Abstract
Komodo dragons (Varanus komodoensis) are large lizards known to take down prey even larger than themselves. They rarely attack humans. A 38-year-old woman was bitten by a Komodo dragon on her hand while cleaning its enclosure. She was transiently hypotensive. The wounds were extensively cleaned, and she was started on prophylactic antibiotics. Her wounds healed without any infectious sequelae. Komodo dragon bites are historically thought to be highly infectious and venomous. Based on a literature review, neither of these are likely true. As in any bite, initial stabilization followed by wound management are the main components to therapy.
Introduction
Komodo dragons (Varanus komodoensis) are the world’s largest lizards. They are native to Indonesia. These large lizards are carnivorous and are known to kill large prey, including water buffalo, deer, and pigs. 1 There have been various theories about how they are able to kill animals that are larger than them. These theories include induced sepsis, envenomation, and blood loss. We present a case of a zookeeper who was bitten by a juvenile Komodo dragon. The animal was a healthy and captive-born female, weighing approximately 5 kg.
There are only a few reports of Komodo dragons attacking humans. There is 1 published case report detailing a zookeeper who was bitten on the hand, causing a mallet finger injury. 2 In the lay press, there have been reported attacks by wild Komodo dragons in Indonesia, including up to 4 fatal attacks over the past 35 years. There was also an attack at the Los Angeles Zoo, during which a journalist was bitten, requiring reconstructive surgery of his foot and ankle.
Case Presentation
A 38-year-old, otherwise healthy female zookeeper was bitten on the right hand by a Komodo dragon while servicing its enclosure at the zoo. According to the patient, the reptile reached from a ledge and bit her hand. She had a moderate amount of pain as well as brisk bleeding. She left the enclosure and attempted to control the bleeding with pressure, with assistance from a coworker. The bleeding could not be stopped, so they called 911 for assistance after approximately 30 minutes. Upon emergency medical services’ arrival, the wound was still bleeding. The patient reported feeling faint. Her vital signs were blood pressure 83/39 mm Hg by automatic cuff, pulse 78 beats per minute, oxygenation saturation 100%, and respirations 16 per minute. Her Glasgow Coma Scale was 15. The paramedics placed an occlusive dressing over the wound. An intravenous line was inserted, and approximately half of a liter of normal saline was infused while en route to the emergency department. She was also given 4 mg of ondansetron for nausea. The transport time was 8 minutes. By arrival to the emergency department, her vital signs were blood pressure 123/70 mm Hg, pulse 78 beats per minute, respiratory rate 24 per minute, oxygen saturation 100% on room air, and temperature 36.7°C. She was given 1 mg of lorazepam intravenously upon arrival for anxiolysis. On examination, there were multiple lacerations on the right hand, including a large 6 cm laceration over the dorsal aspect (Figure 1). There was no active bleeding from her wounds, and she complained only of pain at the site of the bites. She had full active flexion and extension of her digits at both the proximal interphalangeal and distal interphalangeal joints. She had normal sensation to light touch in her ulnar, radial, and median nerve distributions. Her tetanus immunization was up to date. Due to concern for a retained tooth, a radiograph was obtained, which showed a retained tooth in her fifth digit near the distal interphalangeal joint (Figure 2).
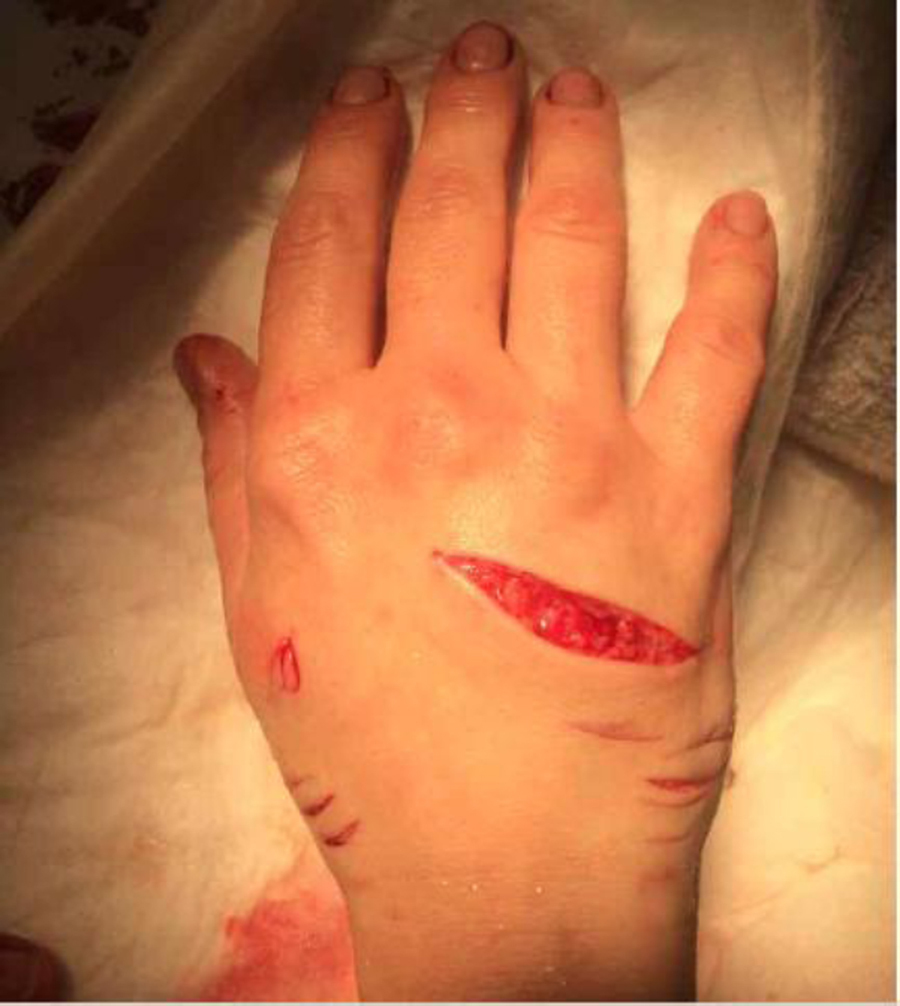
Right hand with large dorsal laceration and multiple smaller lacerations.
