Abstract
Objectives
The study was designed to compare effects of food deprivation (FD) and sleep deprivation (SD) on cognition during survival training.
Methods
In a cross-over design (n=12), the effects of FD (up to 66 hours followed by 500 kcal intake over 24 hours) and SD (up to 50 hours) on cognitive variables, blood glucose, and 3-OH-butyrate were studied.
Results
Food deprivation and SD impaired attention-dependent tasks. The FD impairment of simple reaction time was independent of blood glucose levels, which were normalized by a 500 kcal intake over 24 hours while the reaction time was not. Sleep deprivation and FD impaired maze-solving performance on all variables except rule breaks, which were significantly occurring after 50 hours of SD. Delayed word recall was impaired by SD for 50 hours. On the Balloon Analogue Risk Task, SD was associated with reduced risk-taking. In a gambling task, both SD for 50 hours and FD for 66 hours were associated with a tendency to make early choices when presented with consecutive choices, but the risk-taking was not affected.
Conclusions
Sleep deprivation has multiple cognitive effects, including attention, memory, visual-spatial ability, and risk-taking. Food deprivation had no affect on risk-taking, while the other tasks were affected in a way similar to SD but were less pronounced. The FD effects on cognition did not appear to depend on blood sugar levels. The need to sleep should be prioritized in survival situations to avoid cognitive impairment.
Introduction
Recommendations concerning emergency or survival situations focus on unexpected, prolonged subsistence without access to resources commonly available in human urban or rural communities. 1 -3 The advice is based on a mixture of personal experience, military survival training, and archaeological and anthropological studies of hunter-gatherer and nomadic societies.
The basic needs relevant to civilian life that are taught in survival training courses are usually: 1) protection from exposure; 2) water; and 3) food. Exposure to cold or heat may be fatal within a few hours, or even less, while dehydration due to lack of water takes several days to develop into a life-threatening level. Deaths from starvation are commonly reported after more than 6 weeks of total lack of food.4,5 In practice, it seems that deaths and serious injuries among subjects exposed to a survival situation often are the result of exposure or risky behavior.6,7 Therefore, factors contributing to poor judgment should be minimized.
The number of studies on the effects of sleep deprivation (SD) and food deprivation (FD) on decision-making is small. There are some studies that were carried out in a military training context that partly address the question. 8 -11 However, data relevant to the civilian survival situation is lacking. Furthermore, while the combined effects of SD and FD and other stressors in a simulated combat situation are severe on cognitive function,8,9 it is not clear what the relative contributions may be from each factor.
There is vast literature on the effects of SD on cognitive functions as well as the risks involved in SD (see review by Harrison and Horne 12 ). Less is known about cognitive effects of food deprivation. Attention deficits were found in children not eating breakfast, 13 but no effect was found in a controlled caloric restriction study. 14 However, the effects of hypoglycemia on cognition in diabetic and nondiabetic subjects have been extensively researched.
This study compares the effects of SD and FD on cognitive functioning with special reference to attention and risk-taking, both of which may be important in survival situations. Metabolic adaptation is monitored in parallel in order to facilitate interpretation of data. The aim of the present study was to directly compare the impact of SD with FD on selected cognitive functions. In order to investigate the effect of a specific recommendation, 2 the ability of a small amount of carbohydrates (500 kcal over 24 hours) to counteract the effects of food deprivation was also tested.
Materials and Methods
Subjects
Five females and 7 males, 21 to 52 years old, non-smokers, weighing 50 to 87 kg, without diabetes or sleep disorders or any other disease considered likely to interfere with the experiment, participated in the experiment that was included as a major part of a 9-day course arranged by the Swedish Survival Guild and the Swedish Civilian Defence League. The subjects' active engagement in survival training ranged from 1–18 years. All subjects were right-handed, except one who was ambidextrous but uses the right hand to control the computer mouse. The study was approved by the Ethics Review Board at Karolinska Institute and the subjects were given oral and written information prior to giving their written informed consent to participate.
Subjects were randomized into 2 different cohorts, with 6 subjects per group, stratified for gender (2 females in one group and 3 in the other): 1) sleep deprivation first and food deprivation second (“group SF”); or 2) food deprivation first and sleep deprivation second (“group FS”) (see Table). On every study day (except training day) the cognition test battery was administered between 8
Overview of activities related to the study protocol
Body weight was measured at the same time each day after emptying the bladder and while wearing standardized clothing. Capillary glucose and ketones were analyzed on-site. Remaining clinical chemistry analyses will be discussed elsewhere.
Computerized cognitive measurements
For this study the computerized cognition test battery was programmed in Delphi (Embarcadero, San Francisco, CA) on a laptop PC by the first author. Simple reaction time, choice reaction time, digit span, and word recall are all based on tests discussed by Lezak et al 15 and were modified to fit the setting used in this study. To avoid a learning effect as a confounding factor, the participants familiarized themselves with the tests for one day before the start of the study.
Simple reaction time
The subjects were instructed to press the left mouse button when a black square on the screen switched to red. There were 30 presentations. Each presentation appeared 4 to 10 seconds (randomly evenly distributed) after the previous response or a time-out error (2 seconds). The mean reaction time, standard deviation, and number of premature (reaction time < 150 ms), and time-out errors were calculated. Paired t-tests on log-transformed reaction times were used.
