4 cm; weight, 81.3 ± 7.6 kg and 59.4 ± 3.6 kg; body surface area, 1.98 ± 0.08 m2 and 1.65 ± 0.07 m2; and body fat percentage, 12.9 ± 2.1% and 19.3 ± 2.0%. All subjects were current smokers, with men and women averaging 19 ± 2 cigarettes and 17 ± 2 cigarettes per day and an average smoking duration of 4.0 ± 0.7 years and 4.7 ± 1.1 years. All subjects smoked cigarettes with a NIC content of 0.8 mg. All subjects were healthy, free of any known disease, and were not taking any medication that may have affected the physiological responses to cold exposure. The Institutional Review Board of Kent State University approved the protocol, and informed consent was obtained in accordance with guidelines set forth by the Board.
Each subject visited the laboratory on 3 separate occasions. During the first visit, anthropometric variables were assessed. On the second and third visits, a cold-air trial (CAT) was performed. One CAT was performed after administration of a NIC dosing, whereas the second CAT was performed without a NIC dosing but with a placebo (PL). The order of the CATs was counterbalanced. All subjects were instructed to abstain from alcohol and physical activity for the 24-hour period immediately preceding each visit to the laboratory. In addition, each subject was instructed to abstain from eating and caffeine for 12 hours and from smoking for 15 hours before each trial. Women were tested during the follicular phase of the menstrual cycle to control for any confounding effects of menstrual phase on temperature regulation. Follicular phase of the menstrual cycle was defined as days 1 through 8 of the menstrual cycle with day 1 representing the onset of menses. Each subject performed all trials at the same time of day and completed testing within a 4-week period. At least 48 hours separated all trials.
Anthropometric Assessment
Anthropometric variables were measured during the first visit to the laboratory. Height and weight were measured with a stadiometer and a balance-beam scale, respectively. Skinfold thickness was measured at 7 sites (chest, tricep, subscapular, suprailiac, abdomen, thigh, midaxillary) with Harpenden skinfold calipers (British Indicators LTD, West Sussex, England). Each site was measured 3 times, with the median of the 3 measurements taken as the skinfold thickness. Body density was calculated by using the equations of Jackson and Pollock, 31 and percent (%) body fat was calculated by using the Brozek equation. 32 Body surface area was calculated from height and weight according to the formula of DuBois and DuBois. 33
Cold-Air Trial
Each subject completed a protracted CAT on the second and third visits to the laboratory. The protracted CAT consisted of a 30-minute baseline (BASE) period and 120 minutes of cold-air exposure. Subjects were dressed only in shorts (men) or shorts and an athletic bra (women). Upon arrival to the laboratory, smoking abstention was confirmed by measurement of tidal volume carbon monoxide levels (BreathCO, Vitalograph, Kansas City, MO). This procedure required that subjects maximally inspire, hold the breath for as long as possible, and then slowly expire into the carbon monoxide analyzer. A carbon monoxide level of less than or equal to 13 ppm was used as an indication of smoking abstention. 34 All subjects for all trials met this criterion.
After the confirmation of smoking abstention, subjects were instructed to void and body weight was measured. Subjects were then instrumented with equipment to measure the thermoregulatory responses to cold exposure. These results have been previously reported, and a complete description of these methodologies may be found elsewhere. 20
After instrumentation of the subjects was completed, the subjects sat quietly for 30 minutes in 25.5°C ambient air for the BASE period. At the end of BASE, a 20-mL venous blood sample was obtained and centrifuged, and the plasma was stored in a −120°C freezer for subsequent analysis.
After completion of BASE, subjects were transported in the semirecumbent chair to the environmental chamber. Subjects sat with arms and legs separated and extended for 120 minutes of exposure to 10°C air. The CAT was terminated if rectal temperature was lower than 36°C; however, no subject met this termination criterion. At the end of the CAT, a second blood sample was obtained for subsequent analysis, and subjects were then escorted to a warm shower for rewarming.
NIC Dosing and Determination
For the protracted CAT, a NIC dosing or a PL was administered 8 hours before arrival to the laboratory with a 21-mg transdermal patch (NicoDerm CQ, SmithKline Beecham, Pittsburgh, PA) or a PL patch made of adhesive foam similar in appearance to the NIC patch. Plasma NIC levels have been shown to peak and plateau approximately 8 hours after placement on the body; therefore, subjects would begin the CAT during this plateau period to minimize fluctuations in NIC levels during the trial.35,36 Each subject was given a packet containing a NIC or PL patch, an alcohol pad, gauze, a razor, and an instructional sheet. Eight hours before each CAT, the subjects were instructed to shave an area on the upper arm, clean and dry the area, and affix the patch to the upper arm, making sure to attain maximal adhesion. The patch remained on each subject until termination of each CAT.
