Abstract
Shear stress plays a critical role in regulating platelet adhesion and thrombus formation at the site of vascular injury. As such, platelets are often examined in vitro under controlled shear flow conditions for their hemostatic and thrombotic functions. Common shear-based platelet analyses include the evaluation of genetic mutants, inhibitory or experimental compounds, matrix substrates, and the effects of different physiological and pathological shear forces. There are several laboratory instruments widely used for studying shear flow, including cone and plate viscometers and parallel plate perfusion chambers. These technologies vary widely in the types of samples, substrates, blood volumes, and throughput that are involved. Here, we describe a microfluidic system for platelet analysis under shear flow. We used the devices to study thrombus formation on collagen I and von Willebrand factor. The system was also used to investigate dose response to the antiplatelet compound, Abciximab, under shear flow conditions with an emphasis on maximizing the number of data points per single patient sample. The presented method confers multiple advantages over conventional approaches. These include the ability to assess up to 24 conditions simultaneously in real time, maintain identical physical conditions across experiments, and use extremely low donor volumes.
Introduction
Analysis of platelet function under flow is requisite to understanding the complex biological relationships leading to hemostasis and thrombosis. 1 The function of platelet receptors and the eventual biological outcome are strongly influenced by fluid shear stress generated by the partially laminar flow of blood in the circulation (reviewed in Refs. 2 and 3). Common in vitro methods to study platelet biology under conditions of shear flow include light transmission aggregometers, cone and plate viscometers, perfusion chambers and more recently, microfluidic devices.
The standard instrument for real-time highresolution data capture while flowing blood over a relevant substrate is the perfusion chamber. 2,4 Common perfusion configurations include parallel plate-flow chambers (PPFCs) or glass capillary tube-flow chambers. Typically, the flow is driven by a syringe or peristaltic pump placed alongside the chamber to generate wall shear stress that is similar to blood flow in the vasculature. These configurations are flexible in terms of shear rates applied and substrate in the flow path. One of the distinct disadvantages of perfusion chambers is that a relatively large volume of blood must be used. These volumes typically fall in the 10–30 mL range per each experimental condition, although there are smaller volume chambers available. 5 The flow rates in PPFCs rapidly deplete the available human donor blood and are prohibitive for using murine and pediatric clinical samples. Multivariant analyses and replicates from the same blood sample are largely impossible. The assembly of the chamber and apparatus is inherent in the use of PPFCs. To assemble the chamber, pumps and tubing must be prepared that takes an average of 20–45 min to complete. When the time sensitivity of patient draws is considered, the number of individual experiments that can be performed within 3 h of sample collection is limited to the setup time plus the experimental time; the total time required per condition is set at a minimum of 30 min. Given that the working lifetime of one blood draw can be as short as 3 h postcollection, only a small and limited number of conditions (maximum of six conditions) can be examined from the same donor sample using conventional flow chambers.
To address the limitations posed by PPFCs, many laboratories have devised microfluidic devices that can be used in lieu of PPFCs for platelet studies. 6 –9 These devices generally provide workflow advantages over conventional technologies, which include laminar flow, compatibility with realtime microscopy, and microliter-scale fluid requirements. The latter benefit enables multiple experiments from single blood draws or murine samples. Although the microfluidic format has provided several key advantages over conventional platelet assays, many of these devices do not address the need for a simple, reliable, and repeatable fluidic interface. For instance, some of the limitations include nonstandardized fabrication conditions and lack of an integrated or standard fluid-chip interface. As such, these devices have limited capacity for widespread use outside of the developer's laboratory.
Here, we present a well-plate microfluidic system and novel method for in vitro platelet behavior analysis. The system was designed to provide precise control over shear stress to emulate physiological conditions encountered in the vasculature. It addresses critical limitations with conventional platelet assay technologies by providing real-time highresolution microscopy data in a low-volume microfluidic format. Compared with other microfluidic technologies, the system presented here provides integrated control over critical experimental conditions and an integrated fluidic interfacing mechanism. 10
Methods
Blood Samples
Blood samples were collected from five healthy donors using sodium citrate as anticoagulant (0.38% final concentration) (Analytical Biological Services, Wilmington, DE or Baylor). Before use, collected blood was held at room temperature in the collection tubes. Whole blood was stained with Calcein AM (Invitrogen, Carlsbad, CA) at a final concentration of 4 μM for a minimum of 15 min. Abciximab (Centecor/Eli Lilly, Horsham, PA) was dosed directly to whole blood and incubated at room temperature for a minimum of 5 min before application of shear force.
