Abstract
The complete blood count (CBC) has by and large remained confined to the traditional laboratory setting since its inception. Used in a variety of diagnostic assessments, the CBC has essentially become limited to clinical laboratories because of reliance on large automated hematology devices. With many potential uses at the point of care and clinical settings, as well as the research laboratory, a portable low-cost hematology analyzer could aid in earlier detection of a wide variety of medical conditions. Using smaller sample volumes, inexpensive polymers, and low power consumption, microfluidic devices present one such route toward miniaturization of the traditional flow cytometer-based hematology analyzers. This review focuses on challenges for development of cost-effective portable analyzers, potential areas for point-of-care clinical usage, current commercial systems with increasing portability, and recent research in improved miniaturization and automation, including developments in acoustic and inertial focusing techniques and novel detection methodologies.
Introduction
The field of hematology, the study and analysis of blood, has proven to be a vital and necessary tool for health care and medical diagnostics. In the clinical setting, a complete blood count (CBC) is recommended annually, often as part of a regular physical examination. In the emergency room (ER) setting, more than 50% of ER patients have at least one laboratory blood test during a typical ER stay. 1 With such a high frequency of use, a simple blood test, particularly the CBC, ranks among the most relied on tests in medical diagnostics. Through quantitation of erythrocyte, leukocyte, and platelet levels, a CBC can be used to detect a wide variety of diseases and medical conditions, which may otherwise be difficult to recognize. table 1 outlines several components of the CBC and lists many of the conditions for which blood counting may be ordered.
Applications of the CBC in Clinical and ER Settings

CBCs are used to diagnose and detect a wide variety of conditions, diseases, and anomalies.
With the advent of the flow cytometer and automated hematology analyzers, automated blood cytometry has risen to become the “gold standard” in the field and for good reason. Before the analyzers of the modern day, blood counting was commonly performed under a microscope by manual enumeration of stained blood components. This laborious task proved tedious and often inaccurate. Built on principles of flow cytometry, automated analyzers soon emerged capable of systematically quantifying the various blood components. Cytometry has yet to be surpassed in terms of reliability and high throughput, with machines capable of sorting and analyzing 10,000–25,000 cells in a given second. 2 Most modern cytometers can even measure several parameters simultaneously, providing comprehensive feedback to the technician. 2 –5
Despite these virtues, modern flow cytometers carry several drawbacks. These expensive and cumbersome machines have dominated the arena of hematology analyzers, while sacrificing portability, cost efficiency, and potential for point-of-care analysis. 2 Flow cytometers require trained laboratory technicians or a strong background in clinical science, making them impractical for the everyday health care practitioner. High blood volume requirements, in addition to costly reagents necessary for sample pretreatment, confer several more disadvantages onto these machines, not to mention the high costs associated with laboratory hematology analyzers. 6
Perhaps the most overlooked flaw in laboratory hematology is the time span associated with testing. Depending on location and availability, the blood sample of interest cannot be tested until it reaches the flow cytometer, thus introducing the possibility of contamination and inaccuracy because of the time delay. With the potential for earlier detection and treatment, rapid, yet accurate, testing has become a necessity for CBCs. The different components of the CBC suggest a myriad of time-critical applications in which precision and efficiency are of utmost concern.
An example illustrating the utility of the CBC in a time-critical situation involves dehydration or blood loss (from hemorrhage or general hypovolemia). In 2009, Ohio nursing facility resident Peter Southard died from dehydration because of a lack of fluids. 7 The culpable nursing facility subsequently paid $6.5 million in damages to Southard's family. The mode of death in this case is easily preventable and is also simple to detect. A CBC assessment of Southard's blood would reveal elevated erythrocyte counts, indicative of his medical condition. 8 As is often the case, an early blood count can lead to earlier detection and treatment.
The goal of this review is to discuss current developments in the field of hematology analyzers and to argue for the miniaturization of conventional flow cytometers to the microfluidic level. By operating at the microfluidic level, such blood analyzers would prove advantageous over traditional flow cytometers because of enhanced portability and ease of use. With applications ranging from emergency prehospital care to patient monitoring in hospitals and nursing facilities, microfluidic flow cytometry may soon emerge as the next “gold standard” in clinical hematology.
