Abstract
Objective
The purpose of this study was to determine whether 400 µg/kg oral ivermectin is able to kill
Methods
Ten study subjects each wore 2 ostomy bags, the one containing 24
Results
Eleven percent of the
Conclusions
There were a high number of ticks that died in both groups but the data do not support our hypothesis that ivermectin can kill
Introduction
The deer tick, or blacklegged tick,
Lyme disease is responsible for more than 90% of all human vector–borne disease in the United States, with an estimated 35,000 cases a year and an additional 60,000 cases annually in Europe.1,2 Lyme disease is most common in the northeastern and midwestern United States where
The
Current recommendations for preventing tick-borne disease are often impractical and have limited effectiveness. They focus on reducing deer and mice populations, avoiding tick habitat, wearing light-colored clothing to aid in identifying the ticks, tucking socks into your pants, and applying insect repellents and insecticides.
7
The US Centers for Disease Control and Prevention (CDC) recommends using insect repellents containing 30% to 50% N,N-diethylmetatoluamide (DEET) or picaridin, but both require frequent reapplication and are only effective on areas of the body and clothing where they are applied.
7
Ticks may walk across a surface containing DEET to attach to skin where DEET is absent.
3
Permethrin acts more as an insecticide than a repellent and will kill ticks after prolonged exposure. Permethrin is applied to clothing and is not indicated for application on skin.
3
Prophylaxis after exposure with 200 mg oral doxycycline within 72 hours of tick attachment has been shown to reduce the risk of infection from
Ivermectin is one of the most ubiquitous antiparasitic drugs, with more than 5 billion doses of ivermectin-containing products sold worldwide. 8 A single 150 to 200 µg/kg dose of ivermectin has shown effectiveness against several human parasites, including the ectoparasites lice, scabies, mosquitoes, and bed bugs. 9 –12 Ivermectin has been used extensively in the control of Onchocerciasis (river blindness) and filariasis and is administered to an estimated 50 million people annually.13,14 As the only approved endectocide for human use, approximately 1 billion ivermectin treatments have been given to humans over the past 30 years.10,13
Ivermectin acts on the glutamate-gated chloride channel, which is lacking in humans, and to a lesser extent the γ-aminobutyric acid (GABA)-gated chloride channel, causing hyperpolarization of invertebrate nerve and muscle cells.8,14 In humans, ivermectin is widely distributed in the body, with a peak plasma level occurring approximately 4 hours after taking the drug.14,15 The peak concentration of ivermectin in sebum, sweat, and squames is approximately 8 hours after drug consumption, and levels drop after 24 hours. 15 The antiparasitic effects of ivermectin appear to persist longer than the 18 to 22 hour half-life.14,15
Although ivermectin has been used in veterinary medicine to kill ticks on animals, there is only a single previous report about the use of ivermectin to kill ticks on humans.
11
Our study was designed to determine whether a single dose of oral ivermectin could be used to kill
Previously it has been shown that ivermectin is effective at killing several different human parasites at 200 µg/kg, but 400 mg/kg was shown to be more efficacious for lice with few additional side effects. 8 –15 We chose to use 400 µg/kg in our study with the rationale that a single large dose of the drug would be the ideal option should ivermectin ever be adopted for wider use in the prevention of tick-borne disease. Hypothetically, ivermectin could be taken shortly after a person has a potential tick exposure and would kill the tick when it starts to feed but before transmitting tick-borne disease. Because ivermectin has such a long half-life, it even could potentially be effective if the tick wandered on the body for several days before attaching and consuming a lethal dose of ivermectin.