PPFC Studies
PPFCs were assembled as described previously. 11 Briefly, a glass coverslip was coated with 10 μg/mL of type I fibrillar collagen from equine tendon (Helena Laboratories, Beaumont, TX) and incubated for 1 h at room temperature. Unbound collagen was then removed by washing the coverslip with 0.9% of NaCl and uncoated surface was blocked with 1% bovine serum albumin (BSA). Four milliliters of blood were perfused through the chamber at a flow rate of 2 mL/min, which generated a calculated wall shear stress of 10 dyn/cm2 for 2 min using a peristaltic pump (Ismatec, Glattbrugg, Switzerland). Images of thrombus formation were captured with a Nikon TS100 and CCD camera (QICAM Fast, Surrey, BC) in five locations in the flow chamber and analyzed using BioFlux Montage Software (Fluxion, South San Francisco, CA). Thrombus formation was expressed as average surface coverage for experimental over control.
Microfluidic Device Studies
Well-plate microfluidic devices (Fluxion Biosciences, South San Francisco, CA) were used for thrombosis experiments. 10 Each device consisted of 24-individual microfluidic flow channels assembled in parallel onto an Society for Biomolecular Sciences-standard 48-well plate. The devices were coupled to an airtight lid, called an interface, and flow was controlled using pneumatic controller, called the BioFlux controller (Fluxion). 12 Two device configurations were used: a pathological, high shear (HS) variation that runs from 0 to 200 dyn/cm2 (0–5000 s−1 at 4cP), and a physiological, low shear (LS) variation that runs from 0 to 20 dyn/cm2 (0–500 s−1 at 4 cP) (Fig. 1). LS channels were prepared by coating with 10 μg/mL of type I fibrillar collagen from equine tendon (Helena Laboratories, Beaumont, TX) or 100 μg/mL bovine collagen I, sheared (Invitrogen) and HS channels were coated with human von Willebrand factor (VWF) at 100 μg/mL (Haematologic Technologies, Inc., Essex Junction, VT) in phosphate buffered saline (PBS, containing Ca2+ and Mg2+) for 1 h at room temperature. Channels were washed with PBS for 5 min at 5 dyn/cm2 and blocked with BSA 1% (w/v) in PBS for 15 min. Blood was treated with Abciximab at 26, 53, 100, 200 (315, collagen bovine and VWF), and 420 nM. Controls of no coating and no Abciximab were included in each experiment. Fibrillar collagen I experiments were performed for 2 or 5 min (dose response) at 10 dyn/cm2. VWF experiments were performed at 200 dyn/cm2 for 5 min. Micrographs were captured using a CCD camera (QICAM or Retiga, Surrey, BC) on a Nikon TS100 (for comparison to PPFC) or BioFlux 1000 imaging system (Fluxion). Data analyses were performed using Montage (Fluxion). Dose—response curves were prepared using Graph Pad Prism (Graph Pad Software, La Jolla, CA).