Choice reaction time
The subjects were shown 3 differently colored squares on the screen to indicate action (blue = click left mouse button, magenta = click right mouse button, green = do not click a mouse button for at least 1.5 seconds). A large, black square was shown on the screen. There were 30 presentations, randomly ordered, with 10 of each color. Each presentation appears 2 to 10 seconds (randomly evenly distributed) after the previous response or a time-out error (2 seconds). The mean reaction time, standard deviation, and number of premature reactions (reaction time < 150 ms), false reactions, failed inhibitions, and time-out errors were calculated. Paired t-tests on log-transformed reaction times were used.
Digit forward and backward recall
A successively increasing number of digits in sets, starting with 4 digit sets (forward recall) and 3 digit sets (backward recall), were presented on the screen at 1 second intervals. The subjects typed the digit set in forward or backward order. Two attempts for each number of digits were allowed, and the test stopped when both failed.
Word recall
A series of 15 words was presented with 1 second per word. Recall was collected by letting the subjects type as many words as they could recall. A new list was used for each day. The same proportion of 1-, 2-, 3-, and 4-syllable words and of nouns, verbs, and adjectives was used for each day. The list was presented 3 times, with immediate recall testing after each presentation and a delayed recall after about 45 minutes. The immediate recall test was the first in the test battery and the delayed recall testing was done at the end of the test battery.
Milner maze
The method was a computerized version of the test used by Milner. 16 A maze consisting of a 10 × 10 square grid was shown on the screen. The maze started at the lower left corner and ended in the diagonal corner. Subjects were required to find the hidden maze, locating the next correct square using the computer mouse. An incorrect response forced the subject to go back to the last correct square. Only north-south and east-west single steps were allowed. Three consecutive successful trials were required for task completion. The time to completion, the number of trials, the number of errors, and the number of illegal moves were measured. The maze path was standardized to 28 squares of length and 11 turns, and there was a different path for each day. A very similar test is commercially available under the name “Groton” maze by CogState (Melbourne, Australia). 17
Cambridge gambling test
This test is very similar to the commercially available test by CanTab (Cambridge, UK) 18 and measures risk-taking with known risk. The test person was initially given 100 points. On each game, 10 small squares were shown on the screen and, systematically (but in random order) 1 to 9 of the squares were blue, and the remaining were red. One square is randomly selected as the hidden winning square. The test person first had to guess if he/she thought the selected square was a blue or a red square. Next, the test person would bet on whether or not the guess was correct. Choices were 5, 25, 50, 75, or 95% of the points available. The choices were presented in strictly ascending or descending order, with 3 seconds to make a decision or the next bet size was presented. No choice resulted in the final alternative being used. In one test session, a total of 36 games were run (ie, one game per number of blue squares, run under ascending and descending bet choice conditions [which adds up to 18 games] and this was run in duplicate). The test is sensitive to frontal lobe damage and measures perception of risk and utilization of risk information. To increase motivation on this task, the 2 subgroups (see above) competed to get the highest sum of scores each day.
Balloon Analogue Risk Task
The implementation of the Balloon Analogue Risk Task (BART) was based on the test developed by Lejuez et al. 19 The BART has been shown to have acceptable test-retest stability 20 and correlates with self-reported risk-taking in adolescents 21 and differentiates between smokers (who were more risk-prone) and non-smokers. 22 In the test, a balloon depicted on the screen was inflated by clicking the mouse, maximally 100 clicks. For each of the 30 trials, the number of clicks that result in explosion of the balloon was randomly determined. If the test subjects decided to stop inflating before explosion, the points (1 per click) were added to the total score, or else the points were lost. The BART results are presented as the average percentage of theoretical maximum number of inflations (100) of non-exploded balloons.
Karolinska Sleepiness Scale
The Karolinska Sleepiness Scale (KSS) is a 9-point verbally anchored scale developed by Åkerstedt and Gillberg. 23 The following scale steps are verbally described: 1 = extremely alert (mycket pigg in Swedish); 3 = alert (pigg); 5 = neither alert nor sleepy (varken pigg eller sömnig); 7 = sleepy, but no difficulty remaining awake (sömnig, men ej ansträngande att vara vaken); and 9 = extremely sleepy, fighting sleep (mycket sömnig, kämpar mot sömnen); intermediate steps 2, 4, 6 and 8 are not verbally described.
Edinburgh Hypoglycemia Scale
The Edinburgh Hypoglycemia Scale (EHS) is constructed as a 7-point scale of 11 items as proposed by Deary et al. 24 The items are sweating, palpitation, shaking, hunger, confusion, drowsiness, odd behavior, speech difficulty, incoordination, nausea, and headache (svettig, hjärtklappning, skakig, hungrig, förvirrad, trött, konstigt beteende, svårt att prata, okoordinerad, illamående, huvudvärk), and the scale goes from 0 to 7. Subscales for central nervous system (CNS) symptoms (confusion, drowsiness, odd behavior, speech difficulty, incoordination), autonomic nervous system (ANS) symptoms (sweating, palpitation, shaking, hunger), and general symptoms (nausea, headache) were analyzed.
Statistics
Data are presented as arithmetic or geometric means with corresponding SEM (standard error of the mean), depending on the distribution of the data (symmetric and skewed upward, respectively). Hypothesis-testing was carried out by repeated measures ANOVA, followed by paired Student t-tests without correction for multiplicity. The last day of the study was used as the control because most variables displayed a training effect despite one day of training. The only exception is simple reaction time, where no such effect was observed and the mean of the first and the last day was used as a control.
