Abstract
An efficient system for screening for the octanol/water distribution coefficient (logD) was developed. We previously developed a 96-well—based high-speed logD assay system (HSlogD) using a water-plug aspiration/injection method by auto liquid sampler (HTS-PAL) to analyze the water phase without contamination of the upper octanol phase. In this report, we introduce an automated liquid-handling system (Biomek2000) into the previously reported HSlogD system to dilute the octanol phase automatically. As a result, throughput is increased, allowing the analysis of 400 compounds per week with a range of 0 < logD < 5.
Introduction
During the early stages of drug discovery and development, the activity of promising lead compounds was optimized by synthesizing a large numbers of derivatives and testing the resulting compounds. Unfortunately, many of the lead compounds identified by high-throughput screening were highly hydrophobic and poorly water soluble. The logD value, the logarithm of the distribution coefficient between octanol and water, is often used as a parameter of hydrophobicity. Optimization of hydrophobicity and solubility is very important in the drug-discovery process, because they are closely associated with absorption, 1 distribution, metabolism, 2 and excretion of the compound, including blood–brain-barrier penetration 3 and clearance, 4 –6 as well as other important pharmacological characteristics. Specifically, poor physicochemical properties can identify compounds that may be difficult and time-consuming to develop due to problems with formulation, process development, and safety. Therefore, a high-speed method for logD (HSlogD) evaluation in the early stages of drug discovery would be a valuable tool for pharmaceutical research and drug discovery.
Many methods have been reported to measure logD, such as the conventional shake-flask method. 7 There have been other methods based on the 96-well plate format, 8 pH measurement, 7,9 UV spectroscopy, 7 chromatographic retention times, 7,10 –12 counter-current chromatography, 13 microemulsion electrokinetic chromatography, 7,14 –16 immobilized artificial membranes, 17 and the dialysis tubing method. 18 Predictive software also has been used to estimate hydrophobicity. 19
Previously, we reported a high-speed logD assay system (HSlogD) 20 and high speed solubility (HSSOL) assay system. 21 In the HSlogD system, a water-plug aspiration/injection method of the auto liquid sampler (HTS-PAL) can analyze the water phase without contamination of the upper octanol phase. 20 For example, during logD analysis of a highly hydrophobic compound, the concentration of the compound in the octanol phase is much higher than that in the water phase, making it difficult to automatically separate the water phase from the octanol phase without contaminating the water phase. To resolve this problem, we developed a water-plug aspiration/injection method that allows the water to be taken into the needle before sampling of the water phase, enabling a decrease in contamination.
In the previously reported HSlogD system, the octanol phase was manually diluted to the proper concentration for LC–MS assay. In this report, we introduce an automated liquid handler (Biomek2000) to dilute the octanol phase automatically. This increases throughput dramatically, allowing the analysis of 400 compounds per week with a range of 0 < logD < 5.
Experimental
Chemicals (Materials and Reagents)
All generic drugs—chlorpromazine, imipramine, clozapine, diltiazem, bifonazole, niflumic acid, and flufenamic acid—were purchased from Sigma-Aldrich Japan (Tokyo, Japan). Compounds were dissolved in UV spectral grade dimethylsulfoxide (Dojindo Laboratories, Kumamoto, Japan) at 10 mM. Guaranteed reagent grade 1-octanol, and HPLC grade formic acid and ethanol (EtOH) were obtained from Wako Pure Chemical Industries (Osaka, Japan). Water was purified using a MilliQ Gradient A10 system (Millipore, Billerica, MA). HPLC grade acetonitrile (MeCN), NaH2PO4, and Na2HPO4 were obtained from Junsei Chemical Co. (Tokyo, Japan). Octanol and 10-mM phosphate buffer (pH 7.4) were mutually presaturated.