The current research is a follow-up study to one conducted in 2011 suggesting that ivermectin might be able to kill ticks. However, too few ticks attached to study subjects to reach statistically meaningful conclusions. 11 In this study, we increased the number of study subjects, ticks, and attachment time to increase the number of attached ticks. 11
Methods
This study was approved by the Eastern Virginia Medical School and Old Dominion University Institutional Review Boards. The US Food and Drug Administration granted investigational new drug approval for the use of 400 µg/kg of ivermectin against
The study was a randomized, double-blind, placebo-controlled, phase 2 clinical trial to determine whether a single oral dose of 400 µg/kg ivermectin could kill feeding
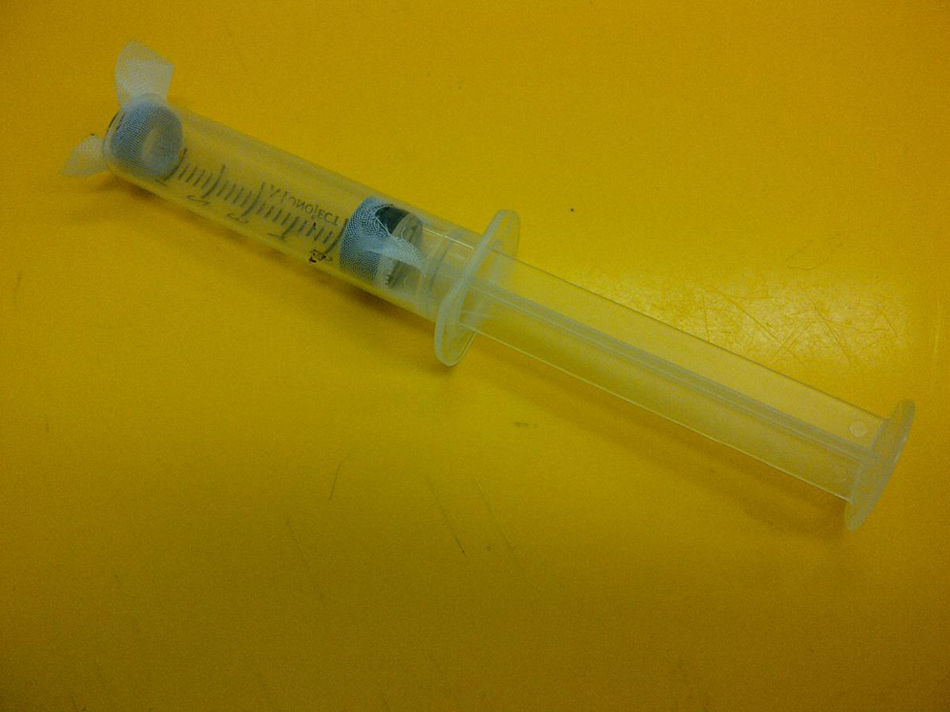
Modified syringe for injecting ticks into the ostomy bags. At the time of injection, the rubber washer and cheesecloth at the end of the syringe is removed and the plunger is depressed. Image courtesy of Dr Michael Levin.
Each study subject had two Securi-T (Genairex, Largo, FL) 6-inch, one-piece ostomy system bags cut to 2-3/8 inch internal diameter applied to their abdomen using Stomahesive paste (ConvaTec, Skillman, NJ). Osto-Bond (M.O.C., Vaudreuil, Quebecc, Canada) skin bonding latex adhesive was then applied around the perimeter of the ostomy bag where it attached to the skin. Some study subjects also applied tape around the perimeter of the ostomy bag to assist in securing the bag to the abdomen (Figure 2). A syringe containing the ticks was then injected into each ostomy bag before it was manually sealed. Nine of 10 study subjects received super glue (Loctite, Rocky Hill, CT) to seal the ostomy bag closed. All study subjects also used tape or a manual locking closure device or both for the ostomy bag. Each study subject received 24 nymph and 24 adult female
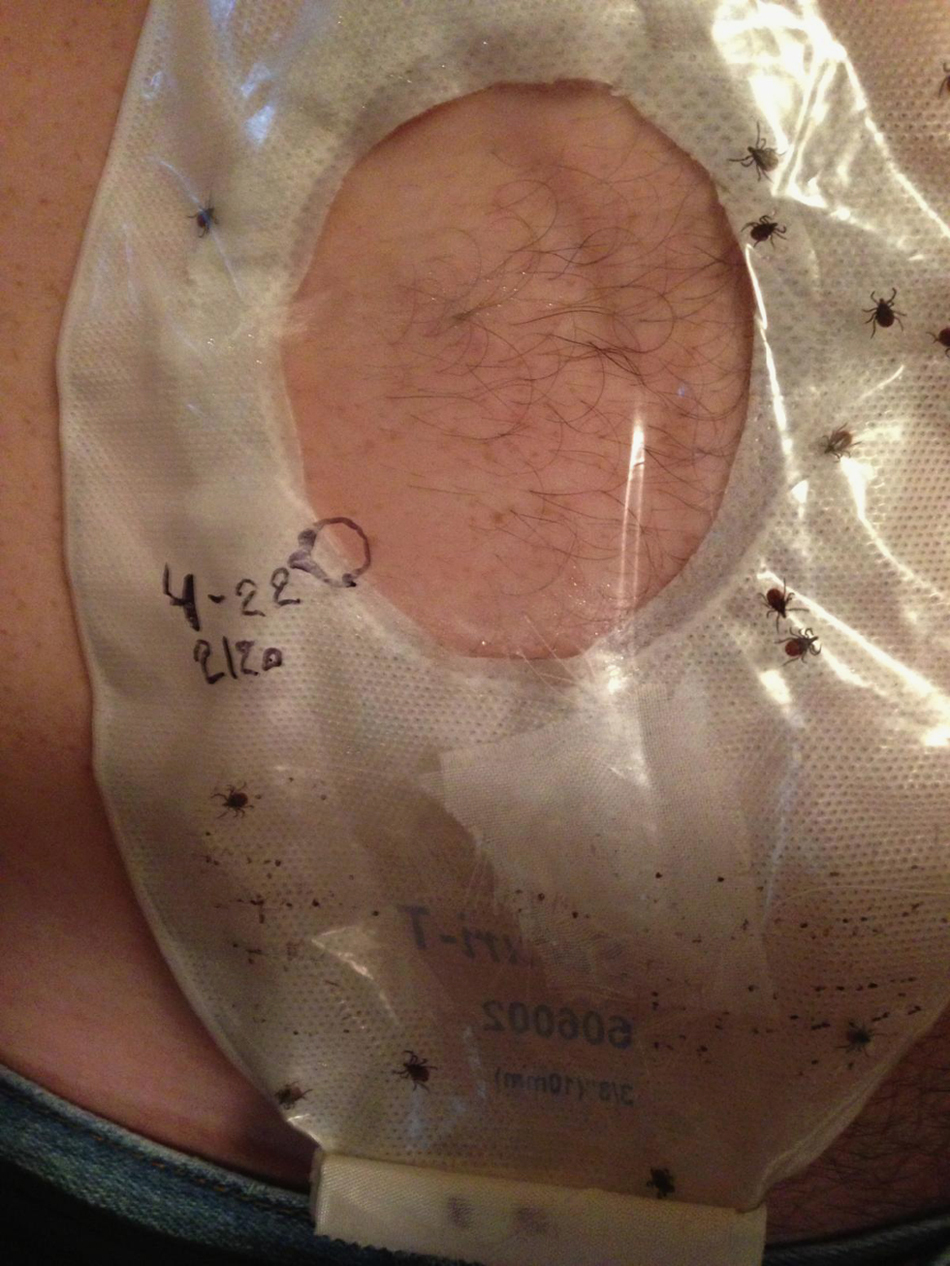
Ostomy bag with
