Abstract
Objective
The interaction of 15 variables representing physical characteristics, previous altitude exposure, and ascent data was analyzed to determine their contribution to acute mountain sickness (AMS).
Methods
Questionnaires were obtained from 359 volunteers upon reaching the summit of Mt Whitney (4419 m). Heart rate and arterial oxygen saturation were measured with a pulse oximeter, and AMS was identified by Lake Louise Self-Assessment scoring. Multiple logistic regression analysis was used to identify significant protective and risk factors for AMS.
Results
Thirty-three percent of the sample met the criteria for AMS. The odds of experiencing AMS were greater for those who reported a previous altitude illness (adjusted odds ratio [OR] = 2.00, P < .01) or who were taking analgesics during the ascent (adjusted OR = 2.09, P < .01). Odds for AMS decreased with increasing age (adjusted OR = 0.82, P < .0001), a greater number of climbs above 3000 m in the past month (adjusted OR = 0.92, P < .05), and use of acetazolamide during the ascent (adjusted OR = 0.33, P < .05).
Conclusions
The significant determinants of AMS on the summit of Mt Whitney were age, a history of altitude illness, number of climbs above 3000 m in the past month, and use of acetazolamide and analgesics during ascent.
Introduction
Trekkers and mountaineers are often concerned with developing acute mountain sickness (AMS) during their ascents. Acute mountain sickness has been defined as a self-limiting syndrome that commonly occurs in unacclimatized individuals who ascend too rapidly to altitudes higher than 2500 m. 1 The syndrome is characterized by a headache accompanied with other symptoms that may include gastrointestinal distress (eg, nausea, vomiting, and loss of appetite), fatigue or lassitude, dizziness, and sleep disturbances. Hydration, drug treatment, and descent are advised depending on the severity of symptoms. Resolution of AMS typically occurs after a couple days of acclimatization to the new altitude or upon descent to a lower altitude. However, if symptoms are ignored, and ascent continues, AMS can develop into life-threatening high-altitude cerebral edema. The pathophysiology of AMS has been reviewed,2,3 and several researchers have investigated the interaction of risk factors in an attempt to determine what contributes to this syndrome. 4 –8
Mt Whitney (4419 m) is the highest point in the contiguous 48 states of the United States. There are numerous routes of varying difficulty to reach the summit, with no technical climbing or mountaineering skill required for an ascent of the easiest route. The trailhead (2550 m) can be accessed by a paved road. Because of its distinction of being the “highest” summit, being easy to access, and having a nontechnical climbing route, Mt Whitney sees many attempts by both novice and experienced mountaineers alike, thereby providing an ideal setting to study AMS. Furthermore, the mountain is located within a half-day drive of many highly populated California costal cities. Thus, many of the attempts are made by unacclimatized residents who live at sea level.
The aim of our study was to expand upon previous AMS investigations by examining the interactions of numerous variables that might contribute to this syndrome on the summit of Mt Whitney. In addition to analyzing more self-report variables than have previous studies, we also included measurable variables of interest such as heart rate and arterial oxygen saturation (%Sa
Methods
Setting and Subjects
Data collection took place on the summit of Mt Whitney (4419 m) during 5 consecutive days in August 2004. People who reached the summit during this time were invited to participate in this study. After explaining the purpose, objectives, and procedures of the study, informed consent for participation was obtained. Subjects had the opportunity and were encouraged to ask questions about the study, and participation was entirely voluntary. Although a tally of those declining to participate was not kept, the participation rate was extremely high, with nearly all summiteers volunteering for the data collection. The study was approved by the participating institutions and granting agencies, and a special-use permit was granted for this data collection by the Inyo National Forest Service.
People who ascend this mountain from the west are often on treks of long duration and are well acclimatized to high altitude. In contrast, ascents from the east side of the mountain are typically done in 1 to 3 days. Only data from summiteers who ascended from the east side of the mountain (Whitney Portal) were included in the analyses (n = 359). This was the only exclusion criterion.
Questionnaire and Pulse Oximetry
Information about the subjects’ physical characteristics, acclimatization, experience at high altitudes, and ascent was obtained from a questionnaire. Personal data included age, gender, smoking status, and height and weight for subsequent calculation of BMI. In an effort to quantify previous altitude exposure and acclimatization history, subjects were asked about their altitude of residence, the number of times that they had been above 3000 m in the month before this ascent, the approximate number of times they had been above 3000 m in their lifetime, the highest altitude that they had ever hiked to before this ascent, and whether they had ever experienced altitude sickness. The variable of hours per week of training in the month before the ascent was used to quantify physical conditioning. Ascent data included the day and time that the subjects started their ascent (from the Whitney Portal trailhead) and reached the summit for subsequent calculation of ascent time, an estimate of the amount of liquid consumed, and any medications that were taken during this time.
