Abstract
A 24-year-old man presented with acute abdominal pain upon ascent to moderate altitude (3500 m). An immediate evaluation revealed a splenic infarct, and he was evacuated to sea level. Upon recovery, he was sent back to 3500 m without detailed etiological evaluation, whereupon he experienced recurrent episodes of left-side subcostal pain. Imaging suggested autosplenectomy, and workup revealed a negative thrombophilia profile but was positive for sickle cell trait (SCT). Individuals with SCT can be asymptomatic until exposure to severe hypoxia, upon which they can manifest clinically as sickle cell syndrome. We discuss the rare presentation of autosplenectomy in a patient with previously undiagnosed SCT on exposure to high altitude.
Background
Sickle cell trait (SCT) is common in the Indian population, with prevalence up to 13%.1,2 It is a benign condition that goes undiagnosed in the majority of cases unless neonatal screening is done, but neonatal screening is not routine in the Indian subcontinent. SCT is a carrier condition in which the heterozygosity will rarely lead to clinical manifestations. The importance of SCT detection lies in the coinheritance of other hemoglobinopathies and concern of homozygous state in the next generation if the individual’s spouse is also a carrier. Rarely, SCT can present with a vaso-occlusive phenomenon or features of sickle cell disease when the patient is exposed to severe hypoxia-related conditions like high altitude. 3 We discuss the rare presentation of autosplenectomy in a case of SCT on exposure to high altitude and suggest screening for SCT in cases presenting with splenic infarction at high altitude.
Case presentation
A 24-year-old apparently healthy central police force member with no known comorbidities became symptomatic on ascent to moderate altitude (3500 m) as part of military service. He developed high-grade fever, severe pain in the left upper quadrant, vomiting, and weakness 1 day after exposure to high altitude. He had no other associated symptoms suggestive of any other high-altitude illnesses. He gave no history of similar symptoms in the past. Medical and family histories were noncontributory. Military history was noncontributory because he had never served in high-altitude areas before this exposure, and he did not have any similar symptoms on physical exertion during low-altitude training activities. He was a nonsmoker and did not consume alcohol. He was managed as a case of acute mountain sickness with no improvement in his condition.
On initial evaluation, the patient was of average build (height 170 cm, weight 64 kg, and body mass index 22.1 kg·m-2). All vital signs were within normal limits except for mild tachycardia (pulse rate 104 beats·min-1). Local examination revealed no abnormality. Systemic examination revealed tenderness in left subcostal and left lumbar region with rigidity and decreased bowel sounds. No other abnormality was detected.
The patient’s biochemical and hematological tests done at high altitude were within normal limits except for mild leukocytosis (Table 1). Ultrasonographic findings revealed diffusely hyperechoic splenic parenchyma measuring 12.6x3 cm with Doppler showing no flow. A large supracapsular collection measuring 5x9.4x12.9 cm of approximately 320 mL containing tiny echogenic foci was noted. (Figure 1). He was diagnosed with a case of splenic infarction that was confirmed by abdominal contrast-enhanced computed tomography. He was taken to sea level after conservative management with analgesics. He was further evaluated at a nonmilitary tertiary care center at sea level. His evaluation for thrombophilic states (protein C, protein S, antiphospholipid antibody syndrome workup, factor V Leiden mutation analysis, and prothrombin gene mutation analysis) was unremarkable. Evaluation for any malignancy by radiological evaluation and biomarkers was negative. Doppler evaluation of splenic vessels was normal with no splenic cavernomas, thrombosis, or reversal of flow pattern.
Laboratory evaluation of the patient
SCT, sickle cell trait; Hb, hemoglobin; TLC, total leukocyte count; AST, aspartate aminotransferase; ALT, alanine aminotransferase; ALP, alkaline phosphatase.