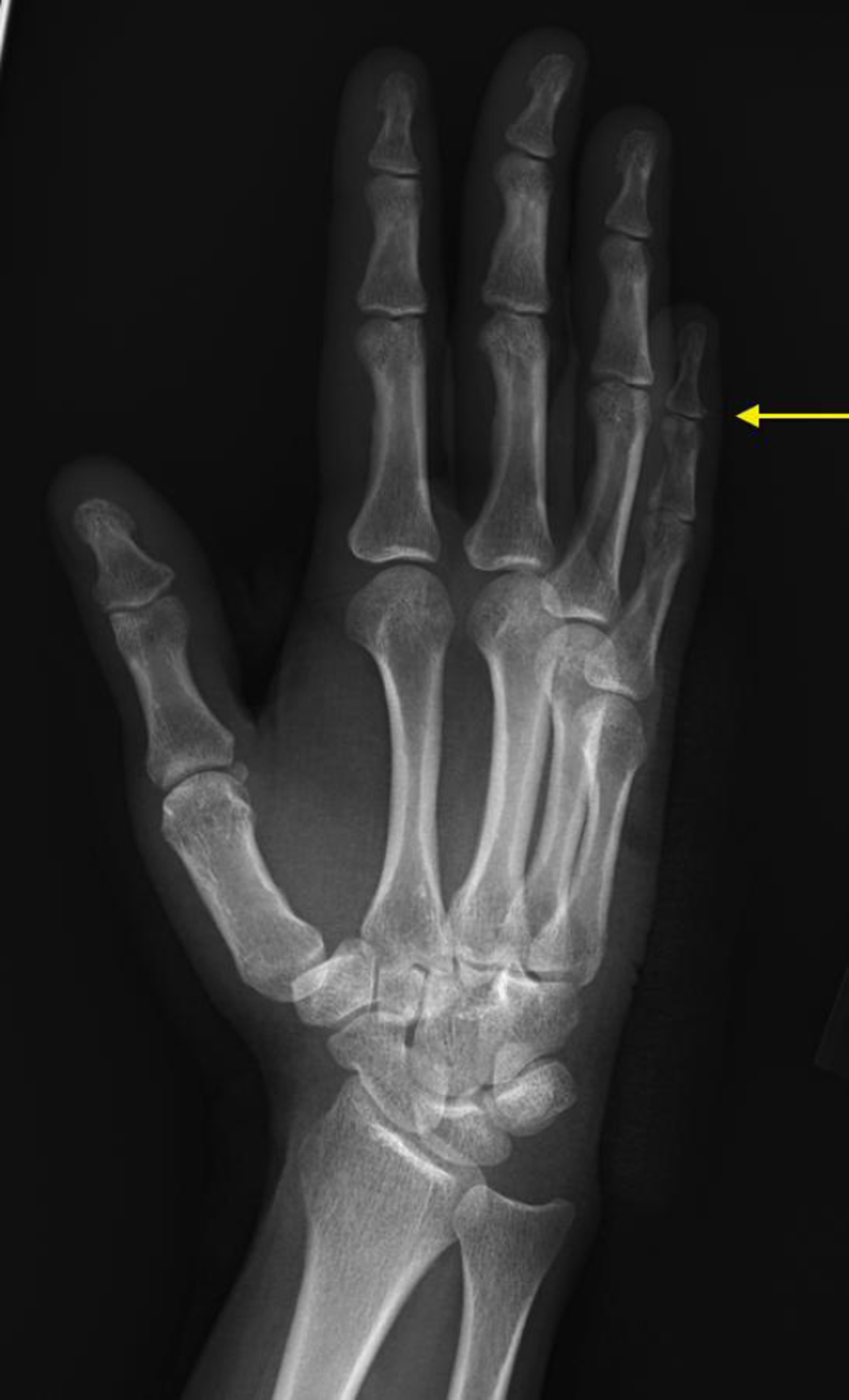
Oblique radiograph of right hand showing small retained tooth near proximal aspect of fifth distal phalanx (as denoted by the yellow pointer arrow).