In addition to self-report data obtained from the questionnaire, heart rate and %Sa
Assessment of AMS
Acute mountain sickness was assessed by the Lake Louise Self-Assessment score. 1 This commonly used assessment includes 5 symptoms: headache, gastrointestinal distress, fatigue or weakness (or both), dizziness or lightheadedness, and difficulty sleeping. Each symptom is scored 0 (not present) through 3 (severe or incapacitating) for a combined minimal score of 0 and a maximal score of 15. Our criteria for AMS were 1) a headache, 2) at least 1 other symptom, and 3) a total score of 3 or more. This constitutes the preferred assessment method and criteria for evaluating AMS. 9
Statistical Analysis
Basic descriptive statistics, including measures of central tendency, variance, and frequency, were computed for all variables measured on the study sample. Multiple logistic regression analysis was used to evaluate potential protective and risk factors for AMS. Presence or absence of AMS served as the outcome variable, and covariates included the following: age (years), gender, smoking status (yes/no), BMI (kg·m−2), use of acetazolamide (Diamox) or Ginkgo biloba during ascent (yes/no), use of any analgesics (eg, aspirin, ibuprofen, naproxen) during ascent (yes/no), history of altitude illness (yes/no), altitude of residence (meters), time to summit (hours), number of ascents above 3000 m during the past month and in one's lifetime, average hours per week of training in the month leading up to the ascent, pulse rate at summit, %Sa
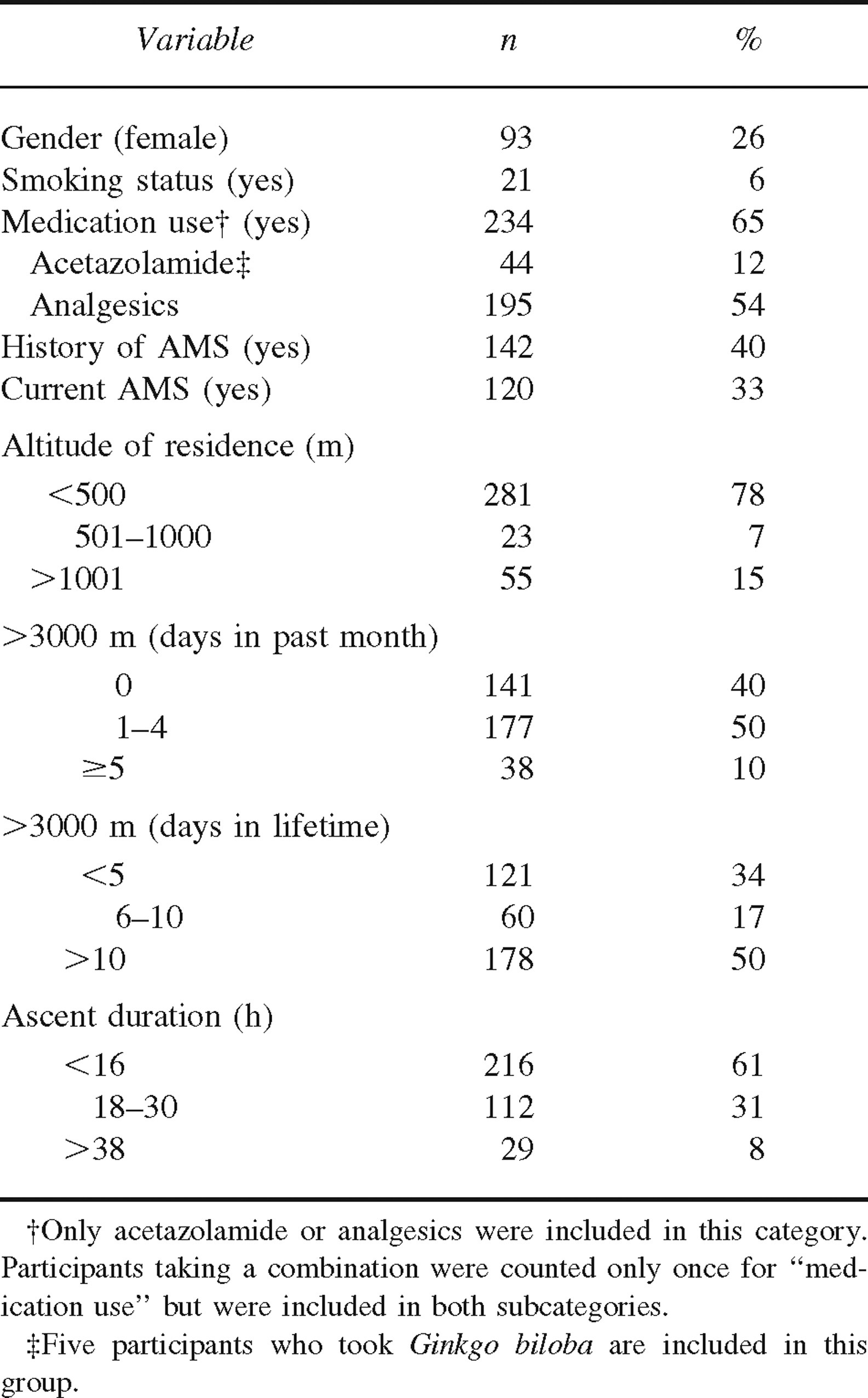
Demographic and ascent-related characteristics of study sample (n = 359)
An iterative model-building procedure using the likelihood ratio test was followed to develop the multiple logistic regression model. 10 First, a univariable analysis of the relationship between each previously mentioned covariate and the presence or absence of AMS was conducted. Second, any variable whose univariable analysis resulted in a P-value of <.25 was included in the main effects multivariable logistic model.11,12 Third, physiologically meaningful 2- and 3-way interactions among the remaining covariates in the main effects model were individually and jointly evaluated for significance by using the likelihood ratio test statistic. Fourth, covariates were iteratively dropped from the final model if they were not part of an interaction term and if the likelihood ratio test statistic was nonsignificant after their removal (P > .05). All analyses were conducted by using SAS (version 9.1).