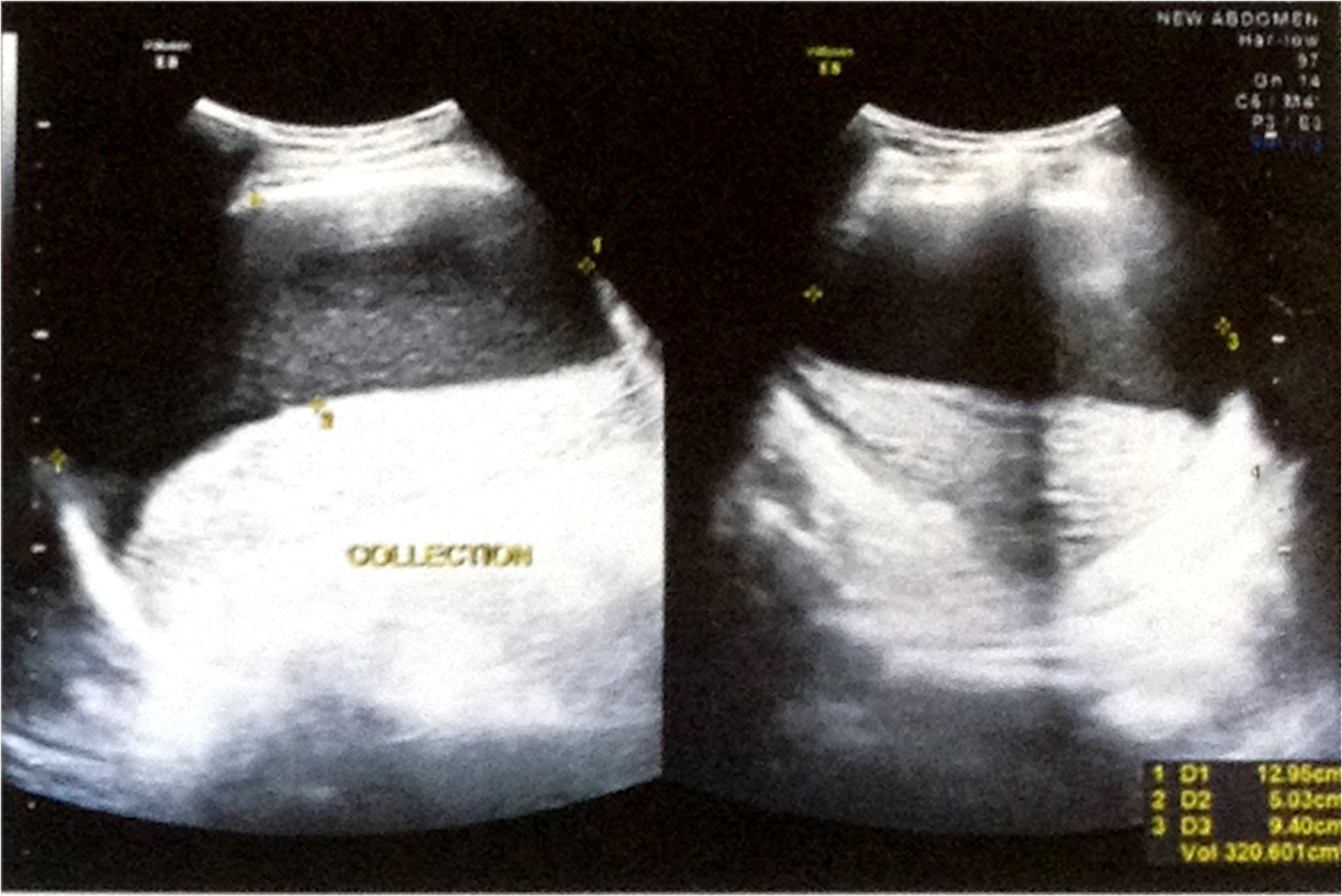
(Left) Ultrasonographic findings revealed diffusely hyperechoic splenic parenchyma measuring 12.6×3 cm in size with Doppler showing no flow. (Right) A large supracapsular collection approximately 320 mL noted with tiny echogenic foci within measuring 5×9.4×12.9 cm.