Results
Blood glucose and 3-OH-butyrate
Blood glucose was significantly influenced by the experimental conditions (F [6,66] = 26.8, P < .001). There was a significant and progressive decline over time during FD. Intake of 500 kcal over 24 hours restored blood glucose to the control level. There was no effect from 26 hours of SD on blood glucose; however, 50 hours of SD resulted in a statistically significant (P < .01) but numerically small decline of about 1 mM (18 mg/dL) (Figure 1A). It should be noted that blood glucose samples were collected after breakfast (when relevant) and shortly before cognitive testing.
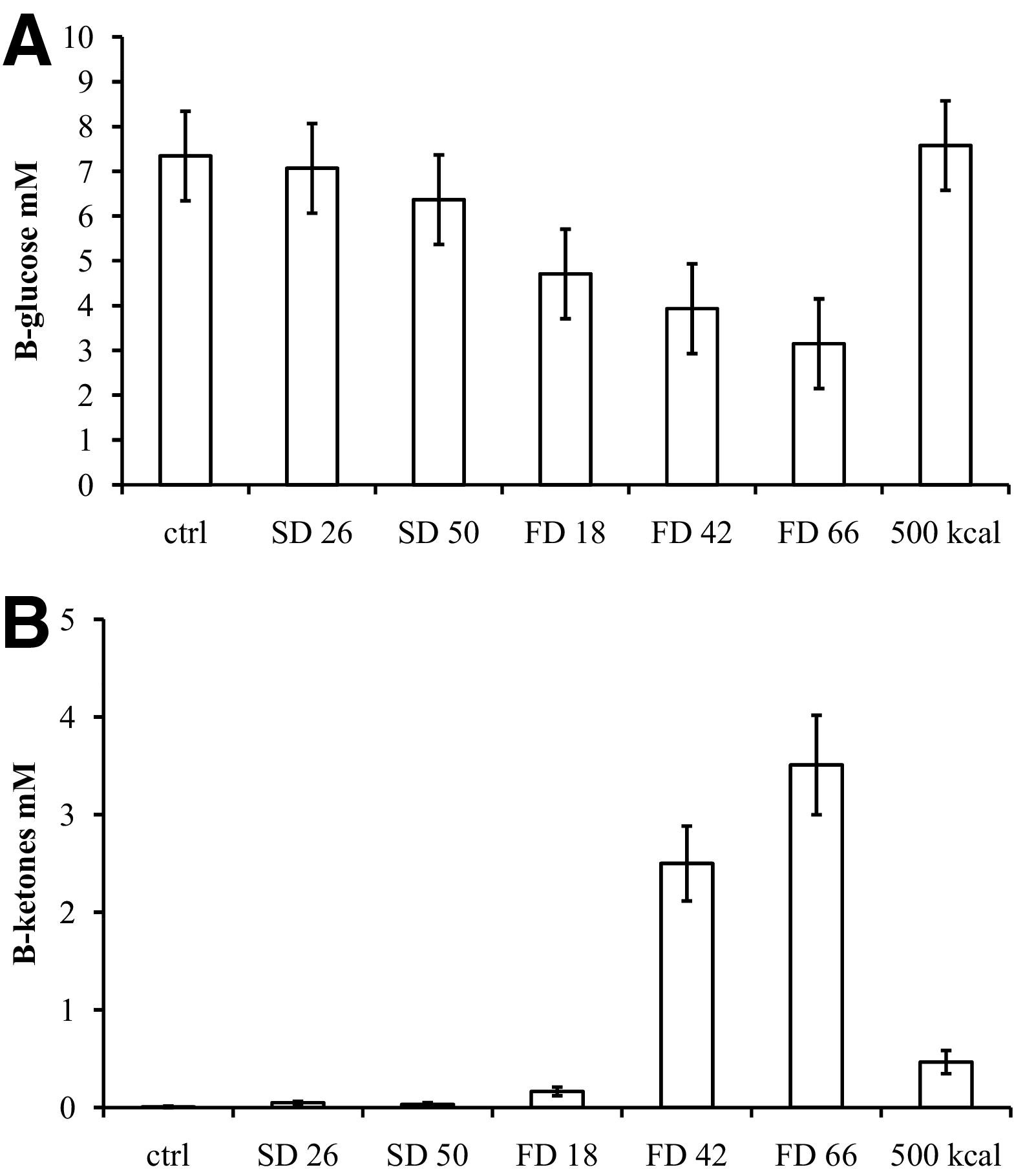
(A) B-glucose (1 mM = 18 mg/dL). Differences to control conditions are significant for FD for 18 to 66 hours (P < .001) and for 50 hours of SD (P < .01). The B-glucose level did not differ after 500 kcal compared to controls but was significantly increased compared to 66 hours of FD (P < .001). (B) 3-OH-butyrate. Differences to control conditions are significant for FD for 18 to 66 hours and after intake of 500 kcal (P < .001) but there was no effect of SD. The 3-OH-butyrate level was significantly lowered compared to 66 hours of FD (P < .001).
The experimental conditions significantly influenced 3-OH-butyrate (F [6,66] = 15.3, P < .001). Only FD increased 3-OH-butyrate, which increased progressively to the highest levels after 66 hours of FD. Intake of 500 kcal over 24 hours reduced 3-OH-butyrate substantially, but the levels were still increased as compared to control conditions (Figure 1B).
Sleep and hypoglycemia scales
The KSS was significantly influenced by the experimental conditions (F [6,66] = 28.6, P < .001). Sleep deprivation was reflected in the KSS (Figure 2A) with an apparent “dose” dependency of SD. Food deprivation also increased KSS significantly (P < .05). Intake of 500 kcal reversed the increase in KSS, compared to 66 hours of FD (P < .05).
