Abstract
Introduction
Wilderness medicine involves the treatment of individuals in remote, austere environments. Given the high potential for injuries as well as the unique treatment modalities required in wilderness medicine, evidence-based clinical practice guidelines are necessary to provide optimal care. In this study, we identify evidence gaps from low-quality recommendations in wilderness medicine clinical practice guidelines and identify new/ongoing research addressing them.
Methods
We included relevant clinical practice guidelines from the Wilderness Medical Society and obtained all 1C or 2C level recommendations. Patient/Problem/Population, intervention, comparison, outcome (PICO) questions were created to address each recommendation. Using 24 search strings, we extracted titles, clinical trial registry number, and recruitment status for 8899 articles. We categorized the articles by trial design to infer the effect they may have on future recommendations.
Results
Twelve clinical practice guidelines met inclusion criteria. From these we located 275 low-quality recommendations and used them to create 275 PICO questions. Thirty-three articles were relevant to the PICO questions. Heat-related illness had the highest number of relevant articles (n=9), but acute pain and altitude sickness had the most randomized clinical trials (n=6).
Conclusion
Overall, few studies were being conducted to address research gaps in wilderness medicine. Heat-related illness had the most new or ongoing research, whereas no studies were being conducted to address gaps in eye injuries, basic wound management, or spine immobilization. Animals, cadavers, and mannequin research are useful in cases in which human evidence is difficult to obtain. Establishing research priorities is recommended for addressing research gaps identified by guideline panels.
Introduction
Wilderness medicine involves the treatment of individuals in remote, austere environments. This field of medicine is increasing in popularity because of heightened interest in adventure tourism and the spread of civilization into a greater variety of environments. 1 As more people participate in such activities, the likelihood for injury in these environments will increase.1,2 Although epidemiological data are preliminary, soft-tissue wounds, strains, sprains, and fractures have been the most frequently reported injuries. 3 –5 The most common causes of death in remote environments are head trauma, cardiac arrest, drowning, and hypothermia. 6 Given the high potential for injuries and the unique treatment modalities required in wilderness medicine, evidence-based clinical practice guidelines (CPGs) are necessary to aid practitioners in providing the best care for individuals injured in austere environments. 7
CPGs are based on the best available evidence and are used by physicians to provide high-quality patient care. For example, the Wilderness Medical Society’s wound management CPG 8 advocates for the use of tourniquets with severe injuries because evidence suggests that tourniquets can stop bleeding in 85% of patients compared with 17% of patients not treated with a tourniquet. 9 Because physicians and patients rely on the recommendations of these guidelines, assessing the literature behind each recommendation has recently become an important area of further research. 10 A grading scale was developed by the American College of Chest Physicians (ACCP) to permit clear identification of the strength of a recommendation and the quality of the evidence supporting it. 11 The authors of wilderness medicine CPGs chose this scale because it allows authors to assign grades for recommendation strength and quality of evidence separately. 7 This scale ranges from strong recommendations with high-quality evidence to weak recommendations with low-quality evidence. Critically evaluating the recommendations at the latter end of the scale is particularly important because they are the least helpful in guiding clinicians. These areas of low-quality or very low-quality evidence are defined as “research gaps,” and they need to be appropriately addressed. 12
Addressing these gaps in wilderness medicine can be extremely difficult because of the potentially isolated locations and unpredictable nature of injuries covered by wilderness medicine CPGs. 7 Recommendations that have already received an A grade for research support do not require as much additional investigation as those with a C grade. Projects that focus on areas with exceptional amounts of research are allocating resources to already well-studied areas, producing waste and leaving research gaps in less-studied topics. 13 Scientific research toward biomedical advancements has seen a substantial increase in annual funding to over $240 billion nationally in 2009.14,15 Four main themes are apparent in wasted research funding: repetitive investigations, research that is not published/reported, lack of access to research data/journals, and research currently underway in clinical areas that are not pertinent to practitioners or patients.14,15 Our project aims to identify the low-quality evidence recommendations in wilderness medicine CPGs that need further research and to identify recent publications that may address them.