The wounds were injected with 0.5% bupivacaine with epinephrine for local anesthesia. She was also given 1 mg of hydromorphone intravenously for further analgesia. The wounds were extensively irrigated with 2 L of normal saline. The large dorsal laceration was loosely approximated with 2 4−0 nylon simple interrupted sutures. The wound overlying the retained tooth was a small puncture wound. This was not probed or enlarged to try to remove the tooth, which was left in place. She was observed for 2 hours and remained asymptomatic. Her blood pressure remained normotensive. She was started on a 1-week course of amoxicillin/clavulanic acid and followed up with an orthopedic hand surgeon. The tooth self-extruded after a few weeks. Her wound healed without any infectious complications. At 8 months postinjury, she still has tingling and numbness in the distal aspect of her right fifth finger. The Komodo dragon remains on exhibit at the zoo, and there have been no further incidents with this animal.
Discussion
Komodo dragon bites are historically thought to be highly infectious. Prey was observed to get lethargic after bites, which was thought to be due to sepsis induced by oral bacterial flora from the bite.1,3 A human death due to sepsis after a bite to the patient’s arm has also been reported, although this case did not have formal medical verification. 1 This theory is known as the “bacteria as venom” model. 3 One study isolated multiple pathogenic species of bacteria from the saliva of wild and captive Komodo dragons. 4 Escherichia coli, Klebsiella sp., Staphylococcus sp., and Strepcoccus sp. were isolated from the saliva of wild dragons. Staphylococcus capitis and Staphylococcus caseolyticus were the most common bacteria isolated from the saliva of captive dragons. In this same study, Pasturella multocida grew from the blood of mice that died after being injected with Komodo dragon saliva. 4 Pasturella multocida is a bacterium common to many infections associated with bites, including from dogs and cats. 5 It is not a common bacterium in the oral flora of other reptiles, and in an additional study on the microbiology of saliva of captive Komodo dragons it was not isolated from any of the dragons. 6 These results do show that bite-induced sepsis is possible, although there is no proof that Komodo dragon bites are actually more infectious than any other animal bite or that their native oral bacterial flora are in fact specifically advantageous to Komodo dragons for killing prey. Multiple authors have cast their doubt upon the “bacteria as venom” model. 3 ,6,7 The bacterial flora isolated from their mouths may reflect bacteria acquired from environmental sources or from feeding on carrion, which is known as the “passive acquisition” model. 3
There are other factors that contribute to the lethality of Komodo dragons. They have relatively weak bites. 8 Despite this, they are still able to take down large prey. Their jaws are best built to bite down on prey and resist pulling forces. 8 In fact, their pulling strength is much larger than their biting strength. 8 They also have serrated teeth. This results in tearing of flesh. A recent study also showed that these lizards have secretory glands in their mandible, which the authors suggest are in fact venom glands. 7 These glands were analyzed and were reported to contain compounds similar to snake venom. 7 It is speculated that these compounds may result in coagulopathy, muscle spasm, and hypotension. 7 Despite not having grooves in their teeth for potential venom delivery, hypothetically, venom secretion over their teeth into open wounds created via biting and pulling could still result in effective venom delivery. Hypotension resulting from the venom could cause lethargy, consistent with the observations that originally resulted in the “bacteria as venom” theory. There is no proof, however, that the oral product of these lizards significantly contributes to prey capture, and thus it remains unclear if it has “venom,” as biologically defined. 9 There are also no reports to suggest any proof of clinical envenomation in humans, including a recent case report in which there were no signs of envenomation. 2
We did see transient hypotension in our patient. There are multiple potential causes including hemorrhagic shock, bacterial endotoxin release, overwhelming sepsis, and vasovagal reaction. Hemorrhagic shock is possible but less likely as the bleeding was controlled at the time of arrival. Sepsis or endotoxin release causing hypotension seems unlikely as she did not develop a wound infection and improved with minimal intervention. We hypothesize this was likely due to a vasovagal reaction. The patient admitted to feeling faint and had a relatively low heart rate while she was hypotensive. The hypotension resolved quickly and with little intervention.
For clinicians, Komodo dragon bites can be approached in a similar manner as other animal bites. Management should focus on bleeding control, assessing hemodynamic stability, and most importantly, wound management. As in any animal bite, extensive irrigation is paramount to preventing infection. Prophylactic antibiotics are suggested to decrease the risk of infection, especially when the bite is located on the hand. There is no evidence to suggest a possibility of clinically important envenomation in humans.
