Abstract
The purpose of this systematic review was to determine if sock, antiperspirant, or barrier strategies were effective in prevention of friction blisters in wilderness and outdoor pursuits. A search of PubMed and EMBASE was conducted. Title, abstract, and full text articles were screened by 2 authors using predetermined inclusion and exclusion criteria to identify prospective controlled trials investigating prevention methods for friction blisters involving the foot. Only blisters associated with wilderness and outdoor pursuits (running, hiking, marching, etc.) were considered. Extraction of a predetermined data set was accomplished using a piloted form. Confidence in effect estimates were determined utilizing the Scottish Intercollegiate Guidelines Network methodology checklist. Literature search resulted in 806 discrete articles. After screening, 11 studies were identified for inclusion in systematic review. Included studies investigated 5 sock, 3 antiperspirant, and 3 barrier strategies. Only 2 articles were determined to have moderate confidence in effect estimate. Clinical and methodologic diversity precluded meta-analysis. Despite the high frequency, discomfort, and associated cost there is a paucity of high-quality quality evidence in support of socks, antiperspirants, or barriers for the prevention of friction blisters. Moderate confidence in effect estimate suggests that paper tape may be an effective form of barrier prevention.
Introduction
Friction blisters involving the foot represent a common injury in active individuals engaged in wilderness and outdoor related pursuits. Among long distance hikers and backpackers, reported blister rates range from 54 to 86%.1,2 Studies involving military personnel report blister rates ranging from 5 to 77%.3,4 Studies involving ultramarathon and adventure racing report blister rates ranging from 26 to 76%.5,6 In fact, foot blisters have been cited as the most common medical complaint among hikers 7 and ultramarathon/adventure racers alike. 8 –10 Although often regarded as a minor injury, the impact of friction blisters is not inconsequential. In competitive single-stage ultramarathons, Hoffman and Fogard reported 40.1% of finishers cited friction blisters as adversely affecting race performance. 5 A study of US Marine Corps recruits participating in initial training evolutions at Marine Corps Recruit Depot San Diego, CA, attributed an estimated annual expense of $690,000 to friction blisters. 4
Friction blisters result from pressure and shear forces causing delamination at the level of the stratum spinosum. 11 Risk factors include but are not limited to moisture, temperature, external load, activity duration, characteristics of footwear, and adaptation/conditioning. 11 –15 Each of these risk factors influence frictional force (Ff), where Ff = μ × Fn. Prevention and treatment strategies therefore seek to reduce the coefficient of friction (μ), pressure or shear force (Fn), frequency of force application, or any combination thereof. Such strategies include any combination of sock fibers, sock piles, sock layers, barriers such as tapes and plasters, antiperspirants, lubricants, and orthotics. Despite a multitude of published studies on the subject, friction blisters remain a common and debilitating injury among participants in wilderness and outdoor pursuits, and there remains no widespread consensus on effective prevention strategies. 14 ,16–18
We conducted a systematic search and qualitative analysis of prospective controlled trials to determine if sock, antiperspirant, or barrier strategies were effective in prevention of friction blisters of the foot in wilderness and outdoor pursuits.
Methods
Study design and methodology was predetermined through collaboration of all authors following guidelines in the PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses. 19 Methods of analysis and criteria were documented in a written protocol in advance of the study.
Search Strategy
Two electronic databases (PubMed, EMBASE) were systematically searched to identify prospective controlled trials investigating prevention strategies for friction blisters of the foot. The search strategy was formulated in collaboration with appointed university librarians from multiple institutions using a peer review process. No restriction on date or language of publication was applied. Details of this search strategy including discrete search terms utilized may be found in Appendix 1 (e-component). Database searches were conducted on May 20, 2016, and duplicates removed using EndNote citation manager software (Thomson Ruters, New York, NY). A secondary search of those references included in identified publications, and a tertiary search involving expert consultation, was subsequently conducted.
