Abstract
Objective
Hemodynamic changes in response to the hypoxic environment of high altitude are vascular bed-specific. The aim of the present study was to investigate diameter and blood flow changes in conduit vessels in response to hypobaric hypoxia.
Methods
Eleven healthy subjects ascending Mount Everest to base camp participated in this study. Vessel diameter and blood velocity for brachial, carotid, common femoral, superficial femoral, and deep femoral arteries were measured by portable Doppler ultrasound. Blood flow was calculated from these values. Measurements were taken at sea level, at increasing altitudes on ascent to base camp (1310 m, 3470 m, 5330 m), and repeated on descent to lower altitude (1310 m).
Results
For all vessels except carotids, both vessel diameter and blood flow decreased between sea level and initial ascent to altitude, with subsequent persistence of these decreased values; there was no further significant change with continued ascent to higher altitude. Blood flow for all arteries (except carotids) increased significantly on descent to lower altitude, with an associated nonsignificant increase in velocity and decrease in diameter.
Conclusions
This study showed that there is vasoconstriction of limb conduit vessels at altitude, which persists upon descent to lower altitude. Blood flow in these vessels also decreases with initial exposure to high altitude, yet increases when returning to lower altitude, reflecting variations in blood velocity. Carotid arteries responded differently to the stimulus of hypobaria than limb conduit vessels; there was no change in diameter seen on ascent or descent, but there was a progressive decrease in blood flow on ascent, with no change on subsequent descent.
Introduction
Exposure to the hypoxic environment of high altitude requires important physiological compensatory mechanisms in order to sustain oxygen delivery to tissues. Among these, circulatory changes related to vessel diameter and blood flow play important roles in physiologic adaptation to hypobaria.
The vascular response to hypobaria is organ- and vascular bed-specific. Several studies have examined specific circulatory beds at high altitude, especially in relation to altitude illnesses. 1 Studies looking at cerebral vascular changes at altitude describe an initial increase in blood flow of the cerebral vascular bed upon rapid ascent to high altitude, followed by return to normal values after days of acclimatization. 2 In addition, pulmonary circulation at altitude has also been the subject of extensive research. These studies describe pulmonary vasoconstriction in response to hypoxia causing the opening of shunts and overperfusion of certain capillary beds. This causes endothelial injury and fluid shifts into the alveoli, which play a key role in the development of high altitude pulmonary edema (HAPE). 2 More recently, the Caudwell Xtreme Everest Research Group examined the microcirculatory blood flow at high altitude and found a decreased microcirculatory flow index between sea level and high altitude in small and medium blood vessels.3,4
Despite the abundant literature on cerebral and pulmonary circulation, and the novel research on microcirculation at altitude, no studies (to our knowledge) have looked at hemodynamic properties of conduit vessels in response to hypoxia at altitude. This is of value for further understanding the complex relation between the hemodynamics of various vascular beds in response to hypobaria, and may further shed light onto the physiological processes involved in high altitude illnesses.
The aim of the present study was to investigate the diameter and blood flow of conduit vessels in the hypobaric environment of Mount Everest. We hypothesized that vessel diameter and blood flow in the brachial, carotid, and femoral arteries would decrease significantly in response to exposure to the high altitude of Everest Base Camp. We further sought to determine if the observed effect of hypoxia on the variables of interest would persist on the descent to lower altitude, secondary to acclimatization.
Materials and Methods
We studied 11 healthy individuals, who were part of a team of climbers ascending to Mount Everest Base Camp, at altitude 5330 m, between March 24, 2009 and April 17, 2009. The initial study cohort included 13 subjects, but 2 did not reach base camp because of illness. One subject had symptoms of acute mountain sickness, and the other one had photokeratitis (“snow blindness”). Both subjects were excluded from the analysis. These 11 subjects (5 men and 6 women) had a mean age of 46.2 (range 23–62) years. One participant was excluded from the comparison of data between ascent and descent since he attempted the summit of Everest after reaching base camp, instead of descending with the rest of the group. The measurements for the common femoral artery (CFA), the superficial femoral artery (SFA), and deep femoral artery (DFA) at Namche Bazaar for one participant were not obtained because of technical difficulties and were, therefore, excluded from the trend analysis. All study subjects were nonsmokers and none of them were taking vasoactive medications during the course of the study.
The trekkers were in Kathmandu for 2 days before beginning the trek to Namche Bazaar that took approximately 2 days. The group was acclimatized to Namche Bazaar for 2 days prior to beginning the slow ascent up to base camp that took over 10 days. A day and a half later, they began the descent back to Kathmandu that lasted 5 days.
