Abstract
Introduction
Acute kidney injury (AKI) occurs in approximately 5% of cases of envenomation by snakes of the genus
The triad of AKI, thrombocytopenia, and hemolytic anemia with fragmented erythrocytes (schistocytes) is the essential criterion for the diagnosis of hemolytic uremic syndrome (HUS) and has been maintained since HUS was first described.25,26 HUS is extremely rare and has only been reported in isolated cases or in a small series of cases of envenomation by
Neither TMA in general nor HUS in particular has been reported in association with
Case Report
A 57-y-old male military professional, while on the banks of the Anchicayá River in the Colombian Pacific region (southwestern Colombia), was bitten on his left foot by an adult

On admission, the patient was in apparent good condition, with blood pressure of 135/82 mm Hg and heart rate of 78 beats·min-1. His head, neck, and sensory organs were normal, with normal cardiopulmonary, abdominal, and neurologic examination. Extremities were normal, except the dorsum of the left foot, where there was a puncture wound corresponding to a bite with a fang, with active bleeding and the development of an adjacent hemorrhagic blister 1 cm in diameter; moderate edema was also observed (Figure 2).
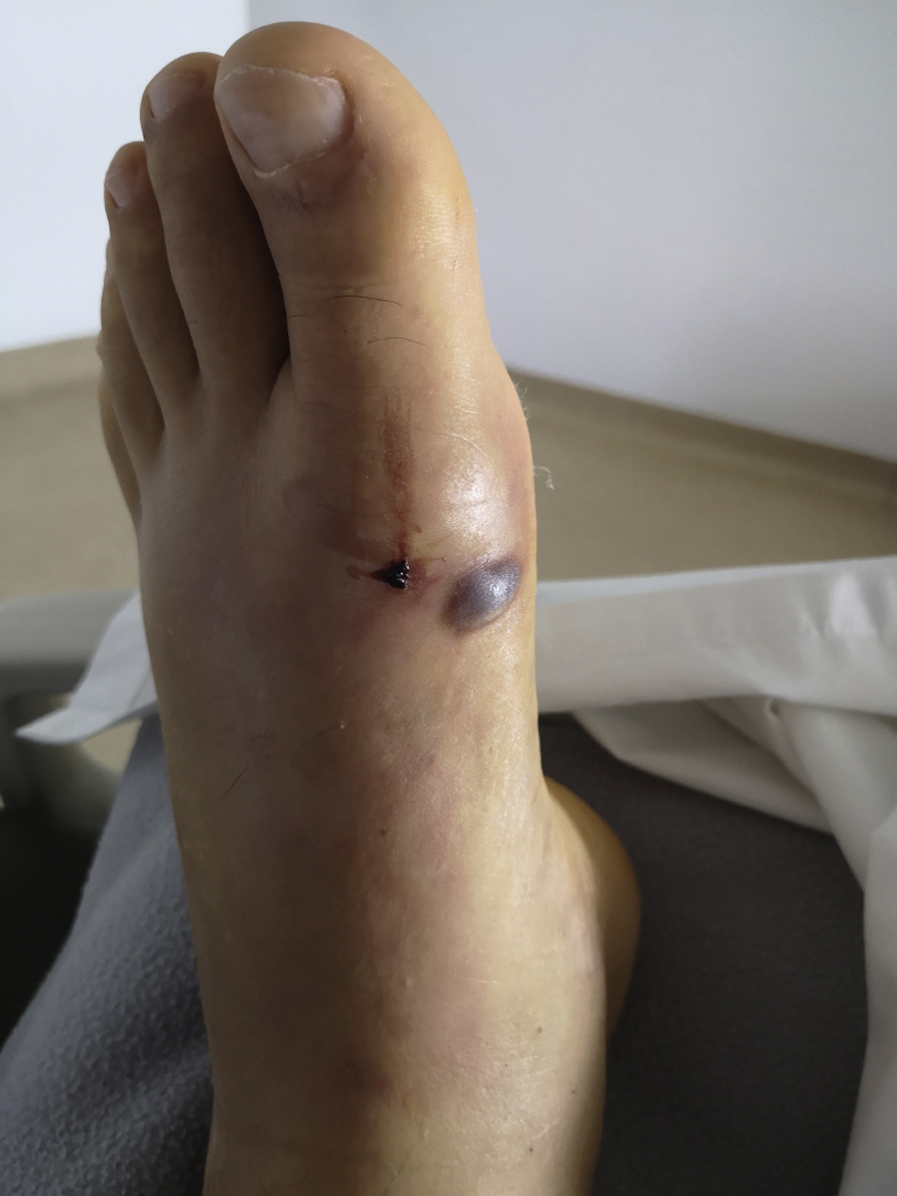
Punctate wound corresponding to a bite with a fang on the dorsum of the left foot, with evidence of bleeding and the development of an adjacent hemorrhagic blister 1 cm in diameter; moderate edema was also observed.
