Abstract
Important hematologic changes can be observed in nonhuman primates with malaria, including inaccurate reticulocyte counts by the ADVIA 2120 hematology analyzer. A 5-year-old male purpose-bred cynomolgus macaque (Macaca fascicularis) imported from a commercial source in Cambodia was enrolled in a nonclinical toxicity study investigating the effects of an immunomodulatory pharmaceutical agent. On study day 22, an increase in large unstained cells (LUCs), due to increased monocytes (2.20 × 103/µl, reference interval: 0.17-0.76 × 103/µl), was reported by the analyzer during a scheduled hematologic evaluation, which prompted blood smear review and revealed that the macaque had a high burden of Plasmodium spp.. The macaque did not have clinical signs for the infection at this time point. Progressively higher parasite burdens and persistently increased monocytes (markedly increased by study day 56, 10.38 × 103/µl) were observed at subsequent hematologic evaluations. New Methylene Blue stain manual reticulocyte counts were performed on study day 43 and at later time points, and showed that the analyzer reported erroneous higher reticulocyte counts (study day 43: +6.7%, +266.2 × 109/L; study day 50: +18.9%, +409.8 × 109/L) compared with the manual reticulocyte counts (pseudoreticulocytosis). The magnitude of regenerative response was considered inadequate for the severity of anemia at these time points. Atypical reticulocyte scatter plot distributions from the analyzer were also observed at time points with high parasite burdens, and combined with increased LUCs, may suggest high burden parasitemia. Verification of automated reticulocyte counts is important in cases with high malarial parasite burdens and the recognition of pseudoreticulocytosis is prudent in assessing appropriateness of the regenerative response. Increases in monocytes correlated with higher parasite burdens and marked increases may be an indicator of advanced disease.
Introduction
Malaria, the Anopheles spp. mosquito-borne disease caused by Plasmodium spp. protozoal infection, is a critical public health problem worldwide. It is a leading cause of morbidity and mortality in many tropical or subtropical developing countries with those who are immunocompromised at higher risk of complications or severe disease.1,2 The individual’s immune status plays a large role in the outcome of infection, and despite a risk of clinical disease with competent immune function, chronic asymptomatic infections are commonly described in humans with competent immune function and low parasite burdens.3,4 When infection does produce clinical malarial disease, the most common manifestations are fever and hemolytic disease, which can be rapid and fatal.1,3,4
Nonhuman primates (NHPs) residing in tropical and subtropical geographic regions are also susceptible to Plasmodium spp. infection (simian malaria). While the prevalence of simian Plasmodium spp. varies by region, the most commonly reported species in wild or captive cynomolgus macaques (Macaca fascicularis) of Southeast Asia have been P. inui, P. cynomolgi, P. fieldi, P. knowlesi, and/or P. coatneyi.5-7 The majority of documented cases in NHPs have been asymptomatic.5-7 Both P. cynomolgi and P. knowlesi have been implicated in human infection as well, with the vast majority of cases also being asymptomatic and only few progressing to clinical malarial disease.8,9
Given the common East and/or Southeast Asian sourcing of NHPs for nonclinical toxicology studies, malaria is one of the most frequent parasitic diseases of NHPs imported from endemic regions.10-12 According to the World Health Organization in 2020, Cambodia accounted for the most cases of human malaria (58%) in the Greater Mekong Subregion, which also includes China (Yunnan province), Lao People’s Democratic Republic, Myanmar, Thailand, and Vietnam. 1 While the prevalence of malaria in wild or captive NHPs in Cambodia has not been established, it is possible that there could be similar representation of NHP malaria cases in the same region. However, simian Plasmodium spp. infection is still considered generally very low in research NHPs, with a prevalence (detection by real-time polymerase chain reaction and independent of clinical status) within Charles River Laboratories ranging from approximately 0.2% to 2.8% 13 in NHPs sourced from Asia. Nonetheless, infection may create challenges in the toxicology setting given its potential to confound study findings and negatively affect the health of the animal.
Although the hematologic effects of malaria have been well described in humans, effects within NHPs have been less well characterized. The present report describes important hematologic changes that may occur in cases of malaria in NHPs, with particular emphasis on pseudoreticulocytosis as reported by a flow cytometry hematology analyzer with fluorescence measurement capabilities.
Case Presentation
An approximately 5-year-old male purpose-bred cynomolgus macaque (M. fascicularis) imported from Cambodia was acquired by the testing facility (Charles River Laboratories, Reno, Nevada) from a commercial source. Prior to importation to the testing facility, the subject macaque had undergone infectious disease screening for tuberculosis (skin test) and viral infections (using serological and/or molecular testing) inclusive of herpes B, simian retrovirus, simian immunodeficiency virus, and simian T-lymphotropic virus and had received vaccination and serological testing for measles and hepatitis A and anthelmintic treatment within 1.5 to 3 months of departure from the commercial source. All screening tests were negative or unremarkable. Upon arrival to the testing facility (6 weeks prior to first dosing) and prior to study initiation, the subject macaque was deemed to be healthy via unremarkable clinical observations, physical examinations, complete blood count (CBC), serum biochemistry, coagulation, and urinalysis measurements. The testing facility was accredited by the Association for Assessment and Accreditation of Laboratory Animal Care and was in compliance with applicable guidelines of the US Department of Agriculture. The study protocol was approved by the testing facility Institutional Animal Care and Use Committee prior to study start. The subject macaque was housed with other cynomolgus macaques designated for a single toxicity study, and was acclimated to the housing and study handling procedures for a minimum of 4 weeks prior to initiation of dosing. All macaques were fed Certified Primate Diet (No. 5048, PMI, Richmond, Michigan) daily in amounts appropriate for the age and size of the animals. Tap water was treated by reverse osmosis and ultraviolet irradiation and provided ad libitum to animals via an automatic watering system. Animals were also provided with psychological enrichment opportunities prior to placement on study and for the duration of the study including treats, fresh fruit, cereal, toys, and social interaction. The study animals were group housed with up to 3 animals of the same sex and dosing group in climate-controlled rooms at 64°F to 84°F with a relative humidity of 30% to 70%. The rooms had greater than 10 air changes per hour with 100% fresh air (no air recirculation) and a photoperiod cycle of 12 hours light/12 hours dark.