Baseline measurements were taken in a vascular laboratory accredited by the Intersocietal Commission for the Accreditation of Vascular Laboratories (ICAVL, Centre de Santé et de Services Sociaux [CSSS] de Gatineau, Hull, Québec, Canada) 5 days prior to departure. Further measurements were taken at 4 different altitudes: sea level (Gatineau, Canada), 1310 m (Kathmandu), 3470 m (Namche Bazaar), and 5330 m (Everest Base Camp). Measurements were repeated on the descent at altitude 1310 m (Kathmandu) for evaluation of the affect of acclimatization on our variables. Altitude measurements were taken by a certified vascular surgeon (CSSS de Gatineau, Hull, Québec, Canada). The examiner was blinded to all baseline data.
Participants rested for approximately 24 hours between arrival at a specific altitude and the time of data collection. Measurements for all participants were taken in a heated shelter, on the same day, by the same evaluator. Clothing was removed on one limb at a time immediately prior to measurements and subsequently replaced.
Using Doppler ultrasound, 2 variables were measured: vessel diameter (cm) and blood velocity (cm/s). The ultrasound system (GE LOGIQ e, GE Medicals) was equipped with 2 linear array transducers operating at an imaging frequency of 7 to 8, and 10 MHz. The vessel diameter was determined at a perpendicular angle along the central axis of the scanned area, where the best spatial resolution was achieved. The blood velocity profile was obtained using the same transducers with Doppler ultrasound frequency of 4.0 to 5.0 MHz, operated in the high-pulsed repetition frequency mode, with a sample volume of 1.5 to 3.5 cm in depth. The proper function of this portable ultrasound system in the cold, hypobaric conditions has been confirmed by Otto et al. 5
Using measured vessel diameter and blood velocity, blood flow (mL/min) was calculated using the following formula: Blood flow (mL/min) = Blood velocity x π x (vessel diameter/2)2 x 60 s/min.
The study protocol was approved by the CSSS de l'Outaouais ethics committee. The informed consent for the study participation was obtained prior to departure.
Statistical Analysis
The mean values (and SDs) of the diameter (in cm), blood velocity (cm/s) and blood flow (mL/min) for each type of artery (ie, brachial, carotid, common femoral, superficial femoral, deep femoral) were calculated for 11 study participants by location/altitude. In order to assess the impact of altitude as a surrogate measure for hypoxia on the parameters of interest, the analysis of variance for repeated measures test was performed and the mean values of each parameter across the 5 different altitudes (ie, 4 different altitudes during ascent and 1 repeated altitude after descent to 1310 m) were compared. The choice of the analysis of variance repeated measures test was based on the consideration that more than 2 repeated measurements were obtained on a single sample (nonindependence assumption; within-subject variability). We calculated 2-tailed P values and used the α = 0.05 level of statistical significance.
For each parameter, the mean values for the 5 locations/altitudes are presented in tabular (Tables 1 through 3) and graphic forms (Figures 1 through 6).
Mean (SD) artery diameter (cm) by altitude a
SFA, superficial femoral artery; CFA, common femoral artery; DFA, deep femoral artery; m, meters.
n=11, except for 1310 m descent for all arteries, and at 3470 m for CFA, SFA, and DFA, where n=10.
Statistically significant vs sea level (P < .05).
Mean (SD) arterial blood velocity (cm/s) by altitude a
n=11, except for 1310 m descent for all arteries, and at 3470 m for CFA, SFA, and DFA, where n=10.
Statistically significant vs sea level (P < .05).
Mean (SD) blood flow of the artery (mL/min) by altitude a
SFA, superficial femoral artery; CFA, common femoral artery; DFA, deep femoral artery; m-meters.
n=11, except for 1310 m descent, and 3470 m for CFA and DFA, where n=10.
Statistically significant vs sea level (P < .05).
Statistically significant vs previous altitude value (P < .05).
Statistically significant between 1310 m on ascent vs 1310 m on descent (P < .05).