Results
A summary of the demographic and ascent-related characteristics of the study sample is presented in Tables 1 and 2. The majority of the sample consisted of male (74%) nonsmokers (94%) who reported using either acetazolamide or analgesics during the ascent (65%). Forty percent of the sample reported experiencing at least 1 previous instance of AMS, and 33% of the sample met the criteria for AMS.
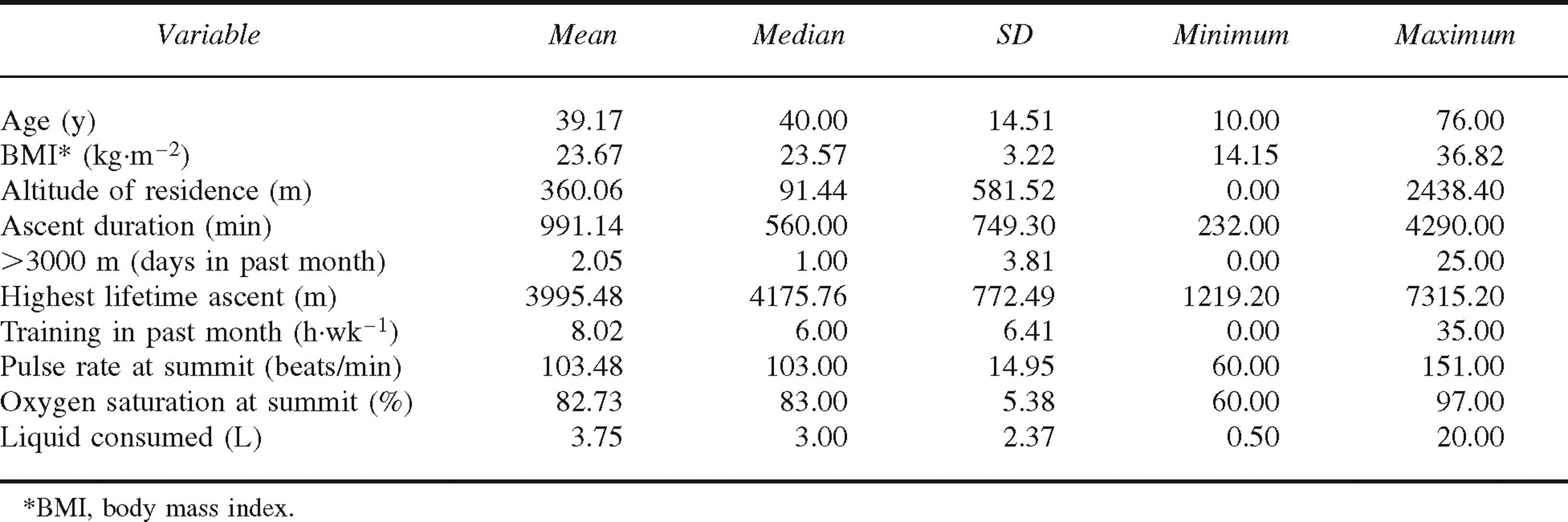
Demographic and ascent-related characteristics of study sample (n = 359)
Results of multiple logistic regression analysis are presented in Table 3. The analysis indicated that there was a significant reduction in the incidence of AMS on the summit of Mt Whitney with increasing age, number of climbs above 3000 m in the past month, and use of acetazolamide. The odds of experiencing AMS were significantly increased in subjects who had a history of altitude illness or were taking analgesics during the ascent. No significant interactions were found. The fit of the final model was significantly better than the null model (likelihood ratio test = 50.96, P < .0001), indicating that the predictors as a set significantly distinguished between subjects who did and did not have AMS (overall classification rate = 66.30%). Diagnostic analyses indicated that no collinearity was present among the main effects included in the final logistic model.