Admission laboratory examination results showed creatinine at 2.28 mg·dL-1 (normal 0.67–1.17), blood urea nitrogen (BUN) at 40 mg·dL-1 (normal 6–20), hemoglobin at 16.5 g·dL-1 (normal 13.7–17.5), and platelets at 225,000 μL (normal 163,000–337,000). Leukocytes were 8940/mm3 (normal 4230–9070) with 6600/mm3 neutrophils (normal 1780–5380), 1840/mm3 lymphocytes (normal 1320–3570), 400/mm3 monocytes (normal 300–820), and 100/mm3 eosinophils (normal 40–540). A noncoagulating prothrombin time (PT) and thromboplastin time (TPT), undetectable fibrinogen levels, and D-dimer concentration of 3.5 μg·dL-1 (normal <0.50) led to the diagnosis of venom-induced consumption coagulopathy (VICC).
A total of 15 vials of polyvalent antivenom was required; the initial dose was 8 vials, according to the recommendations given by national guidelines, depending on the classification of the envenomation,
30
and subsequent doses were added every 6 h until the patient’s coagulation tests were normalized. At 6 h, 3 vials were used (PT: noncoagulating, TPT: noncoagulating, fibrinogen: 50 mg·dL-1); 12 h later, 2 vials (PT: 30 s, TPT: 60 s, fibrinogen: 150 mg·dL-1), and 18 h later, 2 vials (PT: 14, TPT: 34, fibrinogen: 175 mg·dL-1). Each vial of polyvalent antivenom (Laboratorios Probiol, Bogotá, Colombia) contains 10 mL of equine polyvalent antivenom, which neutralizes at least 25, 10, and 5 mg of the venom of
Forty-eight hours after admission, the patient presented with oliguria. He was pale and jaundiced. Erythema and increased local heat began to be observed on the back of the left foot. Given suspicion of the onset of an infectious process, piperacillin-tazobactam at a dose adjusted to his kidney function was initiated. Magnetic resonance imaging of the left foot was performed and showed considerable soft tissue edema without evidence of fluid collection. There was a decrease in hemoglobin to 10 g·dL-1. Lactate dehydrogenase (LDH) was 1100 U·L-1 (normal 135–225), haptoglobin was undetectable, and indirect bilirubin was 17 mg·dL-1 (normal 0.3–1.9). These findings are diagnostic of hemolytic anemia. Peripheral blood smears showed schistocytes, a decrease in platelets to 9000 μL, BUN 78 mg·dL-1 (normal 7–20), creatinine 5.8 mg·dL-1 (normal 0.59–1.04), complement component C3 98 mg·dL-1 (normal 90–180), and C4 8 mg·dL-1 (normal 10–40). The findings of hemolytic anemia in conjunction with thrombocytopenia and kidney injury are highly suggestive of TMA. Due to hyperkalemia (potassium 5.49 meq·L-1), acidosis (pH 7.26 and HCO3 17.1), and edema, we decided to start daily hemodialysis and hemofiltration as needed. Given the suspicion of HUS, we decided to start concomitant therapeutic plasma exchange (TPE) with replacement with fresh frozen plasma. The patient required 4 TPE sessions. The hemolytic anemia was controlled, and his platelet levels gradually increased.
