Abstract
Lyme disease is a multisystem tickborne illness caused by the spirochete
Introduction
Vectorborne diseases, including those spread by ticks, pose a growing risk to outdoor enthusiasts. The 2013 Outdoor Participation Report estimated that nearly 50% of US residents age 6 and older participated in outdoor recreational activity in 2012, accounting for more than 12 billion outdoor excursions. 1 Twelve percent of all US residents reported hiking, 13% reported camping, and 19% reported jogging or trail running. 1 Among hikers there was an average of 18 outings annually with a cumulative 603 million hiking outings in 2012. 1 In the next 30 years, person-days spent hiking, backpacking, and camping are all expected to increase, doubling in some areas of the country. 2
Lyme disease is a multisystem illness caused by the spirochete
History
Lyme disease was first described by Steere et al
4
in 1977 after an investigation of a cluster of arthritis cases in children living near Old Lyme, Connecticut, and in 1981 the causative spirochete
Causative Agent
Transmission
Transmission of Lyme disease to humans occurs through a bite of certain species of infected
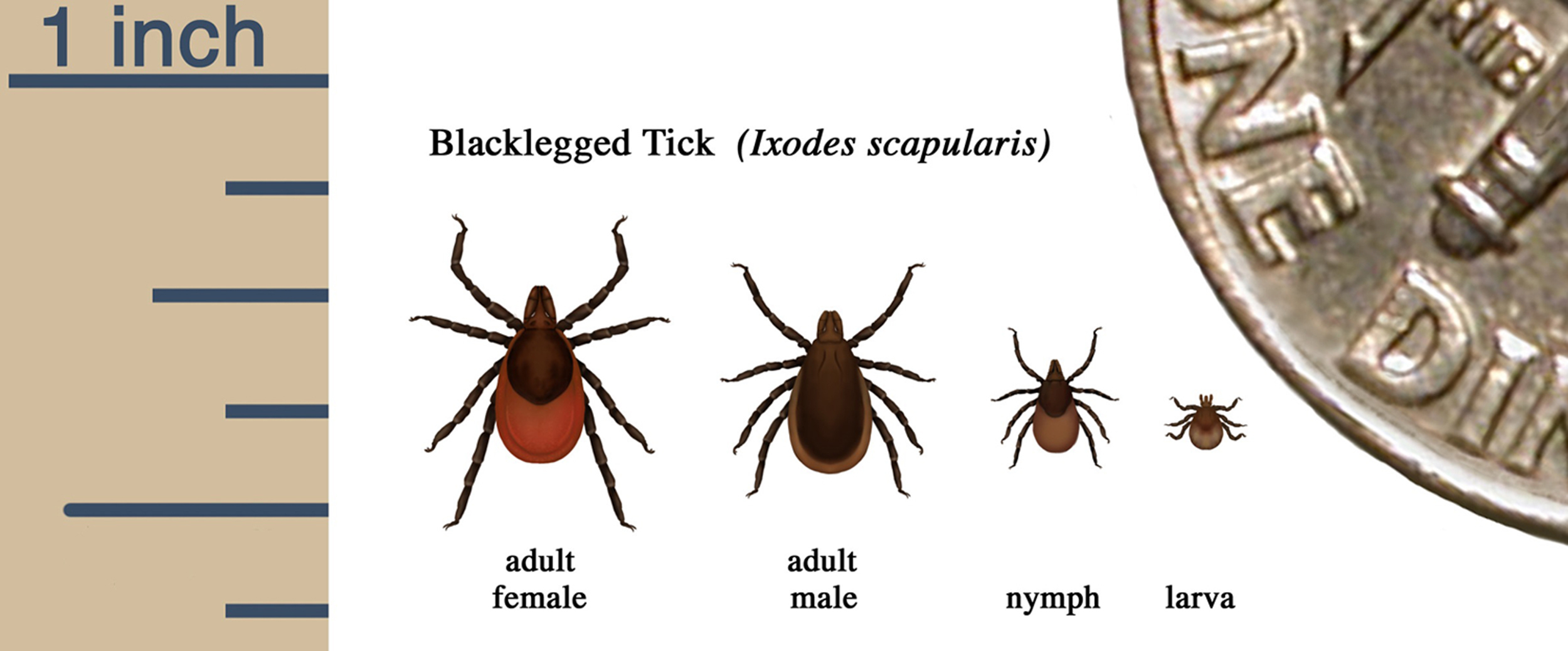
Humans are not part of the natural life cycle of the spirochete. Human infection only occurs when humans enter areas where ticks, infected reservoir hosts, and deer coexist at a level high enough to support a dynamic enzootic cycle (Figure 2). Larval ticks hatch in the spring from eggs laid the previous fall; they are uninfected as the spirochete is not transmitted transovarially. 14 After several days of maturation, the larval ticks climb up foliage and begin to “quest” for hosts (Figure 3), taking their first blood meal in the late summer, typically from small rodents, birds, and deer. 12 This blood meal lasts for approximately 3 days, after which the larva will drop to the ground to digest the meal. 15 This is the first time that the ticks can become infected, most commonly from an infected rodent. It is important to note that although deer can provide the blood meal for the tick, deer are immune to infection and do not transmit the spirochete to the larva.