Blood Analysis
At BASE and 120 minutes of the CATs, blood samples were obtained for the measurement of plasma NIC, norepinephrine, epinephrine, cortisol, glucose, glycerol, and nonesterified fatty acid (NEFA) concentrations. A 20-mL venous blood sample was obtained from a venipuncture and placed into 2 heparinized vacutainers and 1 EDTA vacutainer. One heparinized vacutainer contained 100 μL of glutathione and EGTA for the stabilization of plasma catecholamines. Before centrifuging, a 0.5-mL blood sample from a heparinized vacutainer was removed for the measurement of hemoglobin and hematocrit levels, which were used to correct blood concentrations for changes in plasma volume. All blood samples were then centrifuged at 1500 g (Centra-7R, International Equipment Company, Needham Heights, MA), and the plasma or serum was separated and frozen at −120°C until analysis at the completion of the study.
To measure hematocrit, blood samples were collected in microcapillary hematocrit tubes (∼30 μL) in duplicate and subsequently centrifuged (Model CL, Arthur H. Thomas Company, Philadelphia, PA) for 9 minutes to separate red blood cells and blood plasma. Hemoglobin concentration was measured in duplicate using the cyanmethemoglobin procedure (Kit #525-A, Sigma Diagnostics Inc, St Louis, MO). 37
Plasma NIC concentration was measured by an independent laboratory (Labstat Inc, Kitchener, Ontario, Canada) using a nitrogen gas chromatography technique based on the methods of Jacob et al.38,39 This technique involves liquid-liquid extraction of the sample matrix together with structural analogues of NIC, which are used as internal standards. Analysis of NIC concentration was determined by Varian 3400 gas chromatographs (Varian Inc, Palo Alto, CA), which couple capillary column technology with split-splitless injection and thermionic specific detection (nitrogen selective). Chromatographic editing and results were handled by Varian Star software.
Plasma catecholamine concentrations were also measured by an outside laboratory (Walter Reed Army Institute of Research, Washington, DC). Isolation of plasma norepinephrine and epinephrine from human plasma was accomplished by alumina extraction using a Chromsystems reagent kit (Alko Diagnostics, Holliston, MA). Once extracted, plasma norepinephrine and epinephrine concentrations were quantified by high-performance liquid chromatography (Waters HPLC system, Waters Corp, Milford, MA).
Plasma cortisol concentration was measured in duplicate by radioimmunoassay (Corti-Cote, ICN Pharmaceuticals, Orangeburg, NY).
Plasma glucose concentration was measured in duplicate by the Trinder enzymatic method (Kit #315-100, Sigma Diagnostics Inc). 40 Glucose concentration was calculated based on the absorbance of the sample, a standard (300 mg·dL−1), and a reagent blank (deionized water).
Plasma glycerol concentration was measured in duplicate by using a modification of the enzymatic method described by McGowan et al 41 (Kit #337-A, Sigma Diagnostics Inc). Glycerol concentration was calculated based on the absorbance of the sample, a standard (2.82 mmol·L−1), and a reagent blank (deionized water).
Plasma NEFA concentration was measured in duplicate by an enzymatic method (Kit #994-75409, Wako Chemicals USA Inc, Richmond, VA).
Statistical Analysis
Differences in anthropometric variables and smoking status between men and women were assessed by an independent samples t test. A 3-way analysis of variance with repeated measures was used to determine statistical differences in the metabolic and hormonal variables as well as the plasma NIC concentrations. The factors used in the analysis were sex (men vs women), treatment (PL vs NIC), and time, with treatment and time being repeated measures factors. Significance was set a priori at P ≤ .05. In instances of a significant interaction, a simple effects analysis was used to examine specific contrasts, and statistical significance was determined by the Bonferroni adjustment. All values are expressed as mean ± SE.
Results
Men were significantly greater than women in height, weight, and body surface area (P = .008, .038, and .014). Additionally, men tended to be lower in % body fat (P = .059). There was no difference in average number of cigarettes smoked per day (P = .525) or smoking duration (P = .592), and all subjects smoked cigarettes with a NIC content of 0.8 mg per cigarette. These findings confirmed that there was no difference in the magnitude of NIC intake between men and women.
