Abstract
Superhydrophilic surfaces are investigated for their potential to provide antifogging and antifouling properties for microfluidic devices. Two types of exemplary superhydrophilic surfaces are prepared, including polyester films treated by oxygen plasma and indium tin oxide-coated glasses treated by an electrochemical method. The superhydrophilicity of the treated surfaces presented herein is confirmed by their near-zero water contact angles. Their corresponding antifogging and antifouling capability is examined. The fluorescence microscopic study has confirmed the significantly reduced adhesion of the fluorescein and fluorescent proteins after the surfaces are treated to be superhydrophilic, indicating their potential for antifouling applications. The degradation of the superhydrophilicity under different humidity conditions is also investigated.
Introduction
Superhydrophilic surfaces have attracted increasing interest for several of their remarkable properties, such as self-cleaning, 1 antifogging, 2 and enhancement of boiling heat transfer. 3 Various applications of superhydrophilic surfaces have been proposed especially for household appliance. 4 However, much less investigation has been reported to use superhydrophilic surfaces in the biomedical devices, where the surfaces are usually in direct contact with DNAs, proteins, or cells. It is well known that cell adhesion and protein adsorption onto a substrate are highly sensitive to the wettability and the chemical nature of the surface. Nonetheless, current studies are usually limited to relatively smooth surfaces with medium wettability. 5 There is both fundamental and practical interest in extending such studies toward surfaces with extreme wettability, such as superhydrophilic (contact angle θ < 5°) and superhydrophobic (θ > 150°).6-8 For example, new insights may be obtained into the influence of extreme wettability on the physiological activities of cells, such as morphology of colony, anisotropic growth, migration, proliferation, differentiation, adhesion, and signaling activities. 9 Information in surface-specific cellular physiological characteristics could open up new possibilities in areas, such as biological diagnosis, antimicrobial or hemocompati-ble surfaces, and biomaterials for tissue engineering applications or microfluidics.
Between the two extreme cases of wettability, superhydrophobic surfaces have received more attention because of the well-known lotus effect. 10 Recent progress on these extremely nonwetting surfaces include giant liquid slip to reduce flow resistance. 11 However, the antifouling capability of superhydrophobic surfaces is limited by the strong hydrophobic interaction between the surface and the hydrophobic portion of biomolecules. As a result, the antifouling effect of superhydrophobic surfaces usually requires a constant presence of liquid flow, 12 which is not always available for microfluidic devices, especially when they are paused or stored. Although a precoated layer of proteins (e.g., bovine serum albumin) can be used to reduce the tendency for further biofouling, direct reduction of protein—surface interaction is preferable for microfluidic devices especially when reusability is considered. In this article, we evaluate the application of superhydrophilic surfaces for an-tifogging and antifouling in microfluidic devices filled with stagnant biomedical fluid.
Considerable efforts have been made to obtain superhydrophilic surfaces. For microfluidic devices, coating inside a microchannel 13 is usually difficult because of the increased complexity for fabrication. Generally speaking, the hydro-philicity of many hydrophilic materials can be enhanced by introducing strong polar groups to the surface, increasing the roughness or a combination of the two. The specific methods include exposure to ultraviolet radiation, 14 plas-ma,15-17 and electrochemical potential, 18 to name a few. In general, the treatment strongly depends on the material being used. In this article, polyester films and indium tin oxide (In2. O3—SnO2)-coated glasses (ITO glasses) are chosen because of their potential applications in microfluidic devices. Superhydrophilic polyester films and ITO glasses are obtained by the oxygen plasma treatment and the electrochemical method, respectively.
Sample Preparation
Plasma Treatment of Polyester Films
The polyester film, chosen for this study, is poly(ethylene-terephthalate) transparency from Tri-State Visual Product, Inc., Cincinnati, OH. The film is first cut into 1 × 1 in. square samples and carefully cleaned with deionized (DI) water. The samples are then placed in a reactive ion etching chamber (March Instruments, Jupiter II) for plasma treatment under a pressure of 0.130 Torr and a flow rate of 20 sccm for 5 min. The radio frequency power is 50 W. The water contact angles are measured before and after the treatment. The plasma treatment is known as an effective method to increase the surface energy of polymer by forming hydrophilic functional groups, such as -OH and -COOH. It has been widely used to improve adhesion.15, 16 Polyester films have been reported to turn superhydrophilic after proper plasma treatment. 2 The treated polyester surface has been shown to maintain optical clarity even when the relative humidity (RH) is high because water tends to spread on the surface to form a thin film instead of droplets. Therefore, superhy-drophilicity can effectively provide antifogging properties to the surface.