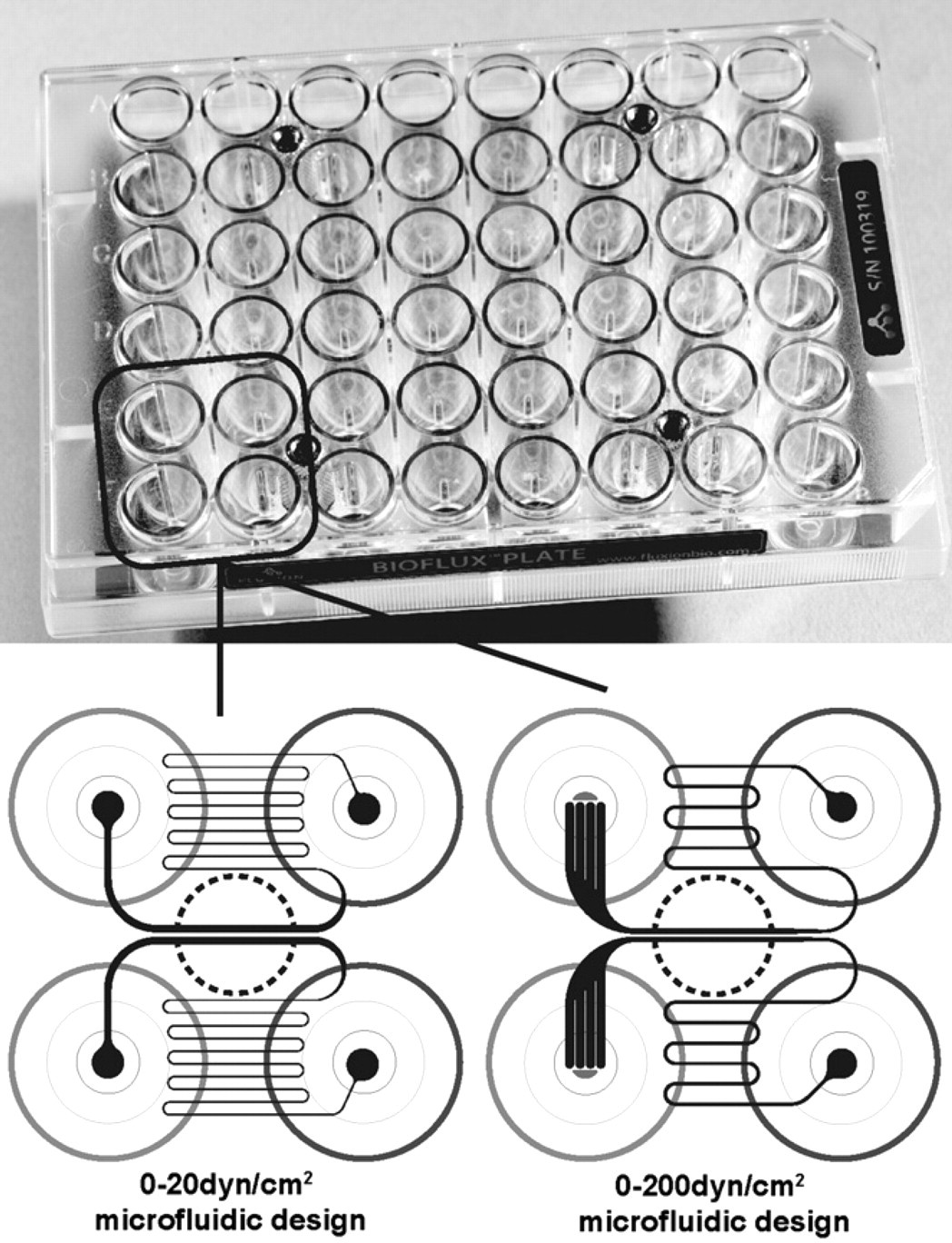
Well-plate microfluidic devices used in this study. The microfluidic channels are located below the wells of the microplate. Sample is added to the well of the plate and perfused through the channel. Microfluidic channel geometries (bottom) are shown in black, each channel requires two wells of the well plate. The experimental zone, where data are captured, is located between the wells (circle, black dashed line).
Results
The study of dynamic platelet behavior under flow is typically executed using a PPFC. We have designed microfluidic devices with 24-microchannels (MCs) coupled to well plates to perform such experiments with high-throughput and lower-sample volumes. To determine that the platelet response to shear was as expected, thrombus formation was investigated concurrently using a PPFC and the described MC device. A 5-mL sample from a single donor was used for both experimental instruments. Type I fibrillar collagen, a major component of subendothelial matrix and a potent signal for thrombus formation, was immobilized on the bottom surface of the chamber and the microfluidic channel as a prothrombic substrate 13 and was intended to model an area of exposed subendothelium induced by vascular injury. Thrombus formation was evident in both devices after 2 min of flow at 10 dyn/cm2. Surface coverage was similar in both devices; in the MC, a surface coverage of 4.1% (±0.75%) was observed compared with 3.5% (±0.95%) in the PPFC (Fig. 2). The PPFC required 4 mL of whole blood for the 2-min run, whereas the MC exhausted < 10 μL for the 2-min period under flow.