Study Selection
Two reviewers independently conducted title, abstract, and manuscript review using predetermined inclusion and exclusion criteria. Inclusion criteria consisted of the following: publications were original, prospective controlled trials (randomized controlled trial, prospective controlled cohort, case crossover, etc.); subjects were recreational or professional, military or civilian, engaged in wilderness and outdoor related activity (military evolutions, hiking, running, etc.); the intervention was for prevention of friction blisters (sock systems, liquid and powder antiperspirants, tapes, etc.); and outcome measures included incident data. All settings (desert, tropical, laboratory, etc.) were accepted. Exclusion criteria consisted of the following: case reports, case series, cross-sectional or descriptive studies; publications limited to abstract only; field- and water-based sports (soccer, basketball, rowing, etc.); blisters not of the foot; blisters created by artificial means or secondary to identifiable pathology (diabetes, vascular stasis, dermatologic conditions, etc.); and cases involving complication (infection, ulceration, chronic wounds, etc.). Reviewers remained blinded to results during review. A third reviewer subsequently mediated any dispute.
Data Collection
To assess diversity, a summary description of included full text articles was prepared utilizing a piloted form. Extracted data included the following: primary author and year of publication; study design; population (including mean age where provided); setting (including distance/time where provided); number of subjects; description of the intervention; outcome measures; and reported results (including incident data). In preparation for quality assessment, study design was determined using the Scottish Intercollegiate Guidelines Network Study Design Algorithm. 20 Studies were organized alphabetically within 3 groups based on intervention (socks, antiperspirants, or barriers).
Assessment of risk of bias and confidence in effect
Assessment of risk of bias and confidence in effect estimate was performed in the context of blister risk ratio with reference to the Cochrane Handbook for Systematic Reviews of Interventions 21 and utilizing the Scottish Intercollegiate Guidelines Network Methodology Checklist 2: Controlled Trials. 20 The checklist includes 10 elements for assessment of internal validity, including identification of a clearly focused question, method of randomization and concealment, blinding, similarity of treatment and control groups, outcome measure, dropout rate, intention to treat analysis, and comparability of results across sites where applicable. Minimization of bias in study design was coded as high, acceptable, or unacceptable (low) based on rater interpretation of relative risk, direction, and magnitude of bias effect. Only those studies determined to be of high or acceptable bias control were assessed for confidence in effect estimate. Risk ratio, confidence interval, and statistical significance for individual studies were calculated using MedCalc (MedCalc Software, Ostend, Belgium). A confidence in effect estimate was then determined through consideration of risk of bias, directness of evidence, consistency and precision of results, publication bias, and other identifiable methodological limitations. Confidence in effect estimates were coded as high, moderate, low, or very low in accordance with Grading of Recommendations Assessment, Development and Evaluation methodology. 22 Two reviewers who remained blinded to results independently assessed all studies. A third reviewer subsequently mediated any disagreements.
Results
A total of 806 titles in 12 languages were identified for screening. Dates of publication ranged from 1950 to 2016. Title and abstract review resulted in elimination of 786 studies. An additional 9 of the 20 remaining studies were excluded on manuscript review based upon previously outlined criteria. A total of 11 studies underwent data extraction and analysis (Figure).