Mechanism of Operation: Conventional Flow Cytometers
Pretreatment Procedure
Before cytometry, a blood sample is typically pretreated with a wide array of reagents specific to the flow cytometer and the quantification being performed. A reagent universal to any cytometer is one to prevent coagulation of blood; this is typically performed during venipuncture using an anticoagulant, such as ethylenediaminetetraacetic acid (EDTA), heparin, or citrate. Other reagents can function to add cell markers (antigen—antibody coupling with fluorescent tags) to specific particles within the blood. Still other reagents are often necessary, primarily to ensure proper maintenance of the flow cytometer. 6
A representative example of an automated hematology analyzer is the Coulter AcT 5diff Hematology Analyzer produced by Beckman Coulter (Brea, CA). Capable of performing a CBC with five-part white blood cell differential (WBC-diff), the AcT 5diff incorporates five reagents to pre-treat its samples as follows. 9 To begin, the sample is divided into several chambers, where each portion is mixed with a diluent reagent (Act 5diff Diluent). In general, diluents primarily consist of a normal saline solution (0.9%) to simply dilute the blood to more manageable concentrations. 10 After dilution, each chamber is treated separately, depending on the parameter desired. To prepare a sample for an erythrocyte count (red blood cell [RBC]), the sample is heavily diluted with the diluent. The erythrocytes can then be quantified on the basis of impedance measurements (Coulter principle). 11 To prepare for hemoglobin measurements, another chamber is treated with a second reagent (Act 5diff Hgb Lyse), which functions to lyse erythrocytes. 9 The lysing reagent invariably contains a cyanide compound, which on binding with hemoglobin produces the optically active cyanmethemoglobin. 10 By using spectrophotometry, the concentration of the cyanmethemoglobin, and therefore the hemoglobin level, can be determined.
Often, reagents play multiple roles in hematology analyzers. In the case of the AcT 5diff analyzer, one particular reagent (Act 5diff Fix) is used to quantify monocytes, eosinophils, and neutrophils. This reagent is applied to a separate blood-containing chamber and acts to stain these particular leukocytes with Chorazole Black E stain. 9 In addition, this staining reagent induces lysis of erythrocytes, leaving tagged leukocytes capable of being distinguished from platelets using optical methods. Another reagent plays a similar role in distinguishing basophils from other leukocytes in the AcT 5diff analyzer. Applied to yet another chamber, this reagent (Act 5diff WBC Lyse) tags basophils for enumeration and also induces erythrocyte lysis. 9 The final reagent (Act 5diff Rinse) simply acts as a rinsing agent to maintain function of the device. In general, the hematology analyzer sequentially delivers precise amounts of each reagent to the sample in question in a process known as sequential dilution. 9 This prepares the blood sample by dividing it into different chambers, diluting each sample, and adding specific reagents based on the parameters desired.
Hydrodynamic Focusing
After the pretreatment steps, the sample in each chamber is injected into the actual analysis region of the device. As illustrated in Figure 1, the flow cytometer essentially consists of an inner glass capillary coaxial to a surrounding sheath. Each sample individually proceeds through an inner capillary while a focusing reagent flows within the outer sheath. In this way, the sheath fluid flow confines the sample and aligns each particle in a process known as hydrodynamic focusing. At the sheath—sample interface, each medium flows with a low enough Reynolds number; as a result, laminar flow prevails and mixing does not occur. By adjusting the sheath fluid flow, hydrodynamic focusing serves to create a narrow stream of particles in single file. This allows for serial analysis of each individual cell by ensuring that each cell proceeds down the same path through a specific detection region and that cells do not travel in aggregates. 12,13
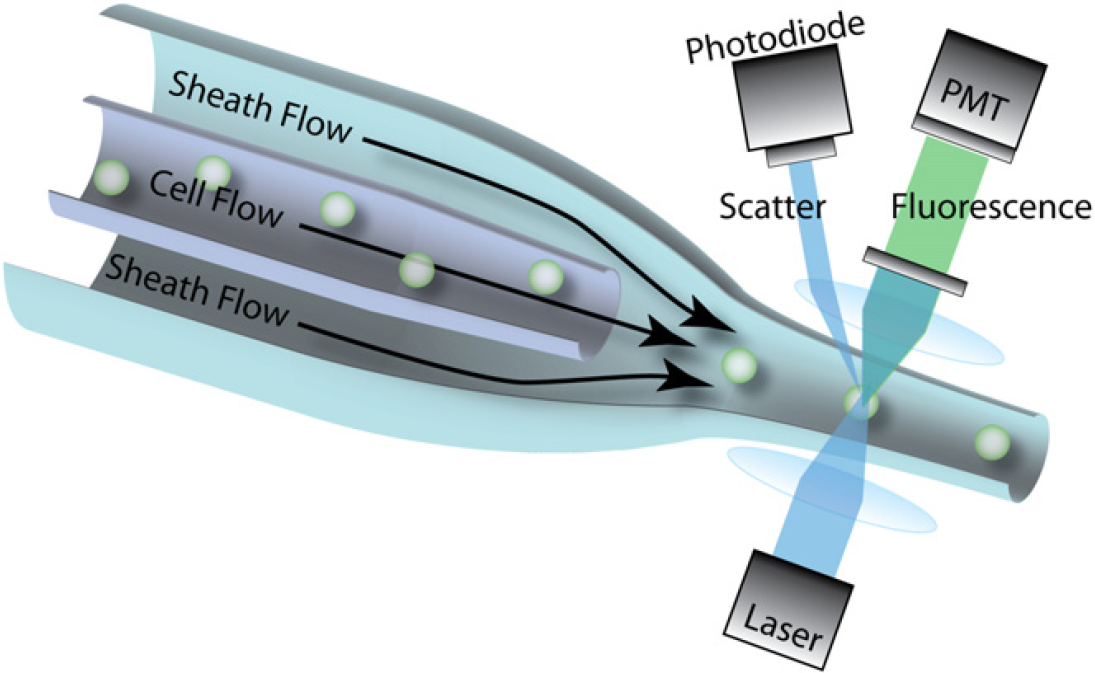
Mechanism of operation for a typical flow cytometer. The sample passes through an inner glass capillary, surrounded by sheath fluid flow. The sheath fluid runs parallel to the sample in the open space between the coaxial capillaries. When the sample particles emerge from the capillary, the sheath fluid acts to confine the particles, resulting in single-file ordering. The particles pass through a focused laser or LED light source, resulting in measurable fluorescence emissions and light scattering. PMT = photomultiplier tube.