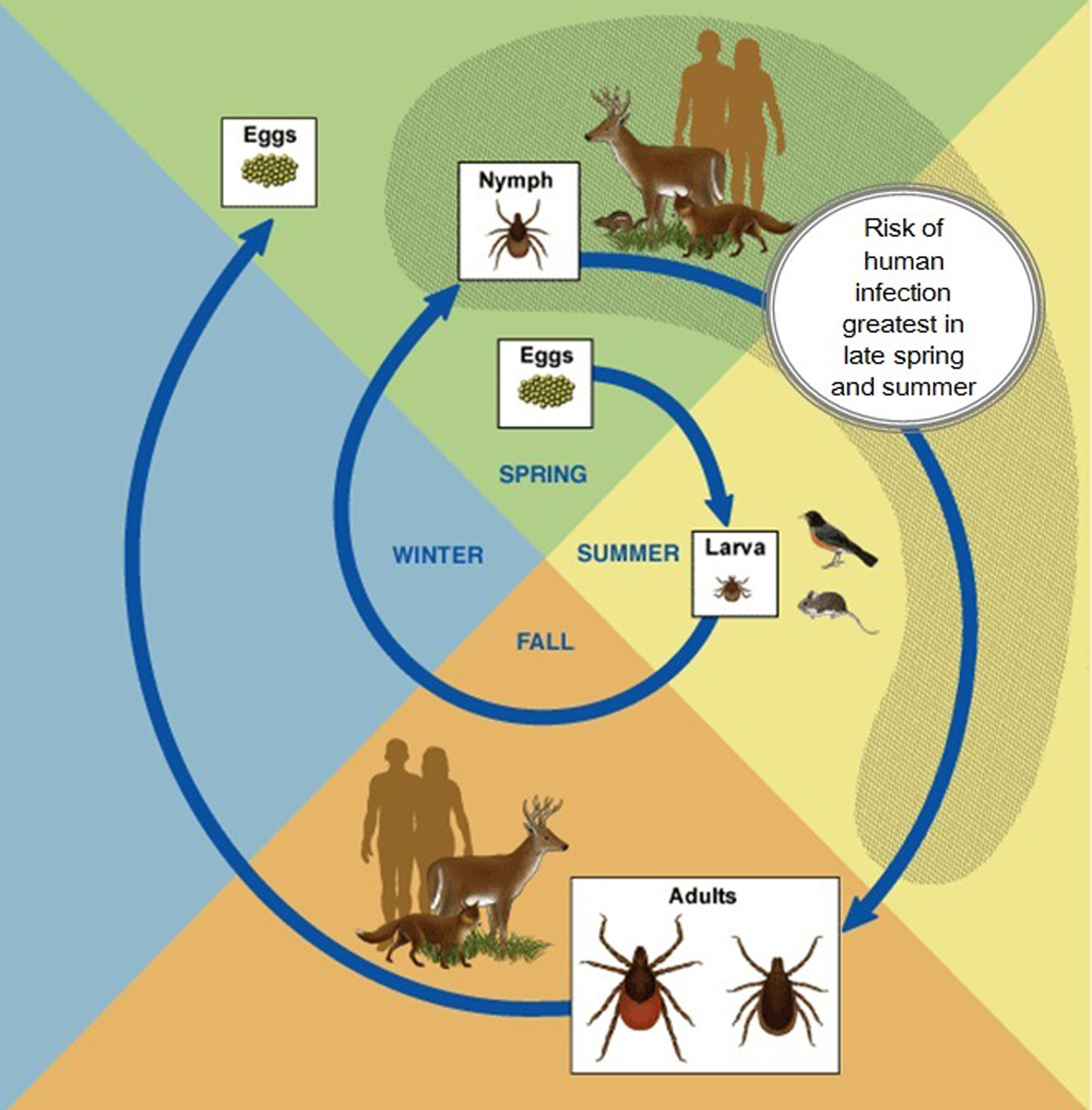
Lifecycle of the

Adult
The larval ticks molt into nymphal ticks that quest again, searching for a blood meal; humans can be incidentally fed on during this time. If the nymph was infected as a larva, then the infected nymph can transmit the spirochete. Nymphal ticks typically feed during the late spring and early summer, with meals lasting approximately 5 days, after which the nymph disengages and falls to the ground. 15 Although both nymph and adult ticks can transmit the spirochete, nymphs are believed to be the principal source of human infection owing to their greater abundance and smaller size, which makes detection more difficult.
The nymph molts to become an adult tick that feeds in the fall, most commonly on deer, although humans can also serve as blood meal sources. Again, the spirochete can be transmitted transstadially from nymph to adult. Adult female ticks consume more than male ticks, reaching more than 200 times their prefeed body weight. 15 Adult ticks feed for approximately 1 week and then drop to the ground.13,15 If the adult tick feeds on humans, spirochetes can be transmitted. After feeding, adult female ticks can lay approximately 1000 to 10,000 eggs before dying. 12
