Abstract
A simple low-cost yet versatile microtechnology platform is demonstrated as capable of performing a variety of microfluidic actuation functions including on-chip pumping, mixing, cell/particle sorting, and sample extraction. This technology termed air-liquid cavity acoustic transducers (ALCATs) uses trapped air bubbles as the functional elements. When an external acoustic energy source is applied to the device, the trapped bubbles oscillate, generating acoustic streaming within the fluid. By controlling their relative position and angle with the liquid channel/chamber, the ALCAT can push away or pull in the surrounding liquid contents and its contained particulates. In this report, we provide a general introduction of the ALCAT microtechnology platform and its application to enhancing biomolecular assays.
Introduction
A versatile microtechnology platform termed air-liquid cavity acoustic transducers (ALCATs) enables enhanced biomolecular assays by enabling faster detection, multiplexed and multistep assays, and the sorting of cells and beads (for bead-based assays). The ALCATs can potentially perform sample preparation steps such as analyte extraction and pre-concentration. The ALCATs can also be designed to function as micropumps enabling the integration of different bioassay functions on common chip-scale platforms to miniaturize and automate laboratory steps that normally require tedious manual operations. The ALCATs are easily microfabricated using various polymer materials that are widely used and compatible with traditional biomolecular assays.
The ALCATs technology exploits a phenomenon called acoustic microstreaming to manipulate fluid flow within a microfluidic environment. Acoustic microstreaming has been previously demonstrated as capable of achieving high degree fluid transport and mixing1,2 using gaseous bubbles as the actuation source. The principle of bubble-induced acoustic microstreaming is based on the fact that bubbles trapped within a liquid phase oscillate when excited by an acoustic energy source. This oscillatory motion of the air-liquid interface leads to a first-order periodic flow within the liquid phase. The magnitude of which is at a maximum at the air/liquid boundary and is given by 
where Schematic of VCAT-based acoustic bubble-array micromixer. The black streamlines denote the second-order acoustic streaming that result from the oscillation of the air/liquid interface of the bubble by an external acoustic source.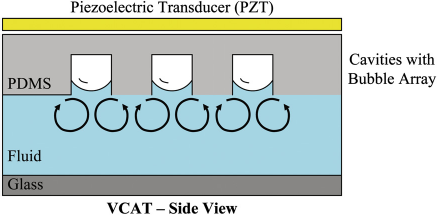
ALCATs are air cavities that form naturally within deadend side channels in hydrophobic devices filled with liquids. The interfacial membrane between the air and fluid created on the bubble surface can be resonated or vibrated with an external acoustic energy source such as a simple piezoelectric transducer (PZT). Depending on its relative position and angle with the liquid chamber/channel, the ALCAT can push away or pull in the surrounding liquid contents and its contained particulates. Two types of ALCATs are described in this report: lateral cavity acoustic transducers (LCATs) and vertical cavity acoustic transducers (VCATs).
ALCAT Fabrication and Experimental Setup
ALCAT-based microfluidic devices are fabricated using standard soft lithography methods. 7 First, SU-8 (MicroChem Corp., Newton, MA) is spun onto a silicon wafer as per manufacturer's instructions to obtain the desired thickness. The wafer is then prebaked at 65 °C and soft baked at 95 °C on a hotplate. It is then exposed through a high-resolution transparency mask using an ultraviolet flood exposure system to cross-link the exposed SU-8. This is followed by a postexposure bake at 65 and 95 °C. The wafer is then developed using an SU-8 Developer until all unexposed SU-8 has been washed off resulting in an SU-8 master mold.