Enumeration Using Optical Methods
After focusing, modern hematology analyzers can automatically enumerate the various components of the blood sample. The mechanisms used for enumeration can be broadly categorized as either being optics based or impedance based. Optical methods usually proceed by directing a beam of light (from laser or light-emitting diode [LED] source) at the stream of particles. Flow focusing secures the cells within a narrow stream, so each cell passes under the focal spot for examination. Using the principles of light scattering, fluorescence, and absorbance, cytometers can predict the sizes and shapes of the particles within the stream. Light incident on a particle can result in any combination of forward scattering, large angle scattering, and fluorescence, all of which can be detected by the cytometer using optical instruments. 6 These instruments include high-gain photodiodes or photomultiplier tubes (PMT), which function on the principles of the photoelectric effect and secondary emission to convert electromagnetic radiation into a measurable current or voltage. 14
Enumeration Using Impedance-Based Methods
Impedance-based methods usually invoke the Coulter principle for direct current (DC) electrical conductance measurements. The sample is directed through a small aperture with two electrodes positioned on either side of the focused stream. As the stream passes between the two electrodes, a current can be conducted through the stream because of the electrolyte nature of the diluent and blood plasma. 10 As cells pass in sequence through the current path, each cell produces a pulse of increased impedance that is directly proportional to the size of the cell. 10,11 By modifying the alternating current (AC) frequency for the impedance measurement, information concerning internal cell structure and dielectric properties can also be obtained. 15 Detection of these pulses provides a means for distinguishing between different particles based on slight differences in size or dielectric properties. However, the Coulter principle presents several limitations; when two different cells have similar sizes and dielectric properties, as is the case for various leukocytes, differentiating between the two cells can prove difficult. For this reason, impedance-based methods are often combined with optical methods, which can be more adept at differentiating between cells based on specific cell markers.
Both optical and impedimetric methods rely on the processing of electromagnetic signals to report pertinent information regarding the size, shape, and biomolecular composition of any particle in the sample. Sample processing can occur at a rate anywhere between 10,000–50,000 cells s−1, depending on the parameters and accuracy desired. table 2 compares three commonly used hematology analyzers (flow cytometers), varying in complexity, cost, and output. Depending on the various parameters tested, each analyzer requires its own particular set of reagents. In all three, the Coulter principle plays a significant role in enumeration. 3 –5
Side-by-side comparison of three common hematology analyzers used in clinical and research laboratories

Note: Unless otherwise noted: data for Sysmex XE-2100 from ref 3; data for Medonic CA-620 from ref 4; and data for Beckman Coulter LH750 from ref 5.
Estimates based on online marketplace resale data.
Data only found from 2002.
Reagents required for sample analysis only.
Challenges to Achieving Miniaturization
To achieve miniaturization of the traditional hematology analyzer and ultimately portability and point-of-care testing, several obstacles must be overcome. A smaller device suggests dealing with highly reduced volumes of blood; at the point-of-care level, blood samples can potentially come from a finger stick, with a typical volume of 50–100 $mUL. Although this eliminates the need for any real phlebotomy or venipuncture, the miniaturized hematology analyzer must be capable of mixing the sample with anticoagulant and diluting the small sample to a more manageable volume. Portability necessarily implies a highly reduced footprint and size, as well as minimized power consumption. From a practical standpoint, a handheld device capable of performing a CBC would need to weigh less than 5 kg and be capable of operating on a small ideally rechargeable power source, such as a 9-V battery. In contrast, the Sysmex XE-2100 (Sysmex Corporation, Kobe, Japan), a traditional laboratory-confined hematology analyzer, weighs more than 100 kg with a power consumption of 770 VA. 3 Clearly, this device would be ill-suited to run on a power source as small as a 9-V battery (capacity of 625 mA-h) and is best suited for an AC outlet in the laboratory setting.
