Abstract
Objective
The aim of this study was to investigate the associations between alleles of the hypoxia-inducible factor 1A (HIF1A) C1772T polymorphism and several physiological responses to hypoxia, including the hypoxic ventilatory response (HVR), and serum erythropoietin (EPO), arterial oxygen saturation (Sa
Methods
A total of 76 males participated in the study; 52 participants completed an 8-hour exposure to 12.7% oxygen, during which time Sa
Results
The magnitude of the hypoxic responses was highly variable between individuals. The increase in participants' EPO responses ranged from 89% to 388% of baseline values following hypoxia, while Sa
Conclusion
In this study, the HIF1A C1772T polymorphism does not appear to influence EPO, Sa
Keywords
Introduction
Exposure to hypoxic conditions, such as that found at high altitude, induces adaptations in various physiological systems that aid the maintenance of oxygen delivery to metabolically active tissue, and thereby mitigates the impact of the fall in atmospheric partial pressure of oxygen (P
The transcription factor hypoxia-inducible factor-1 (HIF-1) is the key regulator of the cellular response to hypoxia. 9 HIF-1 activates more than 100 genes, which encode proteins that facilitate cellular and systemic responses to hypoxia and promote the restoration of cellular oxygen homeostasis; these proteins are linked to the regulation of angiogenesis, 10 erythropoiesis, 11 and energy metabolism. 12 HIF-1 is a heterodimer that is comprised of α and β subunits. Under hypoxic conditions, HIF-1β protein levels remain unchanged, while HIF-1α levels increase exponentially as oxygen tension decreases within the cell. 13 HIF-1 DNA binding activity follows a similar trend to HIF-1α protein levels, indicating that the HIF-1 transcriptional response is predominantly controlled by HIF-1α protein concentration. 13 In order for HIF-1 to activate transcription of a target gene, it must bind to a transcription regulating domain located within the promoter regions of target genes called the hypoxic response element (HRE). 14
Studies conducted on HIF-1α “knock-out” mice have illustrated the influence of HIF-1α on acclimation responses to hypoxia. Homozygous knock-out (Hif1a−/−) mice have an embryonic lethal phenotype due to malformations of the heart and vasculature; however, heterozygous knock-out (Hif1a+/−) mice, which are partially deficient in HIF-1α, are indistinguishable from wild-type mice in normoxic conditions. 15 Compared to wild-type mice, the Hif1a+/− mice have impaired acclimation responses to hypoxia; these include impaired carotid body function and ventilatory responses, 16 reduced right ventricular hypertrophy, 15 an absent EPO response, 11 and delayed erythropoiesis. 15
The human gene that encodes HIF-1α, HIF1A, contains a cytosine (C) to thymine (T) polymorphism at position 1772 (C1772T), which causes a proline to serine amino acid change at residue 582 of the HIF-1α protein. 17 In the COS 7 cell line, the serine variant has more than 2-fold greater HRE binding activity than the proline variant. 17 In addition, the level of HIF-1α binding to the HRE during exposure to severe hypoxic conditions (1% oxygen) was approximately 20-fold higher than baseline values in the serine variant compared to the approximately 7-fold increase observed for the proline variant. 17 This polymorphism has also been shown to influence several human phenotypes, demonstrating the functional effect of the polymorphism in vivo. These phenotypes include, but are not limited to, greater susceptibility to various forms of cancer 18 –20 and reduced coronary collateral formation in patients with ischemic heart disease. 21 All of the phenotypes previously studied in relation to the HIF1A C1772T polymorphism take a long time to progress and the polymorphism interacts with the environmental conditions associated with the phenotype (eg, hypoxia) over a long period. At this time, it is not known whether the HIF1A polymorphism influences acute responses to hypoxia. However, in mice, HIF-1α deficiency has been shown to alter responses to hypoxia after only 1 hour. 11 Furthermore, the C1772T polymorphism influences HIF-1α activity in normoxic conditions, 17 which may subsequently alter physiological responses to acute hypoxia. Therefore, it is plausible that the HIF1A C1772T polymorphism will have an impact on acute physiological responses to hypoxia in humans.