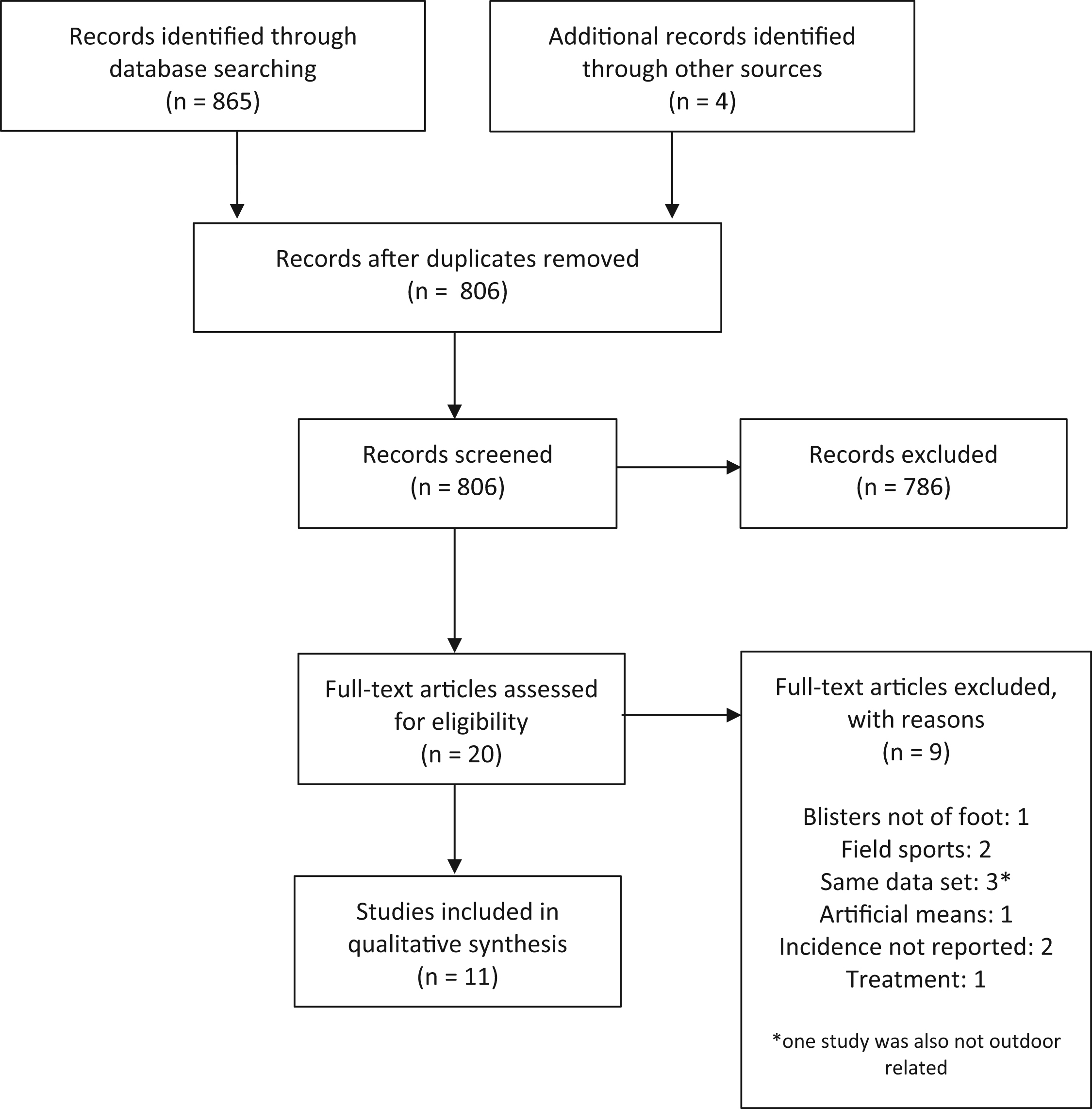
Flowchart outlining study selection for systematic review.
Results of data extraction are summarized in Table 1. 23 –31 Studies included 5 controlled clinical trials and 6 randomized controlled trials with varying degrees of blinding. A total of 1838 subjects were represented. Populations were largely military from the United States, Europe, and China. Long distance recreational runners and endurance athletes were also represented.
Summary description of studies
RR, relative risk; PFPS, patellofemoral pain syndrome; ITB, iliotibial band.