Equipment and Experimental Conditions of LC/MS
Analyses were performed using an Alliance 2690 HPLC system with a quaternary pump (Waters, Milford, MA), a HTS-PAL liquid handler (CTC Analytics, Zwingen, Switzerland), and a ZQ2000 quadrupole mass detector (Waters) equipped with electrospray ionization. This system was controlled with Masslynx and Quanlynx ver 4.0 software (Waters). Chromatography was conducted using a Develosil RP aqueous C30 column (2.0 × 35 mm; particle size 5 μm; Nomura Chemical, Aichi, Japan). The mobile phase was composed of a mixture of 0.1% formic acid in water (solvent A) and MeCN (solvent B). The gradient was delivered at 1.0 mL/min as follows: 0 min, 0% solvent B; 4.0 min, 90% solvent B; 4.5 min, 90% solvent B; 4.6 min, 0% solvent B; and 5.5 min, 0% solvent B; loop time = 5.5 min. The flow was split postcolumn as follows: 0.4 mL/min to MS inlet, 0.6 mL/min to waste. The column was maintained at room temperature. The PAL injector system used a microliter syringe (Hamilton, Bonaduz, Switzerland; 25 μL) with a 22S-G needle (internal diameter, 0.15 mm; external diameter, 0.72 mm; needle length, 51 mm; needle volume, 0.77 μL). The mass spectrometer was operated with an electrospray source. All compounds were analyzed in positive ionization mode. The ionization source conditions were capillary voltage of 3.5 kV, source temperature of 120 °C, and desolvation temperature of 350 °C. The cone and desolvation gas flows were 50 and 400 L/h, respectively, and were obtained from an in-house nitrogen source. Under these conditions, the compounds were analyzed by selected ion monitoring (SIM) with a dwell time of 0.20 s.
Water-Plug Aspiration/Injection Method by HTS-PAL
The customized injection program for the PAL liquid handler was performed as follows.
Wash syringe twice with EtOH to clean the needle.
Draw 2.5 μL of water (water plug).
Draw 2.5 μL of sample (sampling).
Place the syringe in the EtOH wash port (rinse).
Place the syringe in the water wash port (rinse).
Load sample onto the LC–MS.
Wash valve twice with EtOH to prevent sample carryover.
Wash valve twice with water to prevent sample carryover.
Equipment and Experimental Conditions of Automated Liquid Handler
The workstation of sample preparation used a Biomek2000 (Beckman Coulter, CA) unit as an automated liquid handler. The system included the eight-channel pipette tool (MP-20, MP-200). To avoid nonspecific binding, a low-binding polymer tip (Nippon Genetise, Tokyo, Japan) was used in this system.
Sample Preparation
Compounds were dissolved in DMSO at a concentration of 10 mM. A volume of 10 μL of each compound at a concentration of 10 mM in DMSO was placed in a 96-well plate (Agilent Technologies, Palo Alto, CA, Parts No. 5042–6454). Next, 300 μL of octanol was added to each well using an automated liquid handler (Biomek2000, Beckman, Fullerton, CA) with a low-binding polymer tip (Nippon Genetise, Tokyo, Japan), and the plate was sealed with a silicone preslit well cap. The plate was then agitated on a plate shaker (Taitec M36, Taitec, Saitama, Japan) for 5 min at 14 g. After 5 min of centrifugation at 950 g in a swinging-bucket centrifuge (Himac CF-8DL, Hitachi-Koki, Tokyo, Japan), the seal was removed. A volume of 600 μL of 10-mM potassium phosphate buffer (pH 7.4) was added to each well using an automated liquid handler (Biomek2000, Beckman). After sealing, the plate was vigorously mixed on the plate shaker for 1 h at room temperature. After 1 h of shaking at 14 g, the plate was centrifuged at 950 g for 5 min, and the seal was removed. A volume of 10 μL of the octanol phase was transferred into a new plate and 490 μL of 50% EtOH was added using the automated liquid handler (50-fold dilution of octanol phase). Then, the sample was mixed using the Biomek2000 mixing mode. The octanol layer was then diluted 50-fold using the same method, resulting in a final 2500-fold dilution of the octanol phase. The 2500-fold diluted octanol phase plate was sealed and placed in the PAL injector for analysis of the octanol phase. The plate that was initially shaken for 1 h was also placed in the PAL injector without a seal for analysis of the water phase. The water phase was directly sampled and analyzed using the water-plug aspiration/injection method without manual separation of the octanol phase. The 2500-fold diluted octanol phase also was analyzed by the same method. The logD value was calculated using the following equation: logD = log{[octanol phase]/[water phase]} = log {[2500-fold dilution of octanol phase] × 2500/[water phase]}.