Poly(dimethlysiloxane) (PDMS) (Dow Corning Corp., Midland, MI) is mixed with a 10:1 ratio of PDMS primer with PDMS curing agent. The mixture is then degassed under vacuum for 1 h before being poured onto the SU-8 master mold. The SU-8 mold with uncured PDMS is placed in a 60 °C oven for 4 h to allow the PDMS to cure. The PDMS is then peeled off the SU-8 mold, cut into individual devices, and inlet and outlet holes are punched. A 150-μm thick glass cover slip and
These microfluidic devices are placed on the PZT manifold as shown in Fig. 2. Ultrasound gel is used as a coupling agent between the PZT and the microfluidic device. The PZT is actuated using a function generator (Agilent 33220A) and voltage amplifier (Krohn-Hite 7500) at the appropriate frequency. The PZT manifold and microfluidic device are placed under an upright microscope (Nikon Eclipse L200), and videos are captured using a high-speed camera (Photon Fastcam PCI).
An illustration of the typical experimental setup used.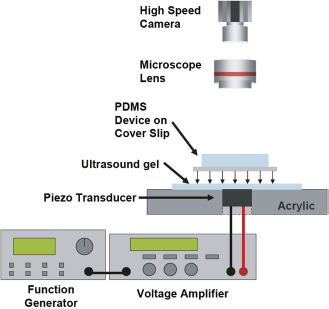
As described above, ALCAT actuators can be easily fabricated. They simply require the appropriate cavity features to be integrated on the same plane as the fluidic channels without additional fabrication steps. Furthermore, the acoustic energy source required to activate the (A) Image of poly(methyl methacrylate) (PMMA) substrate with hot-embossed ALCAT device features. (B) The features of a single LCAT pump device on PMMA. (C) A complete PMMA LCAT pump device fabricated using cellophane tape.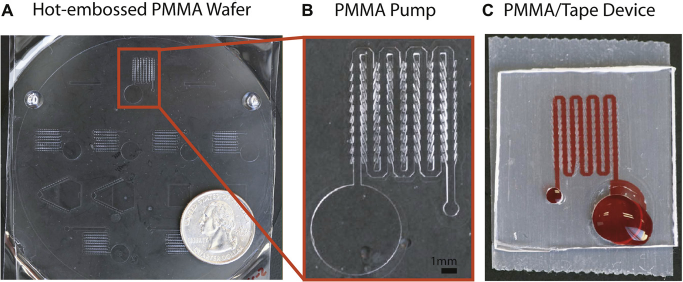
LCAT for Microfluidic Pumping and Recirculation Assay
The LCAT microfluidic platform can enable microfluidic pumps for integrated on-chip molecular assays. One configuration of the micropump is illustrated in Figure 4. A series of LCATs generate sufficiently large pumping pressures to move reagents and samples from chip to chip.
9
Design concept for LCAT for pumping. The image demonstrates how pairs of angled cavities enable the pumping of bulk fluid in the direction indicated when an external acoustic energy source is activated.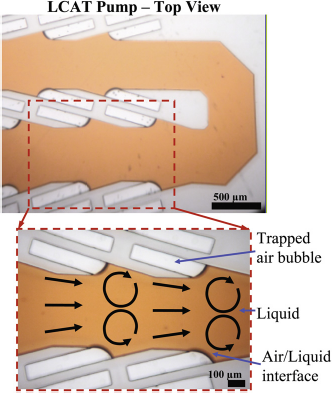
An LCAT pump with 80 pairs of cavities has achieved pumping pressures of ∼ 350 Pa. Using the LCAT micropump, a multistep protein array binding assay has been demonstrated to enable the sequential delivery of reagents and samples for an automated multistep bioassay system.