Plasma NIC concentration at BASE and 120 minutes in both men and women during PL and NIC treatments is presented in Table 1. As expected, plasma NIC concentration was lower during the PL treatment compared with the NIC treatment (P < .001). During the NIC treatment, both men and women exhibited greater plasma NIC concentrations at BASE compared with 120 minutes of the CATs (P < .001). Although plasma NIC concentrations tended to be greater in men compared with women during the NIC treatment, this difference did not reach statistical significance.
Plasma nicotine (NIC) concentration (ng·mL−1) at baseline (BASE) and 120 minutes of cold exposure in men (n = 6) and women (n = 5) during placebo (PL) and NIC treat ments (mean ± SE)
Plasma Norepinephrine
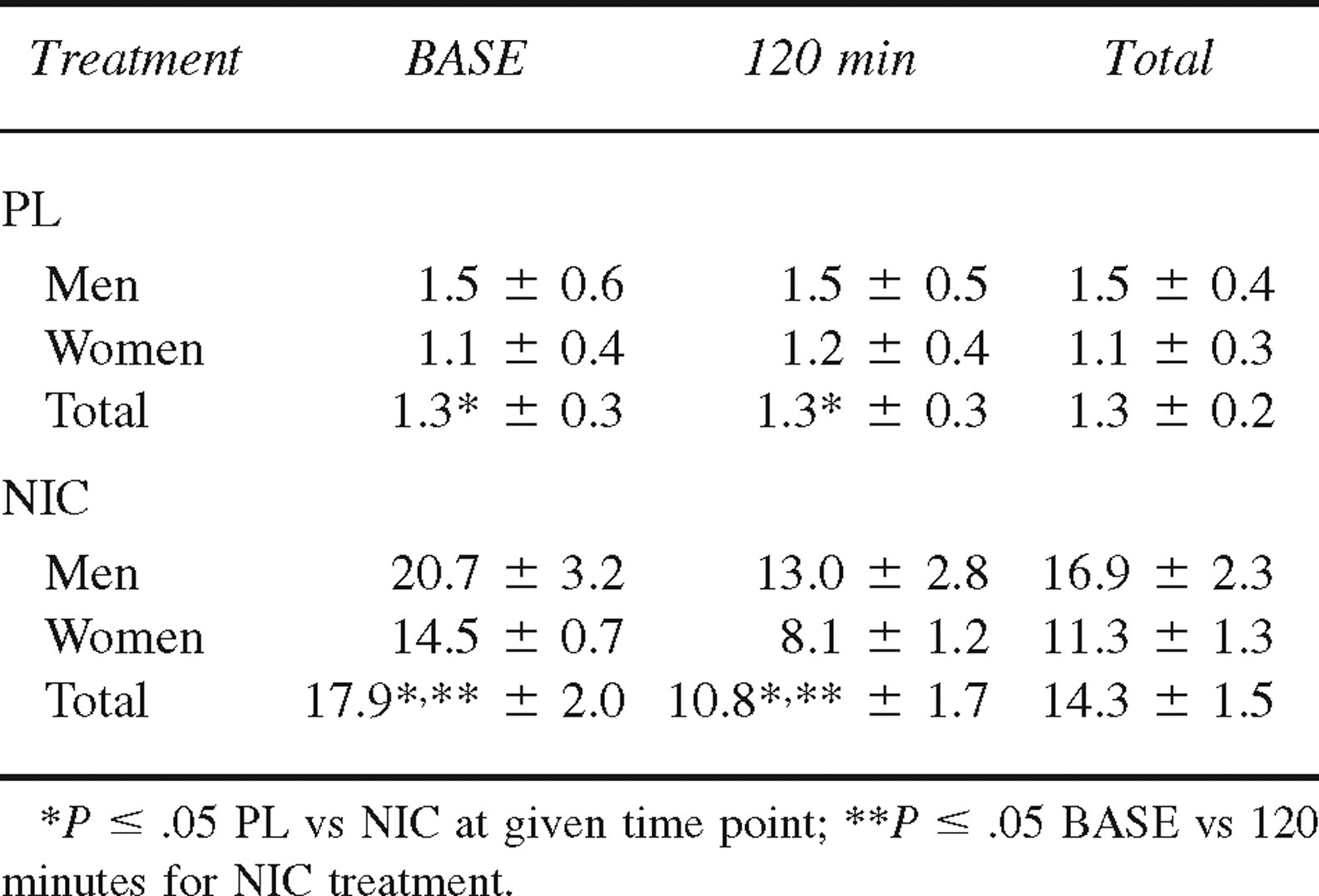
Plasma norepinephrine concentration at BASE and 120 minutes in both men and women during PL and NIC treatments is presented in Table 2. There was no overall difference in plasma norepinephrine concentration between men and women during the CATs (4.18 ± 0.57 nmol·L−1 and 4.94 ± 0.81 nmol·L−1; P = .254). In addition, there was no difference in plasma norepinephrine concentration between the PL and NIC treatments (P = .472) when grouping the sexes together. However, when examining the sexes separately, the sex-by-treatment interaction approached statistical significance (P = .066) (men: PL = 4.73 ± 0.87 nmol·L−1, NIC = 3.62 ± 0.74 nmol·L−1; women: PL = 3.68 ± 1.09 nmol·L−1, NIC = 5.21 ± 1.26 nmol·L−1). From BASE to 120 minutes of the CATs, the increase in plasma norepinephrine concentration was significant (P < .001), and this response was similar between the PL and NIC treatments.