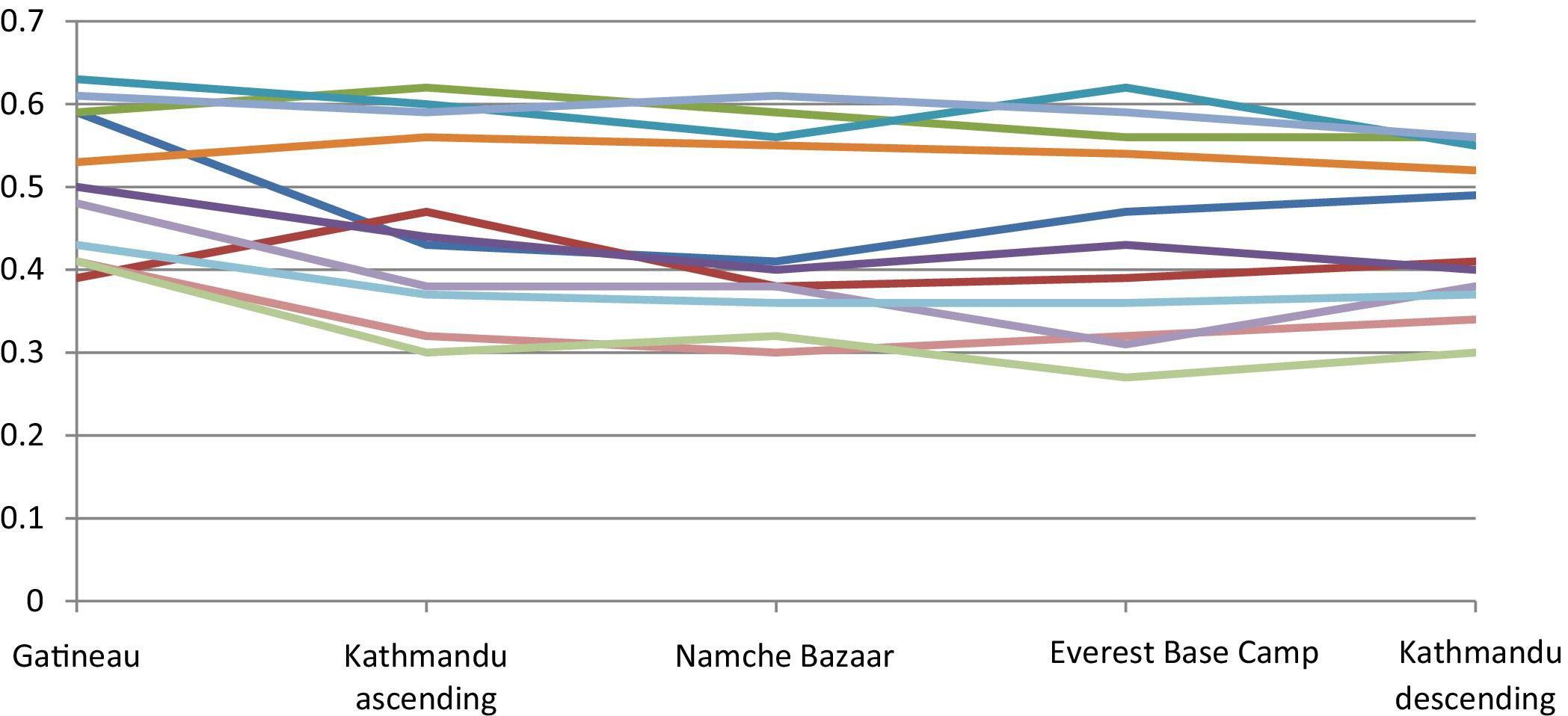
Brachial artery diameter (cm) by location/altitude.
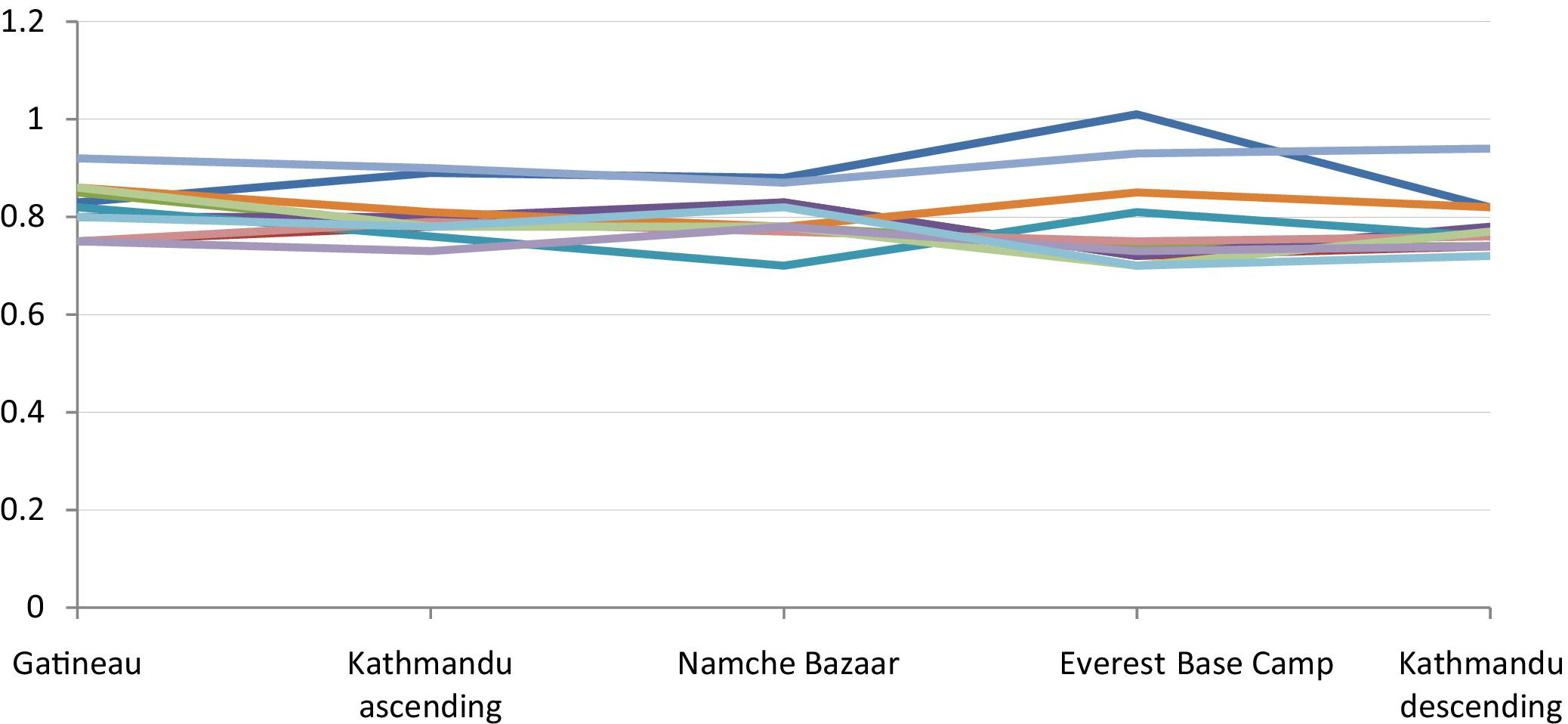