Twenty days after admission, the patient had significant improvement of the left foot injury. He presented with normal urine output and improved laboratory parameters: hemoglobin 9.8 g·dL-1; leukocytes 4752/mm3, with 2450/mm3 neutrophils, 1250/mm3 lymphocytes, 50/mm3 monocytes, 2/mm3 eosinophils, and 234.000/mm3 platelets; BUN 24 mg·dL-1; creatinine 1.6 mg·dL-1; LDH 110 mg·dL-1; indirect bilirubin 0.9 mg·dL-1; sodium 139 meq/L-1; and potassium 4.7 meq·L-1. Complement components were C3 102 mg·dL-1, C4 14 mg·dL-1, creatine phosphokinase (CPK) 98 U·L-1, aspartate aminotransferase (AST) 24 UI·L-1, and alanine aminotransferase (ALT) 18 UI·L-1.
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. This report was approved for publication by the ethics committee of Fundación Valle del Lili.
Discussion
VICC is a coagulopathy that occurs in envenomation by snakes whose venom contains proteases that act by stimulating coagulation factors or simulating their structure and function. Clotting factors are rapidly consumed, clotting times are prolonged, hypofibrinogenemia develops, and D-dimer increases.
31
In the specific case of snakes of the genus
Our patient presented with classic VICC with fibrinogen consumption, prolonged coagulation tests with clinical evidence of a bleeding tendency based on bleeding from the bite orifice, and the presence of hemorrhagic blisters. The patient responded adequately when treated with Colombian polyvalent viper antivenom, and his coagulation tests were normalized within a few hours of treatment. After recovery from VICC, the patient presented low urine output with uremia, his platelet levels began to decrease, and severe hemolysis developed, with evidence of schistocytes in the peripheral blood smear. This indicated the presence of TMA in the absence of criteria for thrombotic thrombocytopenic purpura that met the criteria for HUS. Similar cases have been reported that initially presented as VICC and were followed by the development of thrombocytopenia, TMA, and AKI. 14 The predilection toward kidney involvement seems to be characteristic of TMA associated with HUS caused by snake envenomation, a finding that should be differentiated from occasional initial prothrombotic phenomena in the presence of envenomation by vipers, which tends to affect the cerebral arteries, specifically those at the base of the skull. 38
In HUS, there is endothelial damage, which in the kidney manifests as thickening of the vascular walls with detachment of the endothelial cells of the glomerular basement membrane and the formation of microthrombi with variable degrees of vessel occlusion, a condition that causes erythrocytes to rupture when passing through this partially occluded microvasculature.
39
Two forms of HUS have been described: typical and atypical. The most frequent (90% of cases) is typical (or classic) HUS, which is associated with a diarrheal syndrome caused by