Methods
Oversight and Reporting
This study was not subject to institutional review board oversight because it did not meet the regulatory definition of human subject research as defined in 45 CFR 46.102(d) and (f) of the Department of Health and Human Services’ Code of Federal Regulations. We applied relevant statistical analyses and methods in the published literature reporting guidelines for reporting descriptive statistics. 16
We located the latest CPGs for wilderness medicine, found in Figure 1. Recommendations from these guidelines are rated based on the quality of evidence described by the ACCP Clinical Guideline (Table 1). 11 For each grade 1C or 2C recommendation, we constructed one or more research questions using the patient/problem/population, intervention, comparison, outcome (PICO) format. 16 This method is used to identify clinical components for systematic reviews and is endorsed by the Cochrane Collaboration. 17 It was chosen over the participants, intervention, comparator, outcomes, study design and the sample, phenomenon of interest, design, evaluation, and research methods because evidence suggests that this method produces searches with greater sensitivity. In addition, PICO questions are widely used and are the best framework to identify research gaps and investigate the reasons that they exist. 18 Board-certified emergency medicine physicians constructed all initial PICO questions independently and then convened and reconciled any differences for accuracy before drafting the final questions. 16
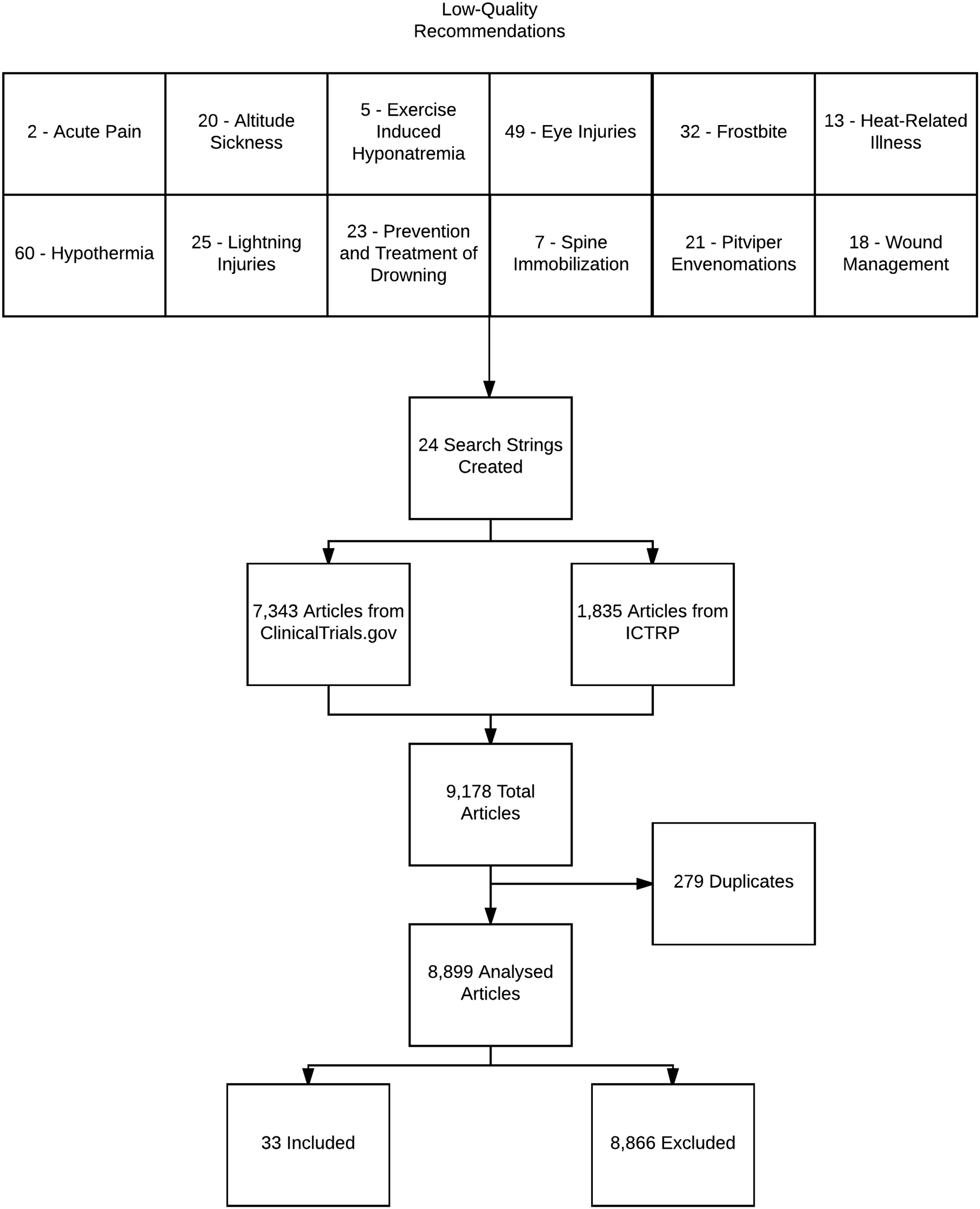
PRISMA diagram showing data abstraction and breakdown.