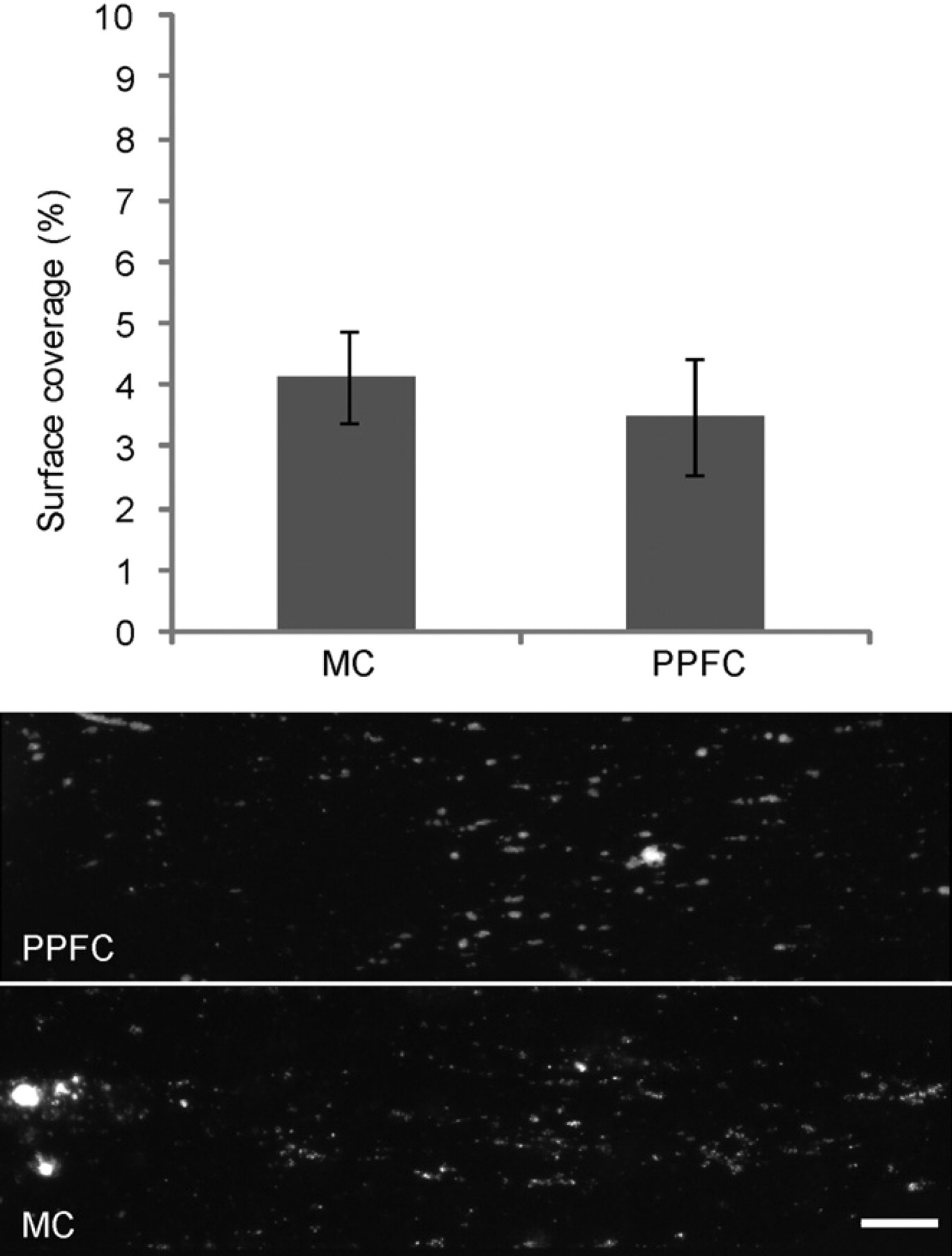
Thrombus formation in parallel plate-flow chambers (PPFC) vs microchannel (MC). Whole blood from the same donor was perfused through either a PPFC or an MC. Data were captured on the same microscope for both types of chamber. Platelet adhesion data were collected after 2 min of flow at 10 dyn/cm2 in five distinct fields of view. The surface coverage above threshold was determined for all the data and expressed as % of total area queried. Error bars indicate standard deviation. Representative fields of view are shown (below) for PPFC and MC. Scale bar denotes 100 μm.