A variety of interventions for blister prevention were investigated. Five studies investigated socks with different fiber content, sock knits, and combinations of sock systems (layered socks) with and without powdered antiperspirants. Four antiperspirant formulations were investigated in 3 trials, 2 of which included placebo comparison. Three studies evaluated the prophylactic effect of barriers including paper tape and a proprietary plaster. Both subjective and objective measures were used across studies. Subjective outcome measures included perceived padding, comfort, dampness, temperature, subject preference, and satisfaction. In addition to blister incidence, objective outcome measures included blister severity, sweat accumulation, other foot injuries, musculoskeletal overuse injuries, skin irritation, and number of “sick visits.”
Results of risk of bias and confidence in effect estimate assessments are summarized in Tables 2 3 ,18,23–31 and 3, 18 ,25–27,29–31 respectively. Identified prospective controlled trials included both positive and negative results, suggesting publication bias is unlikely. Of the 11 studies evaluated, 4 were noted to be of low bias control and excluded from further assessment. Of the 7 remaining studies, 4 were determined to be of acceptable bias control and 3 of high bias control. Calculated risk ratios met statistical significance (P < .05) for 2 sock and 2 barrier studies. No antiperspirant studies met statistical significance. Confidence in effect estimate was determined to be moderate for 2 studies involving a paper tape barrier method. In the Prevention Trial Assessing Paper-Tape in Endurance Distances, 30 the authors reported no significant difference in blister incidence between prerace application of paper tape compared with the untreated contralateral foot (calculated relative risk 1.23; 95% CI 0.97–1.68; P = .08). On the experimental foot, fewer blisters were noted around the tape compared with under the tape. A substantial number of participants (44%) did not complete the study. In the Prevention Trial Assessing Paper-Tape in Endurance Distances II, 18 the same authors modified the comparator such that uncovered areas of the experimental foot served as the control. This second study was incompletely randomized, with the experimental area chosen by convenience in those racers that reported prior blisters (92%), and at random in those racers who did not report prior blisters. The authors reported a 40% absolute reduction in blister formation (calculated relative risk 0.49; 95% CI 0.37–0.66; P < .01). The primary author reports a potential conflict of interest involving the promotion of the barrier method of study in 2 for-profit wilderness publications. Confidence in effect estimate was low or very low for the remainder of the studies. A detailed narrative of assessments for each study may be found in Appendix 2 (e-component).
Risk of bias assessment
Y, potential source of bias was addressed; NA, not applicable; N, source of bias that was not addressed; –, low quality of risk control (high risk of bias); UNK, inadequate information available to determine whether potential source of bias was addressed; +, acceptable risk; ++, high quality risk control (low risk of bias).
Categories assessed are as follows: 1) focused question; 2) randomization; 3) allocation concealment; 4) blinding; 5) similarity of control and treatment groups; 6) difference in treatment among experimental group(s); 7) outcome measure; 8) dropout rate; 9) intention to treat analysis; 10) compatibility across sites.
Assessment of confidence in effect estimate for individual studies
RR, risk ratio; COI, conflict of interest.
Very low indicates true effect is likely to be substantially different from the estimate, Low indicates true effect may be substantially different from the estimate, Moderate indicates the true effect is likely to be close to the estimate of effect, but the possibility of substantial difference remains.
Table 4 summarizes clinical and methodological diversity identified among grouped studies. Diversity in study design, intervention, and comparator were common. Diversity was substantial and precluded meaningful multivariant meta-analysis with a priori grouping.
Summary of diversity among grouped studies
RCT, randomized controlled trial; NRCT, nonrandomized controlled trial.
Discussion
The existing body of literature addressing friction blisters includes a number of narrative reviews. Most notable among these narratives is that published by Knapik et al in 1995, who included a detailed evidence-based review of pathophysiology, factors influencing blister formation, and recovery. 11 Additionally, there was at least 1 attempt at the formation of evidence-based clinical practice guidelines for the prevention and treatment of friction blisters, 16 where the subject was addressed in the greater context of wound management in austere environments. This approach limited the review methodology, accuracy of results, and applicability of resultant guidelines. 17 The present systematic review followed standardized methodology consistent with Appraisal of Guidelines for Research and Evaluation in Europe II and Grading of Recommendations Assessment, Development and Evaluation. To our knowledge, this is the first systematic review using evidence-based methodology dedicated to friction blister prevention to date.