ACCP classification of grading evidence for recommendations in CPGs
ACCP, American College of Chest Physicians; CPGs, clinical practice guidelines; RCT, randomized controlled trial.
Development of the Search Strings
PICO questions were reviewed to identify high-yield keywords. These keywords were then used to design search strings for the questions. Search strings are part of a search strategy for finding information in databases. A search strategy is the process used to translate a clinical query (ie, research question in PICO format) into a format that can be correctly understood by the search engine. 19 The goal of a search string is to strike a balance between retrieving relevant studies and excluding irrelevant ones. For this study, we used a highly sensitive search strategy. Our searches retrieved a large number of false-positive results to ensure that important studies were not missed.
The keywords were compared with Cochrane systematic reviews, Medical Subject Headings, and PubMed automatic term mapping to determine relevant synonyms, entry terms, and variant word forms. A search string was formulated leveraging Boolean operators (eg, OR, AND) and parenthetical groupings to optimize the use of key terms to retrieve as many relevant records as possible in the clinical trial registries. Although both
Searching the Trial Registries
Using the search strings for
Using the search strings for ICTRP, we retrieved studies using the basic search function. By performing a basic rather than advanced search, we were able to achieve a more sensitive search.
20
Studies returned from the ICTRP search were also downloaded as a Microsoft Excel file. These studies were then added to those identified through
Screening Studies for Eligibility
Three investigators screened studies for relevance. First, they evaluated whether studies retrieved from the searches were relevant to wilderness medicine based on the title and listed objective. Studies that were not relevant were immediately excluded. Studies relevant to the PICO questions were retained, and studies that were unclear were reviewed for relevance by 2 emergency medicine physicians. To qualify for inclusion, a study had to fit the PICO question. Second, the original investigators screened relevant studies by completion date. Only studies completed after each respective guideline composition and studies still in progress were included. After screening, studies were mapped to their corresponding recommendation. We excluded 2 CPGs from our analysis. The first was “Prevention and management of avalanche and nonavalanche snow burial accidents,” 22 because it was published in March 2017, which did not allow adequate time for research gaps to be addressed. The second guideline, “Use of epinephrine in outdoor education and wilderness settings: 2014 update,” 23 was excluded due to the lack of an ACCP-graded guideline recommendation.
Results
We identified 14 CPGs from the Wilderness Medical Society after exclusion of 2 for being published in 2017 and another for not providing recommendations with evidence notations. Two hundred and eighty-two low-quality evidence recommendations were identified with a total of 446 recommendations being made (63.0%). From the 282 recommendations, 275 PICO questions were formed. Acute pain, exercise-associated hyponatremia, and wound management were the only CPGs for which less than 50% of the recommendations were based on low-quality evidence. The percentage of low-quality evidence recommendations ranged from 16.7 (acute pain) to 96.0% (lightning strikes) (Table 2). 8 ,21,24–33 Search strings for each CPG identified 8899 articles. Of the articles identified, only 33 provided support for 29 individual PICO questions (Table 3). Heat-related illnesses had the most research completed since the release of the CPG (n=9), followed by acute pain (n=8) and acute altitude sickness (n=7). There were no studies addressing eye injuries, basic wound management, or spine immobilization. A list of all recommendations, corresponding PICO questions, and the number of studies addressing these recommendations is provided in Table 3. 8 ,21,24–33
Low-quality evidence recommendations by clinical practice guideline
CPG, clinical practice guideline.