As the platelet response to collagen and shear in the MC was comparable to the PPFC, a model set of experiments was then performed in the MC. Abciximab treatment was used as the test condition for the MC devices. Platelet binding to glycoprotein VI and integrin a2b1 initiates intracellular signals, which activate the major platelet integrin αIIbβ3 (GPIIb/IIIa) to aggregate platelets by fibrinogen. 14 Abciximab blocks fibrinogen binding to activated αIIbβ3 preventing platelet aggregation. Microfluidic flow-channel devices were used to study the Abciximab dose response at 10 or 200 dyn/cm2 (250 or 5000 s−1), using fibrillar collagen I (10 dyn/cm2), collagen I (bovine, sheared) (10 dyn/cm2), or VWF (200 dyn/cm2) as adhesion matrices. Dose responses to Abciximab for three donors were examined simultaneously from 0 to 420 nM of Abciximab on fibrillar collagen I followed by a single donor for collagen I (bovine, sheared) and VWF. Data were collected for 40 experimental conditions within 1 h using the MC devices. On fibrillar collagen I, the IC50 value for Abciximab was less than 26 nM with maximum inhibition at 95% of the control (Fig. 3A). On collagen I (bovine, sheared), the IC50 was approximately 26 nM with a surface coverage maximized at 90% of the control (Fig. 3B). Abciximab-mediated inhibition for platelet adhesion on VWF matrix had a measured IC50 of lower than 26 nM; complete inhibition (100%) was achieved with VWF (Fig. 3B). For all the treatments, the maximum level inhibition was reached at 53 nM (Fig. 3). The fundamental difference between the two types of substrates can be attributed to individual platelet adherence to collagen I at a basal level without interference from Abciximab, which in this case blocks thrombus formation at the secondary step of platelet—platelet aggregation. In contrast, primary platelet adhesion to VWF is blocked by Abciximab. Therefore, inhibition of platelet surface coverage of the chamber surface is likely to be more pronounced with VWF. 3
