Abstract
Splenic infarction is extremely rare in human immunodeficiency virus-infected populations. We report a rare case of splenic infarction involving Mycobacterium avium complex infection in a patient with acquired immune deficiency syndrome with immune reconstitution failure. A young man was initially admitted with cryptococcus meningitis and found to be infected with human immunodeficiency virus. He had anti-cryptococcosis treatment performed in combination with placement of an Ommaya capsule because of persistent intracranial hypertension, and first-line therapy followed by second-line anti-retroviral therapy were performed. Although there was an absence of immune reconstitution, the patient refused to take prophylactic sulfamethoxazole/trimethoprim, isoniazid, and clarithromycin continuously because of gastrointestinal intolerance. Pneumocystis pneumonia then developed. Finally, the patient developed a fever again accompanied by abdominal pain and splenic infarction. M. avium complex infection was verified by a metagenomic next-generation sequencing test using a whole blood sample. M. avium complex infection should be considered as an etiology of splenic infarction in human immunodeficiency virus-infected patients with an extremely low CD4+T-cell count.
Introduction
Mycobacterium avium complex (MAC) is the most prevalent species of non-tuberculous mycobacterial infections (NTMs) in China.1,2 Splenic infarction is extremely rare in human immunodeficiency virus (HIV)-infected populations.3–9 To date, no MAC infection has been reported to cause splenic infarction in HIV-infected patients. We report a case of splenic infarction involving MAC infection in a patient with acquired immune deficiency syndrome (AIDS) and immune reconstitution failure.
Case report
A 28-year-old man was admitted to hospital in May 2019 owing to intermittent fever for longer than 1 month and an aggravated headache for 10 days. Subsequent examinations showed HIV infection. The HIV viral load was 1.45 × 105 copies/mL, CD4+T-cell count was 4/µL (410–1590), and CD8+T-cell count was 116/µL (190–1140). Cerebrospinal fluid (CSF) and blood culture showed Cryptococcus neoformans. No abnormality was found in a head magnetic resonance imaging scan (Figure 1a). Lung computed tomography (CT) showed inflammation of the upper lobe of the right lung (Figure 1e). Cryptococcal meningitis and cryptococcal pneumonia were diagnosed. Amphotericin B combined with flucytosine was administered for the standard treatment of Cryptococcus. The patient refused to take prophylactic sulfamethoxazole/trimethoprim, isoniazid, and clarithromycin continuously because of gastrointestinal intolerance. CSF culture was negative for C. neoformans 2 weeks later. The time line of the patient’s symptoms is shown in Figure 2. There was continued cranial high pressure (300–600 mmH2O), and brain magnetic resonance angiography (MRV) showed that the lower lumens of the superior sagittal sinus and the sigmoid sinus were slender (Figure 1b, c). An Ommaya capsule was placed for decompression (Figure 1d). The patient took anti-retroviral therapy (ART) with the regimen of tenofovir, lamivudine, and efavirenz. When drug resistance was detected 1 month later, he changed to lopinavir/ritonavir and dolutegravir. The patient was then followed up at a local hospital. His HIV RNA was negative, but he did not achieve immune reconstitution. His CD4+T-cell count was maintained between 2/µL and 13/µL.

(a) Brain magnetic resonance imaging on 3 June 2019 shows no definite abnormality. (b) and (c) Craniocerebral magnetic resonance angiography on 16 July 2019 shows thinning and narrowing of the lower part of the superior sagittal sinus. The right internal jugular vein and sigmoid sinus are slender, and the right transverse sinus cannot be seen. (d) Enhanced magnetic resonance imaging of the brain on 12 October 2019 shows the Ommaya capsule and drainage catheter, and no abnormalities can be seen. (e) Lung computed tomography (CT) on 11 June 2019 shows a ground-glass density shadow in the upper lobe of the right lung and no abnormality in the remaining lobes. (f) On 3 August 2019, lung CT shows that the density shadow of ground glass remains in the upper lobe of the right lung. (g) Lung CT on 11 October 2019 shows ground-glass density patches in both lobes. (h) On 27 December 2019, lung CT show a considerable improvement in the ground-glass density patches in both lungs. (i) An abdominal CT scan on 11 October 2019 serves as a control. (j) and (k) Enhanced abdominal CT on 2 July 2020 shows considerable hepatosplenomegaly, multiple low-density foci in the spleen, and multiple splenic infarctions and (l) CT angiography shows reduced branches of the splenic artery.