Clinical practice guideline with PICO question breakdown
CPG, clinical practice guideline.
We attempted to evaluate whether studies located in the trial registries would upgrade the status of a recommendation if they were included as supporting evidence. Table 4 lists the study designs associated with each study. 21 ,24–26,28,29,33 Of the topic areas, acute altitude sickness and acute pain have the highest likelihood for upgrades to recommendations because 6 studies for each were based on randomized trials. Accidental hypothermia, however, may garner little or no improvement in recommendations because the studies found had either observational or nonrandomized designs. Given that the ACCP task force provided criteria 11 that bases judgments on both study design and the benefit-to-risk ratio, we were hindered in making more conclusive judgements regarding the likelihood for a study to upgrade a particular recommendation.
Studies identified as providing support to guideline recommendation and associated study designs
AMS, acute mountain sickness; HAPE, high altitude pulmonary edema; IV, intravenous; EAH, exercise-associated hyponatremia.
A study that was a crossover, indicating that the same group for the intervention was used to test the control.
Discussion
This study examined recommendations based on inadequate or no supporting evidence in wilderness medicine to understand the extent to which new and ongoing research is being conducted to bolster the confidence in guideline recommendations. Our search yielded 8899 articles; however, only 33 studies were found to provide support for the low-level recommendations. CPGs should be based on strong evidence supported by randomized controlled trials when possible; however, this aim is easier said than achieved in wilderness medicine. Although it is difficult to find evidence for wilderness medicine, each clinical practice guideline attempted to find articles from similar fields of research to bolster the evidence behind each recommendation. For example, the CPG for eye injuries in the austere environment had evidence collected from ophthalmology and emergency medicine literature. Validated CPGs are important because they provide systematic and reproducible methods and serve as a mechanism for the identification of areas that require additional research. 34 Table 4 shows the effect that the clinical trials would have on the low-quality recommendations. Filling research gaps would help clinicians provide more effective evidence-based treatments. 35
The Institute of Medicine has established developmental standards to evaluate the quality of CPGs, which includes items such as attempts to reduce gaps in research needs and minimization of duplicate research and waste. 36 Several practical barriers, such as austere environments and rare injuries, prevent the wilderness medicine CPGs from reducing research gaps for low-quality recommendations in accordance with the Institute of Medicine standards. Options do exist, however, to aid in this process. Classically, injuries and illnesses that are rare or difficult to study can be investigated using animal models and mock human models. One such study reported the use of rats as a proxy for humans to study a novel first aid treatment for snake envenomations. 37 The use of mock envenomations in human patients has enabled the comparison of treatment methods. 38 In that study, investigators injected participants with contrast dye of a consistency similar to that of snake venom and monitored the time of progression to reach a lymph node after application of either cold packs or pressure to the site. This methodology used a novel method of venom injection that did not endanger patients and provided comparable evidence to aid in CPG formulation. Another possibility is technology-enhanced, simulation-based education. Numerous systematic reviews and meta-analyses have been conducted on the effectiveness of simulation-based learning in health professions, 39 –43 with the general consensus being that there are at best only small to moderate effects compared with other instructional methods; however, in comparison with no instruction, there are significant effects. In the field of wilderness medicine, where no instruction is taking place because of the rarity of certain conditions, environments, or injuries, simulation-based learning may provide a plausible means of instruction.