Plasma norepinephrine (nmol·L−1), epinephrine (pmol·L−1), and cortisol (nmol·L−1) concentrations at baseline (BASE) and 120 minutes of cold exposure in men (n = 6) and women (n = 5) during placebo (PL) and nicotine (NIC) treatments (mean ± SE)
Plasma Epinephrine
Plasma epinephrine concentration at BASE and 120 minutes in both men and women during PL and NIC treatments is presented in Table 2. Overall, there was no difference in plasma epinephrine concentration between men and women (340 ± 30 pmol·L−1 and 210 ± 20 pmol·L−1; P = .080) or between the PL and NIC treatments (P = .634). There was also no significant change in plasma epinephrine concentration from BASE to 120 minutes of the CATs during the PL or NIC treatments (P = .515).
Plasma Cortisol
Plasma cortisol concentration at BASE and 120 minutes in both men and women during PL and NIC treatments is presented in Table 2. There was no difference in plasma cortisol concentration between men and women during the CATs (600 ± 30 nmol·L−1 and 470 ± 30 nmol·L−1; P = .077). There was also no difference in plasma cortisol concentration between the PL and NIC treatments (P = .547). Overall, plasma cortisol concentration was significantly higher at BASE compared with 120 minutes of the CATs (P = .036), and this response was similar for the PL and NIC treatments (P = .077).
Plasma Glucose
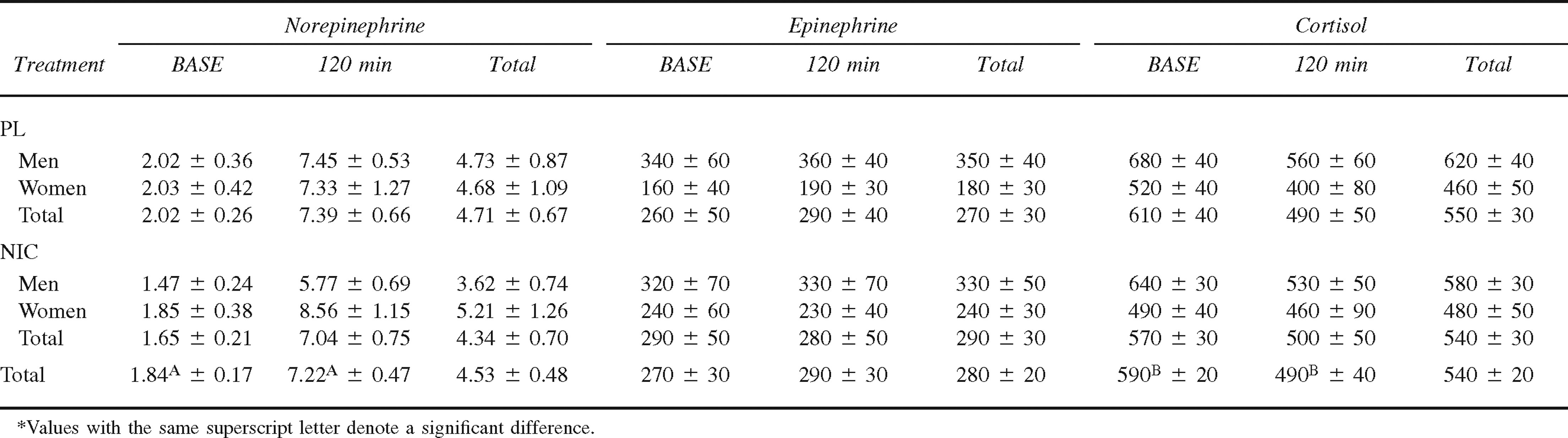
Plasma glucose concentration at BASE and 120 minutes in both men and women during PL and NIC treatments is presented in Table 3. Overall, there was no difference in plasma glucose concentration between men and women during the CATs (5.4 ± 0.2 mmol·L−1 and 5.3 ± 0.2 mmol·L−1; P = .814) or between the PL and NIC treatments (P = .075). Overall, plasma glucose concentration was significantly higher at BASE compared with 120 minutes of the CATs (P < .001), and this response was similar for the PL and NIC treatments (P = .078).