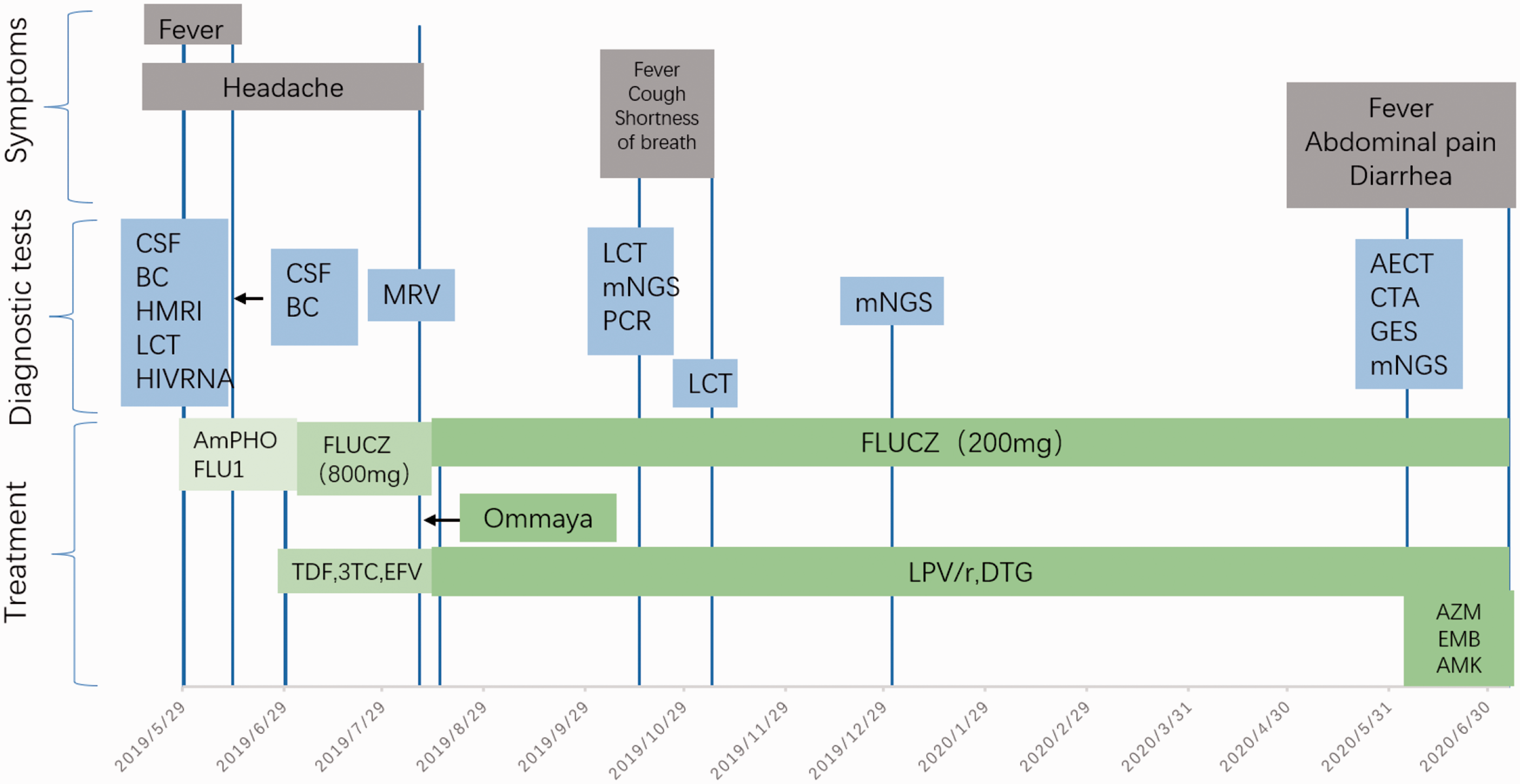
Time line of the patient’s course. The patient’s symptoms, diagnostic tests, and treatment are shown.
The patient developed a fever, cough, and shortness of breath in October 2019. The lung lesions were not improved (Figure 1f), but had gradually expanded (Figure 1g). Polymerase chain reaction and metagenomic next-generation sequencing ([mNGS] BGI, Shenzhen, China) of alveolar lavage fluid showed mono-infection of Pneumocystis jirovecii. Sulfamethoxazole/trimethoprim was administered for 21 days, and the lung lesions showed improvement (Figure 1h). On 31 December 2019, the patient had whole-blood mNGS (BGI, Shenzhen, China) performed because of intermittent fever, but no pathogenic agent was found.