The influence of the HIF1A C1772T polymorphism on human responses to atmospheric hypoxia has only been studied twice previously. Droma et al
22
observed no difference in the frequency of the HIF1A genotypes between Sherpas who were and were not susceptible to acute mountain sickness (AMS). A further study looked to evaluate the influence of the HIF1A C1772T polymorphism on acclimation responses to intermittent hypoxia; however, only the CC genotype was identified in the small study population (n = 26), which limited analysis.
23
At this time no study has been conducted to investigate whether this polymorphism accounts for some of the variability in ventilatory and hematological responses to atmospheric hypoxia. As such, here we have studied whether the HIF1A T allele, which is associated with higher HIF-1α activity and increased transcription of target genes,17,24 is linked to a higher HVR and a greater EPO response, a higher Sa
Methods
Participants
Following local human ethical committee approval and written informed consent, 76 healthy Caucasian males were recruited for the study. The study was separated into 2 trials. In the first trial, 52 participants (age 24 ± 6 years, height 1.77 ± 0.07 m, body mass 76.1 ± 9.6 kg) completed an 8-hour exposure to 12.7% oxygen. In the second trial, 62 individuals (age 24 ± 5 years, height 1.80 ± 0.07 m, body mass 78.8 ± 8.4 kg) completed an HVR trial. Of the study population, 38 participants (age 23 ± 4 years, height 1.79 ± 0.06 m, body mass 77.6 ± 8.7 kg) completed both protocols. The 38 participants first completed the 8-hour exposure trial then, after a “wash-out” period of 1 month, took part in the HVR trial. All individuals were native low-landers who had not been exposed to hypoxic conditions in the 3 months prior to participation. Participants completed a health screen questionnaire prior to the first trial, and individuals were excluded from the study if they reported current or previous cardiovascular, respiratory, or renal problems.
Trial 1: 8-Hour Exposure To 12.7% Oxygen
Participants were instructed to abstain from caffeine and exercise in the 12 hours leading up to the trial and during the trial. On arrival at the laboratory, located at approximately 50 m altitude, participants assumed the supine position and maintained this for 10 minutes before baseline measurements were taken. Baseline Sa
Hematological analysis was conducted on 50 of the 52 participants. From a 15-mL blood sample, 10 mL was aliquoted into a tube that promotes coagulation (Sarstedt, Numbrecht, Germany). After a 20- to 30-minute incubation at room temperature, the coagulated blood was centrifuged to obtain serum, which was stored frozen (−80°C) until used for analysis. From the remaining whole blood, hematocrit (Hct) values were determined in triplicate using microcentrifuge techniques (Hawksley, Sussex, England). The cyanmethemoglobin method of analysis was used to determine hemoglobin (Hb) concentration. Hct and Hb levels were subsequently used to estimate plasma volume changes using equations originally proposed by Dill and Costill. 27 Serum EPO concentration was determined using an EPO enzyme-linked immunosorbent assay (ELISA) (Biomerica, Hannover, Germany) and the values obtained were corrected for changes in plasma volume. The ELISA procedure was performed in duplicate according to the manufacturer's instructions. The intra-assay and interassay coefficients of variation for this method were 5.4% and 6.5%, respectively.
Trial 2: Hypoxic Ventilatory Response
On arrival at the laboratory, pulmonary function [forced vital capacity (FVC), forced expiratory volume in 1 second (FEV1), and FEV1/FVC (%)] was measured and assessed using a portable spirometer (Micro Plus, Micro Medical, Kent, England) in accordance with guidelines published by the British Association of Sport and Exercise Sciences. 28 An exclusion criterion was put in place in which participants would not be allowed to take part in this aspect of the study if either their FEV1 or FEV1/FVC value was below 70% of the predicted value. However, no participants were excluded from the trial according to these criteria.