Electrochemical Treatment of ITO Glass
The ITO glass is purchased from Techgophers Corp. at Los Angeles, CA. A two-electrode setup is used for electrochemical treatment, with the ITO glass as the anode and a stainless steel electrode as the cathode. An aqueous solution of 0.5 M H2SO4 is used as the electrolyte. The two electrodes are immersed in the electrolyte and a voltage is applied with a Keithley 2425 SourceMeter (Keithley Instruments, Inc., Cleveland, OH). Water contact angles are measured before and after the electrochemical treatment. The superhy-drophilicity is observed when a potential of 3 V or higher is applied for 10 min or longer time. This is consistent with Pre-mkumar and Khoo's report, 18 which also suggested that 3 V is an approximate threshold potential to generate superhydrophilic ITO glass in similar conditions. In our experiments, a potential of 50 V is applied for 20 min to obtain superhydrophilic ITO glasses. The formation of superhydrophilicity can be attributed to water oxidation under electrical potential that leads to the formation of hydroxyl groups on the surface of the ITO layer. 18
Adhesion of Fluorescein and Green Fluorescent Protein
Fluorescein isothiocyanate (FITC, molecular weight of 389.2, excitation and emission of 494 and 518 nm, respectively; Pierce Biotechnology, Inc., Chicago, IL) and green fluorescent protein (GFP), extracted from a plasmid of pDEST17 of transformed BL21 cells, are used to inspect their adhesion with superhydrophilic-treated surfaces and untreated surfaces for a comparative study. GFP and FITC are chosen for the antifouling study because they are two kinds of fluorescent particles most commonly used in microfluidic devices.19-21 Samples are first dipped in 10 mM FITC solution for 1 min before they are incubated for 30 min at 37 °C in an oven. They are then rinsed by DI water and dried in air for 30 min at room temperature. A similar procedure is followed for the GFP experiments. A 4 mM GFP in 1 mM Tris—HCL buffer solution is used for the experiments. While dipping in the FITC and GFP solutions, special care has been taken to make sure that only one side of the samples can make contact with the solution.
Testing and Analysis
The superhydrophilic-treated and untreated surfaces are compared by measuring their water contact angles, inspecting their optical images under a high humidity condition for antifogging evaluation and analyzing the fluorescence microscope images after incubating them with FITC and GFP solutions.
Contact Angle Measurement and Antifogging Evaluation
The water contact angle of an untreated polyester film is about 95°. After the plasma treatment mentioned previously, the water contact angle is decreased dramatically to near 0°, indicating a superhydrophilic surface. Similarly, the water contact angle of an ITO glass is about 80°. Superhydrophilicity is obtained after the above-mentioned electrochemical treatment. The samples are also exposed to a highly humid environment by placing them on top of a cup filled with hot water. Figure 1 shows the optical images taken after 5 min, with their corresponding water contact angle measurement images as inserts. It is found that the untreated surfaces are covered by water droplets and fogged, whereas the superhy-drophilic-treated surfaces remain clear because water formed thin films on them.
Antifogging evaluation and corresponding water contact angle (θ) measurements for polyester films and ITO glasses before and after superhydrophilic treatment. The optical images show antifogging characteristics of each sample. Water contact angle measurements are given. (A) Untreated polyester θ = 95°, (B) treated polyester θ = 0°, (C) untreated ITO θ = 80°, and (D) treated ITO θ = 0°.