Pathogenesis
To maintain the enzootic cycle, spirochetes must be viable in both mammalian and tick hosts. There are substantial differences in temperature and pH between mammals and ticks, requiring adaptation by the spirochete.
When the tick takes a blood meal from an infected host, spirochetes travel through the mouth and into the midgut of the tick. The spirochete lays dormant there until the following spring (if a nymph is infected) or fall (if an adult is infected).
10
When the tick attaches for its next blood meal, the spirochetes multiply in the tick midgut and switch from expressing outer-surface protein A (OspA) to expressing outer-surface protein C (OspC).
10
The spirochete then migrates from the midgut to the salivary glands; from there it can be injected into the host. Typically, a feeding period of at least 36 hours is required for the spirochete to make this journey.
16
Consequently, risk of infection with
After leaving the tick, spirochetes disseminate locally at the site of the tick bite, initiating a robust innate immune response. 15 If untreated, spirochetes can disseminate and infect other organ systems, including joints, heart, and nervous system tissue. Systemic clinical manifestations of infection are caused by the migration of spirochetes through host tissue, adhesion to host cells, and the subsequent immune response; the spirochete has no traditional virulence factors.11,15
Epidemiology
There are approximately 30,000 confirmed and probable cases of Lyme disease reported annually to the Centers for Disease Control and Prevention (CDC); the actual number of annual infections is estimated to be 10-fold higher.8,17 Although cases have been reported by residents of all states, 95% of cases occur in high-incidence states or districts in the Northeast (Connecticut, Delaware, Maine, Massachusetts, New Hampshire, New Jersey, New York, Pennsylvania, Rhode Island, Vermont), Mid-Atlantic (Maryland, Virginia), and upper Midwest (Minnesota, Wisconsin; Figure 4;
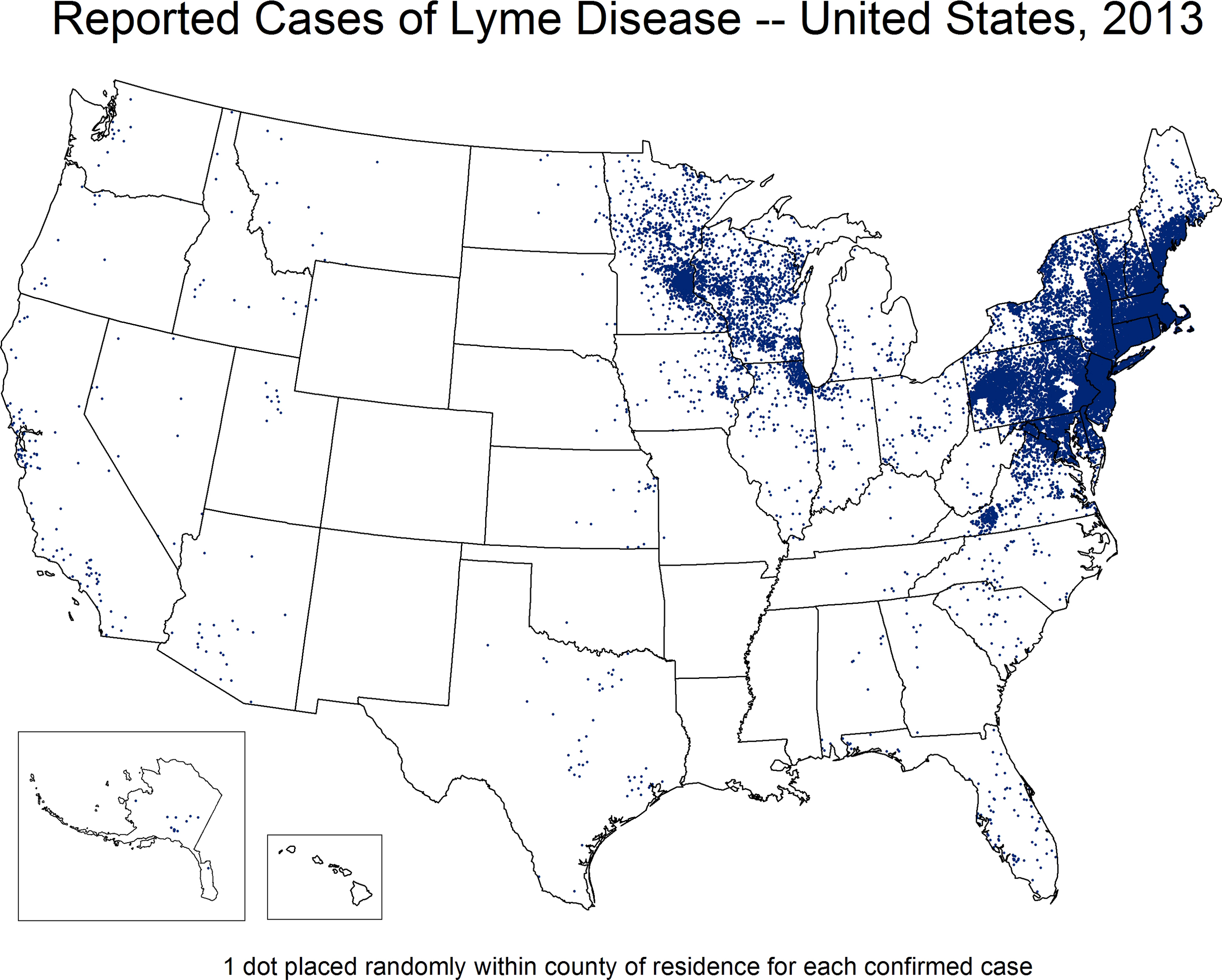
Reported cases of Lyme disease in the United States by county of residence, 2012. One dot is placed randomly within the county of residence for each confirmed case (figure courtesy of CDC).