Plasma glucose (mmol·L−1), glycerol (mmol·L−1), and nonesterified fatty acids (NEFA) (mmol·L−1) concentrations at baseline (BASE) and 120 minutes of cold exposure in men (n = 6) and women (n = 5) during placebo (PL) and nicotine (NIC) treatments (mean ± SE)
Plasma Glycerol
Plasma glycerol concentration at BASE and 120 minutes in both men and women during PL and NIC treatments is presented in Table 3. Overall, there was no difference in plasma glycerol concentration between men and women during the CATs (0.12 ± 0.01 mmol·L−1 and 0.13 ± 0.01 mmol·L−1; P = .634). In addition, there was no difference in plasma glycerol concentration between the PL and NIC treatments (P = .729). From BASE to 120 minutes of the CATs, the increase in plasma glycerol concentration was significant (P = .007), and this response was similar between men and women and between the NIC and PL treatments (P = .628).
Plasma NEFA
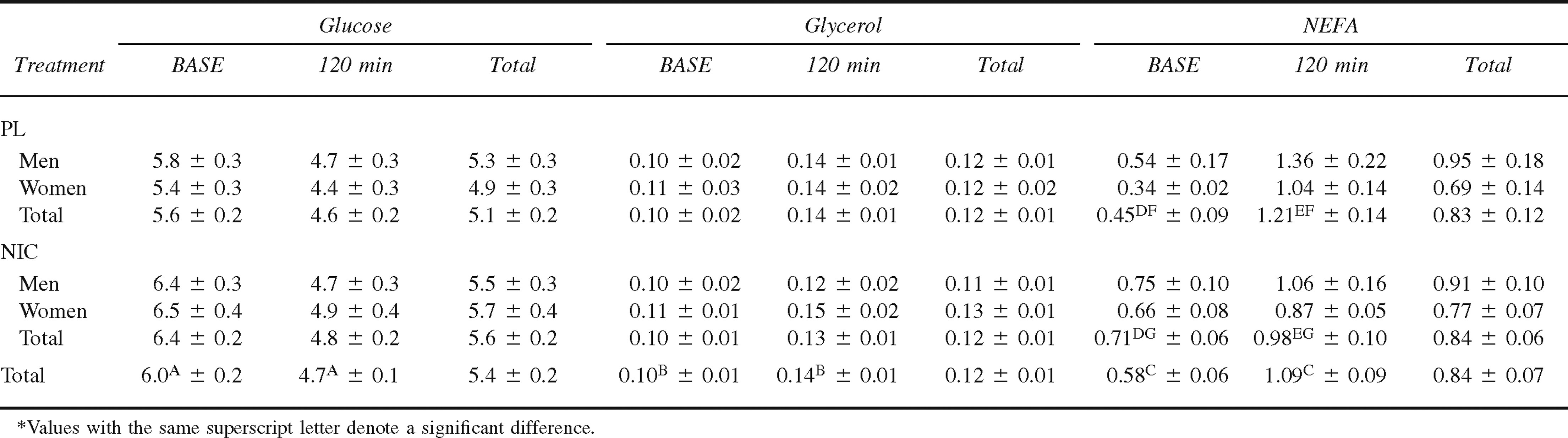
Plasma NEFA concentration at BASE and 120 minutes in both men and women during PL and NIC treatments is presented in Table 3. Overall, there was no difference in plasma NEFA concentration between men and women during the CATs (0.93 ± 0.10 mmol·L−1 and 0.73 ± 0.07 mmol·L−1; P = .240). There was also no difference in plasma NEFA concentration between the PL and NIC treatments (P = .825). Plasma NEFA concentration was significantly lower at BASE compared with 120 minutes of the CATs (P < .001). The treatment-by-time interaction is illustrated in the Figure (P = .002). At BASE, plasma NEFA concentration was significantly lower during the PL treatment compared with the NIC treatment (P = .021); however, at 120 minutes of the CAT, plasma NEFA was significantly greater during the PL treatment compared with the NIC treatment (P = .035). Additionally, plasma NEFA concentration was significantly greater at 120 minutes of the CAT compared with BASE during both the PL and NIC treatments (P < .001 and P = .009, respectively).