The nonclinical study to which the subject macaque was assigned was designed to investigate the pharmacodynamics and any possible adverse effects after dosing with an immunomodulatory large molecule pharmaceutical agent intended to treat lymphoproliferative disease or neoplasia. In previous studies, the pharmaceutical agent resulted in decreased leukocytes and increased cytokines. Administration of the pharmaceutical agent was via intravenous infusion once every three weeks for a total of five doses at a dose of either 0 mg/kg (control vehicle), 3 mg/kg, or 30 mg/kg. The cynomolgus macaque described in this report was assigned to the 30 mg/kg (high dose) group. Blood was scheduled to be collected for serial CBC measurements on the ADVIA 2120 hematology analyzer (Siemens Medical Solutions Inc., Malvern, Pennsylvania) at two time points (fourteen and five days) prior to initiation of dosing (on study day 1) and on study days 2, 8, 22, 29, 43, 50, 64, 71, 85, and 92. In addition, serum clinical chemistry parameters were scheduled to be measured on the Cobas 6000 (Roche Diagnostics, Indianapolis, Indiana) and plasma fibrinogen, prothrombin time, and activated partial thromboplastin time were scheduled to be measured on the STA Compact (Diagnostic Stago Inc., Mount Olive, New Jersey) five days prior to initiation of dosing and on study days 43, 85, and 92.
When analyzer flags or CBC abnormalities were detected, CBCs were performed in duplicate to confirm analyzer results. When reported large unstained cells (LUCs) were ≥5%, blood smear review with manual differential cell counts was performed per laboratory-derived hematology protocol. When reticulocyte counts were ≥10% per the ADVIA 2120 analyzer, manual reticulocyte enumeration was performed on New Methylene Blue (ENG Scientific LLC, New Brunswick, New Jersey) stained blood smears using a Miller’s disk according to previously established methods. 14 If Plasmodium spp. organisms were detected during the course of a blood smear review triggered by laboratory criteria, then the number of organisms were graded using a laboratory-derived semiquantitative grading scheme and a 100× objective, as minimal (1+, present in at least 3 fields) to marked (4+, present in >10 fields). A value of “rare” was reported if organisms were seen in numbers lower than the “minimal” (1+) grading threshold. In addition, parasite burdens were estimated from each blood smear evaluation from day 22 samples onward by counting the number of parasitized red blood cells (RBCs) seen in 5000 RBCs and expressed as a percentage, a modification of a previously documented method. 12 Estimated parasite burden and manual reticulocyte counts were enumerated by the same clinical pathologist in duplicate with the mean value reported (see Tables 1 and 2). In addition, reticulocyte scatter plots generated by the ADVIA 2120 were examined at all hematology sample time points.
Serial CBC and hematology data for the 5-year-old male Cambodian subject macaque with Plasmodium spp. infection.
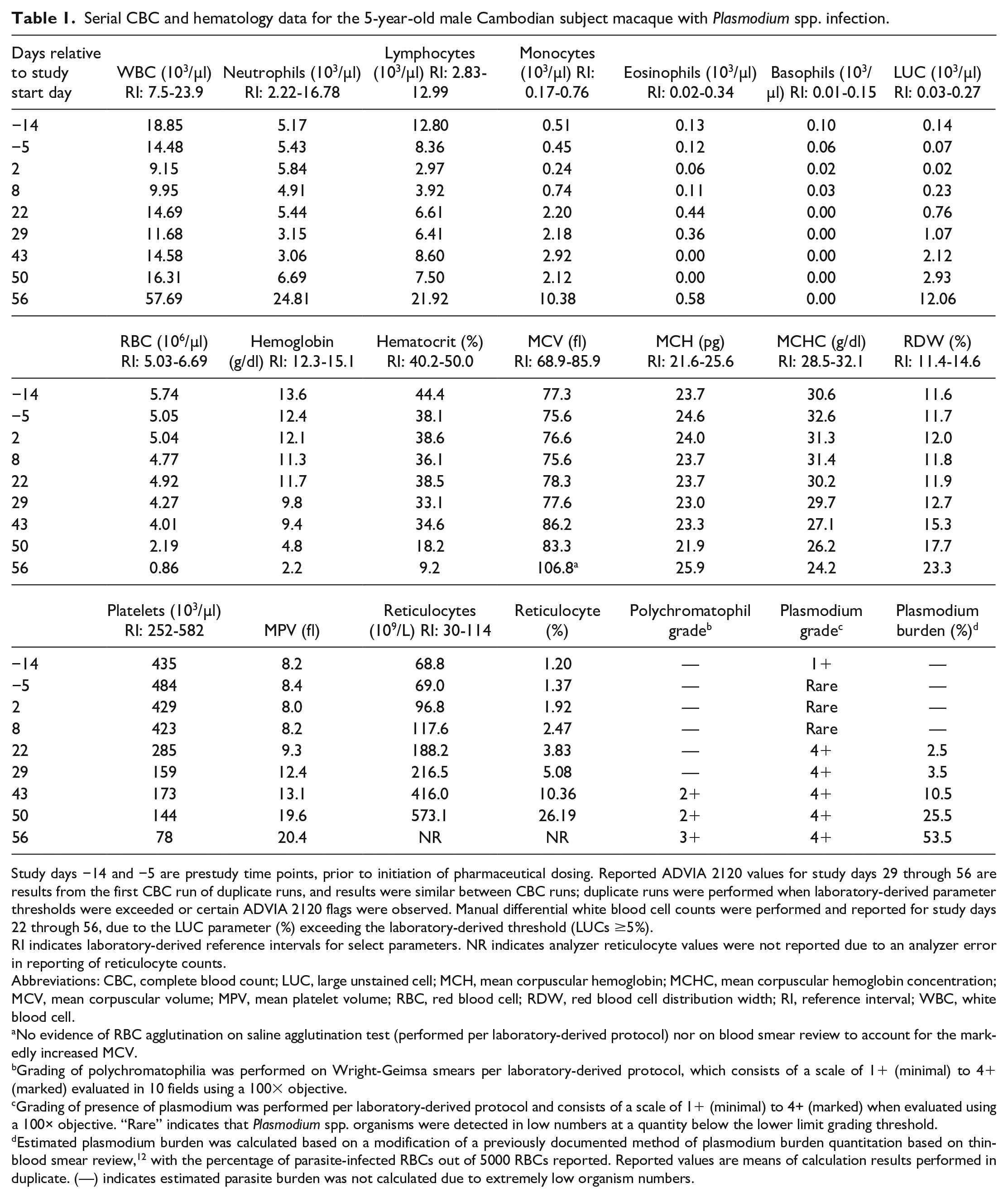
Study days −14 and −5 are prestudy time points, prior to initiation of pharmaceutical dosing. Reported ADVIA 2120 values for study days 29 through 56 are results from the first CBC run of duplicate runs, and results were similar between CBC runs; duplicate runs were performed when laboratory-derived parameter thresholds were exceeded or certain ADVIA 2120 flags were observed. Manual differential white blood cell counts were performed and reported for study days 22 through 56, due to the LUC parameter (%) exceeding the laboratory-derived threshold (LUCs ≥5%).
RI indicates laboratory-derived reference intervals for select parameters. NR indicates analyzer reticulocyte values were not reported due to an analyzer error in reporting of reticulocyte counts.