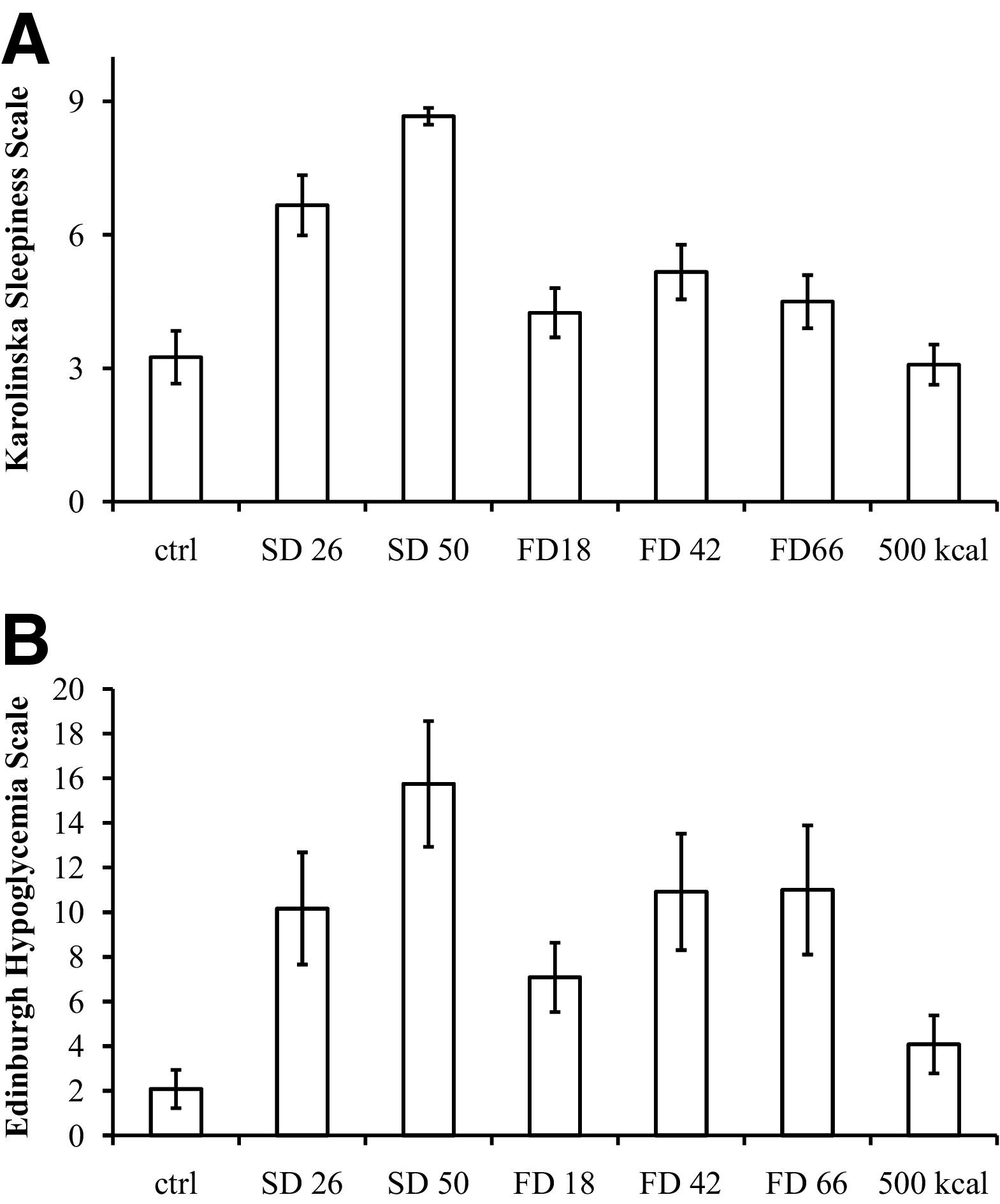
(A) Effects on the KSS. The increase in sleepiness score is significantly different from controls under all conditions except during 500 kcal refeeding. (B) Effects on the EHS. The increase in hypoglycemia score is significantly different from controls under all conditions except during 500 kcal refeeding.
The EHS was significantly influenced by the experimental conditions (F [6,66] = 11.1, P < .001). Both SD and FD increased EHS significantly compared to controls (Figure 2B). Effects on CNS symptoms contributed relatively more to the scores during SD than during FD, where ANS symptoms dominated (data not shown).
Attentional tasks
Simple reaction time and choice reaction time were significantly influenced by the experimental conditions (F [6,66] = 6.35, P < .001 and F [6,66] = 10.9, P < .001, respectively). Both simple reaction time and choice reaction time were impaired by SD, the effect being more pronounced after 50 hours than 26 hours (Figures 3A and B). In the choice reaction test, the total number of erroneous reactions was increased only in the 50-hour sleep-deprived group.