The age distribution for infected patients is bimodal, with a slight male predominance (55% male); boys 5 to 9 years of age account for the largest proportion of Lyme disease cases reported from 2001 to 2010 (
Clinical Manifestations
Lyme disease can be divided into 3 stages: early localized, early disseminated, and late disseminated. Wilderness medical providers will be most likely to encounter patients with early localized infection. The hallmark of early localized disease is the erythema migrans (EM) rash; patients may also have constitutional symptoms such as fever and myalgia. The EM rash is typically described as a macular, erythematous, round or oval skin lesion that expands gradually over a period of days to reach a diameter of 5 to 20 cm (Figure 5). 18 The EM rash is generally not painful or pruritic, but it may be warm to the touch. It usually develops at the site of the infected tick bite, typically 7 to 14 days after tick detachment. Approximately 70% to 80% of patients with Lyme disease will present with an EM rash, and it is the most valuable clinical sign for the wilderness practitioner. In the appropriate clinical situation, the EM rash is the only sign of Lyme disease that is sufficiently distinct to allow clinical diagnosis in the absence of laboratory confirmation. 19 Importantly, the usefulness of the EM rash is strongly dependent on the probability of the patient being exposed to an infected tick. As the potential for exposure to infected ticks decreases, so does the probability that an EM-like rash actually represents true infection; concomitantly the possibility of a false-positive diagnosis of Lyme disease based on EM increases.
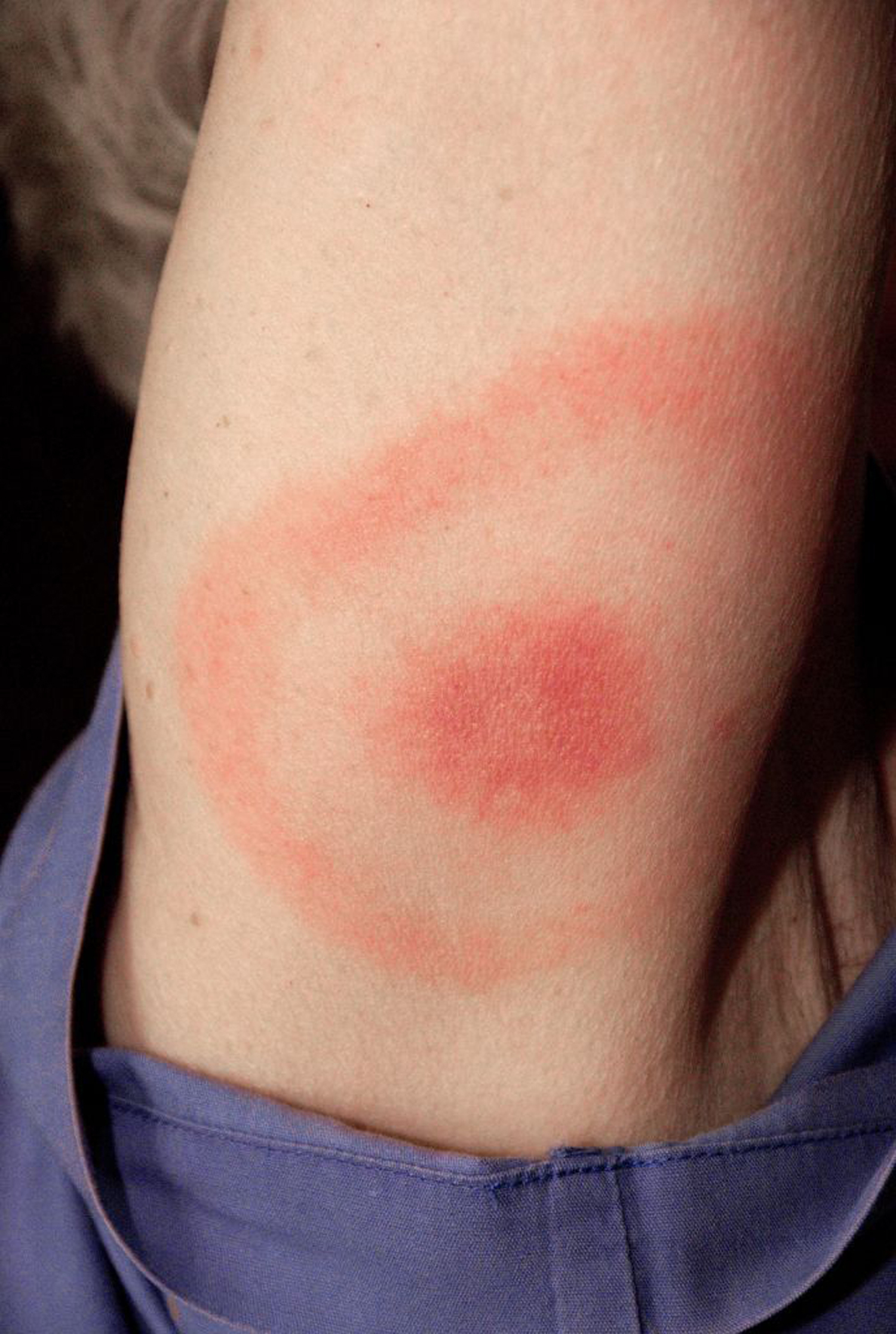
Erythema migrans lesion, left underarm (figure courtesy of CDC).