Abbreviations: CBC, complete blood count; LUC, large unstained cell; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; MPV, mean platelet volume; RBC, red blood cell; RDW, red blood cell distribution width; RI, reference interval; WBC, white blood cell.
No evidence of RBC agglutination on saline agglutination test (performed per laboratory-derived protocol) nor on blood smear review to account for the markedly increased MCV.
Grading of polychromatophilia was performed on Wright-Geimsa smears per laboratory-derived protocol, which consists of a scale of 1+ (minimal) to 4+ (marked) evaluated in 10 fields using a 100× objective.
Grading of presence of plasmodium was performed per laboratory-derived protocol and consists of a scale of 1+ (minimal) to 4+ (marked) when evaluated using a 100× objective. “Rare” indicates that Plasmodium spp. organisms were detected in low numbers at a quantity below the lower limit grading threshold.
Estimated plasmodium burden was calculated based on a modification of a previously documented method of plasmodium burden quantitation based on thin-blood smear review, 12 with the percentage of parasite-infected RBCs out of 5000 RBCs reported. Reported values are means of calculation results performed in duplicate. (—) indicates estimated parasite burden was not calculated due to extremely low organism numbers.
Comparison of mean ADVIA 2120 automated reticulocyte counts (% and absolute counts) with mean manual reticulocyte counts via New Methylene Blue smear evaluation at time points with high parasite burden, for the 5-year-old male subject macaque.

At very high reticulocyte counts with very low RBC mass on study day 56, the analyzer was unable to gate or identify reticulocytes and inaccurately reported 0.00% reticulocytes on the initial and duplicate run. Therefore, NR was assigned to the ADVIA RETIC (%). (—) indicates values were not calculated due to the lack of a valid analyzer result for reticulocytes.
Abbreviations: NMB, New Methylene Blue; NR, not reported; RBC, red blood cell; RETIC, reticulocyte count.
ADVIA 2120 analyzer reticulocyte counts, manual reticulocyte counts, and ADVIA 2120 analyzer RBC counts are reported as average counts from duplicate measurements.
ΔRETIC indicates the difference between ADVIA 2120 analyzer and manual reticulocyte counts (% units or absolute cell count units). The overestimation of analyzer counts compared with manual counts was greater at higher manual reticulocyte counts and higher estimated parasite burdens.
Absolute values of manual reticulocyte count were calculated by multiplying the mean manual reticulocyte count percent by the mean RBC count.
Grading of polychromatophilia was performed on Wright-Geimsa smears per laboratory-derived protocol, which consists of a scale of 1+ (minimal) to 4+ (marked) evaluated in 10 fields using a 100× objective.
Estimated parasite burden (%) was calculated as the percentage of parasite-infected RBCs on thin-blood smear review per a previously documented method, 12 with the modification of assessment in a total of 5000 RBCs. Reported results are mean values of calculations performed in duplicate.
On study day 22 (three weeks after the first dose), the hematology analyzer reported an increase in LUCs >5% for the subject macaque. This prompted a blood smear review, which revealed increased monocytes as the cause of increased LUCs, as well as a high burden of Plasmodium spp. organisms (4+ [marked], estimated parasite burden of 2.5%) (see Table 1). All life stages of the organism were present within RBCs, including gametocytes (micro or macrogametocytes), trophozoites, and schizonts (see Figure 1). The subject macaque did not have any clinical signs of disease at this time point. Retrospective blood smear review from prior time points (pretreatment through study day 8) demonstrated the presence of Plasmodium spp. organisms in very low numbers (reported as either rare or graded as 1+ [minimal]).