In addition, although a low-cost portable device with disposable components would be ideal, the tradeoffs between cost and efficiency must also be considered. In theory, the Coulter principle appears to be more cost effective compared with the more expensive optical methods, yet the Coulter principle can provide information solely regarding particle size, velocity, and dielectric properties. 6 As a result, discrimination between cells of similar size and composition can be difficult without precise detection systems. Optical techniques also suffer from several limitations, including the need for cell tagging during the pretreatment steps as well as large and expensive optical components that can be difficult to miniaturize. 6 As a result, most large-scale analyzers rely on both optical and impedimetric methods, a combination that is difficult to replicate in a miniaturized and more crowded device. If optical methods are the desired means for enumeration, costly lasers can be replaced with less expensive LEDs as the source of excitation. However, excitation by laser has proven more efficient than LED excitation because of the collimated nature and superior power of laser light. 18,19 These characteristics allow for more rapid cytometry in devices using laser excitation. Clearly, a tradeoff exists between fabricating a device cost effectively versus ensuring reliability and accuracy.
Advantages of Using Microfluidics in Hematology
The miniaturization of the traditional flow cytometer to the microfluidic level presents exciting future possibilities for chip-size hematology analyzers. By nature, microfluidic devices offer a substantially reduced footprint and size over conventional devices while using smaller sample volumes. In addition, microfluidic devices allow for simple cost-effective fabrication, which lends such devices to disposability and ease of use. In these ways, microfluidics can help overcome many of the obstacles to miniaturization of traditional hematology analyzers.
Smaller Sample Volumes
Capable of using hydrodynamic focusing to streams on the scale of 10 $mUm, 2 microfluidic flow cytometers require smaller sample volumes and fewer reagents than their bulky counterparts. 6 This comes as a direct consequence of the smaller device dimensions, which necessarily imply reduced “dead” volume and smaller sample volumes. This in turn translates to less blood required from a patient; in this way, the discomforts associated with long blood draws by phlebotomists can be replaced with simple finger-stick methods to extract small volumes of blood. These minute quantities of blood will still require sample pretreatment to add cell markers or antibodies to specific components within the blood. Although pretreatment reagents still appears to be necessary and unavoidable, 6 the smaller sample sizes suggest a proportionately reduced quantity of the expensive reagents. In theory, this would result in a considerable cut in consumable costs and reduced blood counting costs.
Cost Effective and Disposable
In addition, the thermoplastics (such as poly(methyl methacrylate), polystyrene, and polycarbonate) commonly used in microfluidic chips are cheap alternatives to the costly glass capillaries implemented in traditional flow cytometers. 2 By taking advantage of this disparity, the proposed microfluidic cytometers enjoy better cost efficiency without necessarily compromising quality. As noted by Chung and Kim (2007), the fabrication of microfluidic devices can be performed on a mass production scale, allowing for not only lower production costs but also quality control to ensure reliability. 6 The implementation of low-cost mass production also opens the door for a disposable one-time use cytometer. For years, this concept has directed much of the research toward microfluidic flow cytometers, manifesting in models for disposable cartridges and even fully disposable devices. 6 14 20 21 Regardless, the prospect of disposability would eliminate the constant need for expensive maintenance reagents between uses and any possibility of sample contamination from previous runs.
Minimized Footprint and Power Consumption
A microfluidic cytometer would also benefit from a substantially reduced footprint and decreased power requirements, allowing for greater portability. In contrast to a large laboratory flow cytometer, a microfluidic chip boasts a highly reduced size and mass, making for a much smaller footprint. In addition, various microfluidic manipulations can be used that lend themselves directly to passive sample flows within microfluidic chips. One such manipulation involves taking advantage of fluidic surface tension to create passive flow through microchannels. 22 –24 As explored by Beebe, Meyvantsson, and Berthier (2002, 2006, 2007), passive flows driven by surface tension can eliminate the need for onboard sample pumps, thus reducing power consumption and onboard electronics. 22 –24
Multiple studies headed by Thorslund et al. 25 –27 have also demonstrated the viability of microfluidic devices operating without any electrical basis for sample pumping. This protocol involves the addition of a heparin coat to the channels of a polydimethylsiloxane (PDMS) device, which confers hydrophilicity onto its channel surfaces; the hydrophilicity, in turn, allows for channel filling to occur solely on the basis of capillarity without the need for any external pumping mechanisms. 25 28 In this way, the power consumption of a hypothetical microfluidic device can be reduced further still.
Point-of-Care Hematology: Areas of Potential Use
A blood counting device similar in size and portability to a glucose monitor can have major implications in numerous health care fields, especially at the point-of-care level. To meet such an end, the device would need to be sufficiently easy to use to ensure that health care personnel without thorough laboratory training could operate the device consistently. Following the Clinical Laboratory Improvement Amendment (CLIA) of 1988, only certain CLIA-waived devices, such as a blood glucose monitor, can be used by personnel without laboratory training. A hypothetical microfluidic flow cytometer, comparable in simplicity to a blood glucose instrument, could potentially acquire a CLIA-waiver for use in CBC testing by nonlaboratory personnel. Such a device could have far-reaching applications in a myriad of settings.