Results of multiple logistic regression analysis for predictors of AMS (n = 359)*
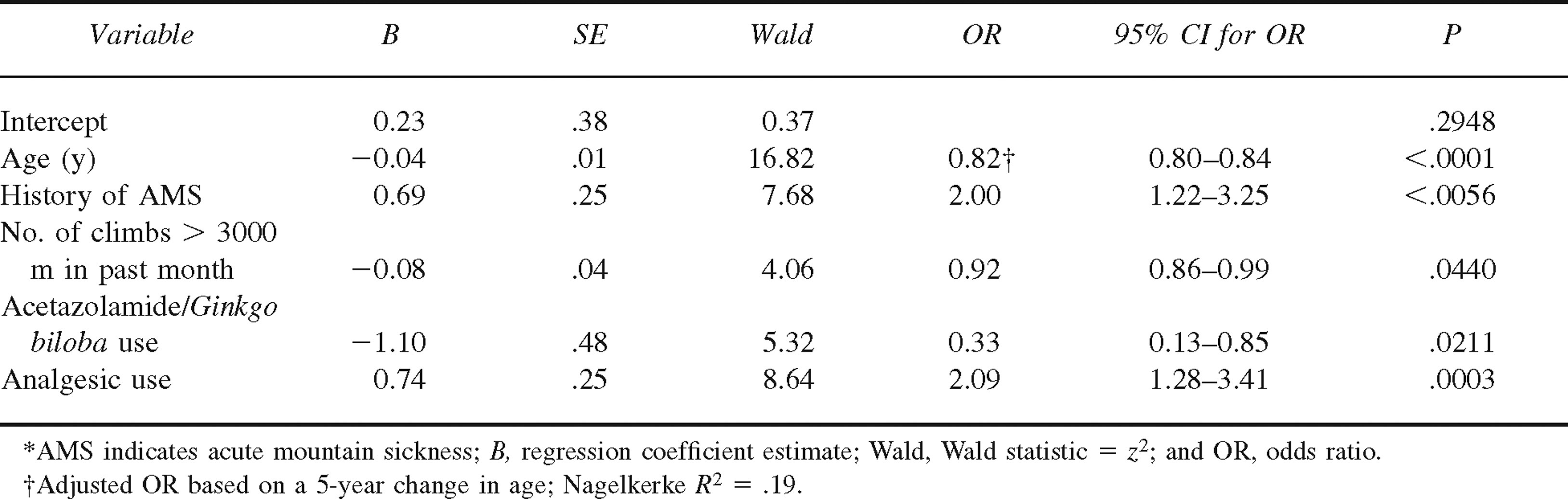
In the present study, variables with adjusted odds ratios (ORs) greater than 1 were considered risk factors for AMS. The odds for current AMS were twice as great in subjects who had previously experienced AMS (n = 142) compared with those who had not, controlling for other factors in the model (adjusted OR = 2.00, 95% confidence limit [CL] = 1.22–3.25, P = .006). Use of analgesics during ascent (n = 195) was also associated with a 2-fold increase in the odds for the presence of AMS (adjusted OR = 2.09, 95% CL = 1.28–3.41).
Variables with ORs less than 1 were considered protective factors for AMS. As age increased, the odds for AMS decreased significantly (adjusted OR for 5-year increase in age = 0.82, 95% CL = 0.80—0.84, P < .0001). The mean age was 41.71 years (SD = 14.71) for subjects without AMS and 34.40 years (SD = 12.81) for those with AMS. However, Figure 1 indicates that the negative relationship between increasing deciles of age and the adjusted ORs for AMS was nonlinear, with decreases in the odds for AMS becoming smaller toward the older range of the age distribution. An increased frequency in climbs above 3000 m in the past month was also associated with a decreased risk for AMS (adjusted OR = 0.92, 95% CL = 0.86—0.99, P = .044) (see Figure 2). Use of acetazolamide (n = 44) during the ascent was also associated with a 3-fold reduction in the odds for AMS (adjusted OR = 0.33, 95% CL = 0.13–0.85, P < .0001).