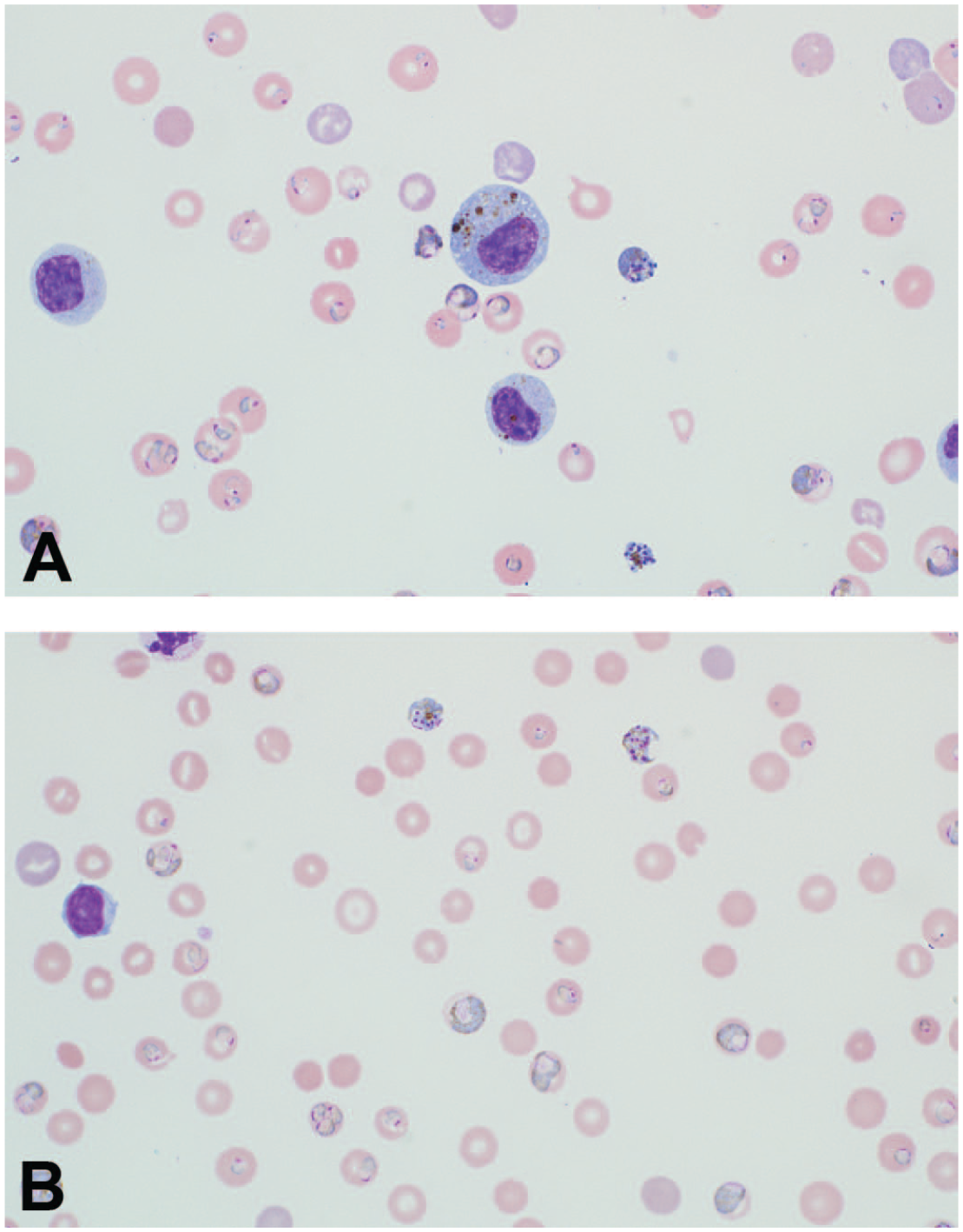
Blood smears from the 5-year-old male subject macaque with a high burden of malarial organisms. (A) Study day 56 and (B) study day 50: high burdens of Plasmodium spp. (within RBCs) are seen and all life stages are present. Some RBCs contain multiple organisms and/or different life stages. (B) Monocytes contain fine to coarse tan or golden-brown malaria pigment granules (hemozoin), which may occur in cases with high parasite burdens. Morphologic findings are similar to those observed on blood smear review from study days 22 to 43. Wright-Geimsa stain, 100× objective. RBC indicates red blood cell.
On subsequent CBC evaluations, the analyzer reported progressive increases in LUCs attributable to progressive increases in monocytes (see Table 1) and decreasing red cell mass parameters (RBC count, hemoglobin [HGB], hematocrit [HCT]) Persistent high parasite grades (4+ [marked]) were observed on all subsequent blood smears, with progressively increasing estimated parasite burdens on study days 29, 43, 50, and 56 (see Table 1).
Progressive increases in analyzer reticulocyte count accompanied the progressive marked anemia which was consistent with regenerative hemolytic anemia due to clinical malarial disease (see Table 1). On study days 43 and 50, the ADVIA 2120 analyzer reported a ≥10% reticulocyte count, and manual reticulocyte counts were performed as previously described. Reticulocytes were distinguishable from organism-infected RBCs (Figure 2); the organisms were present only in mature RBCs, did not take up stain, and often had small golden-brown pigment granules (hemozoin). The ADVIA 2120 analyzer reported reticulocyte counts in excess of those determined via manual reticulocyte counts (see Table 2) on study days 43 and 50, which were beyond an acceptable difference between analyzer and manual counts (see Supplemental Table, regarding laboratory-derived reticulocyte confidence limits), and the discrepancy in reticulocyte counts was greater with higher parasite burdens (see Table 2). ADVIA 2120 reticulocyte scatter plots also showed atypically scattered events in the medium and high fluorescence regions of the reticulocyte scatter plot at time points corresponding with increasingly higher estimated parasite burden (study days 22-50; Figure 3). However, it became difficult to visualize the abnormal scatter at study days 43 and 50, which could be explained by the concurrent increases in estimated parasite burden (percentage of RBCs infected), which may have been misclassified as reticulocytes, and increases in actual reticulocyte count (percent, see Table 2).