After receiving therapy, he recovered well and his pain subsided. After descent to sea level with no recurrence of similar pain, he was reexposed to high altitude after 4 months. On reexposure, he developed recurrent episodes of abdominal pain at a lower intensity with a frequency of 3 to 4 times a week; the first episode occurred within 24 hours of exposure to high altitude. He was reevaluated at high altitude for any additional abnormalities, and no difference in hematological and biochemical parameters was found. He was managed with nonsteroidal anti-inflammatory drugs, which gave temporary relief. He continued with these symptoms for 11 months before being referred to our center for detailed evaluation for repeated splenic infarcts.
Considering a diagnosis of recurrent splenic infarct, etiologies including hypercoagulable state, embolic disease, a myeloproliferative neoplasm, underlying hemoglobinopathy, any condition associated with massive splenomegaly, trauma to spleen or its vascularity, splenic artery torsion, or secondary infectious diseases (such as infectious mononucleosis) were considered. His hematological and biochemical parameters were noncontributory. Viral markers (hepatitis B surface antigen, antihepatitis C, human immunodeficiency virus 1&2) were negative. Repeat thrombophilia profile revealed no hypercoagulable state. Color-flow Doppler assessment of splenic vessels revealed no flow. Considering the possibility of thromboembolic disease, the patient underwent echocardiography, which was essentially normal. An infectious disease screen to rule out infectious causes was negative. Repeat abdominal contrast-enhanced computed tomography revealed a shrunken, cystic, solid lesion replacing the entire spleen, likely resulting from global splenic infarction suggestive of autosplenectomy. With the diagnosis of the autosplenectomy, he was evaluated for hemoglobinopathies. Investigation for coexistent enzymopathy or sickling hemoglobin(s) was negative. High-performance liquid chromatography was indicative of sickle cell trait (Table 2, Figure 2).
High performance liquid chromatography of the index patient