Rashes that develop within 48 hours of tick bite are more likely a tick bite hypersensitivity reaction and do not necessarily represent transmission of the spirochete.
18
One way to differentiate between EM and a local hypersensitivity reaction is to mark the rash and evaluate for 1 to 2 days before initiating antibiotic therapy; the EM lesion should continue to enlarge.
19
Providers should be aware also of another condition, Southern tick-associated rash illness (STARI), which can mimic the EM rash associated with Lyme disease. STARI can occur after the bite of an
Without early treatment,
Late disseminated Lyme disease is characterized by rheumatologic and neurologic manifestations. Intermittent monoarticular or oligoarticular arthritis involving the knee, with effusions that are out of proportion to pain, is typical. 19 Untreated, these episodes will typically resolve over weeks to months, often even without antibiotic therapy. 19 Clinical diagnosis of Lyme disease based solely on rheumatologic symptoms is unlikely to be of benefit to the wilderness provider as many acute and chronic ailments can be similar to Lyme arthritis. Late neurologic manifestations of Lyme disease, such as encephalomyelitis and peripheral neuropathy, are rare and will be the least likely clinical symptoms to be encountered by wilderness providers. 19 Patients suspected of having late disseminated Lyme disease should be evaluated by a physician as soon as possible.
A small proportion of patients (10%–20%) treated for Lyme disease may have persistent symptoms including fatigue, pain, and joint and muscle aches. 27 This manifestation of Lyme disease presentation, described as posttreatment Lyme disease syndrome, currently has no known etiology. Prolonged courses of antibiotics have not shown any improvement in patients compared with those receiving placebo. 28 –30 Additionally, prolonged antibiotic therapy and incorrect diagnosis have been associated with complications, including death. 31 –36 Patients suspected of having posttreatment Lyme disease syndrome should be evaluated by an infectious diseases physician.
Readers are directed to existing reviews for additional information regarding clinical manifestations of Lyme disease.6,19
Diagnosis
Practitioners in wilderness or austere settings are unlikely to have immediate laboratory confirmation for suspected infection. Nevertheless, it is important to have a working knowledge of the recommended diagnostic testing for Lyme disease. There are a variety of laboratory tests developed to detect
Serologic tests are by far the most common means of confirming a clinical diagnosis of Lyme disease. Currently available serologic tests include whole-cell sonicate or C6 peptide enzyme immunoassays and Western blot to assess for immunoglobulin (Ig) M and IgG antibodies to specific spirochete antigens. A 2-tiered approach is recommended in which samples are first tested by enzyme immunoassays and then tested by Western blot only if positive or equivocal results are obtained (
Direct tests include culture, detection of
Prevention
Currently, the best method for preventing infection with Lyme disease or other tickborne diseases is avoiding the bite of infected ticks. Black-legged ticks typically live in moist and humid environments, particularly around the confluence of wooded and grassy areas. Ticks quest for hosts on tall grass and brush, so avoidance of these areas reduces opportunities for exposure. Unfortunately, avoidance of these areas can be impractical for outdoor sport enthusiasts. Additional protection can be obtained by wearing clothing that covers the skin to prevent initial exposure (eg, long pants with complete covering of legs as opposed to shorts) and by applying repellents containing 20%
When outdoors in high-incidence Lyme disease areas, all persons should perform daily tick checks. 42 All areas of the body should be examined daily, and a mirror used to examine difficult-to-view areas (eg, posterior scalp line, gluteal crease, posterior aspect of the scrotum). 44 Areas where ticks frequent include intertriginous areas such as the axilla or popliteal fossae, the periauricular area, in the umbilicus, and in head and body hair, as well as in the inguinal and gluteal creases. Parents should ensure that their children receive daily tick checks while in high-incidence areas. Tick checks should be a mandatory component of the daily routine for all participants in outdoor activities in high-incidence areas. Pets should also be examined daily as ticks on pets can move to the human owner. 45 Owners of pets are advised to use veterinarian-prescribed tick-collars or spot-on treatment for both the health of the pet and prevention of tick exposure to the pet owners.