The patient developed a fever again with abdominal pain, which was dominant in the right upper abdomen, and diarrhea for 1 month in June 2020. A further examination showed the following: white blood cell count, 11.1 × 109/L (3.50–9.50 × 109/L); lymphocyte count, 0.1 × 109/L (1.10–3.20 × 109/L); platelet count, 119 × 109/L (125–350 × 109/L); and hemoglobin concentration, 88 g/L (130–175 g/L). The alkaline phosphatase concentration was 153 U/L (32–126 U/L) and lactic dehydrogenase concentration was 734 U/L (313–618 U/L). The CD4+T-cell count was 9/µL. The C-reactive protein concentration was 63.8 mg/L (0.00–8.00 mg/L) and the procalcitonin concentration was 0.51 ng/mL (0–0.25 ng/mL). The prothrombin time was 13.00 s (11.0–13.7 s), activated partial thrombin time was 33.80 s (25–36 s), thrombin time was 17.00 s (14.00–21.00 s), and fibrinogen concentration was 4.77 g/L (2.00–4.00 g/L). The D-dimer concentration was 5.08 mg/L (0–0.55 mg/L). The protein C activity was 66% (60%–140%), protein S activity was 94.4% (63.5%–149%), and antithrombin III activity was 99% (80%–120%). Three times of performing blood culture were negative. NTM culture was unavailable in this hospital. A routine CSF analysis showed no abnormalities. Lung CT indicated left pleural effusion. Abdominal enhanced CT showed marked hepatosplenomegaly and splenic infarction, and scattered lymph nodes in the abdominal cavity (larger nodes were approximately 1.7 cm in diameter) (Figure 1j, k) in contrast to the scan in October 2019 (Figure 1i). CT angiography showed reduced branches of the splenic artery (Figure 1l). An abdominal ultrasound showed an enlarged liver and spleen, infarction of the spleen, and multiple enlarged lymph nodes in the abdominal cavity and retroperitoneum. Gastroenteroscopy indicated superficial gastritis, duodenitis, and proctitis. Anti-cardiolipin antibody and other autoimmune antibodies were negative, and did not support connective tissue disease. Echocardiography did not indicate valve vegetation and did not support infective endocarditis. An electrocardiogram suggested sinus rhythm, but no atrial fibrillation.
Repeated hospitalizations showed no evidence of a tumor or hematological disease. The patient's coagulation test did not show hypercoagulation, and the activities of protein C, protein S, and anti-thrombin III were normal, with no indication of congenital or acquired coagulation disorder. The patient's high fever persisted and the C-reactive protein concentration rose to 213.12 mg/L. An infectious factor was suspected because of the failure of immune reconstitution. Therefore, mNGS (Beijing CapitalBio Medical Laboratory, Beijing, China) of the whole blood was carried out again and showed MAC infection (Figure 3). The patient was immediately treated with azithromycin, ethambutol, and amikacin. Low molecular weight heparin-calcium was used for anticoagulant therapy. Splenectomy was not performed. After anti-infective treatment for 2 weeks, the patient’s fever subsided, and his abdominal pain and diarrhea were relieved. The C-reactive protein concentration was 192.54 mg/L, alkaline phosphatase concentration was 96 U/L, and lactic dehydrogenase concentration was 536 U/L, which were decreased. A reexamination of abdominal ultrasound showed no progression along with mild regression at the retroperitoneal lymph nodes. He was discharged home (a small village) with continued anti-MAC and ART therapy. Unfortunately, the patient died at home 2 weeks after discharge and we could not obtain details of the cause of death.

Metagenomic next-generation sequencing. The number of sequence reads of Mycobacterium avium was 16,281 (3.187%). The genome coverage of Mycobacterium avium was 23.8%. The reads distribution of the total DNA sequence in the sample excluded a human host.
Discussion
This is the first reported case of splenic infarction involving MAC infection in a patient with AIDS. MAC causes marked splenomegaly, and compression of small blood vessels in the spleen or thrombosis may be responsible for multiple focal splenic infarcts. We had a dilemma as to whether to perform treatment when the patient’s blood culture was negative, and it was difficult to obtain optimal specimens.
Peripheral blood by mNGS is a good method for a rapid diagnosis. As an emerging approach of diagnosing clinical pathogens, untargeted mNGS is well known for its comprehensive detection of microbial DNA and RNA from patients’ samples. In contrast to conventional microbial detection techniques (culture and polymerase chain reaction), mNGS is characterized by simultaneous identification of potential viruses, bacteria, fungi, and parasites in diverse clinical samples, such as blood, cerebrospinal fluid, and sputum. Moreover, increasingly complete databases for pathogen genomes have enabled highly sensitive and specific microbial detection.10,11 Whole blood samples are seldom affected by contamination or colonization compared with other samples, such as bronchoalveolar lavage fluid or sputum. By removing potential background microorganisms using a background database created with mNGS data of in-house control samples, a positive pathogen suggests infection by a microorganism.