Treatment-by-time interaction for plasma non-esterified fatty acid concentration during cold air exposure (M ± SE) (P = 0.002). * Denotes significant difference between placebo and nicotine at specific time point (BASE, P = 0.021; 120 min, P = 0.035). † Denotes significant difference between BASE and 120 min for specific treatment (Placebo, P < 0.001; Nicotine = 0.009)
Discussion
The purpose of this study was to investigate the effects of NIC on the metabolic and hormonal responses during 120 minutes of exposure to 10°C air. As previously stated, the main mechanisms by which the body attempts to maintain temperature homeostasis during cold exposure is an increase in peripheral vasoconstriction and an increase in M, both of which are influenced by sympathetic nervous system activity. Nicotine increases peripheral vasoconstriction and resting metabolic rate by stimulating the sympathetic nervous system. 42 Because of the influence of NIC on the primary mechanisms involved in temperature regulation, it was hypothesized that NIC may also affect the metabolic and hormonal responses observed during cold exposure. To date, research has not examined the influence of NIC on these responses during cold exposure (air or water) in humans.
In response to the cold exposure, plasma norepinephrine concentration increased approximately 4-fold from BASE to 120 minutes of the CATs. This increase in concentration is in agreement with several studies that utilized the same temperature and length of exposure as the present study.7,9 During the CATs, there was no difference in plasma norepinephrine concentration between the sexes, which is also in agreement with previous studies. 43 –45
Overall, plasma norepinephrine concentrations were similar between the PL and NIC treatments. Although some studies have previously shown no change in plasma norepinephrine concentration after smoking,26,46 the consensus is that smoking does increase circulating levels.23,42,47,48 Therefore, the finding of a similar response in plasma norepinephrine concentration between the PL and NIC treatments may be due to the mode of NIC administration or the degree of thermal stress imposed. Studies examining the effects of the transdermal NIC patch on circulating catecholamine levels have reported no change in plasma norepinephrine concentration 49 or a moderate increase after repeated applications. 50 In the present study, plasma norepinephrine concentration at BASE was similar between the PL and NIC treatments. Additionally, the increase in plasma norepinephrine concentration as a result of cold exposure (∼300%) is of a far greater magnitude than that observed as a result of NIC administration (∼30%). Therefore, any changes in plasma norepinephrine concentration resulting from the NIC treatment could have been masked by the severity of the thermal stress imposed.
Although there was no overall effect of NIC on plasma norepinephrine concentration, there was a tendency for a differential response between the sexes (P = .066). In men, plasma norepinephrine concentration was 23% lower during the NIC treatment compared with the PL treatment, whereas women exhibited similar values under both treatments. From the present study, it is impossible to conclude whether or not this difference would be reproducible, and thus future research should reexamine this topic. There are some possible mechanisms that may explain a differential response in the sexes. Nicotine administration increases energy expenditure more during mild physical activity (which cold-induced shivering may mimic) than during rest, especially in men, 51 which may decrease the reliance on sympathetic nervous system activity to maintain temperature homeostasis. In addition, the differential response between men and women may be attributed to dosing differences. It has been reported that plasma NIC concentration in response to the administration of a transdermal NIC patch is strongly correlated to body weight. 52 Therefore, the absolute dose of NIC administered in the present study may have resulted in a smaller dose response in the men and contributed to the differential response, though the men did exhibit higher NIC concentrations from the transdermal patch.
Conclusions on the relationship between NIC administration and sympathetic nervous system activity during cold exposure are difficult to make based on plasma norepinephrine concentrations alone. Blood levels of any hormone are affected by rate of addition and removal from the bloodstream. Therefore, any changes observed could be due to changes in clearance rather than changes in production.