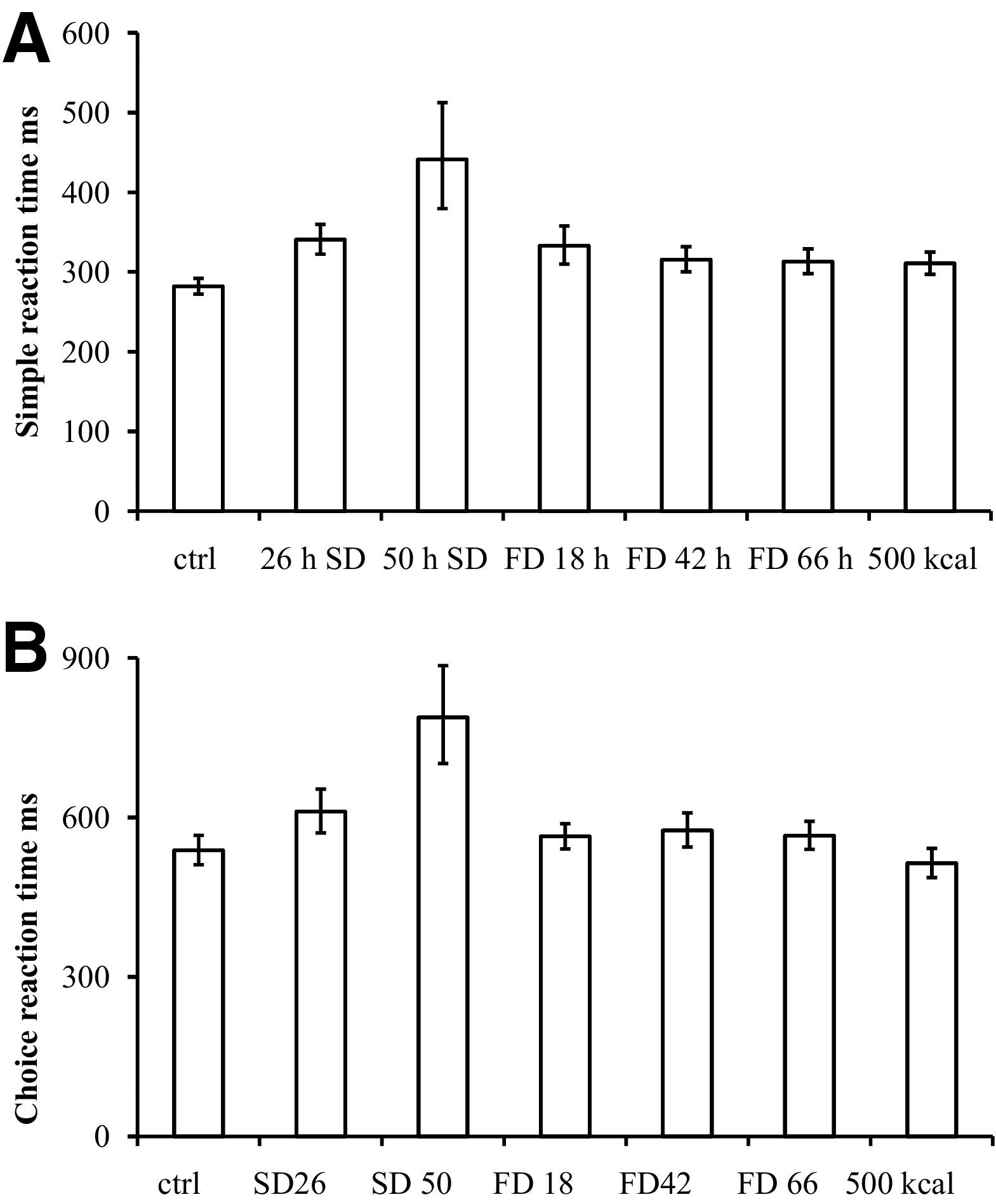
Effects of sleep deprivation (SD) and food deprivation (FD) on attentional tasks. (A) compared to controls, simple reaction time was significantly prolonged by SD for 26 and 50 hours (P < .01) and by FD for 18 to 66 hours (P < .05). Five hundred kcal did not alter simple reaction time compared to 66 hours FD (P > .5). (B) Sleep deprivation for 26 and 50 hours significantly prolonged choice reaction time compared to controls (P < .01) but not FD (P > .05).
Food deprivation significantly prolonged simple reaction time from 18 hours and onward. None of the choice reaction variables were significantly affected. Intake of 500 kcal over a 24-hour period did not restore simple reaction time.
Memory tasks
There was a small but significant influence on digit recall (forward but not backward) by the experimental conditions (F [6,66] = 3.05, P < .05 and F [6,66] = 1.41, P > .05 respectively). The effects of SD and FD were small on forward and backward digit recall. The only group that was impaired in backward digit recall was the SD for 50 hours group; that group's backward digit recall was reduced by an average 1.0 digit from the 7.25 in controls (P < .05).
Delayed word recall was significantly influenced by the experimental conditions (F [6,64] = 8.31, P < .001). Only SD for 50 hours had a significant effect on delayed word recall compared to controls. However, the effect observed was large, with a reduction from an average of 10.3 to 5.7 words (P < .005).
Visual-spatial learning
The performance on the Milner's maze test was significantly influenced by the experimental conditions measured as number of errors (F [6,64] = 8.31, P < .001), number of rule breaks (F [6,64] = 8.31, P < .001), and time to complete the task (F [6,64] = 8.31, P < .001). In fact, 5 of the 12 subjects on SD for 50 hours failed to complete the task, making technically fatal errors that terminated the test. For this occasion, data available up to the time of failure are included and, therefore, represent a systematic underestimate of the impairment (but does not affect the qualitative conclusions).
The performance was strongly affected by SD for 50 hours but much less after 26 hours. All variables (number of trials, number of errors (Figure 4A), number of rule breaks (Figure 4B), and total duration) were impaired after 50 hours of SD. Food deprivation also impaired the maze variables, except the number of rule breaks (compared to controls) and the 500 kcal intake did not restore the performance on the maze.