High performance liquid chromatography (Bioradiant/Variant II system, βThal short program) and hemoglobin electrophoresis (Interlab GenioS system) was indicative of sickle cell trait.
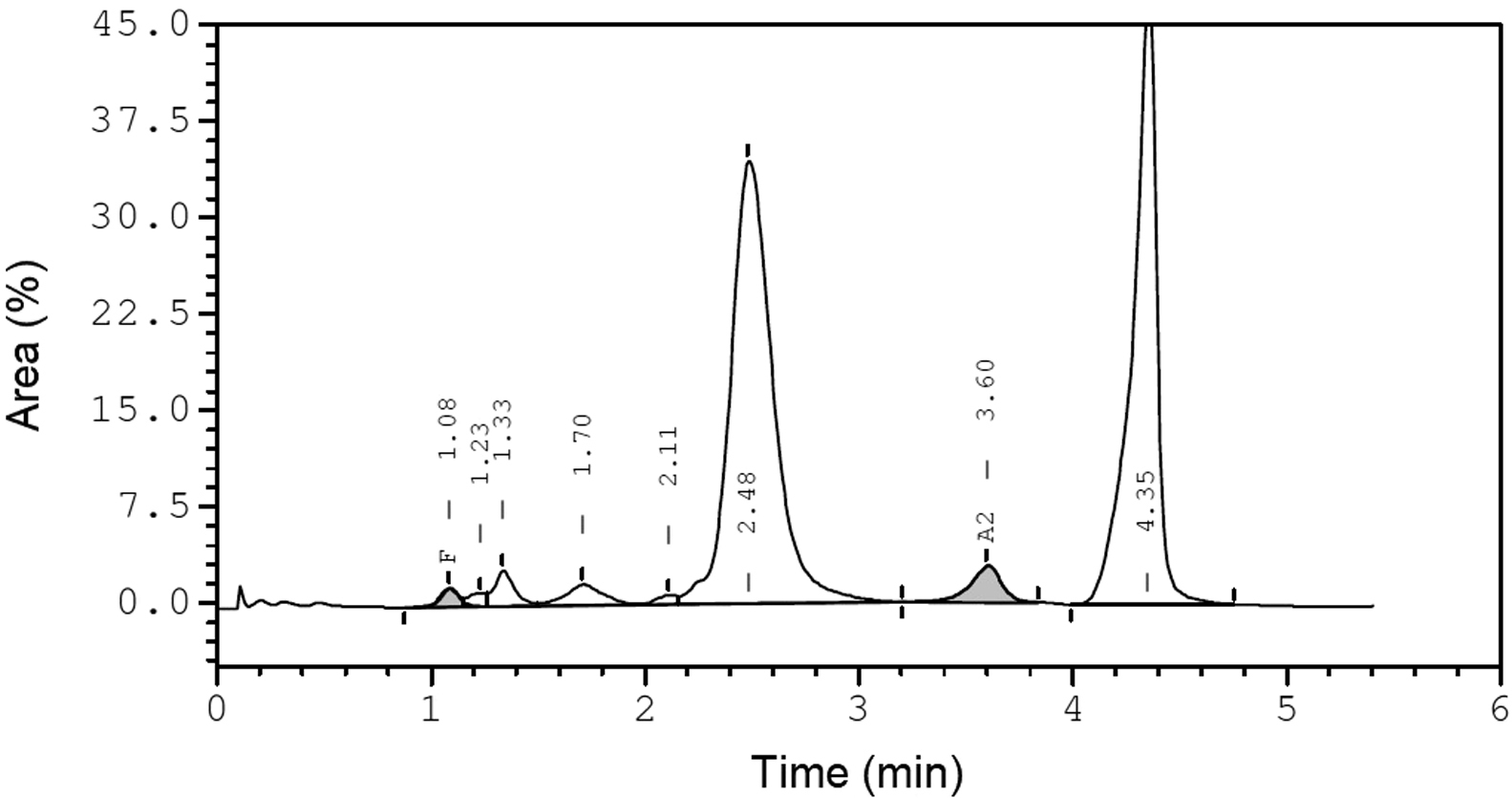
High performance liquid chromatography (Bioradiant/Variant II system, βThal short program) demonstrating elution at F window (HbF)-1%, A2 window (HbA2)-2.9%, S-window peak of 40.3 (retention time of 4.3).
The patient was managed conservatively with analgesics and anti-inflammatory drugs, which provided symptomatic relief. He was advised for genetic counseling and spouse screening. He was instructed to never again travel to high altitude.
Discussion
Sickle cell syndrome is the most common of all hemoglobinopathies. SCT is a genetic mutation of the β-globin chain in which one allele is normal and the other allele has a sickle hemoglobin mutation. About 300 million people in the world have SCT, with prevalence in India of up to 13%.1,2 SCT is often asymptomatic in the general population and goes unnoticed unless a strong family history is present. Even though SCT is generally asymptomatic, the diagnosis of the syndrome is important for the evaluation of rare presentations, as in our case, and as part of preconception counseling.
SCT is associated with specific problems, such as urological and renal disease (renal papillary necrosis leading to hematuria, renal medullary carcinoma seen exclusively in young patients with SCT, increased incidence of urinary tract infection,4,5 and remote risk of chronic kidney disease) 6 and vaso-occlusive phenomenon on exposure to severe hypoxia (splenic infarction, splenic sequestration, priapism,7,8 hyphaema,9,10 venous thromboembolism,11,12 stroke, 13 sudden death during physical exertion, 14 and acute pain syndrome). 15 In contrast to patients with sickle cell disease or severe sickle cell syndromes, the life expectancy of those with SCT is not reduced in comparison to the general population, except in rare cases of sudden death due to strenuous prolonged physical activity (athletes and military personnel), renal cancer, and splenic infarction at high altitude.11,16 It is important for patients to identify symptoms like abdominal pain, hematuria, and hyphaema, especially while traveling to high altitude or areas of low oxygen tension.