There was no change in plasma epinephrine concentration between BASE and 120 minutes of the CATs, which is in agreement with previous research.4,6–9 In addition, the values obtained in the present study were similar to previous studies utilizing the same temperature and length of exposure.7,9 It has been suggested that plasma epinephrine concentration does not increase during cold exposure because cold exposure does not result in a generalized stress response within the body but rather a selective stimulation of the sympathetic nervous system. 8
Overall, there was no difference in plasma epinephrine concentration between the PL and NIC treatments. Previous research has reported smoking-induced increases in plasma epinephrine concentration23,26,46,48 primarily due to the stimulatory effects of NIC on the sympathoadrenal system. 42 However, the lack of change in plasma epinephrine with NIC administration in the present study may be due to the severity of the cold stress masking any change associated with NIC administration.
In the present study, plasma cortisol concentration was significantly lower at 120 minutes of the CATs compared with BASE, with the decrease being approximately 17%. In conditions similar to the present study, Leppaluoto et al 9 observed a 20% decrease after the cold exposure compared with the thermoneutral value. Previous research has also reported a decrease or no change in plasma cortisol concentration in response to cold exposure. 11 –14 It has been suggested that cold exposure elicits a selective stimulation of the sympathetic nervous system rather than a general stress response. 8 Therefore, the stress of cold exposure may not elicit changes in cortisol levels. In addition, though not all CATs were conducted at the same time of day, the majority of the trials were conducted in the morning hours, when plasma cortisol concentrations have been shown to be declining. 9
Overall, there was no difference in plasma cortisol concentration between the PL and NIC treatments. Previous research has reported plasma cortisol concentrations to be similar 22 and higher 23 –26 after NIC administration compared with PL or abstention conditions. The discrepant results in previous studies are most likely due to differences in the amount of NIC administered. The increases in plasma cortisol levels after NIC administration that have been reported may be due to the direct effects of NIC on the hypothalamus and the subsequent increases in corticotropin. 24
The increase in heat production to maintain core temperature during cold exposure requires an increase in the absolute amount of metabolic substrates that are oxidized for energy. In addition to the increases in substrate oxidation, cold exposure may be met with alterations in substrate utilization because of the hormonal adjustments required to maintain temperature homeostasis.
Overall, plasma glucose concentration decreased 22% from BASE to 120 minutes of the cold exposure. Previous research has reported plasma glucose concentration to be higher, 15 lower, 17 and similar 18 between thermoneutral and cold conditions. The mixed results are most likely attributable to differences in the severity of thermal stress and the failure of some studies to correct for cold-induced reductions in plasma volume. The decrease in plasma glucose concentration observed during the present study may be attributed to the large increase in absolute carbohydrate oxidation observed during cold exposure 19 and the hormonal responses observed. During the present study, there was no change in plasma epinephrine concentration, which has been shown to increase glycogenolysis. 53 Additionally, plasma cortisol concentration, which increases blood glucose levels, was significantly lower after 120 minutes of the cold exposure compared with BASE. Therefore, the coupling of an increase in absolute carbohydrate oxidation and the decrease in hormonal stimulation of glucose release or formation most likely led to the decrease over time.
Plasma glycerol and NEFA concentrations increased 40% and 88%, respectively, from BASE to 120 minutes of the cold exposure. Previous research with similar or identical conditions compared with the present study have also reported similar increases.7,9,18 The increases in plasma glycerol and NEFA concentrations observed during cold exposure are likely caused by increases in the rates of lipolysis. 7 The increase in lipolysis observed during the present study may be attributed to the cold-induced increase in plasma norepinephrine concentration because plasma epinephrine concentration was unchanged. Additionally, it has been shown that the increase in NEFA mobilization during cold exposure exceeds the absolute increase in NEFA oxidation. 7 Also, carbohydrate oxidation has been reported to be the major source of energy during cold exposure. 19 Therefore, because lipolysis increases and exceeds NEFA oxidation, and because carbohydrate oxidation is the primary source of energy for shivering muscles, it would seem logical that NEFA concentration would increase during cold exposure.
As expected, there was no difference in plasma glucose, glycerol, and NEFA concentrations between men and women. To date, no research has examined the effects of sex differences on circulating metabolic substrates during cold exposure. However, similar levels of substrate oxidation and plasma catecholamine concentrations during cold exposure have been reported between men and women. 47 Therefore, it would appear to be unlikely that any sex differences with respect to circulating levels of metabolic substrates would be observed.