Study subjects had the ostomy bags secured to their abdomen and then the ticks placed inside on day 1 of the experiment. Twenty-four hours later (day 2) they returned to the laboratory and were randomly allocated to receive either 400 µg/kg ivermectin or placebo (empty gel capsule). There were 5 subjects in each arm. Randomization to study intervention was determined by a random number generator, except for 1 study subject who was unable to return to the laboratory on day 2 because of an emergency unrelated to study participation. This subject was placed in the placebo group. On day 3, study subjects returned 54 hours after the ticks were initially placed (30 hours after ivermectin or placebo was ingested) to have all ticks removed. Dr Sonenshine, one of the study coauthors who is a tick expert with more than 30 years of tick research experience, removed, counted, and divided the ticks into the following four categories: alive attached, dead attached, alive unattached, and dead unattached. At the time of ostomy bag removal, all ticks were placed into test tubes, and after 5 days (day 8) mortality rates were again recorded by Dr Sonenshine.
Results
One hundred twenty
Data summary a
Each group started with 120 ticks, but 1 study subject in the placebo group did not have ticks observed past day 3; all were reportedly dead.
On day 3, 54 hours after the ticks were placed on study subjects, and 30 hours after potential ivermectin exposure, 6 of 11
On day 3, 0% of the
Thirty-eight percent of the
Nine of 10 study subjects received Loctite super glue. The single study subject who did not receive the super glue received ivermectin and had, on day 3, 5 alive unattached nymphs, 1 dead attached nymph, and 14 dead unattached nymphs as well as 2 alive attached adults and 21 alive unattached adults.
There are discrepancies between the total number of ticks that attached during the experiment as recorded on day 3 and day 8 even though all ticks were removed from study subjects on day 3. Only the ticks that were physically attached and feeding on day 3 when they were removed from the study subject’s skin were counted as attached on day 3. On day 8, any tick that showed signs of having been attached and fed were counted as having been attached. A tick will usually not stop feeding and detach.