During the isocapnic HVR trial, respiratory variables were measured breath by breath (ZAN 600USB, Nspire Health, Oberthulba, Germany) and values were displayed continuously on a personal computer allowing constant monitoring of ventilatory parameters, which included the partial pressure of end tidal carbon dioxide (PET
The isocapnic HVR protocol was based on the steady-state methods proposed by Powell,
29
which was designed to bring continuity in HVR methodology between laboratories. As such, this HVR protocol was chosen in the current study so that results would be comparable with other studies in the area. This steady-state method includes a period of hyperoxia before the hypoxic exposure. The use of a hyperoxic period prior to HVR measurement is implemented as it has been reported that at sea level and high altitude an increase in FI
Individuals were asked to abstain from caffeine, alcohol, and exercise for 12 hours prior to the trial, and to fast for 2 hours before the trial, which was performed in a darkened room between 0830 and 1100 hours. The trial was conducted after 5 minutes of rest and with the participants in a semi-recumbent, 30° head-up position. Participants kept their eyes closed and listened to quiet music (the same for each individual) for the duration of the trial. Participants were fitted with a facemask that was attached to the HVR apparatus. Sa
Ventilatory parameters, FI
HIF1A Genotype Identification
Genomic DNA was generally extracted from 300 μL of whole blood using a DNA purification kit (Promega, Madison, WI). However, a mouthwash protocol (Oragene DNA kit, DNA Genotek, Ottawa, Canada) was implemented for 7 participants to obtain buccal cells, from which DNA was subsequently isolated in accordance with manufacturers' guidelines.
A 346-bp fragment of the HIF1A gene containing the C1772T polymorphism was amplified using the polymerase chain reaction (PCR) technique. The 50-μL reactions contained 50 ng genomic DNA, 200 ng of each primer, 5′-AAGGTGTGGCCATTGTAAAAACTC-3′ (forward) and 5′-GCACTAGTAGTTTCTTTATGTATG-3′ (reverse) previously used by Ollerenshaw et al, 30 0.2 mmol/L of each deoxynucleotide triphosphate (Sigma-Aldrich, Gillingham, England), 2 mmol/L MgCl2 (Promega), 10 μL GoTaq Flexi Buffer (Promega), and 2.5 units GoTaq DNA polymerase (Promega). After an initial denaturation at 94°C for 6 minutes, 35 cycles of 94°C for 60 seconds (denaturation), 60°C for 90 seconds (annealing), and 72°C for 120 seconds (extension) were performed. This was followed by a final extension at 72°C for 5 minutes. PCR products were analyzed in 1.5% (wt/vol) agarose gels, which were stained with ethidium bromide. Each 346-bp DNA fragment was then extracted using a commercial purification kit (QIAquick, Qiagen, Crawley, England) and sequenced (MWG Biotech, London, England) using the forward primer.
Statistical Analysis
SPSS 15.0 (SPSS Inc, Chicago, IL) was used for statistical analysis and the level of significance was set at P <.05. Data are presented as the mean ± 1 SD. Chi-square analysis was performed to test whether the distribution of the HIF1A C1772T alleles was in Hardy-Weinberg equilibrium. Because the HIF1A TT genotype was only identified in 2 individuals, data for the CT and TT genotypes were combined to form a single group, and statistical analysis was made between the CC and CT/TT groups. This practice is in accord with previous studies21,31 in which the results for the HIF1A CC genotype were compared with those for the combined CT/TT genotype group. In trial 1, independent-samples t tests were used to test for differences between the HIF1A genotypes in respect to participants' physical characteristics, and environmental conditions. Sa
Results
The distribution of the HIF1A C1772T genotypes in the population that completed the 8-hour exposure to 12.7% oxygen, the HVR population, and the whole study population, are shown in Table 1. In each study population, the frequencies of the HIF1A C1772T genotypes were in Hardy-Weinberg equilibrium. Due to the small number of participants identified with the TT genotype (n = 2), the CT and TT genotypes were combined and analyzed together.
HIF1A C1772T genotype frequencies for the different study populations
HIF1A, hypoxia-inducible factor 1A gene; HVR, hypoxic ventilatory response.