Overall, plasma glucose concentration was similar during the NIC treatment compared with the PL treatment. Plasma glucose has been shown to be similar 23 and higher 27 after NIC administration. The increases in circulating levels of plasma glucose as a result of NIC administration are primarily due to the stimulation of the sympathoadrenal system by NIC and the subsequent increase in circulating plasma catecholamine levels. 23 However, during the present study, NIC administration resulted in similar plasma norepinephrine levels compared with the PL treatment. Therefore, it is not surprising that glucose values were similar between the 2 treatments. Again, the severity of the cold stress may have exceeded any impact that NIC may have on plasma norepinephrine concentrations, and thus there was no impact on glucose. As previously stated, though, the measurement of plasma catecholamine levels is at best an indirect marker of sympathetic nervous system activity.
The response in NEFA concentration over time was different between the PL and NIC treatments. Although both treatments resulted in a significant increase in NEFA concentrations over time, the BASE value was higher and the 120-minute value was lower during the NIC treatment compared with the PL treatment. At BASE, the higher plasma NEFA concentration during the NIC treatment compared with the PL treatment at BASE could be attributed to the influence of NIC on hormonal status. Nicotine has been shown to increase the rate of lipolysis and plasma NEFA concentration. 28 These increases due to NIC are usually attributed to a stimulation of the sympathoadrenal system. However, in the present study, there was no treatment difference in plasma epinephrine or norepinephrine concentrations. Therefore, it is unclear as to the mechanism responsible for the higher NEFA concentration at BASE. Interestingly, plasma glycerol concentration was similar at BASE between the PL and NIC treatments. This observation would seem to indicate that lipolysis was similar between the 2 treatments. Therefore, NIC may have resulted in a shift in substrate utilization or a decrease in NEFA uptake.
Although NEFA concentration was higher at BASE during the NIC treatment, this response was reversed with NEFA concentration being higher at 120 minutes during the PL treatment compared with the NIC treatment. Although the sex-by-treatment-by-time interaction was not significant, men exhibited a plasma norepinephrine concentration that was approximately 23% lower at 120 minutes during the NIC treatment compared with the PL treatment. For women, plasma norepinephrine was approximately 17% higher at 120 minutes during the NIC treatment compared with the PL treatment. Therefore, it is unclear as to why NEFA concentration was higher at 120 minutes during the PL treatment compared with the NIC treatment. The lower plasma norepinephrine concentration at 120 minutes in the men during the NIC treatment compared with the PL treatment may have contributed to an overall statistical decrease in the rate of lipolysis at 120 minutes during the NIC treatment.
Despite the differences in plasma NEFA concentrations noted above, there were no overall differences in plasma NEFA or glycerol concentrations between the 2 treatments. This observation may be because of the lack of an overall difference in plasma catecholamine levels between the PL and NIC treatments.
In conclusion, 120 minutes of exposure to 10°C air resulted in an increase in plasma norepinephrine, no change in plasma epinephrine, a decrease in plasma cortisol, a decrease in plasma glucose, and an increase in plasma glycerol and NEFA concentrations. With respect to the influences of NIC on these responses, plasma norepinephrine concentration tended to be lower during the NIC treatment compared with the PL treatment in men, whereas women exhibited similar values for both treatments. However, as previously stated, this difference did not reach statistical significance, and thus further investigation is warranted. Nicotine administration resulted in a higher NEFA concentration at BASE and a lower NEFA concentration at 120 minutes of the cold exposure compared with the PL treatment. It is also unclear as to whether the differences observed between the PL and NIC treatments during cold exposure with respect to the hormonal and metabolic responses may have any clinical impact on safety and performance during exposure to cold stresses. Further research on the issue of a differential response to NIC administration with respect to the sexes during cold exposure is warranted. Future studies should examine this topic with more subjects and a more homogenous population within each sex.
Footnotes
Acknowledgment
This study was supported by the Herbert N. Hultgren Award from the Wilderness Medical Society.
*
Presented as an abstract at the 2004 ACSM National Meeting, Indianapolis, IN.
**
Use of trade names does not constitute endorsement of products. The opinions or assertions contained herein are the private views of the authors and are not to be construed as official or reflecting the opinions of the Department of the Army or the Department of Defense.