Another example of an unresolved research gap is in the treatment of eye injuries, 27 which includes 49 level 1C and 2C recommendations in the guideline (Table 3). The authors of this CPG had to make recommendations without adequate evidence. In one instance, the authors concluded that “evidence regarding the use of cycloplegics or corticosteroids in traumatic hyphema is lacking,” yet the CPG panel recommended use of such hyphema treatments. Our search resulted in a study concerning the effect of corticosteroids on intraocular hypertension using bovine eyes. 44 Although animal proxies do not correlate with human physiology exactly, this method does provide a way of simulating injuries that are far too rare or unpredictable to be feasibly studied in austere environments.
Spine immobilization in the wilderness is a topic with conflicting opinions regarding the clinical benefit of such techniques being performed prehospital. 32 A recent systematic review reported a lack of evidence either for or against recommending spinal immobilization, citing a dearth of high-quality evidence. 45 This research gap could potentially be filled with studies performed outside the austere environment. As one example, Hyldmo et al used full-body cadavers with an induced globally unstable cervical spine lesion to compare the log-roll technique with a novel lateral trauma position, using electromagnetic tracking to measure mean range of motion produced at the site of the injury. 46 One recommendation of the CPG for spine immobilization states that the lift-and-slide technique has been proven superior to the log-roll, but this recommendation is described as being based on low-quality evidence. The protocol by Hyldmo et al could easily be applied to many recommendations for spinal immobilization to allow for the better collection of evidence in the future.
The field of wilderness medicine does not always lend itself to randomized controlled trials and may be limited to observational studies and case studies, making the acquisition of strong evidence difficult. It may be necessary to draw from other bodies of evidence, such as the prehospital literature, in cases for which insufficient evidence exists in wilderness medicine. Lightning strikes, drownings, and venomous reptile bites are injuries treated in austere environments, and they are relatively rare and disparate from other injuries. Designing robust studies for these injuries is challenging, and these challenges will likely make updating guideline recommendations based on evidence difficult. In such cases, it may be appropriate to consider the higher benefits of the recommendations versus the low evidence-based support underpinning them. 7 For areas more amenable to robust study designs, we recommend the use of prioritization systems for selecting areas of research that will provide the greatest yield to patient care. The Agency for Healthcare Research and Quality’s Effective Health Care program created a system to prioritize research areas to better balance the needs of patients or providers with those of the researching physicians. Whitlock et al found that the best approach is finding an evidence gap as the starting point of the process and then prioritizing research needs by consulting stakeholders. 47 Although this process has been shown to need a few rounds of prioritization to narrow down the important topics, it could prove beneficial to the field of wilderness medicine. 48 This process could help direct efforts toward low-evidence recommendations in fields such as wound care or acute pain in the austere setting, for which evidence could be provided with randomized controlled trials more easily for than others, such as lightning strikes.
Limitations
Our study has the following limitations. Only English-language articles were examined, which may have caused us to miss supporting research published in non-English languages. We used
Conclusions
Our study illustrates that a significant number of CPG recommendations based on low-quality evidence are not being addressed in wilderness medicine. The most deficient areas in need of additional research include spine immobilization, drowning prevention, basic wound management, lightning injuries, and eye injuries, all of which had no new or ongoing research since publication of the CPGs on these topics. These areas will remain deficient unless research stakeholders make a combined effort to directly address these research gaps. Wilderness medicine research is difficult to perform, but it is essential in furthering our understanding of the contributions of specific techniques in improving clinical outcomes.12,50 The use of mannequins, animal models, or cadavers may enhance our understanding of interventions in austere environments when such studies would be difficult or impossible to conduct on humans. Furthermore, implementing a method for prioritizing wilderness medicine research would allow these gaps to be addressed more strategically and is recommended for advancement of evidence-based clinical practice guidelines.
Author Contributions: Study concept and design (JG, MV); acquisition of the data (DT, KD, TB, DS, DG, JC); analysis of the data (DT, KD, TB, JC); drafting of the manuscript (DT, KD, TB, JC); critical revision of the manuscript (MV, DS, DG); approval of final manuscript (MV).
Financial/Material Support: None.
Disclosures: None.