Adhesion Study for Antifouling Evaluation
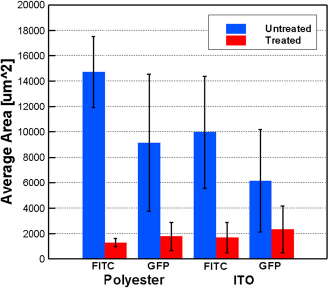
A fluorescence microscopy (Nikon Ti-U inverted microscopy, Nikon, Melville, NY) is used to inspect the samples after they are incubated with FITC and GFP. The images are taken by a 16-bit electron multiplying charge-coupled device (QuantEM; Princeton Instruments, Trenton, NJ). For each combination between the treated and untreated surfaces and FITC or GFP, eight images are taken on spots randomly selected from two different samples. Figure 2 shows the exemplary fluorescent images of various sample surfaces adhered with FITC and GFP after incubation. From the images, it is clearly seen that less FITC or GFP is attached to the superhydrophilic-treated surfaces than the untreated surfaces. The reduction of adhesion by the superhydrophilic surface treatment is quantitatively evaluated by measuring the total sample surface area covered by FITC or GFP molecules. ImageJ software, (National Institutes of Health (NIH), Bethesda, Maryland) is used for image processing and analysis. The raw images are in an 8-bit format, which treats a black pixel as 0, a white pixel as 255, and a gray pixel as a number in between according to its brightness. The threshold brightness value is set to be 93, which generates contours fitting well with the observed bright areas covered by FITC or GFP molecules. This threshold is thus constantly applied for all the images. The total surface area occupied by FITC and GFP is then calculated by using a particle analysis plug-in option from the software. Figure 3 shows the quantitative results that confirm that the number of FITC or GFP adhered to the treated surfaces is lower than that of the untreated surfaces. It is therefore demonstrated that the superhydrophilic-treated polyester films and ITO glasses have the potential to reduce protein or molecule adsorption and decrease biofouling in stagnant liquid. The antifouling effect of superhydrophilic surfaces can be attributed to the highly hydrated layer around the surface, which prevents the adhesion (e.g., by hydrophobic interaction) between the surface and FITC or GFP molecules.
22
The detailed working mechanism deserves further investigation and might depend on the specific molecules in question. However, considering the wide applications of FITC or GFP in microfluidic devices, superhydrophilic-treated ITO and polyester could be considered as very promising antifouling materials.
Fluorescent microscope images of treated and untreated samples after they are incubated in GFP and FITC solutions at 37 °C for 30 min. (A) Polyester and (B) ITO. Protein interaction compared between treated and untreated sample surfaces after they are incubated in FITC and GFP solutions.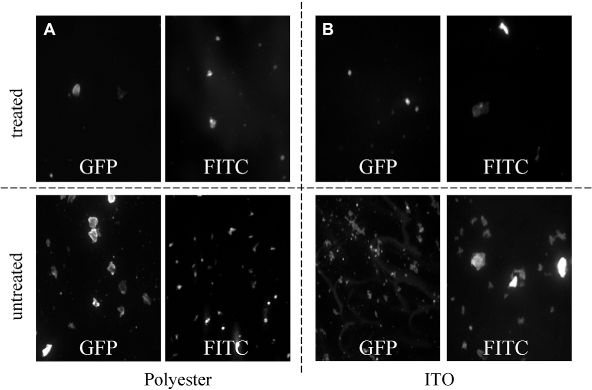
Stability of Superhydrophilicity
The stability of superhydrophilicity is an important aspect when its practical application potential is considered. To find the optimum conditions to minimize the degradation of superhydrophilicity, the changes in water contact angles over time under different environments are investigated for both polyester films and ITO glasses. It was previously reported that humidity has a significant effect on the degradation rate of superhydrophilicity for polyester film.
2
Our further investigation has revealed that the optimum RH to prevent this degradation on polyester films in air is about 20%, under which the increase of water contact angle is slowest. For comparison, in this report, we investigate the degradation of superhydrophilicity under three different environments as follows: (1) dry air environment (RH = 0%) in a desiccator, (2) wet environment in liquid water, and (3) medium humidity (RH = 20%) in air, obtained by a humidity chamber (5503-00 Package D; Electro-Tech Systems, Inc., Glenside, PA). All of the samples are stored under room temperature (approximately 20 °C). Three pieces of polyester film are treated by oxygen plasma and three pieces of ITO glasses are treated by the electrochemical treatment, after the same procedures previously described. They are kept in differently established environments for several days to investigate the effect of humidity on the hydrophilicity of the surfaces. The water contact angles are measured and recorded each day for all samples. The samples kept in water are allowed to dry by air at the room temperature before water contact angle are measured. For the samples stored in dry air and 20% RH environments, the water contact angles are determined within 5 min after removal from the desiccator or the humidity chamber. For each water contact angle data point, four different spots on the sample surface are measured. The average values are calculated and shown in Figure 4.