An early diagnosis and anti-MAC treatment could be crucial to the prognosis. According to sequential sequencing results using peripheral blood in our patient, disseminated MAC infection was considered, and the intestinal tract was the suspected source of infection. Currently, the incidence of NTM is increasing in developed countries and in China.1,2 MAC is the most prevalent species of NTM in China.1,2 The frequency of MAC coinfection in the HIV population is 10.6%. 12 In our case, there was virological or immunological failure, recurrent opportunistic infections, and the patient lacked MAC chemoprophylaxis. Patients with a CD4+T-cell count <50 cells/mm3, HIV RNA levels >100,000 copies/mL, previous opportunistic infections, and previous colonization of the respiratory or gastrointestinal tract are at high risk of developing disseminated MAC disease.
In the general population, splenic infarction is mainly caused by splenomegaly, malignant lymphoma, infectious mononucleosis, infective endocarditis, antiphospholipid antibody syndrome, autoimmune vasculitis, and congenital coagulation disorder. 13 The common infectious factors of splenic infarction are associated with Epstein–Barr virus, cytomegalovirus, malaria, and infective endocarditis. 13 Splenic infarction is extremely scarce in the HIV-infected population. Non-infectious factors of splenic infarction, including anticardiolipin antibodies3,4 and medication-associated unexpected side effects, were reported in several cases.5,6 Because no coagulation abnormalities were found in this case, infection was initially considered. Three cases of HIV infection have been reported as the infectious cause of splenic infarction, with two cases with Mycobacterium genavense infection7,8 and one case with aspergillosis infection. 9 MAC infection as an etiology of splenic infarction has not been previously reported. Mycobacterial cultures are not performed in most comprehensive hospitals in China, and a reliable method of molecular biological identification of NTMs remains a diagnostic challenge. Apart from conventional and time-consuming mycobacterial culture, polymerase chain reaction and 16s rRNA gene sequencing7,8 or reverse hybridization DNA probes are used to identify the specific organisms of NTMs. As a culture-independent method, mNGS provides a rapid etiological diagnosis, especially in critically ill patients.
MAC is an environmental and opportunistic pathogen. The mode of transmission of MAC is thought to be through the respiratory or gastrointestinal tract. There has been a low incidence of colonization and disseminated MAC infection in the effective ART era 14 compared with the pre-ART era, 8 but the recommendation of routine primary prophylaxis for MAC is inconsistent among studies.15,16 However, primary prophylaxis with clarithromycin or azithromycin should be strongly recommended in patients who do not respond to ART. 17 Routine screening of MAC colonization by cultures and smears of sputum and stool in asymptomatic patients is not recommended according to the adult and adolescent opportunistic infection guidelines. In patients with advanced HIV infection, MAC infections usually cause disseminated disease, such as infection in the blood stream, skin, bone, mesenteric lymph nodes, liver, spleen, and intestines. The reporting of this study conforms to the CARE guidelines. 18
Conclusion
We report a case of splenic infarction involving MAC infection in a patient with AIDS. The findings in our case suggest that clinicians should consider MAC as an important etiology of splenic infarction in HIV-infected patients. Chemoprophylaxis for MAC should be recommended in patients with AIDS and immune reconstitution failure.
Supplemental Material
sj-jpg-1-imr-10.1177_03000605221115242 - Supplemental material for Splenic infarction due to Mycobacterium avium complex infection in an HIV-infected patient with immune reconstitution failure: a case report
Supplemental material, sj-jpg-1-imr-10.1177_03000605221115242 for Splenic infarction due to Mycobacterium avium complex infection in an HIV-infected patient with immune reconstitution failure: a case report by Yu Wang, Tao Deng, Yong Wang, XiaoLi Xin and Ying Wen in Journal of International Medical Research
Footnotes
Acknowledgements
We thank Zhou Ying, Liu Sheng, and Wang JinYong (Infectious Disease of the First Affiliated Hospital of China Medical University) for their professional assistance. We thank the patient’s parents for agreeing to publish the case. We also thank the Beijing Genomics Institute (BGI-Shenzhen) for performing mNGS.
Author contributions
Yu Wang, Yong Wang, XiaoLi Xin, and Ying Wen were involved in the patient's treatment. Tao Deng helped perform mNGS. Yu Wang and Ying Wen wrote and revised the case report.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics statement
We obtained permission for publication from the ethical board of China Medical University. Informed written consent for all treatment regimens was obtained from the patient. Informed written consent for publication was obtained from the patient’s father.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by “Double First Class” University and Discipline Construction funds of China Medical University (3110119068 to W.Y.).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