Study subjects were followed up for potential adverse events from the time of consent through the next 8 days. Symptoms developed in 4 study subjects during study participation. Three study subjects had itching, burning, or erythema within minutes of applying the Osto-Bond and ostomy bags and before the ticks were placed inside the bag. One study subject required the ostomy bags to be removed but requested they be reattached without Osto-Bond, and they were taped to the abdomen using athletic tape. Two of these subjects took over-the-counter diphenhydramine that relieved their symptoms. One study subject reported nausea and another dizziness before either received ivermectin or placebo. In the ivermectin arm, 1 study subject complained of chest pain on day 6 and “stomach trouble” on day 7; both complaints resolved spontaneously without medical intervention. Headache developed in 1 subject in each group and resolved without medical intervention. One study subject was unable to return to the laboratory on study day 2 owing to involvement in a minor motor vehicle accident while driving to the study site. To maintain even study arm assignment, this subject was placed in the placebo group and never received any study drug. This study subject received a large amount of Loctite glue to close the ostomy bag and had reported previously that all her ticks died several hours after they were placed inside the ostomy bag and that none of the ticks attached to feed. The ticks from this study subject were discarded on day 3 and not observed on day 8.
Discussion
Tick mortality in both our placebo and ivermectin groups was so high that definitive conclusions about the effects of ivermectin on
Given the large number of ticks that died during the study, some shortly after they were placed in the ostomy bag, we strongly suspect a toxic exposure. The temperature or humidity from inside the ostomy bag would not have killed the ticks. Ticks are very resistant to mechanical trauma, so that is an unlikely cause of their death. Ticks are able to survive in a sealed container for several days without ill effects, so hypoxia would not have contributed to mortality. Lastly, it is unlikely that the ticks died of an infectious process as the ticks were grossly normal before being put into the ostomy bags.
Our study did not look at the ability of ivermectin to prevent
There are few published reports or clinical trials in the scientific literature involving the attachment of ticks to humans. This is the second known attempt at using an ostomy bag attached to the abdomen to secure the ticks to the study subject. Problems with ticks escaping from the bag, as well as premature tick death, mean that other methods of confining ticks to humans need to be developed. Methods of confining ticks to animals have been developed and include gluing (veterinary approved cyanoacrylate-based glues) plastic containers or stockinettes to the animal’s skin, and then when the glue is dry, placing the ticks inside (Figure 3). However, these approaches are less practical for humans. We suspect that the ethyl cyanoacrylate-based glue contributed to our overall tick mortality and would recommend avoiding acute exposure of the ticks to the glue vapor in future experiments. In addition, 1 or possibly even 2 additional days should be added to the tick exposure period before beginning the ivermectin treatment. This would insure a large population of attached feeding ticks at the time of drug administration.

A stockinette glued to a rabbit and tied at the top (left). Untying the stocking allows for direct visualization of the feeding ticks (right). Image courtesy of Dr Michael Levin.
Study Limitations
We had unacceptably high mortality in both the ivermectin and placebo groups was that thought most likely to be related to the Loctite super glue placed at the opening of the colostomy bag. Loctite has an ethyl cyanoacrylate base, and the vapors from the glue drying likely affected the ticks. However, it is possible that the ostomy glue placed around the ostomy bag may have contributed to mortality as well. We believe that the high mortality seen in the placebo group demonstrates a structural flaw in our study design. The ostomy bags were not securely attached to study subjects, as reflected in the large percentage of ticks that had escaped by day 3. The lack of a tight seal between the ostomy bag and the skin allowed water and soap to enter the bag when study subjects showered. Most of the ticks accumulated at the bottom of the ostomy bag. In our experiment, the opening of the ostomy bag was caudal and by orientating the opening cephalad, we would have kept the ticks closer to the skin rather than the ostomy pouch. Study subjects applied athletic tape around the perimeter of the ostomy bag but that was not completely effective at creating a tight seal against the skin.
Conclusions
Our data demonstrate that no definitive conclusion can be made regarding
Footnotes
Acknowledgments
We would like to thank both the Wilderness Medical Society for the Research in Training Award for 2010 and Eastern Virginia Medical School Department of Emergency Medicine for funding this research. We also thank Dr Michael Levin and Lauren McColley in the Medical Entomology Laboratory, Rickettsial Zoonoses Branch, US Centers for Disease Control and Prevention in Atlanta, Georgia, for providing us with the ticks and technical assistance, and for providing the images for ![]() used in the manuscript.
used in the manuscript.
☆
Funding was provided by a Research in Training Award for 2010 from the Wilderness Medical Society and the Eastern Virginia Medical School Department of Emergency Medicine. The authors have no financial disclosures or conflicts of interest to report.