Carotid artery diameter (cm) by location/altitude.
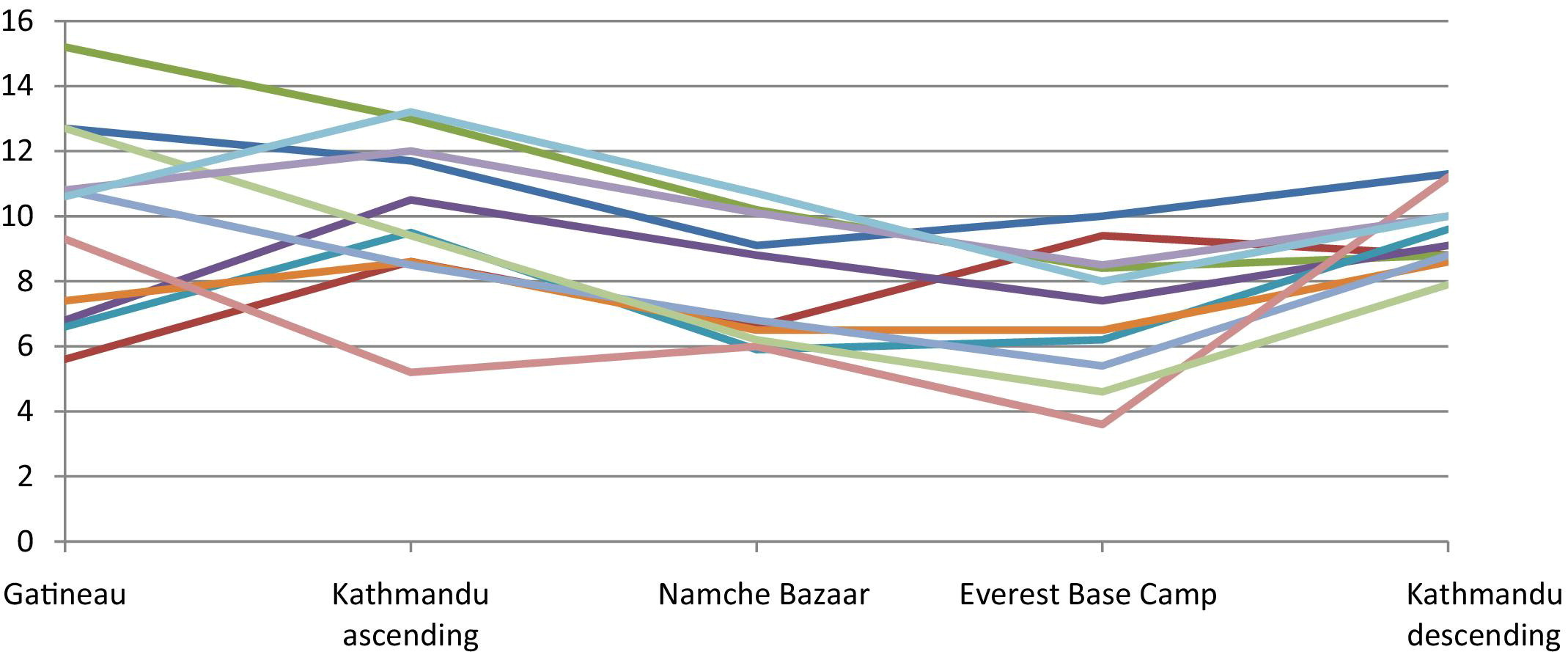
Brachial artery blood velocity (cm/s) by location/altitude.