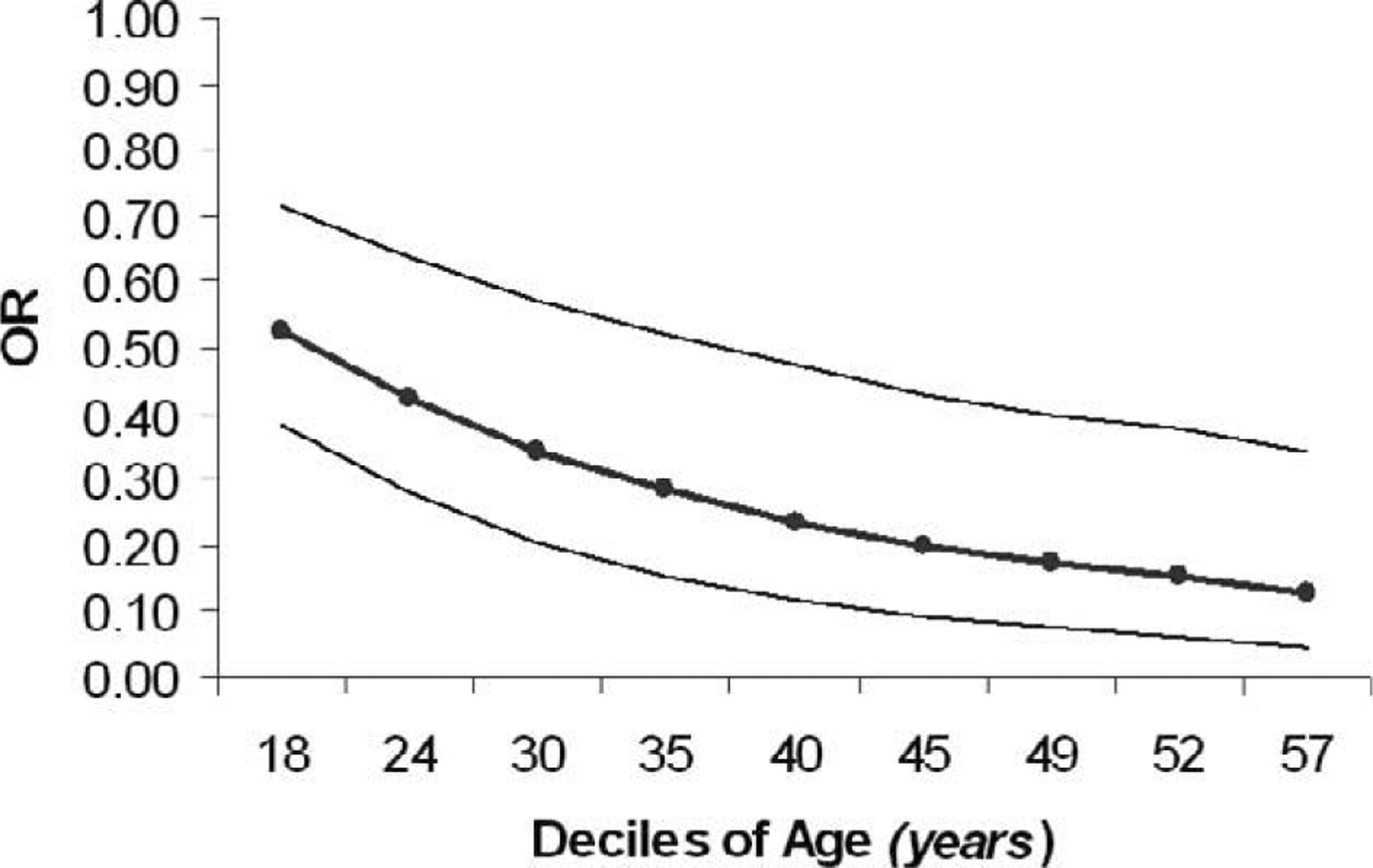
Relationship between adjusted odds ratios (ORs) for acute mountain sickness (AMS) and deciles of age (n = 359). Outer lines represent 95% CIs for ORs.