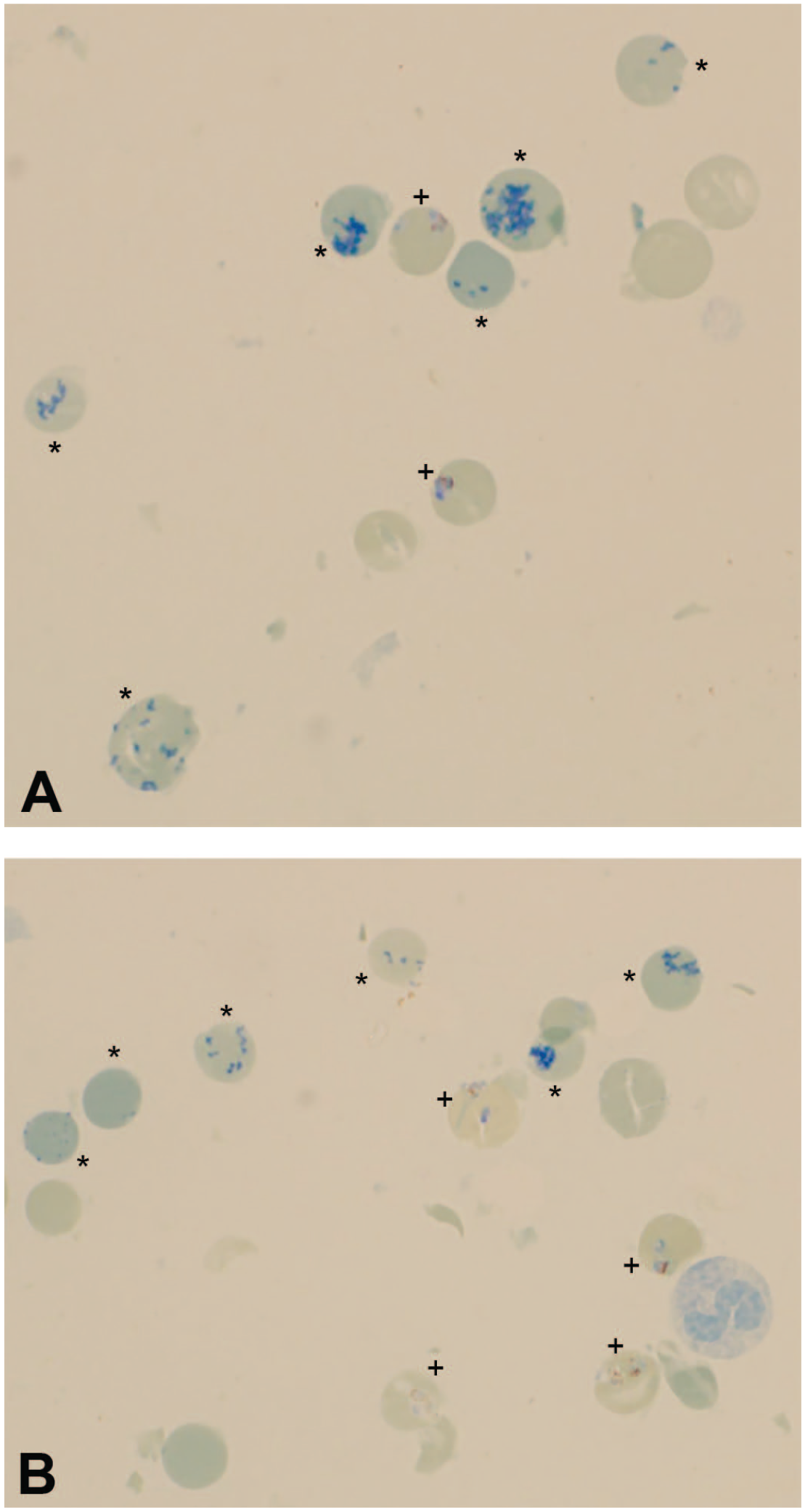
New Methylene Blue stained blood smear in the 5-year-old male subject macaque with a high burden of Plasmodium spp. (A) Study day 56 and (B) study day 50: Reticulocytes are distinguishable from mature RBCs infected with organisms (reticulocytes [*] and mature RBC with intracellular Plasmodium spp. organism [+]). Malaria pigment (hemozoin) is often seen in organisms as tiny golden-brown granules. New Methylene Blue stain, 100× objective. RBC indicates red blood cell.