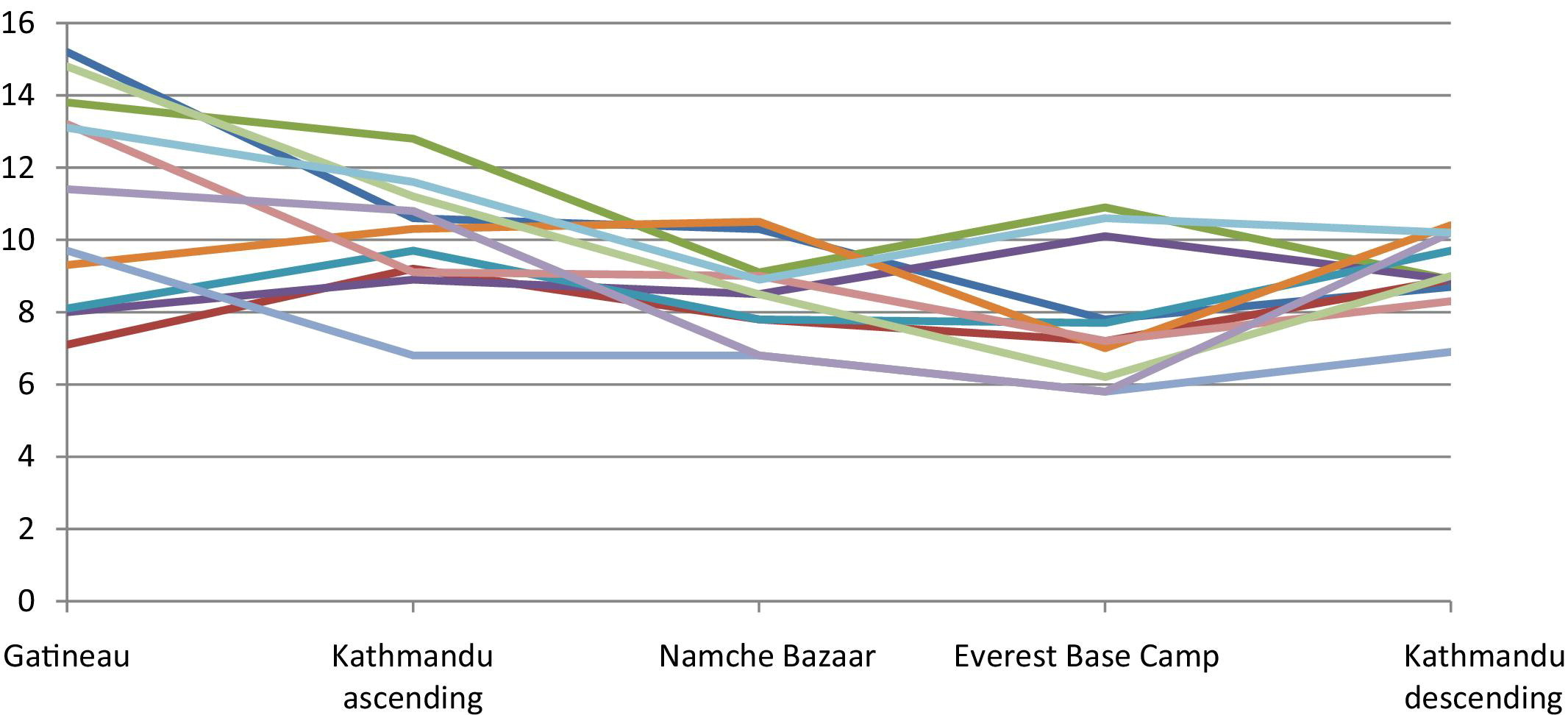
Carotid artery blood velocity (cm/s) by location/altitude.