Trial 1: 8-Hour Exposure To 12.7% Oxygen
Table 2 shows the age, height, and body mass of the individuals who participated in the 8-hour exposure, according to HIF1A genotype, all of which were not significantly different between genotypes. Atmospheric FI
Participants' characteristics and environmental conditions during the 8-hour hypoxic exposure according to HIF1A genotype
Data are mean ± 1 SD; P values are from independent-samples t tests. HIF1A, hypoxia-inducible factor 1A gene; FIo2, fraction of inspired oxygen; FIco2, fraction of inspired carbon dioxide.
Serum EPO was 49% ± 43% higher than sea level values after 4 hours of exposure to 12.7% oxygen (P <.001), and continued to increase and reached values 125% ± 66% greater than baseline after 8 hours (P <.001). The magnitude of the EPO increase was highly variable between individuals; values ranged from 89% to 388% of baseline values after 8 hours. The magnitude of this increase correlated with the individuals' mean Sa
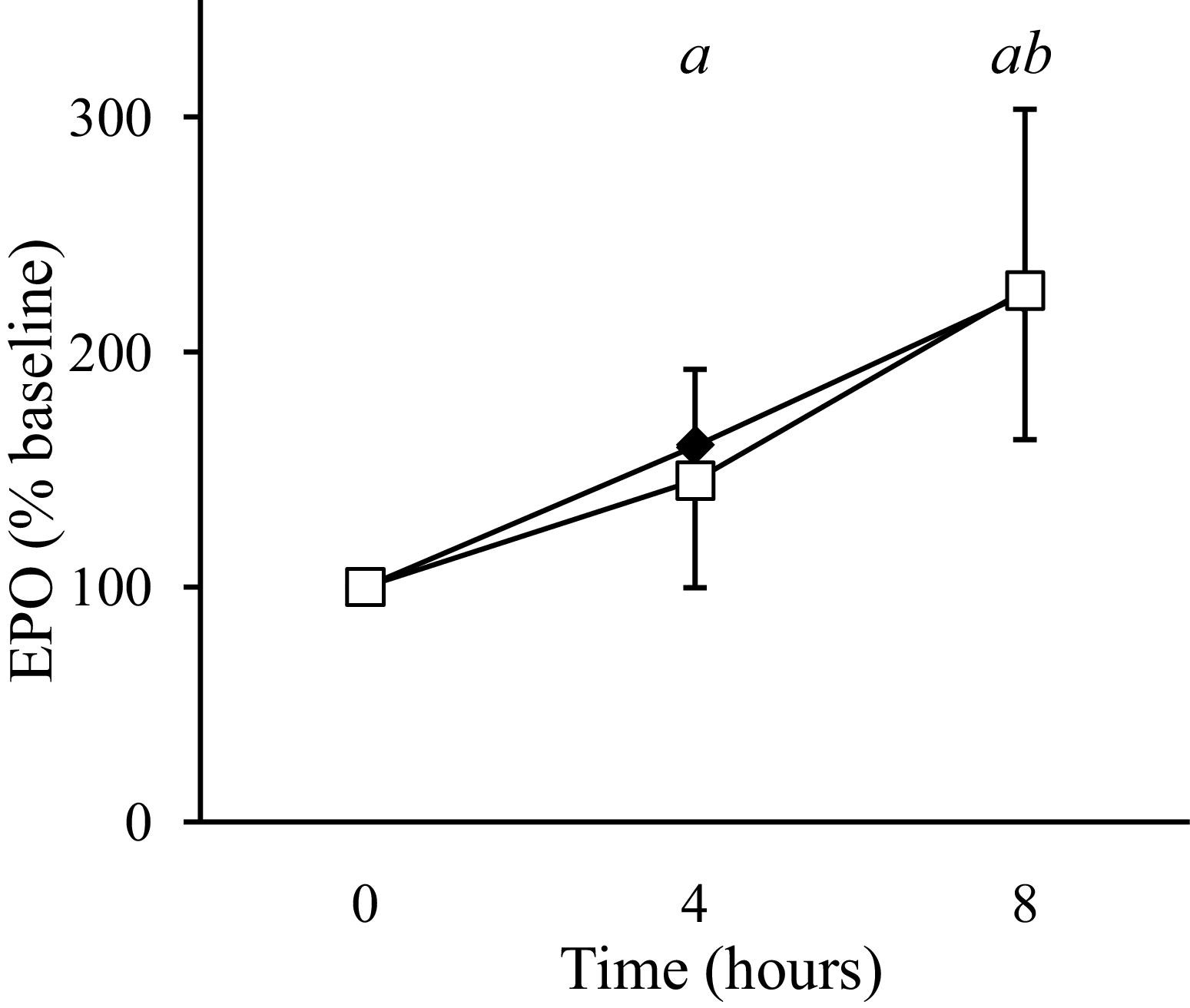
Erythropoietin (EPO) profile according to HIF1A genotype during the 8-hour exposure to 12.7% oxygen. EPO values are represented as a percentage of baseline values; CC (☐), CT/TT (♦). Values are the mean ± 1 SD. a Whole population significantly different from baseline (P <.001). b Whole population significantly different from the 4-hour time point. (P <.001).