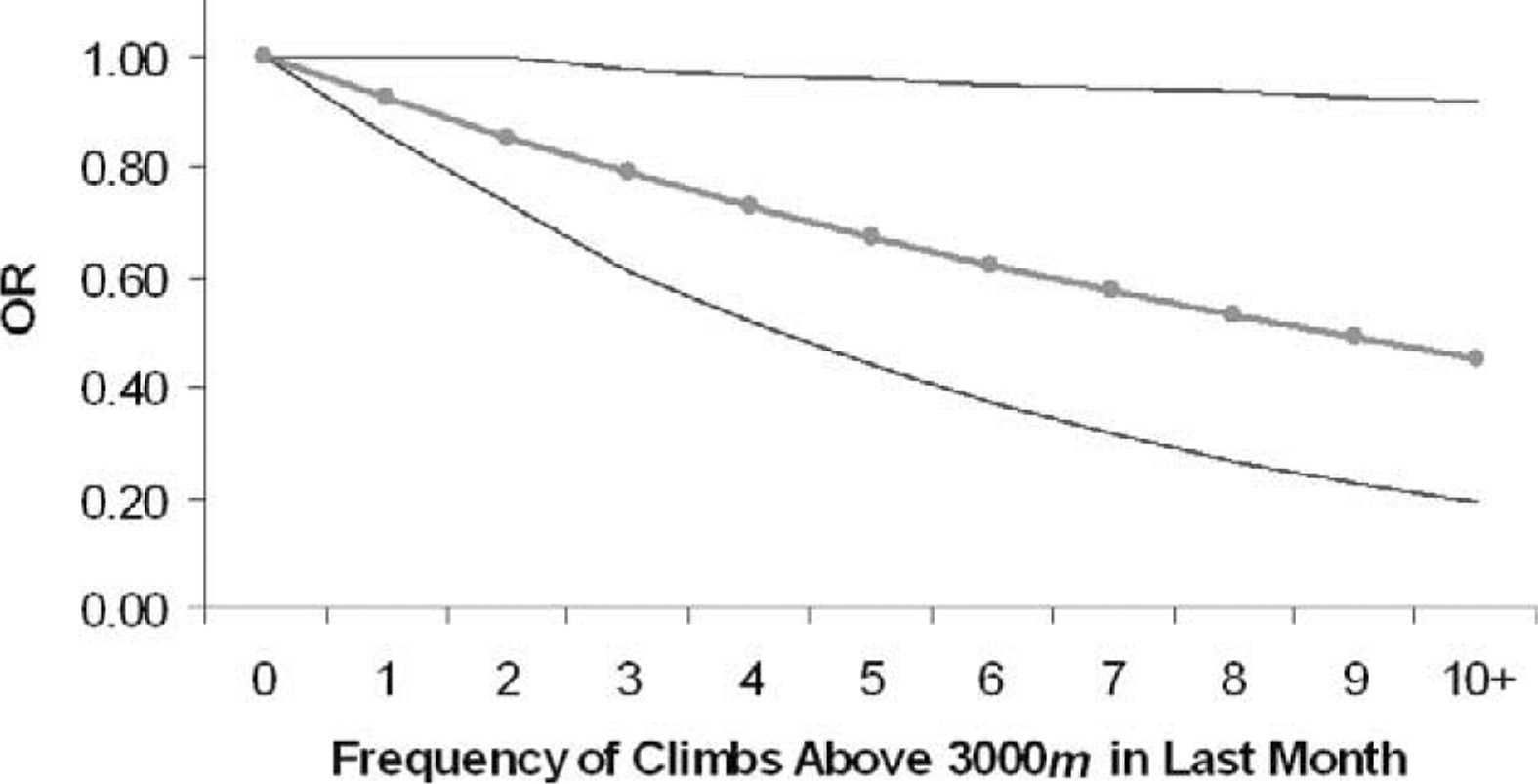
Relationship between adjusted odds ratios (ORs) for acute mountain sickness (AMS) and frequency of climbs above 3000 m in the past month (n = 359). Outer lines represent 95% CIs for ORs.
Discussion
A major reason for selecting Mt Whitney for data collection was the likelihood that our sample would be diverse. Indeed, the participants varied widely on physical characteristics such as age (10–76 years) and BMI (14.2–36.8 kg·m−2). Likewise, variables related to the ascent and previous altitude experience were widely distributed (Table 1). Although many participants had been to the summit of Mt Whitney on previous occasions and some were experienced mountaineers with multiple ascents above 6000 m, for 70% of the sample this ascent was a personal high-altitude record.
Thirty-three percent of the Mt Whitney summiteers met the criteria for AMS. This is very similar to the 28% to 30% prevalence reported by other researchers who evaluated trekkers and climbers at similar altitudes despite using different assessment scales or AMS criteria.4,7 However, our data collection took place on the summit, and thus our study was limited to summiteers rather than everyone on the mountain. It is logical to assume that many of the subjects who turned back before reaching the summit had AMS. Therefore, we hypothesize that the incidence of AMS on Mt Whitney is probably much greater than the 33% witnessed on the summit, and it likely approaches the 53% reported by Maggiorini et al 13 at 4559 m.
Physical Characteristics
Of the physical characteristics entered into the logistic regression analysis, only age was a significant predictor of AMS. Age was a nonfactor in some previous studies4,7,8; however, several other studies support our finding that increasing age may offer some protective benefit against AMS.5,6,14–17 Silber et al 18 reported that both the likelihood of developing a high-altitude headache and the severity of headache were greater in younger subjects. One hypothesis is that AMS is related to brain swelling, and individuals most susceptible to increased cranial pressure (small ratio of cranial cerebrospinal fluid to brain volume) are at greater risk for developing AMS.2,3,19 Ross 19 and Gaillard et al 5 theorized that with advancing age there is a decrease in brain size, leading to an increase in cranial compliance and a reduced risk for AMS.