Emergency Prehospital Care
With such a wide variety of applications and uses, a CBC performed in the prehospital (point-of-care) setting could reveal valuable medical information, which may otherwise remain undetected until the patient reaches a hospital. Trained emergency medical services (EMS) personnel could carry such devices on an ambulance, allowing for on-scene blood counting and more accurate initial impressions; this could not occur with traditional hematology cytometers, as the large and expensive machinery is essentially confined to the laboratory. In addition, a simple on-scene blood count could be a deciding factor for whether patients receive transport to an ER or which services are rendered by EMS personnel.
A variety of medical conditions are detectable via blood count, including anemia, sepsis, and dehydration, which may be difficult to detect otherwise. In suspected cases of anemia, low erythrocyte (low hematocrit) and high reticulocyte counts can quickly confirm suspicions and allow health care personnel to begin the appropriate treatment. 29 In the opposite scenario, polycythemic patients would have abnormally high hematocrit levels because of overproduction of erythrocytes. 30 As a result of the high erythrocyte density, an abnormally high blood viscosity develops, in turn causing a decrease in blood flow as dictated by Poiseuille's law. In the prehospital setting, such a patient benefits from fluid replacement therapy, in the form of normal saline or dextran delivered intravenously to reduce the blood viscosity. However, symptomatically, such a patient would present the signs and symptoms of heart failure, ultimately triggering a conceivably harmful and alternate form of treatment to strengthen the heart. Yet another example is the suspected septic patient, whose blood analysis would reveal abnormally high leukocyte counts. With information from a blood test in hand, EMS personnel can proceed accordingly in treating such a patient. 31 Elevated leukocyte levels can help differentiate septic shock from other varieties of circulatory shock, in which cases, patients often exhibit similar vital signs but markedly different blood compositions. Although specific in nature, each preceding example illustrates the importance and potential use of rapid on-scene blood counting in the prehospital setting.
ER Usage
On a similar note, a microfluidic point-of-care hematology analyzer can play a significant role in time-sensitive hospital ERs. On admission to an ER, patient vital signs are often the first pieces of information collected, along with medical history, but blood work is typically not requested until later stages in the patient's stay. Incorporated alongside a patient's vital signs, a rapid and reliable blood count (obtained from a finger-stick sample) on admission to the emergency department could provide considerable information that may otherwise go unnoticed until a CBC is requested. One study by Bacus et al. 32 evaluated the CBC as a tool to predict the need for an ER stay in patients with undifferentiated abdominal pain (UAP). Not surprisingly, abnormal blood counts, in particular high leukocyte and neutrophil counts, were reliable in predicting whether a patient with UAP would require ER admission. Thus, a point-of-care hematology analyzer capable of providing a prompt CBC on entrance to an ER could serve a prognostic purpose in detecting medical conditions and predicting the need for a costly ER stay.
Partial Blood Counts in Out-of-Hospital Settings
At the very least, a typical CBC reports values for leukocytes, erythrocytes (or alternatively, hemoglobin levels or hematocrit), and thrombocytes. In contrast and for the sake of simplicity, a hypothetical “partial” blood count could focus solely on one component of whole blood. One such device already in use is the glucose meter, which has played a significant role in blood sugar monitoring in diabetic patients. In a similar manner as the glucose meter, a specialized “partial” blood counter could focus solely on leukocytes or erythrocytes, depending on the device and patient needs. In a patient recovering from sepsis, the wide range of parameters reported by complex blood analyzers may not be of primary concern; instead, a simple hematology analyzer capable of performing a straightforward WBC count would be much more practical. In the same way, an anemic patient may only be interested in daily variations in the RBC count and not other parameters. For these reasons, a partial blood analyzer capable of rapid point-of-care analysis could have great utility, particularly in settings geared toward patient recovery. Such a setting can range from a nursing (convalescent) home to a medical clinic to even a personal residence, granted that the device is sufficiently easy to use. A “partial blood count” analyzer would not only be less complex and easier to produce but also cheaper and more cost effective; the device capabilities would be directly pertinent to the patient's needs and extraneous parameters would be omitted from the analysis.
Recent Developments in Microfluidic Flow Cytometry
In general, the field of microfluidics spans many areas of research, with microflow cytometry representing but a small subset of the field. Recent developments in microflow cytometry, particularly those geared toward applications to hematology, have addressed several issues, including focusing methodologies, means of particle detection, channel size, and minimization of onboard electronics.