During the exposure, Sa
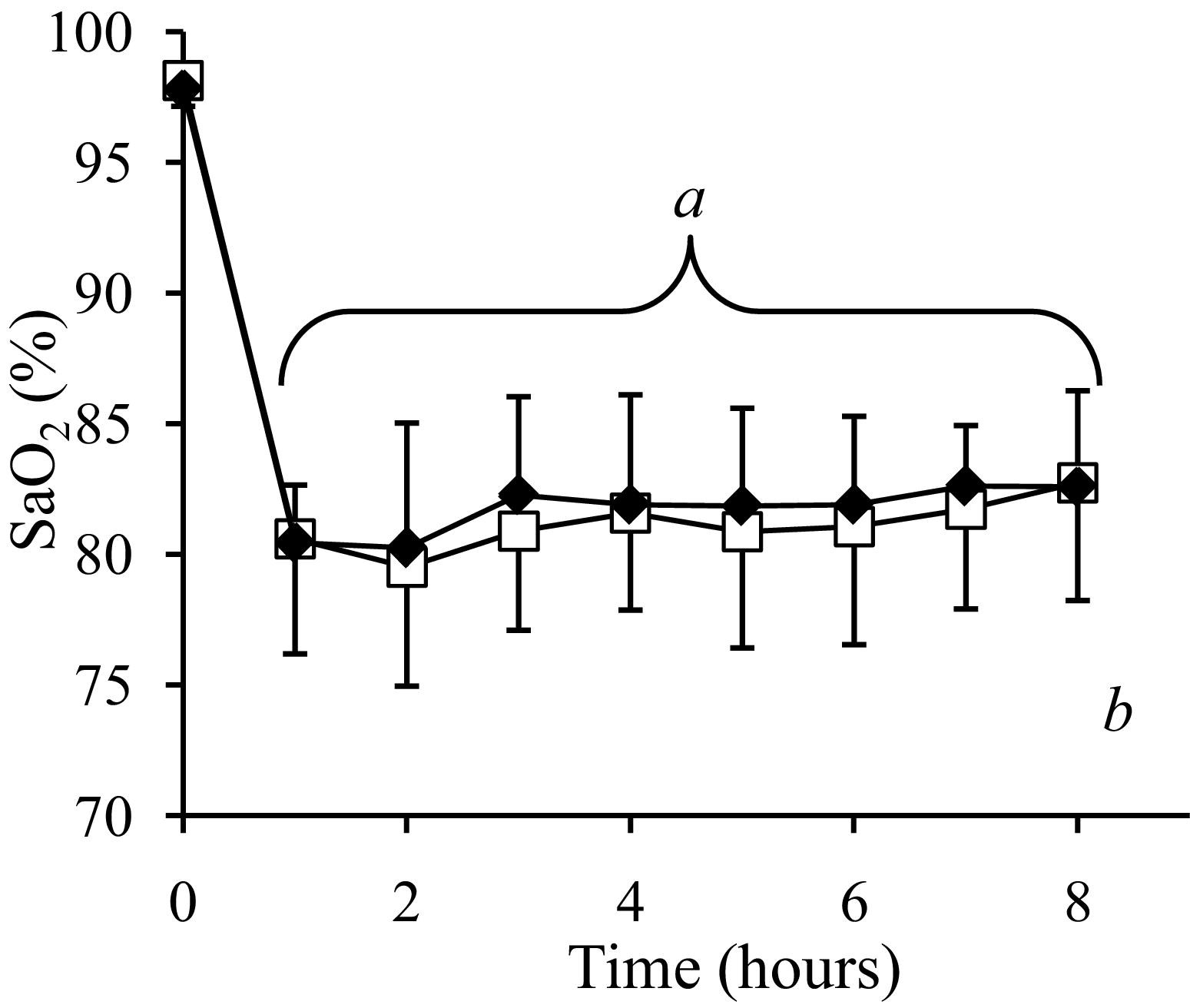
Arterial oxygen saturation (Sa
The symptoms of AMS increased progressively during the 8-hour exposure to 12.7% oxygen, and were significantly greater than resting levels from 2 hours onwards (2 hours, P = .014; 4 hours, P = .002; 6 hours, P = .001; 8 hours, P <.001). The AMS score for the CC genotype group was consistently higher than the CT/TT genotype group during hypoxia (Figure 3), although no significant association between alleles at the HIF1A C1772T loci and AMS symptoms was observed (P = .137).
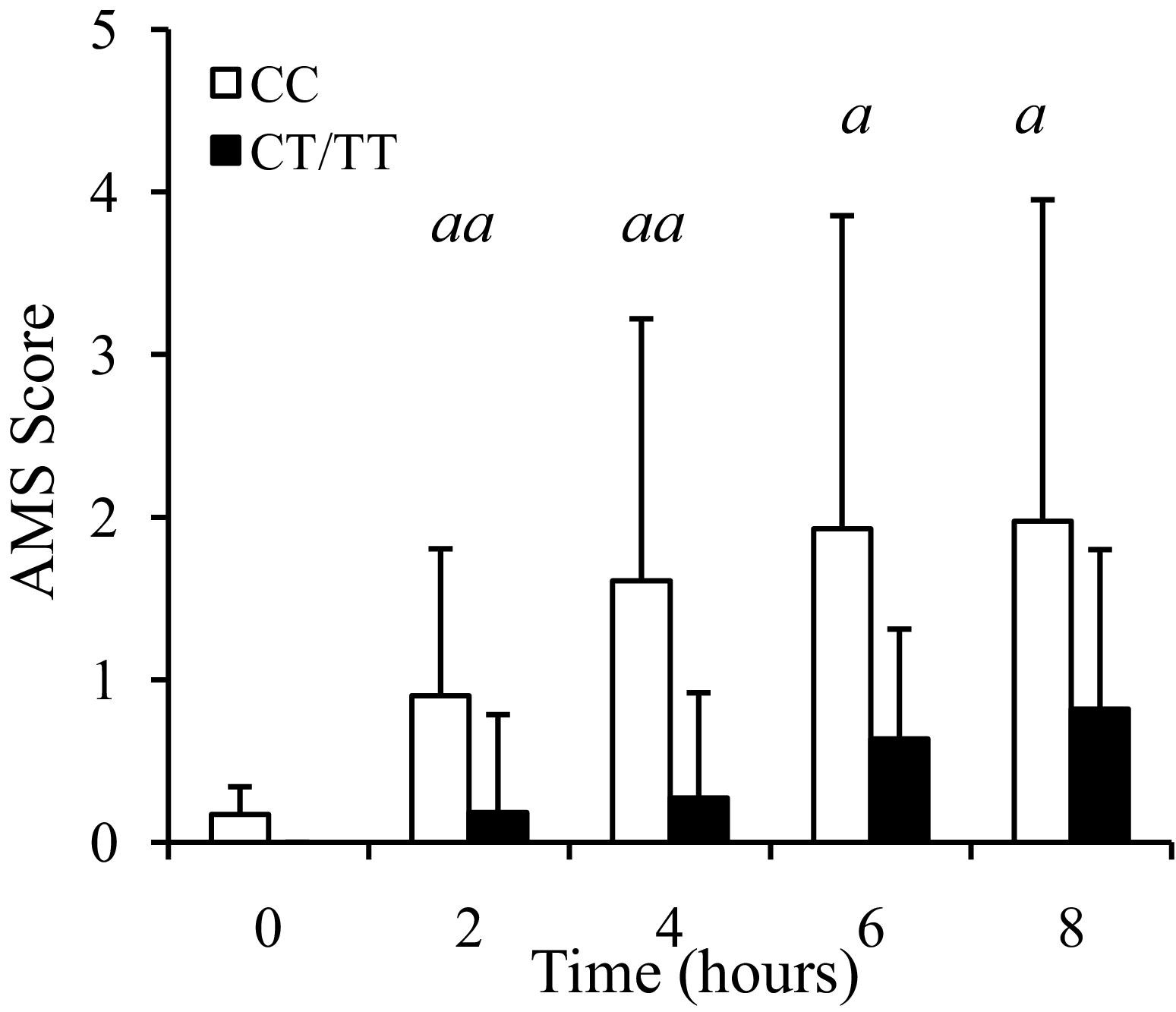
Acute mountain sickness (AMS) scores during an 8-hour exposure to 12.7% oxygen according to HIF1A genotype. Values are the mean ± 1 SD. aa Whole population significantly different from baseline at this time point (P <.05). a Whole population significantly different from baseline at this time point (P <.001).
Trial 2: Hypoxic Ventilatory Response