However, the premise that cerebral edema resulting in increased cranial pressure is the underlying cause for AMS has been questioned. 20 Consequently, the theory that older mountaineers are at less risk for AMS because they might have more room in the skull is highly debatable. Furthermore, Graham and Potyk 21 have cautioned against stating age as a protective factor for AMS, citing methodological differences among studies as a potential confounding factor. Nevertheless, age was the most significant of all the variables included in this analysis, and the age range of our sample was greater than those seen in previous AMS investigations.
None of the other physical characteristics included in the analysis (gender, smoking status, or BMI) were significant predictors of AMS. Although a few studies have reported a greater incidence in AMS among females,5,6,22 most of the previous research is in agreement with our finding of a nonsignificant gender difference.4,7,8,14–16 Likewise, our result that smoking is a nonsignificant predictor of AMS concurs with all previous studies that have included this variable. 5 –8,15,22 Finally, our finding that BMI is not a significant predictor of AMS is also supported by previous research5,7,8; however, in a well-controlled hypobaric chamber study, Ri-Li and colleagues 23 found that average AMS scores increased more with time at altitude for obese subjects than for nonobese subjects. As might be expected, Mt Whitney summiteers have a healthier BMI than does the general US population. By using National Health and Nutrition Examination Survey standards for BMI, 3.6% of our sample were underweight (BMI < 18.5 kg·m−2), 65.5% were in the acceptable range (BMI = 18.5–24.9 kg·m−2), and only 30.9% and 3.3% were overweight (BMI > 25 kg·m−2) and obese (BMI ≥ 30 kg·m−2), respectively, compared with 65% and 31% for the entire US population. 24
Altitude History and Physical Conditioning
The following variables were used to try to quantify our subjects’ previous altitude experience: altitude of residence, number of times above 3000 m in the month preceding the ascent, number of times above 3000 m in one's lifetime, highest altitude ever achieved, and previous altitude illness. Of these variables, previous altitude illness poses a significant risk of AMS. The odds of experiencing AMS were doubled for Mt Whitney summiteers if they had a history of altitude illness. According to our results and those of previous researchers, 6 –8 there seems to be a strong relationship between a self-report of previous altitude illness and the risk of developing AMS subsequently.
It is commonly accepted and well supported with research that acclimatization and recent pre-exposure to altitude offer some protection against AMS. 4 –8 Even intermittent exposure to altitude (4300 m) has been shown to reduce the incidence and severity of AMS. 25 Our study also supports the phenomenon that recent exposure to altitude offers some protection against AMS. Furthermore, the more experiences above 3000 m in the month before the Mt Whitney ascent, the greater the reduction in odds of AMS (Figure 2).
It is likely that a high altitude of residence also offers some protection against AMS; however, our sample probably did not have enough people living at high altitudes to accurately test this hypothesis. Only 8% of the sample lived above 1500 m, and this may not have been a high enough altitude to offer a protective effect. Muza and colleagues
26
reported that subjects residing at moderate altitudes had higher %Sa
The protective benefits of previous altitude exposure appear to be transient. Neither lifetime experience (number of ascents above 3000 m) nor achieving a very high altitude in the distant past (>1 month) offered any protection against AMS. We are unaware of any scientific evidence that previous altitude experience (a lifetime history of ascents or achieving a very high altitude) affects the subsequent development of altitude illness.
Honigman et al 6 noted that attendees to a scientific conference at 3000 m who self-assessed their physical condition as “poor” were more likely to have AMS than those who rated themselves higher, and Ziaee et al 8 commented that there was a decrease in the incidence of AMS among trekkers with “proper physical conditioning” during an ascent of Mt Damavand (5671 m). However, it is widely believed that physical fitness training or conditioning performed at low altitudes does not protect against AMS.5,7,27 Our findings support this school of thought; self-reported hours per week of training was not a significant factor in the development of AMS. Of course, this is a difficult parameter to evaluate with self-report data, as there might be considerable variability in the interindividual interpretation of “training” and “physical activity.”
Ascent Data
By definition, the incidence of AMS should be related to ascent rate. Indeed, research supports the relationship between a rapid ascent and an increased incidence and severity of AMS.5,7,14,28 However, the majority of the participants in these studies were on multiday treks and ascents. There may be an asymptomatic period of 6 to 12 hours after reaching altitude, and symptoms do not reach their peak until 2 or 3 days later. 9 Thus, during ascents of a short duration, the rate of the ascent might not necessarily be predictive of AMS. For example, Ziaee et al 8 reported a nonsignificant difference in the prevalence of AMS between trekkers who took less than 6 hours and those who took longer than 6 hours to ascend from 4200 m to 5671 m. Similarly, the majority of the participants in our study (61%) went from 2550 m to 4419 m in less than 16 hours, and rate of ascent was not significant in the statistical analysis.