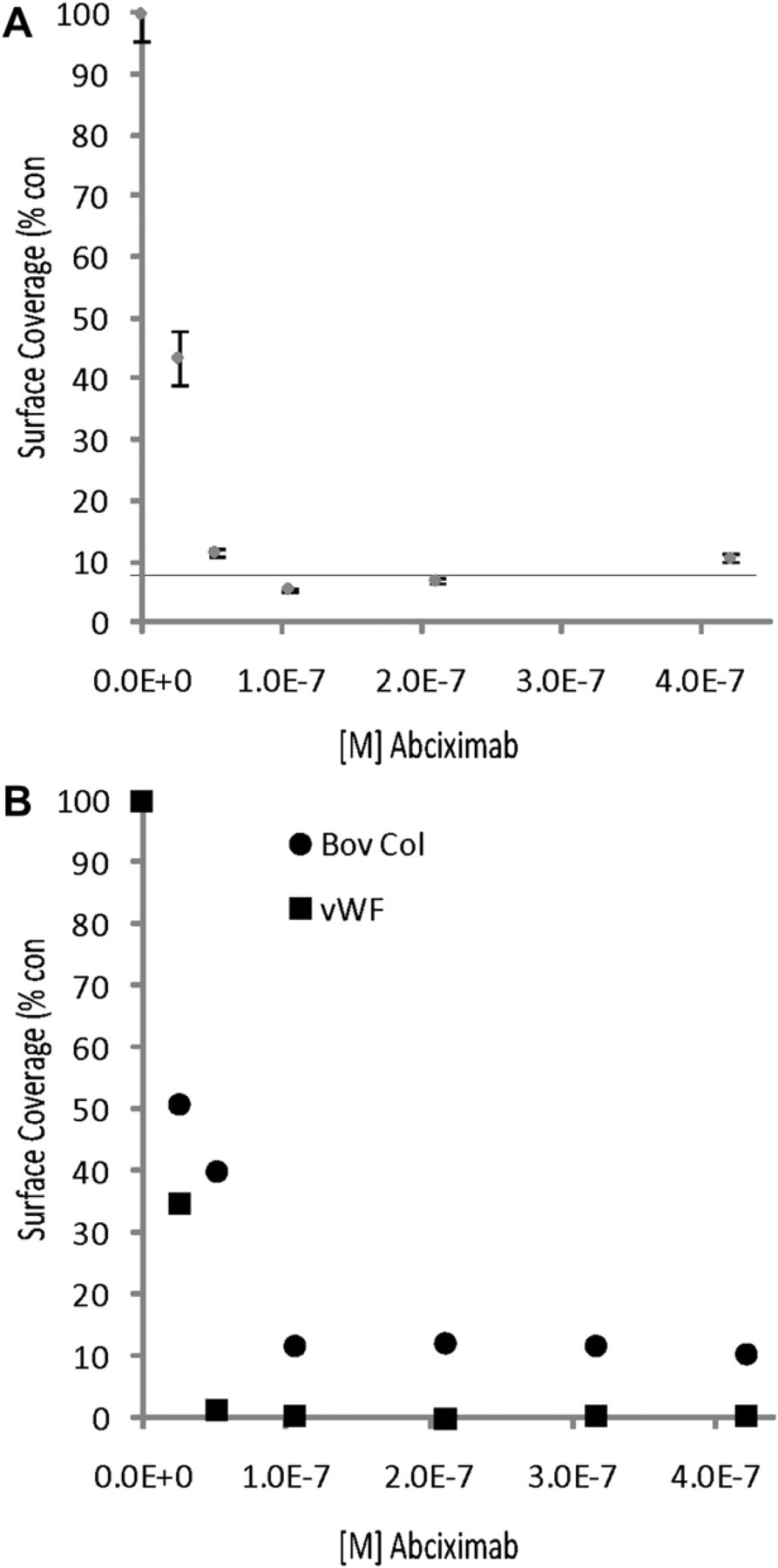
Abciximab dose response using microchannel. Whole human blood was perfused over coated chambers for 5 min at 10 dyn/cm2 or 200 dyn/cm2 (VWF). Micrographs for three fields of view per channel were captured for each condition with an automated microscope. A. Each Abciximab concentration was assessed in duplicate for fibrillar collagen for three donors. Percent thrombus formation was expressed as platelet surface coverage for treatment over control. Error bars denote standard deviation across the three donors. Gray horizontal line indicates platelet response for channel without collagen coating. B. Inhibition of thrombus formation data generated using microfluidic devices at low and high shear on different coatings using a single donor (•) IC50 dose response for Abciximab on collagen 1 using 0–20 dyn/cm2 microfluidic device at 10 dyn/cm2 (▄) IC50 dose response for Abciximab on VWF using a 0–200 dyn/cm2 microfluidic device at 200 dyn/cm2.
Discussion
The method using the MC-containing devices described here enables the full functionality of a PPFC for thrombus formation assays, including the ability to observe real-time platelet behavior under shear stress. However, the MC device and presented method provide significant improvement over the traditional PPFC in the areas of sample volume, throughput, device handling, flow control interfacing, and range of precise physical conditions that can be applied to whole blood.
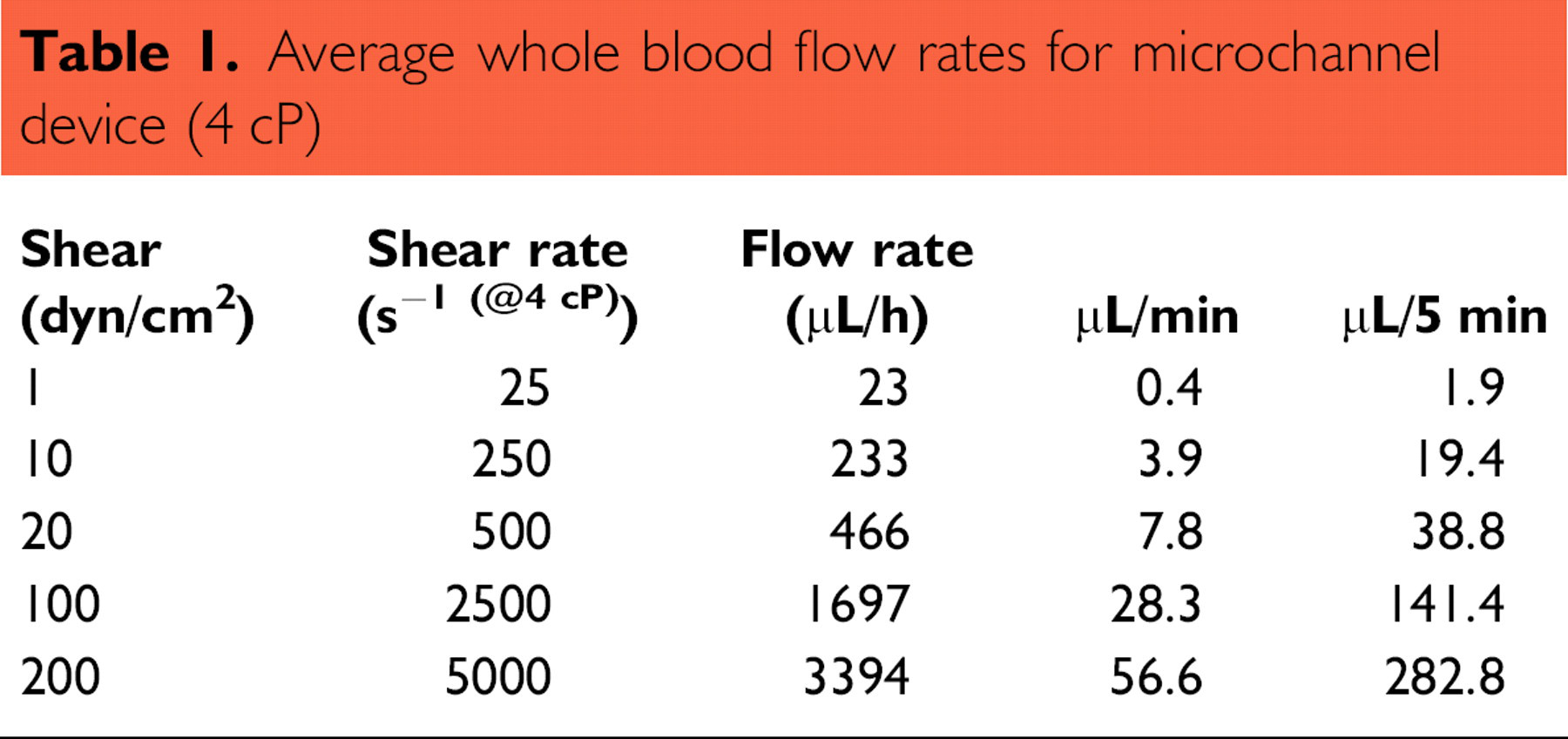
The MCs used in this study require very low blood volumes ≤300 μL per 5-min experiment (Table 1) compared with PPFC. Although commonly consuming 15–30 mL of sample for most flow conditions, even the smallest described perfusion chamber: Sixma et al. (1998), still consumes 16 times more blood volume per assay when compared with the MC running at the same shear rate (1500 for low PPFC vs 90.5 μL for the MC for a 5-min experiment at 1600 s−1). Use of MCs presents the opportunity to study rare clinical samples and murine samples with replicates and different shear or compound conditions and to fully capitalize on freshly collected blood from a single donor.