Novel Focusing Methods
By and large, current research on microflow cytometry centers around two major aspects of miniaturization. The first aspect involves new focusing methods. As pointed out by Chung and Kim, 6 confining blood components within a region of the z-axis in a microfluidic device is difficult because of the fact that microfluidic devices are fabricated from two-dimensional structures. Unlike the miniaturization of other cytometer components from normal scale to microfluidic dimensions, the transition for hydrodynamic focusing has been markedly less smooth. As a result, much research has focused on two- and three-dimensional focusing techniques. Chang et al. 33 have proposed one such method for three-dimensional focusing using two-layer microchannels constructed from PDMS; yet, such a system would require four inlets and considerable geometric complexity, which could present difficulties in fabrication. An alternative design, as conceived by Mao et al., 34 improves on the issue of complexity by using a single-layered PDMS device while still producing a hydrodynamically focused stream in three dimensions. However, such a device still retains four inlets, in addition to a 90° curvilinear segment that functions in vertical focusing.
Despite major progress in the miniaturization of hydrodynamic focusing, much research focuses on alternative approaches to hydrodynamic focusing in efforts to eliminate waste and consumable costs associated with sheath fluid. One such approach to focusing involves the use of surface acoustic waves (SAWs). Demonstrated by Shi et al., 35 this approach uses SAWs generated by two parallel interdigital transducers on either side of a PDMS channel. Constructive interference between the two acoustic waves creates a standing SAW (SSAW) with characteristic pressure nodes and antinodes. By precisely positioning the channel to contain only a single pressure node, focusing can be achieved along the pressure node because of lateral forces imparted by the acoustic radiation pressure. 34 An obvious advantage of SSAW focusing is that such a technique eliminates the need for a sheath fluid and thus reduces waste. On the other hand, the interdigital transducers require an AC power source and expensive piezoelectric substrates, such as lithium niobate, on which to operate. Similar methodologies have been investigated by Goddard et al., 36,37 particularly the viability of ultrasonic acoustic waves in a circumferential arrangement around a cylindrical capillary, and Nilsson et al. 38 and Evander et al. 39 with major emphasis placed on acoustics-based separation and isolation of particles from a sample. An alternative to hydrodynamic and acoustic focusing techniques is electrokinetics, which uses DC or AC to guide particles along a single path. 13,40 However, these techniques generally suffer from highly diminished throughputs compared with hydrodynamic and acoustic methods.
Microfluidic inertial focusing presents yet another alternative to those presented above. As a sheathless focusing method, inertial focusing relies on fluid inertia acting on suspended particles in specially shaped channels, allowing for both focusing to precise positions within the channel and particle sorting. 41 –43 Work done by Gossett and Di Carlo (2009) 42 has elucidated the dependence of focusing on the particle and channel dimensions and fluid velocity, as well as the presumptive role of inertial lift and drag forces. In addition, collaborative work by Di Carlo et al. (2008) 43 has confirmed the feasibility of inertial focusing in isolating platelets from whole blood, a feature pertinent to the design of microfluidic-based hematology devices. The extension of such a principle to leukocytes and erythrocytes could produce a straightforward means for separating and isolating the various cellular components of blood. Preliminary steps toward using inertial focusing for practical flow cytometers have been taken by Bhagat et al. (2009) 44 and Hur et al. (2010). 45 Using a microfluidic device with 256 parallel channels, Hur et al. 45 achieved sampling rates about a million cells per second without sacrificing detection sensitivity of RBCs and WBCs. Overall, inertial focusing is advantageous in that it requires only a single fluidic input and can easily be parallelized to allow for higher throughput measurements. On the other hand, relatively precise control over the channel flow rate is required to maintain consistent focusing.
Novel Detection Methods
The second major focus of current research on microflow cytometry is new detection methods. As outlined earlier, the primary techniques used in detection and enumeration involve either optical methods, including fluorescence flow cytometry and impedance-based methods. In two related studies, Morgan et al. (2006) 13 and Holmes et al. (2006) 46 demonstrated the viability of miniaturized microflow cytometers using both optical and impedimetric methods to quantify and discriminate between fluorescently loaded latex beads. The beads varied in fluorescence intensity (on a relative scale) and diameter (from 2 to 5.5 μm), which approximated the erythrocytes and thrombocytes found in blood. Notably, Holmes et al. 47 later repeated this procedure on human blood samples to perform a WBC-diff count. Blood samples were treated with saponin and formic acid, which served as erythrocyte lysing agents, followed by successful cytometry, based solely on impedimetric methods that were confirmed with optical methods. Holmes et al. 47 could differentiate between dissimilar latex beads, and, critically, different leukocyte subtypes, without cell-specific labels.