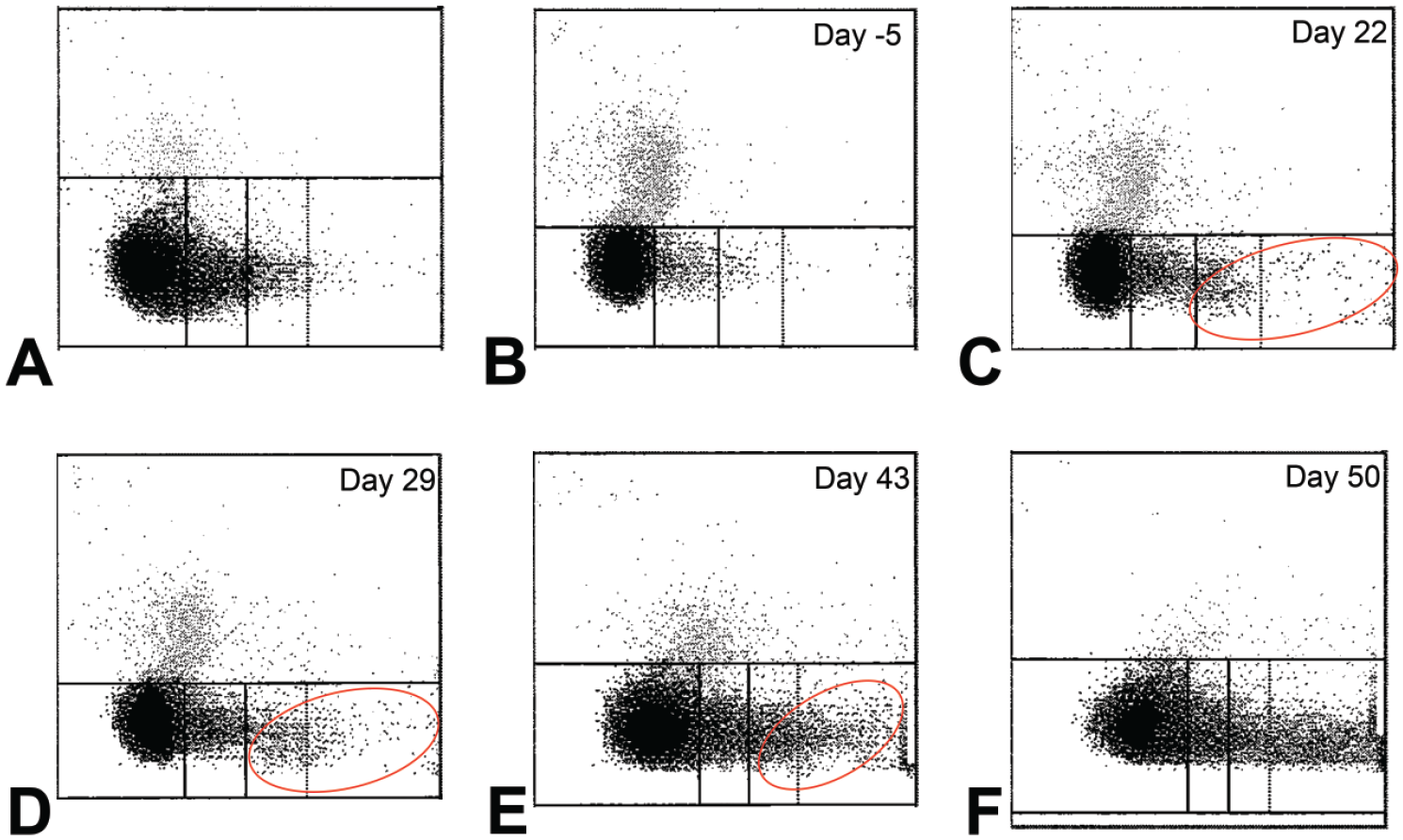
ADVIA 2120 analyzer reticulocyte scatter plots in cynomolgus macaques. (A) A macaque with regenerative anemia without malaria (HGB 9.8 g/dl, HCT 33.0%, reticulocytes 408.2 × 109/L and 10.72%). During regenerative anemia, notice the expected tapering comet tail of events from the right (low fluorescence region) to the left (high fluorescence region), with reticulocyte events densely concentrated in the low and/or intermediate fluorescence regions. (B)-(F) 5-year-old male subject macaque described in this report. At time points with duplicate CBC runs on the ADVIA 2120 (days 29-50), the reticulocyte scatter plot, erythrocyte, and reticulocyte parameter results were reported from the first CBC run and results were similar between runs. (B) Low burden of malarial organisms (rare on blood smear review) 5 days prior to initiation of pharmaceutical dosing (HGB 12.4 g/dl, HCT 38.1%, reticulocytes 69.0 × 109/L and 1.37%). (C) High burden of malarial organisms (2.5% of RBCs) on study day 22 (HGB 11.7 g/dl, HCT 38.5%, reticulocytes 188.2 × 109/L and 3.83%). (D) High burden of malarial organisms (3.5% of RBCs) on study day 29 (HGB 9.8 g/dl, HCT 33.1%, reticulocytes 216.5 × 109/L and 5.08%). (E) High burden of malarial organisms (10.5% of RBCs) on study day 43 (HGB 9.4 g/dl, HCT 34.6%, reticulocytes 416.0 × 109/L and 10.36%). (F) High burden of malarial organisms (25.5% of RBCs) on study day 50 (HGB 4.8 g/dl, HCT 18.2%, reticulocytes 573.1 × 109/L and 26.19%); the atypical scatter cloud is not readily visible which was attributed to concurrent increases in true (i.e., manual) reticulocyte counts and estimated parasite burden.
The subject macaque developed markedly decreased RBC mass by study day 50 (see Table 1) with associated clinical signs of skin pallor and hunched posture, and a veterinary observation and treatment plan was instituted that included daily detailed observations and supplemental food stuffs. By study day 56, an unscheduled blood collection was taken due to clinical deterioration that included progressive weakness, pallor, and weight loss. At this time, serum biochemistry showed mild hyperbilirubinemia (0.60 mg/dl, reference interval [RI]: 0.1-0.4 mg/dl), moderate increases in alanine aminotransferase (644 U/L, RI: 22-80 U/L) and aspartate aminotransferase (566 U/L, RI: 25-125 U/L), and moderate increases in urea nitrogen (90 mg/dl, RI: 16-31 mg/dl) and creatinine (2.4 mg/dl, RI: 0.6-1.4 mg/dl). The decreased RBC mass had progressed to severe (see Table 1), and the highest estimated parasite burden (53.5% of RBCs parasitized) occurred at this time point. In addition, the mean corpuscular volume (MCV) was markedly increased (see Table 1), though there was no evidence of RBC agglutination neither on saline agglutination test (performed per laboratory-derived protocol) nor on blood smear review to account for this marked increase. On study day 56, ADVIA 2120 reticulocyte counts were not reported due to an inability of the analyzer to apply gating to the reticulocyte channel in the presence of severe decreased RBC mass and it was suspected that the analyzer’s upper limit of detection (24.9% reticulocyte count) had been exceeded. Instead, a value of 0.00% reticulocytes was assigned by the analyzer on duplicate runs, despite increased polychromasia observed on blood smear review (3+ [moderate], 11%-20% of RBCs evaluated in 10 fields at 100× objective) and an increased manual reticulocyte count of 15.2% (see Table 2). Thus, comparisons between ADVIA 2120 analyzer reticulocyte count and manual reticulocyte count could not be made. On study day 56, the macaque also had markedly increased monocytes, moderately increased lymphocytes, and mildly increased neutrophils with mild-to-moderate toxic change (presence of cytoplasmic basophilia and Döhle bodies), which were consistent with inflammation and immunogenic stimulation (see Table 1). Platelets were also moderately decreased (see Table 1).