10
Figure 5A illustrates a colorimetric immunoassay chip implemented through the utilization of the LCAT-based recirculation pump platform. A fixed volume of sample analytes (∼10 μL) is recirculated over the binding sites in a closed-loop microfluidic channel to increase the probability of detection. To determine the effectiveness of the recirculation assay platform,
11
it is compared with a traditional assay, where gold-conjugated antibodies (goat antihuman IgG [ (A) Image of colorimetric immunoassay chip. (B) Comparison of standard and microfluidic 1 optical density gold-conjugated secondary antibody delivery. Images were equally adjusted in contrast to aid in visualization.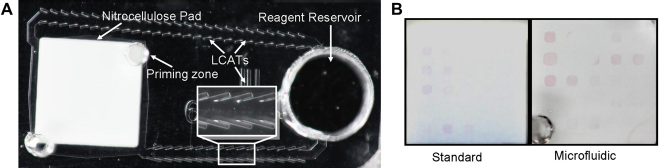
LCAT for Bead or Cell Sorting
The sorting device consists of three inlets that allow the hydro-dynamic focusing of beads within the main channel
12
with the LCAT positioned downstream of the focusing region (Fig. 6). Burst-mode experiments were performed while applying a sinusoidal alternating current signal of 32 (A) Indicates the second-order streaming patterns observed when the LCAT is in the ON state. (B) Stacked images of sequential frames for two different beads showing clearly that an activated LCAT is capable of translating beads in the direction perpendicular to the flow. Frequency - 32 kHz, voltage - 40 Vpp sine wave, flow rate - 5 μL/min, LCAT ON time ∼ 1.6 ms, LCAT OFF time ∼ 4.7 ms.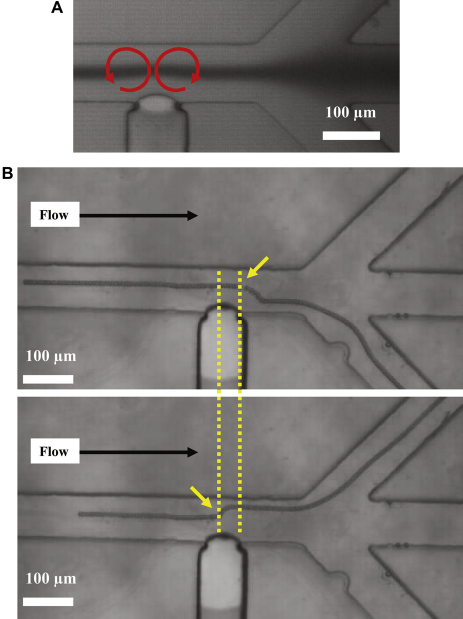
Rapid Protein Binding
Most benchtop array-based biodetection platforms rely on diffusion for biomolecular transport, which can vary detection times to range from hours to days. In the Left: acoustic bubble array mixer fabricated on top of four protein array nitrocellulose pads. Right: The fluorescence intensity signal comparing acoustic microfluidic mixing results with conventional pipetting results.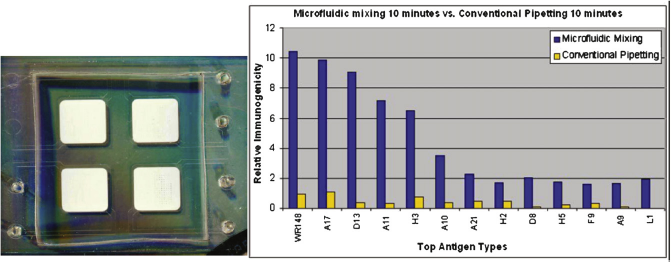
Other ALCAT Microfluidic Devices
We have observed beads recirculating in
Conclusion
In this report, a simple low-cost yet versatile microtechnology platform is demonstrated as capable of performing a variety of microfluidic actuation functions, including pumping, mixing, sorting, as well as sample extraction and purification. This technology termed
Footnotes
Acknowledgments
The authors acknowledge financial support from the DARPA S&T Program (HR0011-06-1-0050) through the Micro/Nano Fluidics Fundamentals Focus (MF3) Center. The authors would also like to acknowledge the Protein Micro-array Laboratory at University of California, Irvine, directed by Dr. Philip L. Felgner for helpful discussions and providing us with the protein spotted nitrocellulose pads for the immunoassay work. The authors would also like to acknowledge Dr. Tianhong Cui and Dr. Tao Zhang from University of Minnesota for providing hot-embossed poly(methyl methacrylate) devices.