The physical characteristics of the 62 participants who took part in the HVR trial, sorted by HIF1A genotype, are shown in Table 3. During the hyperoxic phase of the isocapnic HVR trial Sa
Physical characteristics and spirometry data of the HVR participants according to HIF1A genotype
Data are mean ± 1 SD; P values are from independent-samples t tests. HIF1A, hypoxia-inducible factor 1A gene; FVC, forced vital capacity; FEV1, forced expiratory volume in 1 second.
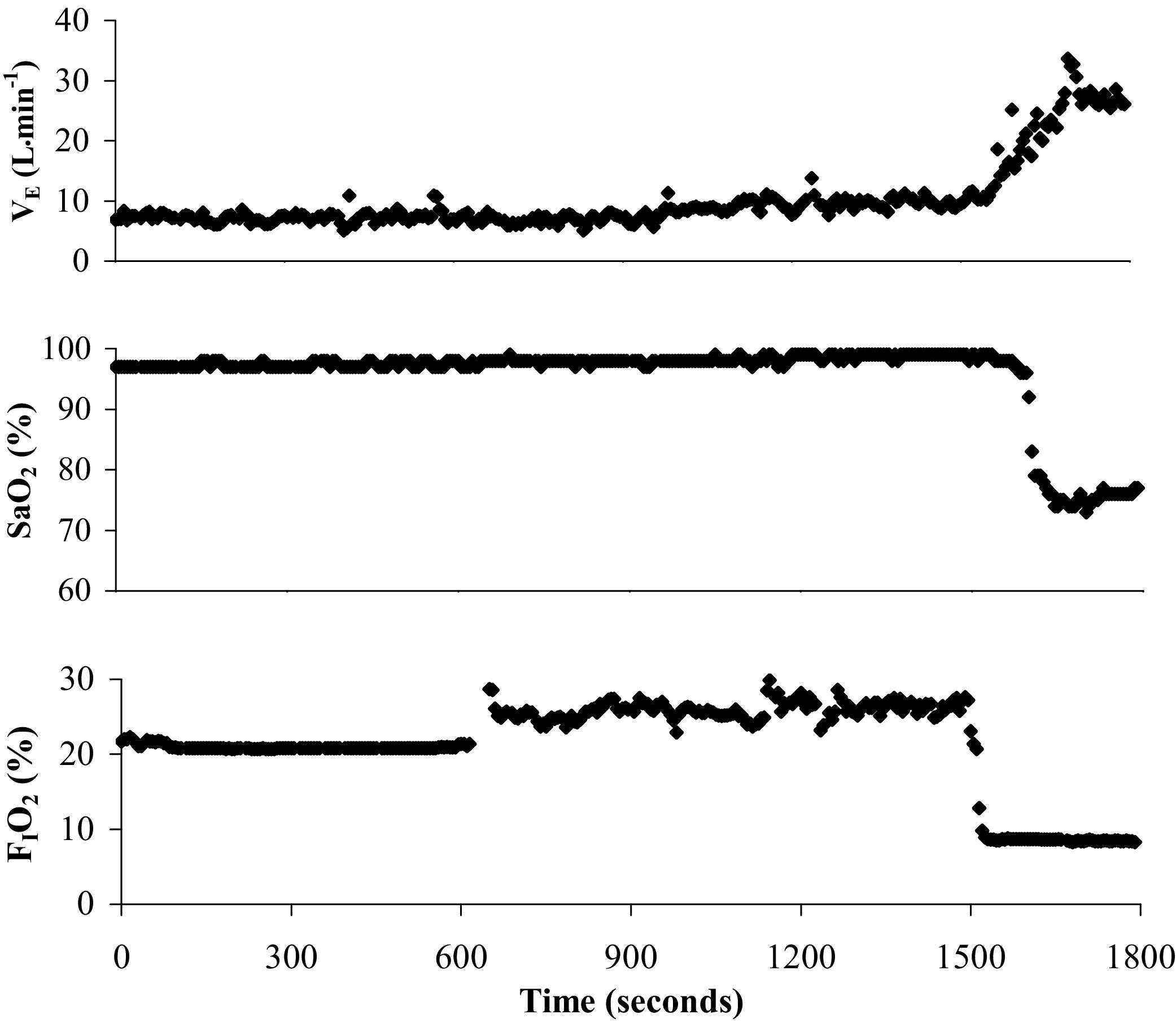
Example of a 30-minute isocapnic hypoxic ventilatory response (HVR) test showing minute ventilation; V̇E, arterial oxygen saturation; Sa
HVR was not influenced by HIF1A C1772T genotype (P = .565). Values were 0.70 ± 0.44 L · min−1 · Sa
Discussion
The main findings of this study were that the HIF1A C1772T polymorphism does not influence isocapnic HVR or acute EPO and Sa
The decrease in Sa
The C1772T variation in exon 12 of the HIF1A gene causes a proline to serine amino acid change at residue 582 of the HIF-1α protein. The serine variant has been shown in vitro to be more stable than the proline variant and the former is associated with greater transactivational activity during normoxic and hypoxic conditions.