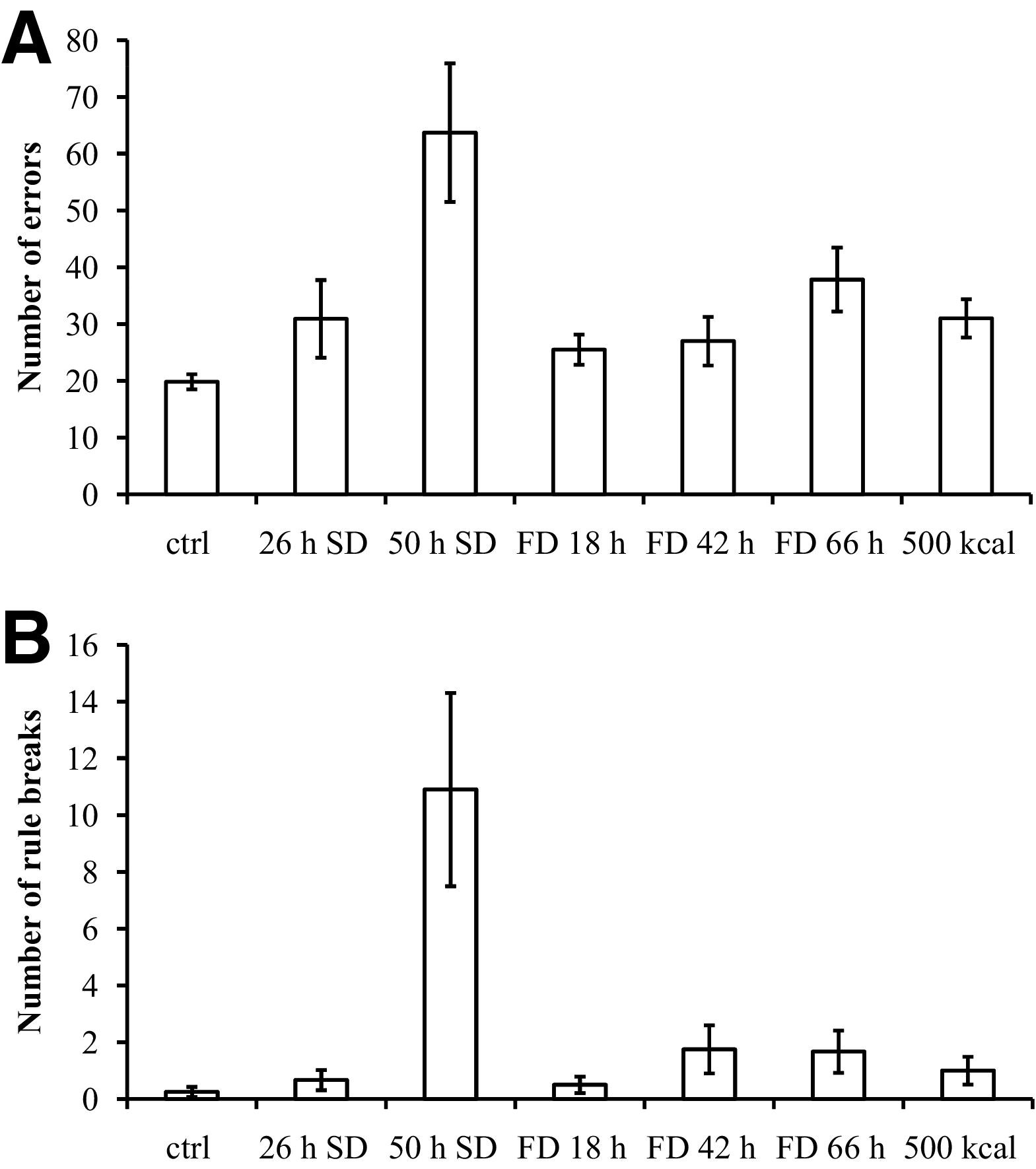
(A) Number of errors in the Milner's maze test. The increase in the number of errors is significantly different from controls under all conditions. (B) Number of rule breaks in the Milner's maze test. Only the increase observed in the 50 hours SD group in the number of rule breaks is significantly different from controls.
Risk-taking tasks
The BART was significantly influenced by the experimental conditions (F [6,58] = 4.38, P < .001). One subject misunderstood the instructions and has been deleted from the analysis. Only SD for 50 hours significantly changed the performance on the BART, with a reduction in risk-taking (Figure 5).