Decreased urine output leading to greater fluid retention is related to AMS29,30; however, the importance of fluid intake as a contributing factor is largely unknown. Thus, we included a self-reported estimate of the volume of liquid consumed during the ascent in our analysis. This variable did not reach significance in our study, but other researchers have reported that dehydration and a water intake of less than 3 L·d−1 increased the OR for AMS in trekkers.4,28 Of course, the volume of fluid consumed is not necessarily an indicator of hydration status, and this is an area of AMS research that merits further investigation.
Neither of the measured variables, heart rate or %Sa
It is now commonly accepted that acetazolamide offers some protection against AMS. Thus, when examining the influence of medication on AMS, we separated medication into acetazolamide and analgesics. No other medications were considered in the analysis. As expected, acetazolamide offered a protective effect against AMS. In a double-blind placebo study, Larson and colleagues 32 showed that acetazolamide users had fewer and less severe symptoms of AMS during a rapid, active ascent of Mt Rainier (4394 m). Since that time, many other researchers have demonstrated that acetazolamide is an effective prophylactic against AMS, and it is the recommended drug treatment to prevent this condition. 20
Five subjects reported taking G biloba (3 took only G biloba whereas 2 took it in conjunction with analgesics). Because G biloba may reduce the severity of AMS symptoms, 33 we included subjects taking this herb in the acetazolamide group in the original analysis. However, the use of G biloba as an effective prophylactic treatment of AMS is debatable, and it is not recommended as a substitute for acetazolamide. 20 Thus, we reanalyzed the data with the G biloba users removed from the acetazolamide group. Because none of the 5 G biloba users met the criteria for AMS, the OR for acetazolamide slightly increased (OR = 0.39, 95% CI = 0.15–1.02, P = .055) when these subjects were removed from the acetazolamide group. This reanalysis did not substantially change the ORs for the other predictors of AMS.
Analgesics use was associated with an increased risk of AMS; however, caution should be applied when interpreting this result. We are not implying that taking an analgesic places one at a greater risk for subsequent AMS. It is likely that many of our participants who used analgesics did so after they started experiencing symptoms associated with AMS in an effort to alleviate those symptoms. Thus, there was a strong relationship between the two, resulting in analgesics use showing up as a positive risk factor for AMS in this regression analysis.
The percentage (65%) of Mt Whitney summiteers who were taking either acetazolamide or an analgesic was surprising. Similarly, Gaillard et al 5 reported that 66% of those trekking around the Annapurnas in 1998 were using medication; this was up from only 17% in 1986. An alarming and disturbing finding in our study was that many of the people taking medication were not certain of the dosage or how much they had consumed and had to check the label during the interview. We echo the concerns of Gaillard et al 5 that trekkers and climbers seem to have a nonchalant attitude toward using drugs during a trek or ascent. Education about the adverse effects, taking analgesics to mask the symptoms of AMS, and the increased risks of self-medicating in a high-altitude wilderness environment seems warranted.
Limitations
Our data should be considered within the context of the limitations in which they were collected. The reader is reminded that this is a study of only summiteers rather than all who attempted the summit. Thus, the prevalence of AMS for all mountaineers (not just summiteers) on Mt Whitney is likely much higher than the 33% observed on the summit. Furthermore, the factors contributing to AMS for those not reaching the summit might be different from what we observed in summiteers. Additionally, other than the measured variables of heart rate and %Sa
Conclusions
We found that advancing age, recent exposures above 3000 m, and acetazolamide use offered some protection against AMS, whereas a previous experience with altitude illness increased the odds of developing this syndrome when on the summit of Mt Whitney. Additionally, there was a positive relationship between analgesics use and AMS, which was likely the result of participants who were already experiencing AMS trying to alleviate their symptoms. Our results should be viewed in the context of other similar studies. Methodological differences such as the large percentage of our sample using medication, the limit of our data collection to trekkers who reached the summit, and the reliance on self-report data likely contribute to some of the differences in results seen between our study and others.
Footnotes
Acknowledgments
Finally, we offer our sincerest thanks to the Inyo National Forest Service for granting us permission to collect data in the Mt Whitney wilderness.
Funding
This research was supported by grants from the American Alpine Club and Mazamas. We are grateful to these organizations for their financial support. Additionally, pulse oximeters were provided by Nonin Medical Inc, Plymouth, MN.
*
This paper has been accepted for a poster presentation on June 3, 2006, at the 53rd Annual Meeting of the American College of Sports Medicine in Denver, CO.
