Abstract
Objective
The purpose of this study was to determine
Methods
Six study subjects, 3 in each group, were randomly assigned to receive either 400 μg/kg ivermectin or placebo in a blinded manner. After consuming either ivermectin or placebo, each study subject had 2 colostomy bags attached to his or her abdomen. One of the colostomy bags contained 7
Results
Fifty-five percent (6 of 11) of the attached
Conclusions
We demonstrate a novel method to confine ticks to human subjects to study tick-borne diseases. While there was a trend toward
Introduction
Ticks are obligate blood-sucking insects with a worldwide distribution. Hard ticks such as
Lyme disease, caused by
The risk of tick borne-diseases in humans can be minimized by reducing populations of deer and mice, avoiding wooded and grassy areas, applying topical insect repellents, wearing insecticide-treated clothing, promptly removing ticks from skin and clothing, wearing light-colored clothing to aid in the identification of ticks, and tucking pant legs into socks. 3 These control strategies have limited effectiveness and are often not practical. Ticks are small, hard to identify, and easily missed on skin inspection. In addition, ticks can wander on a human host for hours to days before they find a suitable attachment site and start to feed.
Ivermectin is one of the most ubiquitous antiparasitic drugs sold worldwide. More than 5 billion doses of ivermectin-containing products have been sold in the past 3 decades. 9 Ivermectin has been extensively used in veterinary medicine to control fleas and ticks but has never been tested for this purpose in humans.6,7 A dose of 150 μg/kg to 200 μg/kg ivermectin has shown effectiveness against numerous human parasites, including strongyloidiasis, ascariasis, trichuriasis, filariasis, scabies, enterobiasis, lymphatic filariasis, and onchocerciasis.6,7 Hundreds of millions of people have been treated during the past 20 years with ivermectin in a global effort to control onchocerciasis. 6 Although a 400 μg/kg ivermectin dose is not approved by the Food and Drug Administration, high doses have been safely used in humans. 10
Ivermectin acts on the glutamate-gated chloride channel, and to a lesser extent, the γ-aminobutyric acid (GABA)-gated chloride channel, causing hyperpolarization of invertebrate nerve and muscle cells. 6 ,7,9 Ivermectin causes paralysis of tick somatic and pharyngeal muscles, causes damage to tick dermal glands, dorsoventral muscles, and inhibition of epidermal cell activity. 6 –8 In humans, ivermectin is widely distributed in the body, with a peak-plasma level occurring about 4 hours after drug consumption. 6 –9 Ivermectin has a half-life of approximately 18 to 22 hours, although antiparasitic effects appear to persist much longer. 6 The peak concentration of ivermectin in sebum, sweat, and squames is about 8 hours after drug consumption, and drops after 24 hours. Ivermectin is extensively metabolized by P4503A4 and excreted primarily in the feces. Humans lack glutamate-gated chloride channels, and ivermectin only minimally crosses an intact blood-brain barrier. 6 Overall, ivermectin is a very safe drug with only rare and mild side effects, which can include gastrointestinal upset, fatigue, abdominal pain, dizziness, rash, somnolence, transaminitis, and leukopenia.
The objective of the study was to determine whether ivermectin is able to cause morbidity and mortality in ticks feeding on humans. Ticks with morbidity and mortality are not likely to transmit diseases to humans.
Methods
The Institutional Review Boards of Eastern Virginia Medical School and Old Dominion University approved this study. All study subjects were adults between the ages of 18 and 30 years, were not pregnant or trying to get pregnant, were not breastfeeding, were not taking daily medications, and were able to visualize the ticks. All women had a negative urine pregnancy test on the day of the experiment. Study subjects were excluded if they had ever had a seizure, orthostasis, an abnormal heart rate, Stevens-Johnson syndrome, toxic epidermal necrolysis, human immunodeficiency virus/acquired immunodeficiency syndrome, acquired or congenital immune deficiency, known malignancy, were on a regimen of immunosuppressive drugs, or had asthma, vision loss, conjunctival hemorrhage, hepatitis, edema, or a history of headaches. Moreover, taking the following medications precluded study participation: warfarin, benzodiazepines, barbiturates, sodium oxybate, valproic acid, clotrimazole, amprenavir, rifampin, ritonavir, St. John's Wort, phenothiazines, amiodarone, erythromycin, verapamil, tamoxifen, quinidine, ketoconazole, cyclosporine, or carvedilol owing to theoretical potential interactions with the metabolism of ivermectin. All study subjects were compensated $300 at the completion of the study.