Our analysis provides moderate confidence that the calculated risk ratio for paper tape versus no treatment is close to the true effect. This suggests that paper tape may be effective in preventing friction blisters among participants in wilderness and outdoor pursuits. There is low confidence in effect estimate for prospective controlled trials evaluating sock, antiperspirant, and alternative barrier strategies for friction blisters of the foot. Methodological flaws created potential for bias in all studies evaluated. Studies of lower quality bias control frequently experienced lack of blinding and randomization, high rates of dropout and noncompliance, and lack of intention to treat analysis. Studies with low confidence in effect estimate additionally experienced inclusion of co-interventions and imprecision of effect estimate. Studies determined to be of moderate confidence in effect estimate experienced similar methodological flaws, but when direction and magnitude of bias were considered in the context of the statistical power of the study, the overall effect remained likely due to the study intervention. We note a trend toward improved quality with more recent publication dates, correlating with the evolution of guidelines on conduction and reporting of controlled trials such as CONSORT. 32 This is reflected in the relative quality of methodology and/or reporting displayed in the manuscripts of Van Tiggelen et al 26 and Lipman.18,30
Limitations exist to the present review. Although criteria and methods of analysis were documented in a written protocol, our study design was not formally registered before implementation. The study was completed in a timely manner relative to search date, and automated monthly searches were utilized where available to ensure additional relevant publications did not appear during its execution. However, data from unpublished or ongoing studies was not considered, nor were findings in retrospective, case report, case cohort, or alternative designs. Inadequate reporting complicated risk of bias assessment in multiple studies. Finally, substantial clinical and methodological diversity prevented quantitative analysis despite use of P – patient, problem or population, I – intervention, C – comparison, control or comparator, O – outcome question format. It is critical to recognize this review is limited to determining the confidence in effect estimate within each study, and does not comment on the cumulative body of evidence for or against friction blister prevention strategies. Current clinical practice guidelines may not reflect the confidence in effect estimates outlined in this systematic review.
It is our opinion that, despite nearly 80 years of research, the existing body of literature does not yet provide sufficient evidence to support the formulation of robust, meaningful clinical practice guidelines. It is our intent that the present systematic review serve as a “call-to-arms” for the performance of additional clinical trials evaluating prevention strategies for friction blisters of the foot using high quality, up-to-date methodology and reporting compliant with relevant guidelines such as CONSORT. Further studies evaluating sock systems should seek to eliminate co-interventions such as antiperspirants and evaluate single variables (fiber content, pile, layering, etc.). Any further study of antiperspirants should consider the potential for adverse effect and involve larger cohorts. Independent validation of prior results through a randomized controlled trial evaluating paper tape versus no treatment would be of particular use in informing further systematic review and guideline development. As the body of evidence grows, homogeneity across studies will allow for quantitative meta-analysis and, ultimately, use of up-to-date methodology such as Grading of Recommendations Assessment, Development and Evaluation for the synthesis of evidence-based clinical practice guidelines.
Author Contributions
All authors contributed to study design and manuscript preparation. Drs. Worthing and Percy were responsible for independent analysis of included studies. Dr. Joslin served as mediator for any dispute.
Financial/Material Support
None.
Disclosures
None.
Footnotes
Acknowledgments
Mr. Mark Ingram, University of Kentucky Medical Center Library, Mr. David Rothman, SUNY Upstate Medical University, and Ms. Brandy Tuttle, Duke University Medical Center Library, for assistance with search strategy. Dr. Oscar Ortiz-Vargas for assistance with Spanish language translation. Dr. William D Grant, SUNY Upstate Medical University, and the University of Kentucky Applied Statistics Lab for guidance regarding statistical analysis.
Supporting information
Supplementary data associated with this article can be found in the online version at
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