17
The HIF1A C1772T polymorphism has also been shown to influence a number of disease phenotypes that relate to hypoxia,
18
–20 demonstrating a functional effect of the polymorphism in vivo. As HIF-1α has been shown to have an important role in ventilatory, cardiovascular, and hematological acclimation responses to atmospheric hypoxia,15,16 we hypothesized that the higher HIF-1α activity associated with the HIF1A T-allele would be associated with greater acclimation to hypoxia, which would include an increased EPO response, a higher level of Sa
Because this is the first study to investigate the associations between the alleles at the HIF1A C1772T loci and the HVR, EPO, and Sa
A limitation commonly found in human genetic studies is having low power to detect differences between genotypes due to limited participant numbers, and the current study is not an exception. In the present study, the lack of associations between the alleles of the HIF1A C1772T polymorphism and the various responses to hypoxia may in part be due to the moderate number of participants studied, despite participant numbers being comparable with other published studies that have investigated the influence of other genetic polymorphisms on these phenotypes.3,36 Future studies could overcome this limitation by genotyping a large number of individuals before measuring a particular phenotype and preselecting individuals to take part in the physiological aspect of the study based on their HIF1A genotype. This would ensure a larger number of TT individuals would be included in the study and would increase the power to detect differences between the genotypes.
In conclusion, we observed no association between the alleles of the HIF1A C1772T polymorphism and either HVR or acute acclimation responses to an 8-hour normobaric hypoxic exposure (12.7% oxygen).
Footnotes
Acknowledgments
The authors thank Julie Rainard and Ashley Belsham for help with molecular biological techniques, and the participants for giving up their time for this study.