The recommended method for removing a tick involves grasping the tick as close to the skin as possible using fine-tipped tweezers and removing the tick using gentle, outward pressure (
Wilderness providers, and medical directors of wilderness programs, should consider ensuring participants in outdoor activities in high-incidence Lyme disease areas are educated to appropriate primary tick bite prevention methods and have supplies available to prevent infection.
Importantly, there may be copathogens carried by ticks with
Treatment
Wilderness providers are most likely to encounter patients presenting immediately after a tick bite with early localized or early disseminated disease and need to be knowledgeable about treatment recommendations. Medical directors of outdoor programs with participants active in high-incidence Lyme disease areas should consider establishing treatment guidelines based on the Infectious Diseases Society’s guidelines, tailored to the individual needs of the program. 19
Patients with recently attached ticks are commonly encountered in wilderness medicine practice, particularly during spring and summer months. A tick can be found on a human immediately after successfully questing, as it travels on the human to a favorable feeding location, while feeding during its blood meal, or immediately after detachment.
Ticks that have not yet attached to the host are incapable of transmitting Lyme disease, so no antibiotic prophylaxis would be recommended if a tick were found at this stage. There is little risk of infection with recently (≤36 hours) attached ticks
19
because, as discussed previously,
Routine prophylactic therapy is not recommended for all patients who experience a tick bite. Prophylaxis against Lyme disease should be given only if 1) the tick can be reliably identified as an

Degree of
Recommended antibiotic treatment regimens have been extensively documented by the Infectious Diseases Society of America. 19 A summary of recommended regiments can be found in the Table. On clinical diagnosis of Lyme disease based on the presence of the characteristic EM rash in a high-incidence Lyme disease area, wilderness providers should provide early and appropriate antibiotic therapy. If early or late disseminated Lyme disease is suspected, prompt evaluation by a medical professional in a setting in which parenteral therapy is available should be considered. Any patient treated for Lyme disease in austere settings should be offered follow-up with a physician after returning from the backcountry.
Recommended therapy for patients with Lyme disease in austere environments a
Adapted from tables in Wormser et al. 19
A single dose of doxycycline may be offered to adult patients and to children ≥8 years of age when all of the following circumstances exist: 1) the attached tick can be reliably identified as an adult or nymphal
Maximum should be adult dosing recommendation.
Finally, although Lyme disease is the most common tickborne disease in the United States, there are many other pathogens carried by ticks. 13 Patients should consider being evaluated by an infectious diseases physician if they develop signs or symptoms inconsistent with Lyme disease after a tick bite.
Conclusions
Lyme disease is the most common vectorborne disease in the United States. Human infection is a byproduct of a robust enzootic cycle dependent on pathogen, vector, and host biology. Although most cases resolve with appropriate and early antibiotic therapy, death has been reported. Wilderness medical providers, particularly those practicing in high-incidence Lyme disease areas, should be aware of recommended Lyme disease preventive measures, common clinical presentations, and established therapeutic regimens to ensure optimal patient care.
Footnotes
Acknowledgments
The authors would like to thank Marc Dolan (CDC) and Martin Williams (CDC) for the image of fed
Disclaimer: The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention.