Results and Discussion
Previously Reported HSlogD System
A HSlogD system using the water-plug aspiration/injection method has been reported previously. 20 Figure 1 shows the scheme of this method. During logD analysis of a highly hydrophobic compound, the concentration of the compound in the octanol phase is much higher than that in the water phase. When the needle passes through the upper octanol phase to sample the lower water phase, a small amount of octanol phase contamination may occur, which could affect sampling of the water phase. To avoid this problem, the water-plug aspiration/injection method was developed. In this method, the needle is aspirated with a few microliter of water as a plug before being passed through the octanol phase to sample the water phase. This should prevent contamination, because the water in the needle repels octanol. This led to the development of the water-plug aspiration/injection method, which enables automatic analysis of the water phase in the range of 0 < logD < 5. 20
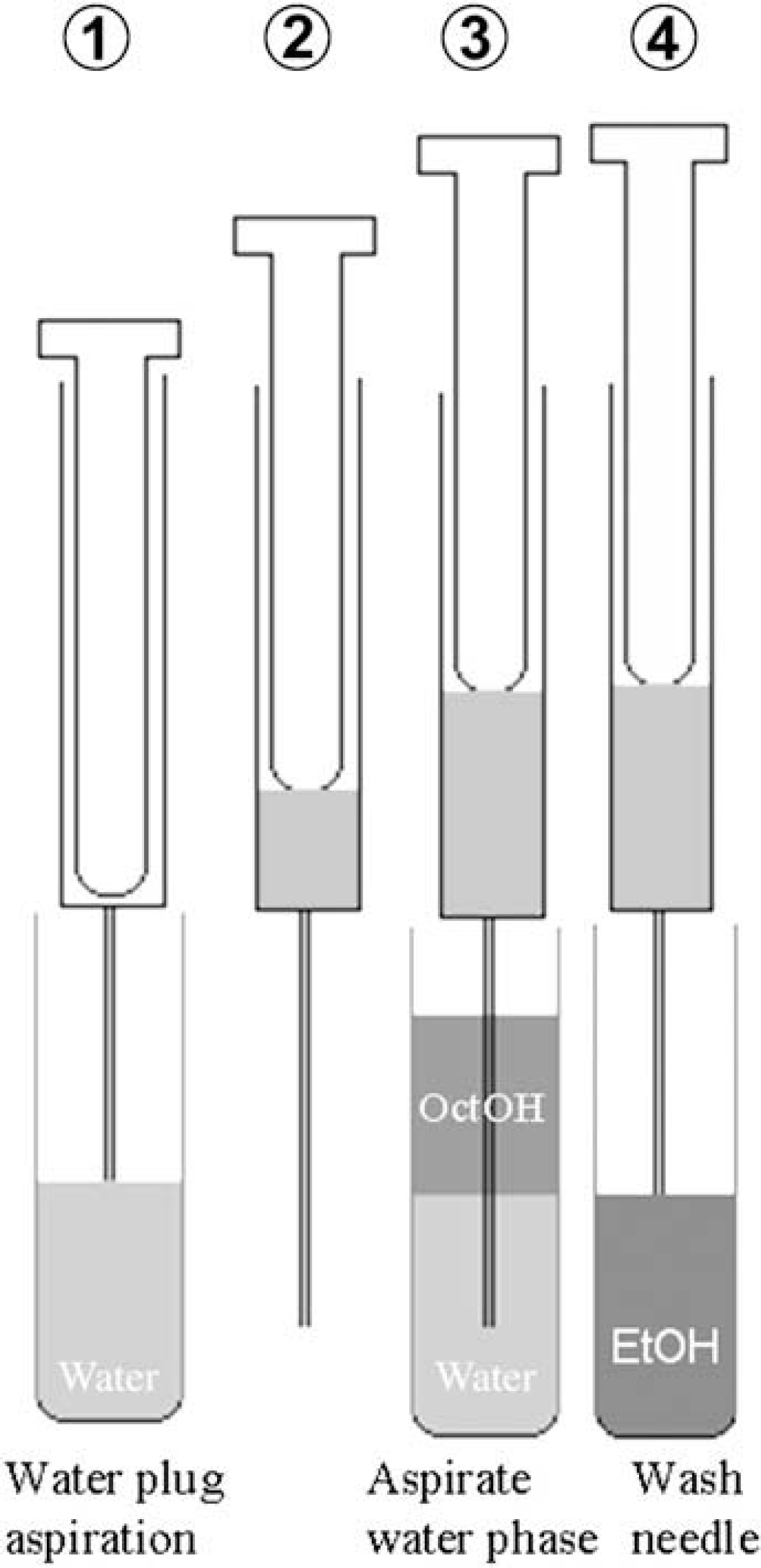
Scheme of the water-plug aspiration/injection method.
Figure 2a shows the scheme of the previously reported HSlogD system. In the previous system, the octanol phase was manually diluted to the concentration of LC–MS assay. To increase throughput, we introduced the automated liquid handler (Biomek2000) to dilute the octanol phase automatically (Fig. 2b).