Due to the rapid decline in clinical condition associated with malarial disease, the macaque was humanely euthanized on study day 56. Prior to euthanasia, the macaque had received a total of three doses of the pharmaceutical agent under study (on study says 1, 22, and 43).
Discussion
Plasmodium spp. infection can have important impacts on hematologic parameters during asymptomatic or symptomatic infection, with the most notable effects observed during the latter scenario.
Chronic asymptomatic infection has been commonly described in humans with low parasite burdens, while higher parasite burdens are typically reported during exacerbation of clinical disease, which most often occurs in those who are immunosuppressed or stressed.1,2,4 As illustrated in this report, Plasmodium spp. infection was present asymptomatically at a low grade at the time of import of the subject macaque from Cambodia, with the development of high parasite burdens and clinical malarial disease following initiation of the study. The present case shows (1) unexpected detection of infection on blood smear review during verification of analyzer flags or after exceeding hematology parameter alert thresholds (e.g., increased LUCs and/or monocytes) and (2) progression to severe clinical malaria after receiving doses of a pharmaceutical agent with immunomodulatory properties and/or after possible physiologic stress during the study period. Due to the presence of organisms prior to the initiation of dosing, the infection was deemed pre-existing, although either immunomodulatory properties of the drug or stress associated with the study (resulting in immunosuppression) may have caused or contributed to the progression of the parasitemia and clinical outcome.
Erroneous increases in reticulocyte counts, or “pseudoreticulocytosis,” have been reported by hematology analyzers using laser flow cytometry technology with fluorescence measurement capabilities in cases of malaria in humans with high parasite burdens.15-20 Analyzers previously demonstrating this finding include the Cell-Dyn CD4000 and Sysmex XE series analyzers.15-20 Similarly, the ADVIA 2120 hematology analyzer uses laser flow cytometry with fluorescence technology for the identification and quantitation of reticulocytes using Oxazine 750, which stains nucleic acids. As has been proposed in other studies,15-20 the mechanism for the pseudoreticulocytosis in the present case is thought to be due to the uptake of the fluorescent reticulocyte dye by the malarial organism’s nucleic acids resulting in misclassification and inaccurate enumeration of organisms as reticulocytes. Aside from Plasmodium spp. organisms, other possible causes of automated reticulocyte count interference (e.g., increases in nucleated RBCs, RBC basophilic stippling, Heinz bodies, Pappenheimer bodies, Howell-Jolly bodies, cytoplasmic fragments) were not present in this case based on blood smear review and/or associated ADVIA 2120 data output.
Pseudoreticulocytosis in dogs with large Babesia spp. protozoal infection has also been documented when using laser flow cytometry analyzers with fluorescence capabilities,21,22 with the findings attributed to reticulocyte stain uptake by the organism. Similar to the current case, abnormal reticulocyte scatter plots have been described in other studies with high burdens of malarial organisms in humans15-19 or in dogs with large Babesia spp. infection.21,22 In the present case, atypical reticulocyte scatter plots were evident on study days 22 to 43 which coincided with high organism burdens.
To the authors’ knowledge, the existing primary literature has not described pseudoreticulocytosis in NHPs with malarial disease nor when performing CBCs using the ADVIA 2120 hematology analyzer. Recognition of pseudoreticulocytosis is clinically important to assess adequacy of the regenerative response to malaria-induced decreases in RBC mass. In the present case, while there was evidence of some regeneration as the RBC mass progressively decreased, the manual reticulocyte counts verified that the magnitude of reticulocyte increases reported by the ADVIA 2120 analyzer was not only inaccurate, but considered inadequate when examining absolute reticulocyte counts on study days 43, 50, and 56 (Table 2) given the duration and severity of decreased RBC mass, and based on the clinicopathologic expertise of the authors and an internal laboratory-derived RI for automated absolute reticulocyte count in male Cambodian cynomolgus macaques (RI: 30-114 × 109/L).
The mechanism of anemia in malarial disease is multifactorial with a significant contribution of inadequate erythropoiesis or erythropoietic suppression documented in severe or acute cases.23,24 Classically, the anemia of malarial disease has been described as arising from hemolysis induced by parasite replication as well as increased RBC membrane fragility of parasite-infected RBCs due to alteration of cytoskeletal and membrane proteins.23-25 In addition, increased removal of both infected and noninfected RBCs by macrophages occurs via increased expression of outer RBC membrane phosphatidyl serine and subsequent IgG-mediated RBC removal.23,24,26 However, both in vitro and in vivo studies in humans, NHPs, and mice have demonstrated an important contributing component of inappropriate responsiveness to erythropoietin and erythropoietic suppression during Plasmodium spp. infection.23,24,27-29 An association exists between blood and/or bone marrow hemozoin (i.e., malarial pigment) concentrations in cases with high parasite burden and decreased proliferation of erythroid precursor cells in the bone marrow.23,30 Hemozoin, a byproduct of the parasite’s metabolism of hemoglobin, has been correlated with parasite burden and is thought to cause both direct damage to erythroid precursor cells via stimulation of apoptosis 30 and secondary oxidative damage via release of 4-HNE (a lipid peroxidation product) from macrophages or monocytes that have accumulated the pigment.23,24,27,30 In addition, the accumulation of hemozoin decreases the responsiveness of erythroid precursor cells to erythropoietin within the bone marrow and stimulates the production of erythropoietic suppressive and inflammatory cytokines by macrophages/monocytes.23,27,30 In vivo and in vitro studies in humans have documented decreased bone marrow erythroid proliferation and inadequate reticulocytosis during severe malarial disease, followed by an appropriate reticulocyte response once parasite burdens decrease or once infection resolves.23,30,31
As demonstrated in the present case, the erroneous increases in reticulocyte counts associated with high parasite burdens could be misleading in cases of clinical malaria with concurrent inadequate erythropoiesis. However, given the subject macaque was allocated to the high dose group within the toxicity trial, a component of a pharmaceutical effect on erythropoiesis could not be entirely excluded in the present case.