In atypical HUS (less than 10% of HUS cases), the disease develops with no relation to infection with
In the particular case of snake envenomation leading to the development of atypical HUS, we are not aware of a study of any mutations in the related complement cascade proteins. The outcomes of these patients and the results of different proposed treatments, such as the use of fresh frozen plasma
10
and TPE,
54
have been reported mainly in observational studies,
55
such as that of Wijewickrama et al. in a series of 103 patients with
Thus, we present a patient with
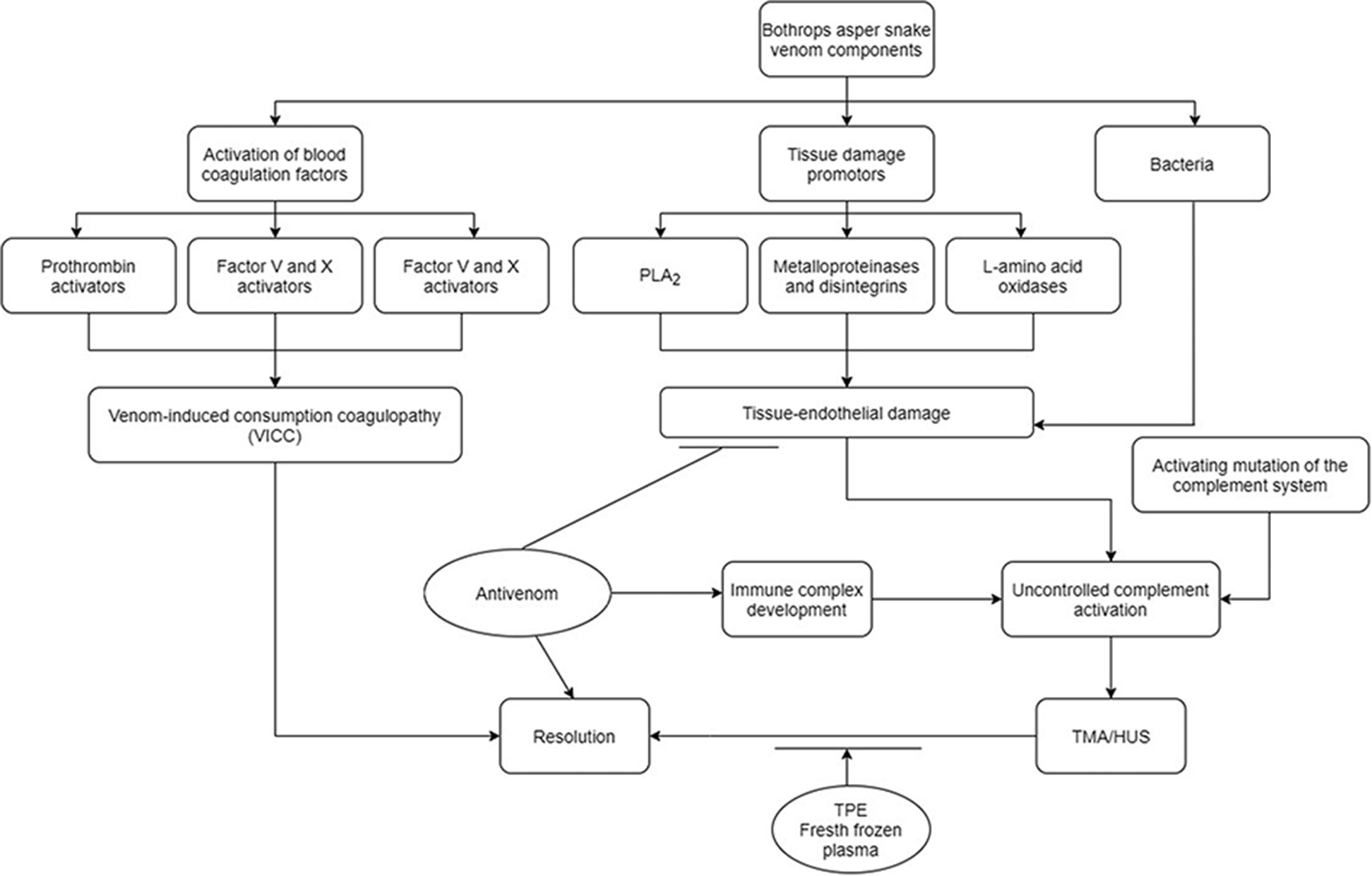
The possible sequence of events that occurred in our patient. Treatment actions are indicated by horizontal line. Typical venom-induced consumption coagulopathy (VICC) developed due to the effect of various proteins that activate clotting factors. He improved with the application of antivenom. The patient then developed thrombotic microangiopathy (TMA), probably as a consequence of a defect in the regulation of complement that could be activated by the effects of endothelial damage caused by other protein elements of the venom, the presence of an infection, and the use of antivenom. TMA with the presence of clinical and laboratory elements consistent with hemolytic uremic syndrome (TMA/HUS) was successfully controlled with hemodialysis and therapeutic plasma exchange (TPE) with fresh frozen plasma.
VICC in our patient was managed with antivenom, and he had an adequate clinical response. The TMA and HUS were managed with TPE with fresh frozen plasma despite the fact that there is no evidence of a consistent response in this type of case. The disease resolved 3 weeks later. Piperacillin-tazobactam was added to his treatment after suspicion of the onset of a local infection. To the best of our knowledge, this is the first case of
In 2009, cases of VICC were retrospectively analyzed 63 and showed that the use of coagulation factors (fresh frozen plasma and/or cryoprecipitate) after the use of antivenom is associated with earlier improvement of coagulation function. However, it is important to note that there is evidence that the use of fresh frozen plasma did not reduce coagulopathy in Russell’s viper envenomation, 57 and some low-quality studies (especially in animal models) 64 show that the use of fresh frozen plasma can be deleterious, increasing the presence of thrombotic complications; for this reason, antivenom is the mainstay of the treatment of VICC and should always be administered.
Footnotes
Acknowledgements
Author contributions: CAC, MJ-V, and IP-O wrote the manuscript and contributed to the medical observations. All authors read and approved the final manuscript.
Financial/Material Support: This work was supported by the Fundación Valle del Lili.
Disclosures: The authors declare that they have no competing interests.