Average whole blood flow rates for microchannel device (4 cP)
In the case of the well-plate devices, an array of MCs coupled together affords the opportunity to perform 24-simultaneous experiments. In this study, 40 experiments to characterize dose response were collected within 1 h. In comparison, the same experiments in using PPFC would take days to complete. Automated microscopy hardware such as motorized stages, focus devices, and CCD cameras are typically used to scale-up the overall throughput capability of the microfluidic flow cells. In this manner, full-chip parallelization (24 flow channels) of dynamic time-lapse data collection can be accomplished with relatively little user intervention.
The interface is unique in that there is no liquid in the MC system outside of the wells of the well plate, circumventing the routines involved with assembly of PPFC and academic microfluidic devices, running fluids through lengths of tubing, and setup of the chambers. The combination of the well-plate chassis and the interface confers a no set-up and no clean-up operating scenario, which further enables the throughput and ease of use of the system. The self-containment of the MC device and pneumatic controller also offers an advantage for performing experiments where using a disposable, contained device is necessary, for instance when using blood from an unknown origin or samples with other blood-borne pathogen situations.
The microfluidic devices presented here offer the ability to perform multiple thrombus formation assays under flow with very low-sample volumes. The ability to perform experiments under these conditions is applicable to other areas of research, which benefit from flow such as studies of endothelial cells, stem cells, and immunology.
Competing Interests Statement: The authors disclose that they are all employees and stockholders of Fluxion Biosciences.