The markedly increased MCV on day 56 correlated with both the highest parasite burden and the highest manual reticulocyte percent in the subject macaque. Therefore, and in the absence of evidence for RBC agglutination, the markedly increased MCV may have been due to a combination of increased reticulocytes (percent) and RBC enlargement due to cytoskeletal alterations in mature parasite-infected RBCs that may have resulted in increased cell volume. 25
On study day 56, when severely decreased RBC mass and clinical disease were present, leukocyte changes were consistent with inflammation and markedly increased monocytes (Table 1). Inflammation associated with severe malaria is well described, and monocytes/macrophages play a significant role in both the mitigation and progression of clinical disease.23,32-34 While monocytes are imperative to the immune response against the infection via beneficial cytokine production and phagocytosis or removal of parasite-infected RBCs, they may also play a large role in the pathogenesis of advanced disease. The latter is thought to occur due to excessive pro-inflammatory cytokine production that can be damaging to various tissues.32-34 In addition, the magnitude of increased monocytes in blood has been proposed to reflect disease severity with some studies documenting increases in monocytes in advanced cases of malaria.32,34 However, this is considered somewhat controversial as other studies have reported decreases in monocytes in severe malarial disease.35,36 Interestingly, in the present case increased monocytes was a notable change that coincided with time points of high parasite burdens (study days 22-56), and the magnitude of increase correlated with more severe clinical disease. As such, increases in monocytes could be a hematologic marker of high burden Plasmodium spp. infection and an indicator of advanced malarial disease when monocyte increases are marked. However, since few other macaques in the high dose group had mild to moderate increases in monocytes at various time points in the nonclinical trial, a contributing component of pharmaceutical-related effect on monocytes could not be entirely excluded in the subject macaque.
Moderate thrombocytopenia was also observed on study day 56, after a steady decline beginning at the initiation of the study. Multifactorial thrombocytopenia has been associated with clinical and asymptomatic Plasmodium spp. infection, with as many as 89% of malaria cases having some degree of thrombocytopenia.37,38 Potential mechanisms include inflammatory consumption, splenic sequestration, and/or increased destruction or early clearance. 37 Platelet counts may have prognostic value with the magnitude of thrombocytopenia correlating with disease severity.37,38 In the present case, platelet decreases of similar magnitude were observed in the remaining study animals (without plasmodium infection), and thus, this change was considered likely to be an effect of the pharmaceutical agent and considering the subject macaque was allocated to the high dose group.
As illustrated in this report, Plasmodium spp. infection in NHPs used in nonclinical toxicity trials has the potential to negatively impact the health of the animal and confound interpretations of various important hematologic end points. With sourcing of NHPs for nonclinical trials occurring from malaria endemic regions, pathologists and scientists should be mindful of possible challenges imposed by pre-existing malarial infection on data quality and accurate interpretations. In addition, proactive screening for Plasmodium spp. infection (e.g. polymerase chain reaction testing) prior to import of animals from commercial sources in malaria endemic regions could be considered to mitigate these potential challenges.
In conclusion, Plasmodium spp. infection with high parasite burden may cause erroneously high reticulocyte counts (pseudoreticulocytosis) when blood is evaluated by hematology analyzers with fluorescence measurement capabilities. To the authors’ knowledge, the present case is the first documentation of such findings in an NHP using the ADVIA 2120 hematology analyzer. These findings are corroborated by similar reports in the human and veterinary literature. Recognition of erroneously increased reticulocytes or pseudoreticulocytosis is important to assess the adequacy of the regenerative response to decreases in RBC mass, especially given the possibility for infection-associated inadequate erythropoiesis in advanced malarial disease. Careful examination of reticulocyte scatter plots may assist in initial screening of whether pseudoreticulocytosis is present. Regardless, the possibility for pseudoreticulocytosis in cases with high parasite burden should prompt verification of automated reticulocyte counts by performing manual reticulocyte counts. In addition, increased LUCs and monocytes may serve as a hematologic indicator of high-burden Plasmodium spp. infection and marked increases in monocytes may indicate advanced disease.
Supplemental Material
sj-docx-1-tpx-10.1177_01926233221083217 – Supplemental material for Pseudoreticulocytosis by the ADVIA 2120 Hematology Analyzer and Other Hematologic Changes in a Cynomolgus Macaque (Macaca fascicularis) With Malaria
Supplemental material, sj-docx-1-tpx-10.1177_01926233221083217 for Pseudoreticulocytosis by the ADVIA 2120 Hematology Analyzer and Other Hematologic Changes in a Cynomolgus Macaque (Macaca fascicularis) With Malaria by Diya Sharma, Heather Priest and Angela Wilcox in Toxicologic Pathology
Footnotes
Acknowledgements
The authors wish to thank John Trousdale, Catherine Brandoff, and the clinical pathology medical technologist staff at Charles River Laboratories (Reno NV site) for their assistance with data acquisition and reporting of results for this case. The authors also thank Darrin McCarthy for his assistance with image formatting.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material is available in the online version of the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