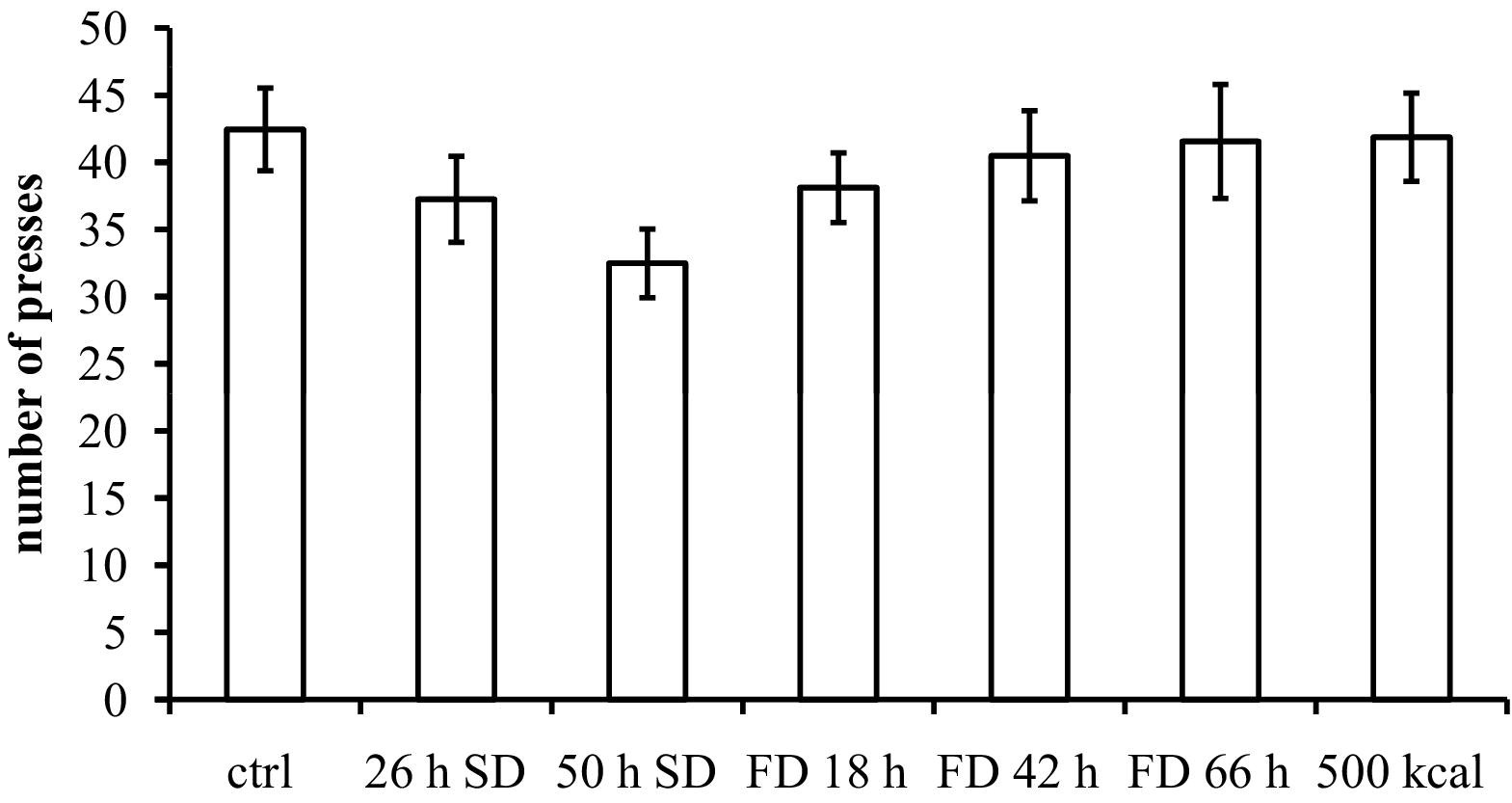
Effects on the Balloon Analogue Risk Task. The reduction in number of presses on non-exploded balloons is significant after 50 hours of SD (P < .005).
There were no significant differences among groups in the Cambridge Gambling Task in terms of the amount wagered. One observation was that the difference under ascending and descending conditions, with respect to bet size, was higher under both SD for 50 hours and FD for 66 hours (21% and 20% compared to 10% in controls) but the ANOVA did not indicate an overall difference among groups (F [6,66] = 1.49, NS) despite significant differences according to paired t-tests (P < .05 for both comparisons). On average, the difference between ascending and descending sequences was 16.2%. There was a trend to an increased betting size with time by about 1.7%/day in both groups.
Discussion
As expected, SD increased KSS in a “dose” dependent manner. Food deprivation also increased KSS, although not to the same extent as SD. Intake of 500 kcal over 24 hours reversed the increase in KSS.
The EHS scores were increased during both SD and FD, with a strong contribution from CNS symptoms to the former. The EHS was developed to capture insulin-induced hypoglycemia, and the maximum value possible is 66 on the EHS. In this study, the largest mean was about 16. Clearly, the scale is not intended to discriminate between SD and FD.
The CNS symptom scores on the EHS after 66 hours of FD, with a mean blood glucose of 3.15 mM (56.7 mg/dL), were comparable to those reported by McCrimmon et al, 25 who induced a hypoglycemia of 2.6 mM (46.8 mg/dL) experimentally while there was a discrepancy in the intensity of autonomic symptoms, which were less pronounced in the present study.
The effects of SD and FD on attentional tasks are consistent with published data. The simple reaction time more sensitively detected effects of both SD and FD. In SD, it is well-known that monotonous tasks are more sensitive than more challenging ones.12,26 Consistent with this observation, simple reaction time was prolonged by both SD and FD, while only SD prolonged choice reaction time.
The slightly more demanding memory tasks were not strongly influenced by SD and FD. Only SD for 50 hours consistently impaired digit span backwards and delayed word recall. Since there was a significant effect of SD on the attentional tasks, it is possible that the observed effects on memory are secondary to the attentional deficit. From a more practical point of view, the effects observed on memory variables by severe SD suggest that retention of information is likely to be impaired, indicating that SD may have consequences for planning and execution of activities in a survival situation.
A technical point on the digit span task is that, when using this computerized test, more digits are recalled compared to what is expected from the standardized clinical testing procedure, 15 for backward recall in particular. It is unlikely to be a selection effect of the participants, since we have used the standardized test during other courses with expected performance (unpublished data). However, presenting the digits visually on a computer screen, instead of reading them aloud to the subjects, theoretically allows the subjects to combine visual sketch-pad and phonological loop capacities.