We performed a randomized double-blinded, placebo-controlled clinical trial to determine whether orally administered 400 μg/kg ivermectin is able to kill
Study investigators could not find other published reports on specific methods to attach ticks to humans. We used pathogen-free
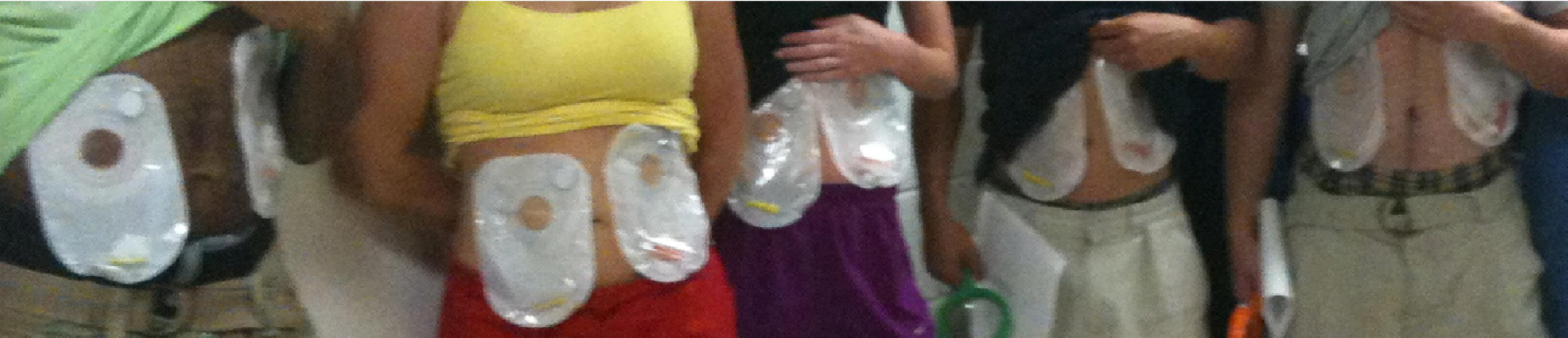
Study subjects showing off their tick-filled colostomy bags at the start of the experiment.
Each colostomy bag had small pinholes placed to facilitate gas exchange. The bags remained attached to study subjects for a total of 24 hours, after which all ticks were removed, examined, and killed. Study subjects recorded the number of ticks that were attached and dead, attached and alive, unattached and dead, and unattached and alive at 1 hour and at 2, 3, 4, 5, 6, 12, and 24 hours. Definitive tick morbidity and mortality was determined at 24 hours by Dr Sonenshine, a tick scientist with more than 30 years' experience, who was blinded to whether the ticks were attached to study subjects receiving placebo or ivermectin. Morbidity was defined as a severely lethargic and premorbid.
Aside from minor skin irritation where the ticks fed, no study participant reported any side effects from study participation, except for 1 woman receiving the placebo who reported intermittent and self-resolving dizziness, lightheadedness, anxiety, restlessness, itching, tingling, and constipation during the 24 hours of study participation. Follow-up communication demonstrated that all her symptoms resolved.
Results
Twenty-four hours after colostomy bags containing ticks were placed on human study subjects, we observed in the
In the
The
Ticks in the
A majority of the ticks that did attach to feed did so at the end of the experiment. Table 1 summarizes our data at 24 hours, and Table 2 shows how many ticks were attached in each group at various times after the colostomy bags were attached.
Tick morbidity and mortality at 24 hours
Number of ticks that attached at different times during the experiment
The nymphal stage of this species is not known to feed on humans, and it is unclear whether these ticks were truly attached or just on the skin.
Discussion
We demonstrate a novel method for containing ticks on human study subjects. During our experiment, 2% of ticks (3 of 156) escaped from the colostomy bags, and 5% of the unattached ticks (8 of 156) died (7 of which were in placebo groups). Our study was limited by the low number of ticks that attached in the allotted time frame. Ticks feed slowly, and most ticks attached late in the experiment, thus limiting their exposure to ivermectin. There may be increased morbidity and mortality among the
It would be unethical to allow
It is unclear why
Limitations
Our study is limited by the low number of ticks that attached during the 24-hour period of observation, limiting broad conclusions about the ability of ivermectin to cause tick mortality within 24 hours. Even though we had more
Conclusions
It may be possible that a single dose of ivermectin, taken within 24 hours of being at risk for tick exposure, can prevent tick-borne diseases, including Lyme disease. If that is shown to be the case, it could be of interest to hikers, backpackers, soldiers, loggers, and outdoor enthusiasts who may find that taking a pill to prevent tick-borne diseases is easier and more effective than using repellents or promptly finding and removing ticks. Ivermectin has many attractive features—it is inexpensive and safe, and has a long half life. More research needs to be done to determine whether ivermectin can be used to prevent tick-borne diseases in humans.
Footnotes
Acknowledgments
We would like to thank the Wilderness Medical Society for funding this research with the Research in Training Award for 2010. We would like to thank Yueqin Zhao formerly at Eastern Virginia Medical School, and Bhaskara Ravi, Department of Mathematics and Statistics, Old Dominion University, for help with our biostatistics and grant writing. We appreciate the support and assistance of Dr Frank Counselman, Chair, Department of Emergency Medicine, Eastern Virginia Medical School. Lastly, we would like to thank Dr Michael Levin and Liz Studer in the Medical Entomology Laboratory, Rickettsial Zoonoses Branch, US Centers for Disease Control and Prevention in Atlanta, Georgia.
Disclosures: Funding for the research was provided by a Research in Training Award for 2010 from the Wilderness Medical Society. The ticks used were purchased from Dr Michael Levin in the Medical Entomology Laboratory, Rickettsial Zoonoses Branch at the US Centers for Disease Control and Prevention in Atlanta, Georgia. The authors have no financial disclosures or conflicts of interest to report.
Presented at the 6th World Congress on Wilderness Medicine, July 13–17, 2012, Whistler, British Columbia, Canada.