Research on novel detection methods has also centered around new sources of excitation in optical cytometers. Large portions of the costs associated with conventional flow cytometers are because of the high quality of the lasers used, with costs easily surpassing thousands of dollars. One study by Habbersett et al. 18 demonstrated that a simple $160 diode-pumped solid-state (DPSS) laser pointer operating at 532 nm could potentially replace the more expensive conventional lasers in traditional flow cytometry without sacrificing accuracy. However, such lasers require slower sample flow (“transit time”) through focal spots because of the modest excitation power of these less expensive lasers. Blue diode lasers can provide another potentially inexpensive source of excitation if configured properly and are at the proper wavelength to excite common dyes. Such lasers, with powers up to 100 mW, are commonly found in Blu-ray DVD players, which have been mass produced and sold for only a few hundred dollars. As a low-cost alternative to lasers of comparable power commonly found in laboratory flow cytometers, such lasers may play large roles in the future of miniaturized flow cytometry.
Early Miniaturization Attempts
Although much research and study is still dedicated to the development of the microflow cytometer, several early attempts toward miniaturization have proven successful. To test the viability and accuracy of two flow cytometers based on microfluidic principles, Simonnet and Groisman 14 used fluorescent beads varying in fluorescence and size (0.75–2.5 μm). The devices were inexpensively cast using ultraviolet-lithography-machined molds and PDMS, rendering them disposable. Using the beads as sample particles, an argon ion laser as an excitation source and photomultiplier tubes to detect and capture the emitted fluorescence, Simonnet and Groisman 14 demonstrated the ability of the microfluidic devices to enumerate fluorescent beads with low coefficients of variation. Such a device may also prove viable in the field of blood counting, as the average erythrocyte measures 7.8 μm and thrombocytes measure at 2–3 μm. 30,48
Effect of Channel Dimensions on Cell Sorting
The sizes of the various cellular components of blood are vital determinants in the design and fabrication of hematology microfluidic cytometers. With large discrepancies in size between platelets (2–3 μm) and macrophages (21 μm), 49 constructing appropriately sized channel walls can be a nontrivial matter; channel walls that are too small will lead to clogging, whereas larger walls may allow smaller particles to move in groups, defeating the purpose of focusing. 19 One solution may involve cell sorting based on size before cytometry and enumeration. Toner and Irimia 50 outline several mechanisms for cell sorting in lab-on-a-chip microfluidic devices, based on mechanical, electrical, magnetic, and even optical principles. Various noninertial separation techniques, particularly dielectrophoretic, optical, magnetic, and acoustic force fields, have also been reviewed by Tsutsui and Ho. 51 After cell sorting, uniformly sized populations of cells can undergo cytometry through channels that are just the right size for the particular cell type. This concept of multiple channels and valves has received much attention from research groups worldwide, most notably from Thorsen et al. (2002) 52 and Unger et al. (2000) 53 such research demonstrates the viability of microfluidic chips containing a complex network of channels, each capable of maintaining a unique internal environment because of large number of valves. Such a feature is key in hematology, which essentially involves isolating different blood components to perform quantitation. With the ability to house different valves, channels, and chambers on one device, microfluidics presents an ideal medium for performing blood counting whenever cell sorting is necessary.
The Current Frontier
Among the leaders toward disposable microflow hematology for point-of-care use is the Chempaq (Farum, Denmark) 54 series of blood analyzers, scheduled for release in early 2010. Designed by Chempaq specifically for complete blood counting, this new line of hematology devices appears to offer considerable improvements over traditional laboratory flow cytometers. Measuring just over a square foot in footprint, the Chempaq XDM701 is listed as weighing six pounds; this certainly allows for portability, although not to the extent of highly portable pocket-sized glucose meters. This device operates via small disposable cartridges, into which a drop of blood (from finger stick) is deposited. Designed for single use, the cartridges are based on microfluidic design, containing several chambers for dilution, separation, and enumeration of blood components. The blood-loaded cartridge is inserted into the analyzer, which displays the results of the CBC within 3 min. table 3 compares the Chempaq XDM701 to the Sysmex pocH-100i, a similar device manufactured by Sysmex (Kobe, Japan).