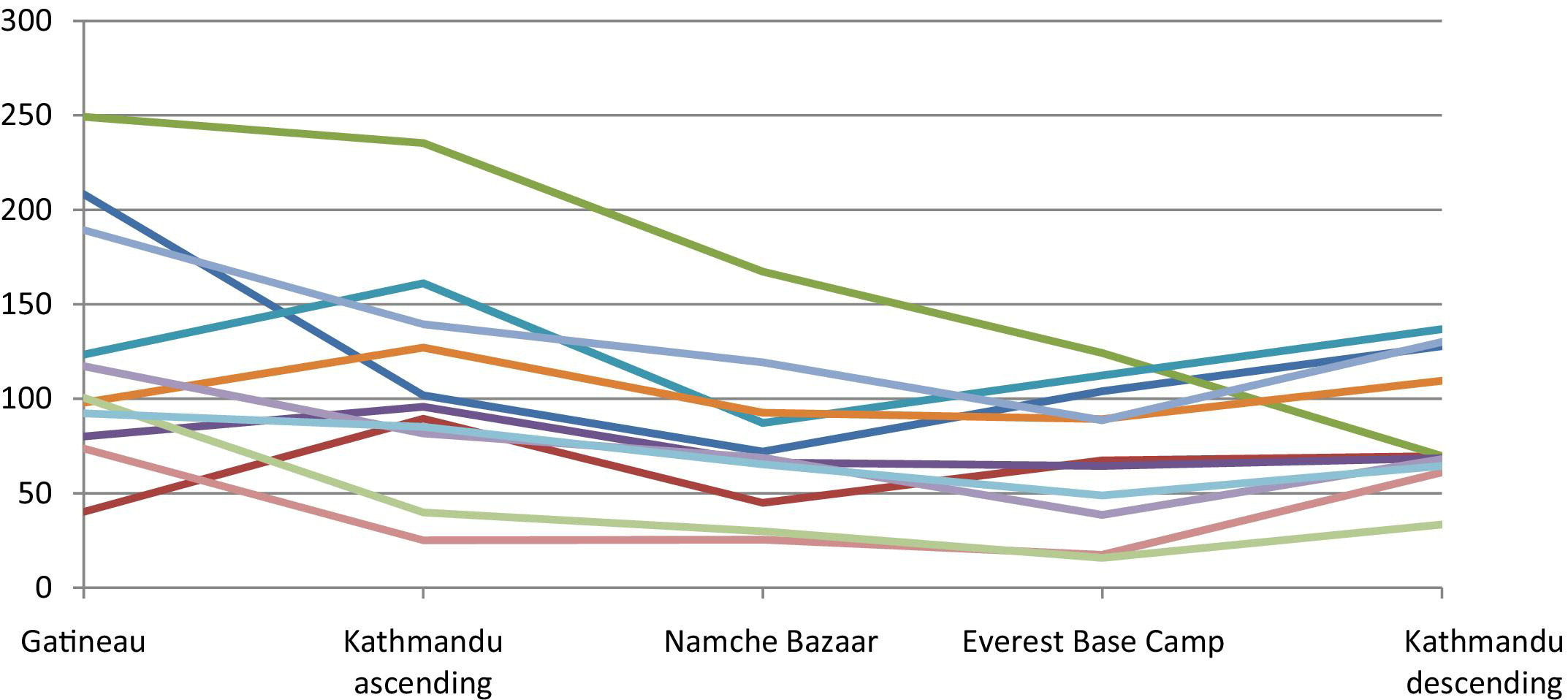
Brachial artery blood flow (ml/min) by location/altitude.
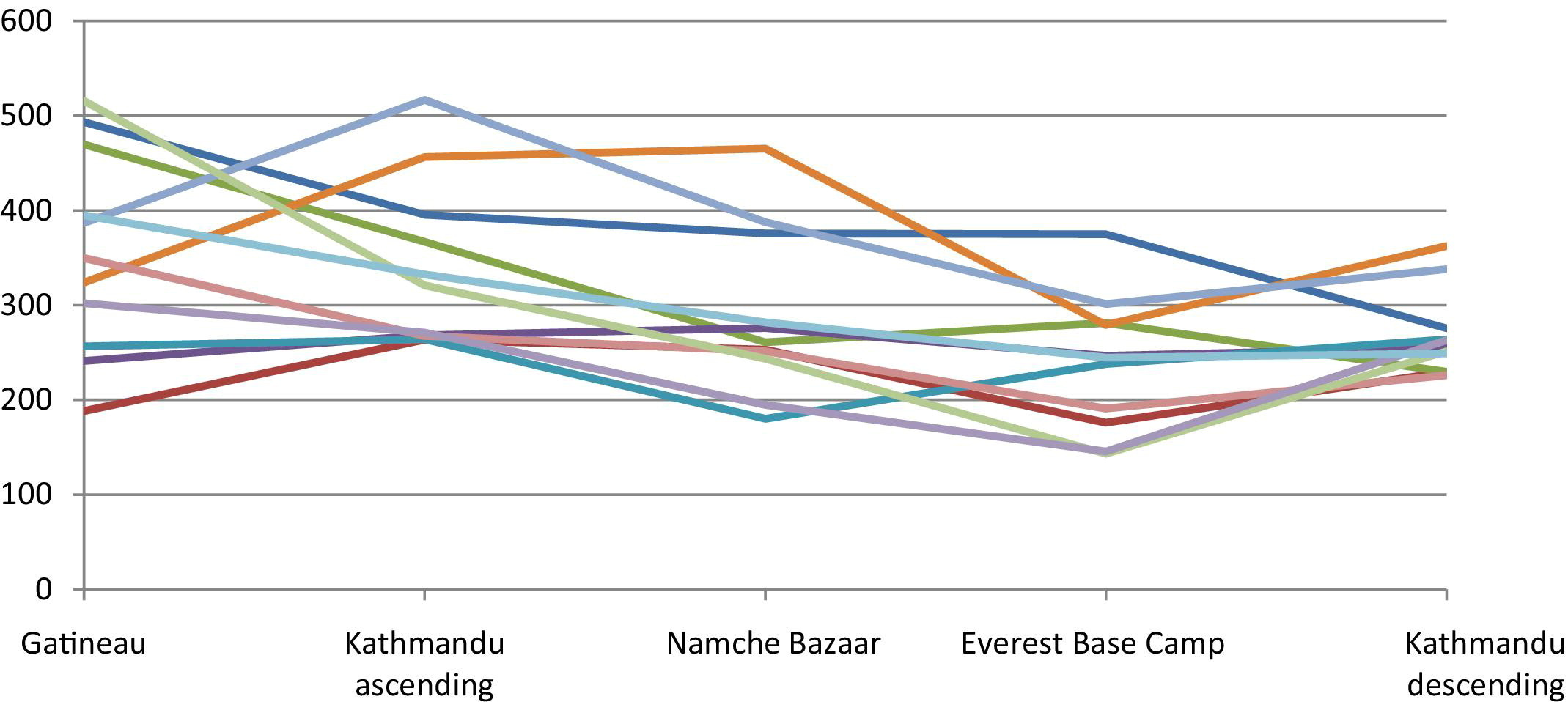
Carotid artery blood flow (ml/min) by location/altitude.
All analyses were performed using SAS (version 9.1; SAS Institute Inc, Cary, NC).
Results
Arterial Diameter—Ascent
As shown in Table 1, the arterial diameter decreased significantly between Gatineau (sea level) and Kathmandu (altitude 1310 m) for the brachial, CFA, and SFA. The decrease in diameter of the DFA was not significantly different between sea level and 1310 m, but was significantly decreased between sea level and the next altitude of 3470 m (Namche Bazaar). After this initial decrease in diameter for the brachial, CFA, and SFA, there was a persistence of this decreased diameter but no further significant decrease with increasing altitude. In contrast to the other vessels studied, the diameter of the carotid artery did not change with changes in altitude.
Arterial Diameter—Descent
On descent back to lower altitude, there was persistence of the initial decrease in diameter but no further decrease between base camp (5330 m) and Kathmandu (1310 m descent) for brachial, CFA, SFA, and DFA. The diameter of the carotid arteries also did not change significantly between these 2 locations on descent. There was no difference in diameter between Kathmandu (1310 m) on ascent and descent for all arteries. Figures 1 and 2 show the pattern of changes in arterial diameter for each individual subject.
Velocity—Ascent
As shown in Table 2, the only significant differences observed for velocity on ascent were between sea level and the altitudes 3470 m and 5330 m for both the SFA and carotid arteries. The velocity for the other arteries remained stable with increasing altitude.