Degradation of hydrophilicity polyester films and ITO glasses in different environments. (A) Polyester and (B) ITO.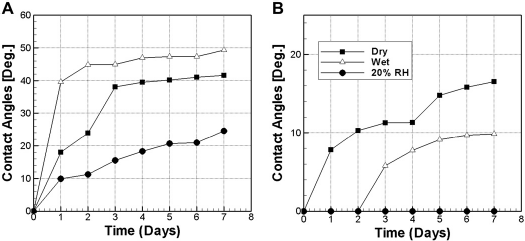
Polyester Films
Figure 4A shows the degradation of the hydrophilicity for the polyester films. Under extremely wet or dry environments, the hydrophilicity degrades quickly. Within 5 days, water contact angles rise to above 40° in both cases. A dramatic decrease of the degradation rate is observed when RH is changed to 20%, resulting in a water contact angle around 20° after a week. It is obvious that humidity plays a major role in degradation of hydrophilicity. The reason for the accelerated degradation under high humidity can be attributed to the plasticizing effect of water. 14 The excess water molecules absorbed into the polyester film can increase the space between the polymer chains and thus favor their mobility because of larger free volume. As a result, the induced polar groups can easily reorientate into the bulk of the material, resulting in a more pronounced aging effect. 23 It is noted that the completely dry environment also leads to quick loss of polar functional groups, although the degradation is slightly slower than that of the wet environment. The dehydration effect and the conformational change of the functional groups are conjectured to be the major aging factors under the dry condition although further investigation is still needed for clarification. As a result, medium RH (e.g., 20%) turns out to be beneficial for the stability of hydrophilicity.
ITO GLASS
Figure 4B shows the degradation of the hydrophilicity for ITO glasses. Different trends from the polyester cases have been observed. Generally speaking, the hydrophilicity of ITO glass degrades slower than that of the polyester films. Again, humidity plays a very important role in the case of ITO glass. The degradation is effectively suppressed under the 20% RH condition for more than 7 days. In contrast with the polyester film, hydrophilicity degradation of ITO glass in the wet environment is slower than the dry environment. It can be attributed to the reduced water absorption by the ITO glass substrate and the absence of the plasticizing effect. Because microfluidic devices are usually filled with an aqueous solution, the slow degradation of superhydrophilic ITO glasses in the wet environment represents an advantage over polyester films in this regard. In addition, the electrochemical treatment method for ITO glasses is relatively simpler than the plasma treatment used for the polyester films. Providing an appropriate environment, the ITO glass can even be treated in situ by applying voltage. Therefore, ITO glasses may represent a more promising material for superhydrophilic applications in microfluidic devices.
For both polyester film and ITO glass, the degradation rate under different conditions reflects the stability of polar functional groups. The different behavior for the two cases may originate from the differences on both the nature of the functional groups and their interactions with the substrates. Further investigation is required to clarify the degradation mechanism and provide the optimum conditions to maintain the superhydrophilicity.
Conclusion
Superhydrophilic surfaces are evaluated for their potential antifogging and antifouling applications in microfluidic devices. Polyester films and ITO glasses are treated to be superhydrophilic by oxygen plasma and an electrochemical method, respectively. They both demonstrate antifogging ability and reduced adhesion with FITC and GFP. The stability of the hydrophilicity is examined for both materials, showing slower degradation in the 20% RH environment. Between the two materials, the superhydrophilic ITO glass is considered to be more promising for microfluidic applications because of its simple process and high stability of hydrophilicity, especially in water.
Footnotes
Acknowledgments
Acknowledgment is made to the donors of the American Chemical Society Petroleum Research Fund and Michigan Tech Faculty Startup Fund for support of this research. The authors thank Ms. M. Nie, Mr. R. Lemmens, Mr. N. Kroodsma, Mr. S. Santhagopalana, Mr. X. Geng, and Dr. F. Teng for discussions, and Mr. Bill Knudsen for help with plasma facilities. The authors also thank Dr. Doktycz at Oak Ridge National Laboratory for providing enhanced GFPs.
Competing Interests Statement: The authors certify that they have no relevant financial interests in this manuscript.