The performance on the maze-learning test was strongly impaired by SD for 50 hours, which affected the number of trials, the number of errors, the total time spent on the task, and the number of rule breaks. The latter was probably due to the inability of the subjects to remain focused on this lengthy task while sleep-deprived rather than an impairment of frontal lobe function as described by Milner. 16 However, sleep deprivation does impair executive functioning, 27 and performance in the maze-learning test depends on executive functioning to an extent and is reflected in total errors and rule breaks. 17 Visual-spatial working memory is also dependent on central executive function, as discussed by McAfoose and Baune. 28 In this connection, there are the interesting observations that sleep-deprived subjects display a reduction of MRI signal strength in the left prefrontal cortex (together with the thalamus and the parietal cortex) 29 and in cerebral metabolism in the frontal cortex and in other brain regions. 30 Further studies are needed to explore the various aspects of executive function 31 in more detail to determine what specific effects SD has on prefrontal cortical functioning, or what effects are simply secondary to impaired attention.
Interestingly, FD for 18 to 66 hours had about the same effect as 26 hours of SD on the maze-learning task, and supplying 500 kcal over 24 hours did not prevent this impairment. Thus, the sensitivity to this task is very similar to that observed on simple reaction time.
In the BART test, there was a significant reduction in risk-taking after SD for 50 hours. This result is consistent with the data published by Killgore, 32 who found that 23 hours of sleep deprivation reduced risk-taking in the same test. His study was larger (n=54), which may explain why no effect was observed in the present study after 26 hours of SD. In fact, numerically, there was a decline (see Figure 5) but this was not statistically significant.
The Cambridge Gambling Task did not show any unambiguously significant effect on any of the conditions. The only systematic observation is the increased difference between betting size in ascending and descending conditions after the most prolonged SD and FD conditions. Thus, there was a tendency to make quicker decisions under both conditions. This is a pattern consistent with the behavior found in the BART but only after SD, not after FD. In future studies it may be interesting to investigate if SD and/or FD influence the balance between patience and risk-taking.
The overall picture is that sleep deprivation consistently impairs performance in psychometric tests of attention, memory, and visual-spatial function. The memory effects may well be secondary to impaired attention, and this may also be true for visual-spatial function. There was a clear “dose”-response to sleep deprivation, and the effects observed were consistent over the 2 experimental sequences (SD first and SD last), supporting the idea that the effects are due to the condition rather than being a period effect. It is, however, not easy to explain the effect on risk-taking observed in the 50 hours of SD group as being secondary to attentional impairment. It may be argued that the subjects were less patient while sleep deprived, but the lack of effect in the Cambridge Gambling Task is inconsistent with such an explanation. We suggest that the reduced risk-taking observed is a physiologically independent effect of SD.
It is the authors' conclusion that sleep deprivation is to be avoided, and being well-rested is a prerequisite for optimal decision-making. As bad decisions are likely to explain at least some unfortunate outcomes of survival situations, it seems reasonable that spending time on arranging for good sleep should be a highly prioritized activity in a survival situation.
The effects of food deprivation were also consistent in several psychometric tests, notably simple reaction time and Milner's maze. The effects are less pronounced than those observed after 50 hours of SD, but of the same order of magnitude as 26 hours of SD. Of particular interest, as discussed above, is the observation that an intake of 500 kcal over 24 hours did little to improve performance in the psychometric tests while, in the symptom scales for sleepiness and hypoglycemia, intake of 500 kcal counteracted the effects of FD. Interestingly, the blood glucose levels here were dissociated from effects on cognitive functions. It may be argued that the residual amounts of ketones observed after the intake of 500 kcal explain cognitive impairment, but this is not consistent with the effects observed after 18 hours of FD despite low levels of ketones. Furthermore, a ketogenic diet has previously shown no effect on attentional tasks but did impair performance on the trail-making task. 33 Also, patients treated for epilepsy with a ketogenic diet are reported to improve in cognitive function, but these effects are primarily longterm and are also confounded by factors such as reduced antiepileptic pharmacotherapy and improvement of the condition. 34 As this is the first time dissociation between blood glucose and cognitive performance during caloric restriction is reported, it needs to be corroborated.
Given that our results hold, there appears to be little support (from the perspective of cognitive functioning) for spending resources and energy on collecting food in a survival situation. However, there may be other reasons to try to find and consume even small amounts of food: 1) sleeping is probably easier with even small amounts of food in the stomach; 2) physical endurance may be improved with food consumption; and 3) it is possible that simultaneous lack of food and sleep amplify the effects on cognition. Also, in hypothermic conditions, even minor caloric intake may be of critical importance for survival. Nevertheless, the present findings emphasize that sleep is to be prioritized in prolonged emergency situations.
Financial Disclosures
Lars Ståhle is an employee of AstraZeneca R&D and is a shareholder of AstraZeneca and Medivir. Peter Annas is an employee of AstraZeneca R&D and is a shareholder of AstraZeneca.
Ewa Ljungdahl Ståhle, Elisabeth Granström, Sven Isaksson, and Harry Sepp have no interests to declare.
The contributions to this study by Lars Ståhle and Peter Annas are unrelated to their employment at AstraZeneca.
Footnotes
Acknowledgments
The authors wish to thank first and foremost the study participants who contributed in many ways to the study. The authors also wish to thank the Swedish Homeguard School in Vällinge, Sweden, for the use of their training ground on which the study was conducted; the School's practical help was invaluable. Additionally, the authors extend much thanks to Research Nurse Marta Stenberg at Clinical Pharmacology, Karolinska Institute, Huddinge, Sweden, who helped with many practical aspects of the sample handling. Financial support has been provided by the Swedish Civilian Defence League, Karolinska Institute, and by Abbott Sweden (analysis kits for capillary glucose and ketones were generously provided).