Velocity—Descent
On descent from Everest Base Camp (5330 m) to Kathmandu (1310 m), there was no difference in blood velocity for any of the arteries. Also, there was no observed difference between Kathmandu (1310 m) on ascent and descent. Figures 3 and 4 show the pattern of changes in blood flow velocity for each individual subject.
Blood Flow—Ascent
As shown in Table 3, there was no significant decrease in blood flow for any of the arteries between Gatineau (sea level) and Kathmandu (altitude 1310 m) on ascent. However, the blood flow for all arteries decreased between 1310 m and the following altitude of 3470 m. Additionally, at this altitude of 3470 m, the blood flow for brachial, CFA, and SFA decreased significantly from sea level. At the next altitude of 5330 m, the decrease in blood flow for brachial, CFA, and SFA persisted but was not significantly different from the previous altitude values. The trend in carotids for blood flow was similar to the other conduit vessels.
Blood Flow—Descent
The blood flow of the arteries (except for carotid) increased significantly after descending from the Everest Base Camp (5330m) to Kathmandu (1310m). These values for blood flow at 1310 m on descent are not significantly different from the blood flow at equal altitude on ascent. The change in blood flow for carotid arteries was not significantly different between 5330 m and 1310 m. However, this value at 1310 m on descent was significantly decreased from the same altitude on ascent. Figure 5 and 6 demonstrate the pattern of blood flow for each individual subject.