Splenic infarction at high altitude generally occurs when an individual who has SCT travels to hypoxic altitudes. 17 –19 In these cases, shortly after ascent to high altitude, the patient presents with abdominal pain localized to the left upper quadrant with guarding, rigidity, and rebound tenderness. This occurs due to sequestration of red cells resulting in an enlarged spleen, followed by splenic infarction caused by vaso-occlusion due to sickle cells. Thus, this creates a vicious cycle of sickling, vaso-occlusion, and infarction. 17 Addae et al demonstrated that SCT leading to splenic infarction is seen more commonly in lowlanders who ascend to high altitude compared with native highlanders.20,21 Goodman et al retrospectively studied the features of 25 cases of splenic infarcts with SCT in Colorado. Interestingly, all were men and symptomatic in the first 24 h on ascent to altitude higher than 2300 m (7500 ft). The results suggest a high likelihood for splenic syndrome in men of any ethnicity with left upper quadrant pain after exposure to moderate to high altitudes. 19 Altitude-related sickling is triggered by conditions secondary to hypoxia at altitudes above 1300 m, such as decrease in pH and fall in venous oxygen partial pressure to around 30 mm Hg associated with a 1 to 2% of sickling of red cells. The response is more noticeable at altitudes of around 4000 m. 22 It is difficult to diagnose a case of SCT with tests of hemoglobin, red cell indices, reticulocyte count, or peripheral blood smear because they may not show abnormalities, as in our case. Advanced techniques like hemoglobin electrophoresis, isoelectric focusing, and high-performance liquid chromatography should be performed. In this index case, we saw a rise in hemoglobin from the initial hemoglobin of 13.2 g·dL-1 to 16.9 g·dL-1, which could have been secondary to autosplenectomy or to raised erythropoietin resulting from high-altitude exposure.
Military/Paramilitary personnel are predisposed to a higher risk for sickle cell syndromes as an occupational hazard owing both to strenuous exercise and placement at high altitudes, as in this case. 23 Although similar cases have been reported in the past, this case aids in raising awareness of splenic syndrome among healthcare providers at moderate to high altitude and could improve recognition of this condition in the future. 18 –20 Early recognition and immediate descent are crucial to avoid life-threatening complications. In this case, the patient was managed at a nonmilitary medical center, the diagnosis was delayed, and he was allowed to re-ascend. This could have been avoided if the health care providers had identified the risk of reascent and were aware of potential sickle syndrome complication at high altitude. In a military medical environment, any patient with left upper quadrant pain is given nothing orally and immediately evaluated with ultrasonography. The reliance on ultrasonography is commonly a result of limited diagnostic facilities at high altitude. The most common ultrasonography findings include irregularly delineated or wedge-/round-shaped, hypoechoic/anechoic lesions. 24 Contrast-enhanced computed tomography is not essential for diagnosis. If the patient has symptoms of splenic infarction, an urgent peripheral blood assay to rule out sickling and a Doppler ultrasonography and D-dimer to rule out thrombosis should be completed. On confirmation of sickle syndrome, the individual should be hydrated, prescribed adequate pain relief, and evacuated to low altitude at the first opportunity.
Furthermore, it is important to rule out coexisting hemoglobin mutations leading to decreased hemoglobin solubility (Hb-Quebec-Chori; HbS-Antilles; Hb Jamaica Plain; or HbS-Oman) or enzyme disorders such as pyruvate kinase deficiency when patients with SCT develop sickling syndromes. 15 ,25,26 These were not detected in our case. 15 ,25,26 This case emphasizes the importance of detecting SCT in cases with splenic infarction as one of the important differential diagnosis.
Conclusions
SCT is an important differential diagnosis for splenic infarction at high altitude. Military personnel are at a potentially higher risk of sickling syndromes due to high altitude and strenuous training. It is important to evaluate for hemoglobinopathies in patients presenting with sickle cell syndrome on exposure to high altitude despite unremarkable history.
Author Contributions: Managing the case (UY, PM, SV); helped in diagnosing the case (RD); prepared the manuscript (UY); and approval of final manuscript (UY, PM, SV, RD).
Financial/Material Support: None.
Disclosure: All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