Comparison of Chempaq XDM701 to the Sysmex pocH-1001
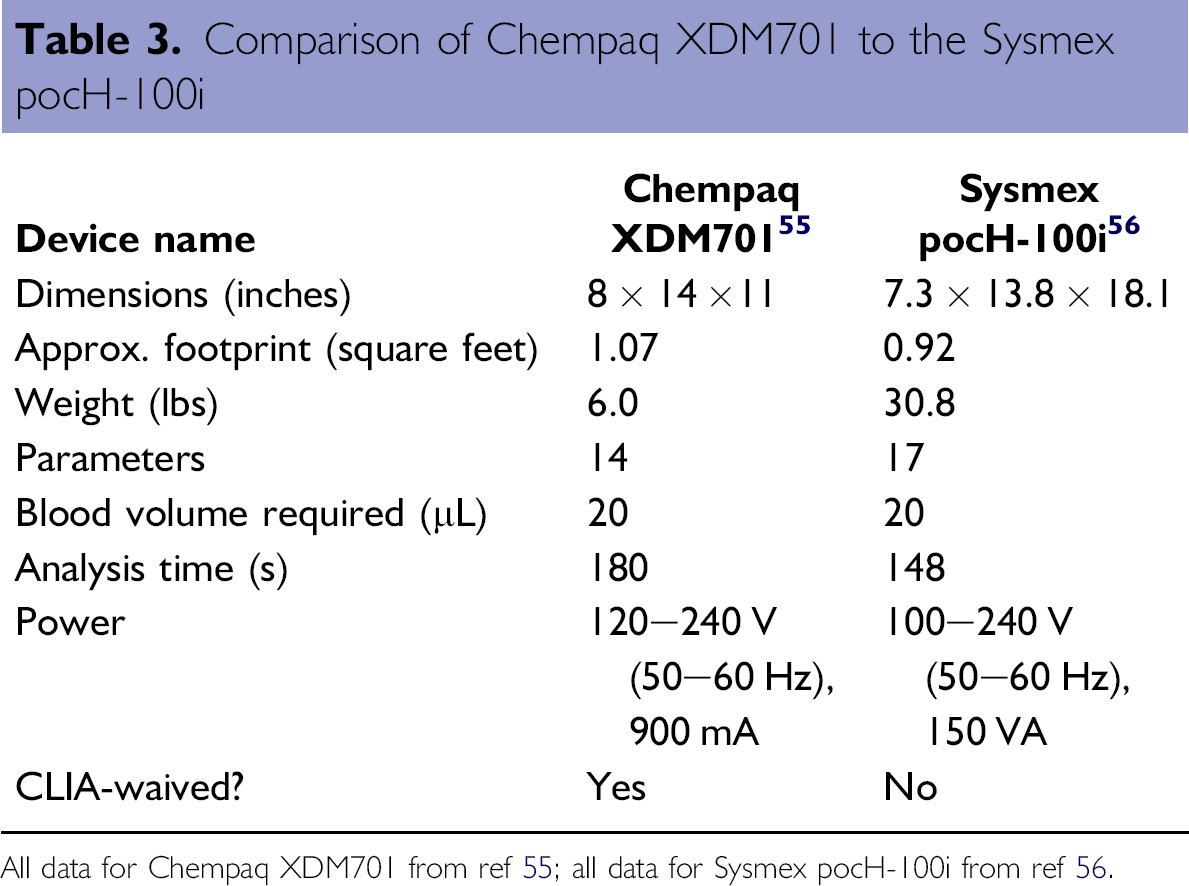
All data for Chempaq XDM701 from ref 55 alldata for Sysmex pocH-100ifrom ref 56.
Although further research is still necessary to assess the efficacy and accuracy of the Chempaq XDM701, one early study, conducted by Rao et al. 56 , demonstrated the precision and reliability of the Chempaq XBC when compared with a traditional flow cytometer. In this side-by-side study comparing blood counts performed by the Chempaq XBC and the Beckman Coulter LH750, the Chempaq XBC performed favorably, with variations less than 7% for leukocytes and less than 5% for hemoglobin. These correlate strongly with manufacturer-reported imprecision values of 4% for leukocytes and 3% for hemoglobin. 54 Based on early evidence, the Chempaq line of blood analyzers appear to fare well in terms of precision and reliability against their laboratory-confined precursors.
Despite the breakthroughs in miniaturization, disposability, and portability, arguably the greatest breakthrough for the Chempaq XDM701 comes in the form of a CLIA-waiver; as a testament to the device's ease of operation, the CLIA-waiver allows for device usage by nonlaboratory personnel, a unique feat in the field of hematology. This achievement opens the door for many future hematology analyzers and the potential for widespread implementation of blood counting in various clinical and nonclinical settings.
Conclusions
As outlined by Ateya et al. 19 , the ideal cytometer should be small, easy to fabricate, and automated. In addition, it should be simple to operate such that primary care physicians, EMS personnel, and nonlaboratory personnel may partake in its use. As the basis for many lab-on-a-chip diagnostic instruments, microfluidics presents one such path toward miniaturization of the conventional flow cytometer to the microscale level. By working on minute sample volumes, a microfluidic device would necessarily require proportionately smaller reagent quantities, thus optimizing cost efficiency. The ideal hematology analyzer would be comparable in size and complexity to a typical blood glucose meter. At a small fraction of the cost of a laboratory flow cytometer, such a blood counter could find its way into homes, hospitals, ambulances, providing rapid yet reliable CBC testing. Unlike current hematology analyzers, a microfluidic analyzer epitomizes the idea of portability, accessibility, and point-of-care testing. Through further research and development of microflow cytometers, hematology may soon emerge from its niche as a laboratory science and expand to become a more widespread tool in medical diagnostics.
Acknowledgment
The authors acknowledge support from the National Science Foundation, Grant # 930501.
Competing Interests Statement: The authors certify that all financial and material support for this research and work are clearly identified in the manuscript.