Discussion
Our data has shown that the diameter of limb conduit vessels decreases upon initial ascent to higher altitude, with a persistence of this decreased diameter during further ascent to increasing altitudes up to the Everest Base Camp. We have also observed a persistence of this decreased vessel diameter when descending back to lower altitude. This simply shows expected vasoconstriction in response to stimulation of the sympathetic nervous system as well as the direct effect of hypoxia on smooth muscle. The stimulation of the sympathetic nervous system and release of catecholamines is an acute response to hypobaric hypoxia and is not sustained for more than a few days at altitudes. 1 This explains our observed plateau in vasoconstriction after initial exposure to high altitude.
Velocity in the limb vessels, except for SFA, has remained relatively consistent with ascent to Everest Base Camp and with descent to lower altitude. This suggests that limb conduit vessel blood velocity is largely unaffected up to the altitude of Everest Base Camp.
Blood flow decreased in all the limb vessels on ascent to high altitude, mainly because there was a decrease in the vessels' diameter (except in the SFA, where both velocity and diameter decreased). On descent back to lower altitude, blood flow increased in the limb vessels mainly because of an increase in velocity. Although mean velocity changes were not significantly different due to individual variability, this general pattern of change is evident in the figures of the brachial artery and was similar in other limb vessels.
The increase in velocity and blood flow when returning to lower altitude may be related to relative hemodilution secondary to the resolution of dehydration and fluid shifts responsible for early volume contraction seen on initial exposure to high altitude. Because neither hematocrit nor cardiac output was measured during this study, direct causality cannot be established. Also, these interpretations are limited by the fact that the observed differences in velocity were not statistically significant. It is possible that a larger sample size could clarify these differences.
The responses in vessel diameter of carotid arteries and limb conduit vessels to the stimulus of hypobaria differed. Specifically, in the carotid vessels, blood flow decreased at high altitude due to decreased blood velocity, whereas in the limb vessels diameter was the primary determinant. However, the progressive decrease in blood flow on ascent and subsequent increase on descent was similar to the other conduit vessel trends. This suggests that there are different physiological mechanisms at play in the regulation of carotid vessels at altitude. Because carotid blood flow is intimately related to cerebral blood flow, it may be that our observations would correlate with cerebral blood flow changes had they been measured. As previously mentioned, studies of cerebral blood flow at altitude describe an initial increase with rapid ascent to high altitude, followed by a return to normal values after days of acclimatization. 2 Because all of our data collection occurred after at least 24 hours of acclimatization to each altitude, we would likely not have captured an initial increase in blood flow of the carotids if it had occurred as described for cerebral blood flow. Additionally, there is no consensus in the literature as to the observed changes in both blood flow and velocity of the cerebral vasculature at altitude. In fact, studies using transcranial Doppler measures observed important individual variation in the response of the cerebral blood flow to hypoxia. 2 The process of cerebral autoregulation, in which cerebral perfusion is maintained despite systemic blood pressure variations, 2 may also play a role in the observed differences between carotid and limb vessels. Future studies measuring cerebral, carotid, and limb vessel blood flow concomitantly may further reconcile our observations with those previously described for the cerebral vasculature.
Study Limitations
The main limitation to the study is its small sample size. There are obvious difficulties in recruiting participants to ascend to the 5330 m base camp of Mount Everest. Nevertheless, the small number of participants and the inter-subject variability limit the generalizability of our results. Furthermore, the ultrasound device used for taking baseline measures at sea level was not the same as the one used at altitude (portable equipment). Also, measurements at baseline were taken by a certified vascular technician, and those at altitude were taken by a vascular surgeon who was also a certified vascular technician. However, all measurements were taken using identical technique, and this made it possible to blind the principal investigator to baseline data. Finally, the baseline fitness and physical condition of our participants was not measured prior to departure. It has been shown that regular physical activity affects vessel structure as well as vasoreactive properties leading to greater capacity for vasodilation in response to exercise in trained muscles. 6 This could have blunted the vasoconstrictive response to altitude in the physically fit individuals, as their conduit vessels would have responded to the concomitant stimulus of exercise with a more important vasodilation. However, this was minimized by taking measurements 24 hours after resting at a specific altitude.
Future Research
The mechanisms through which acclimatization to the hypobaric environment of high altitude occurs are poorly understood. The complex relationship among the multiple factors at stake warrants further research on this subject. This study represents one more step towards better understanding of vascular physiologic responses at high altitudes. Further research with larger sample size combining measurements of vessel diameter, blood velocity, and blood flow with hematocrit, cardiac output, and cerebral blood flow would bring additional insight into conduit vessel reactivity to hypobaria.
Footnotes
Acknowledgments
First and foremost, the authors would like to extend their sincere gratitude to the 13 individuals who participated in this study. The authors would also like to thank Karina Audet, RN, for her assistance with data collection during the expedition. Finally, the authors are grateful to GE Medicals for supplying the GE LOGIQ e ultrasound Doppler system.
Presented at the Entretiens Vasculaires XXVIII, Montebello, May 2010.