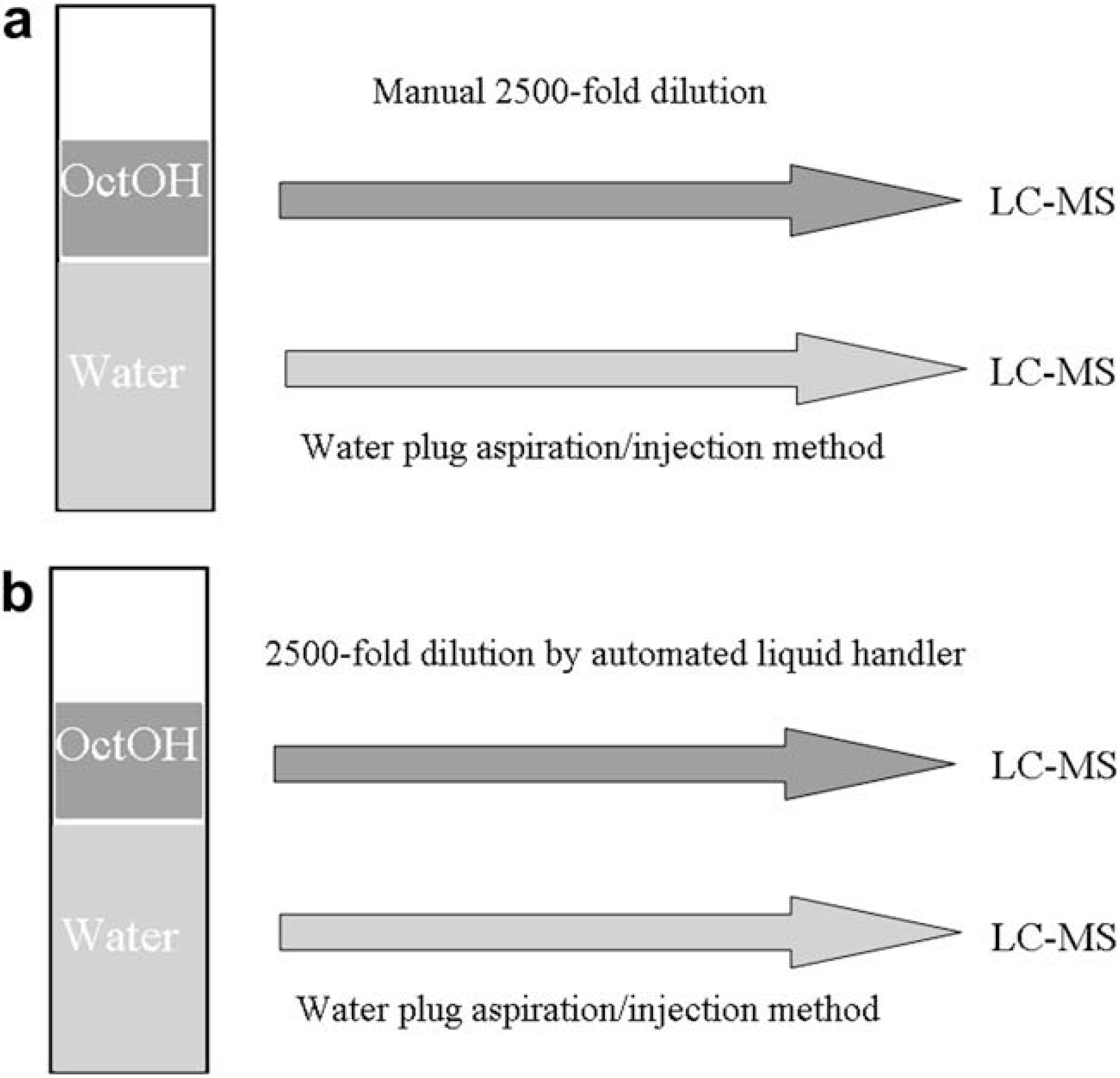
Scheme of the (a) previously reported HSlogD system, and (b) new HSlogD system introducing the automated liquid handler to dilute the octanol phase automatically.
Introduction of the Automated Liquid Handler into the HSlogD System
We introduced an automated liquid handler to dilute the octanol phase automatically. Initially, a 10-μL sampling volume from the octanol phase was used. Figure 3 shows the cross-section of 96-well plate with 300 μL of octanol and 600 μL of water. At the 10 μL sampling point, a height of 65% was marked for the automated liquid handler. A 10-μL aliquot of the octanol phase was collected and diluted 2500-fold with 50% EtOH using the workstation of the automated liquid handler.
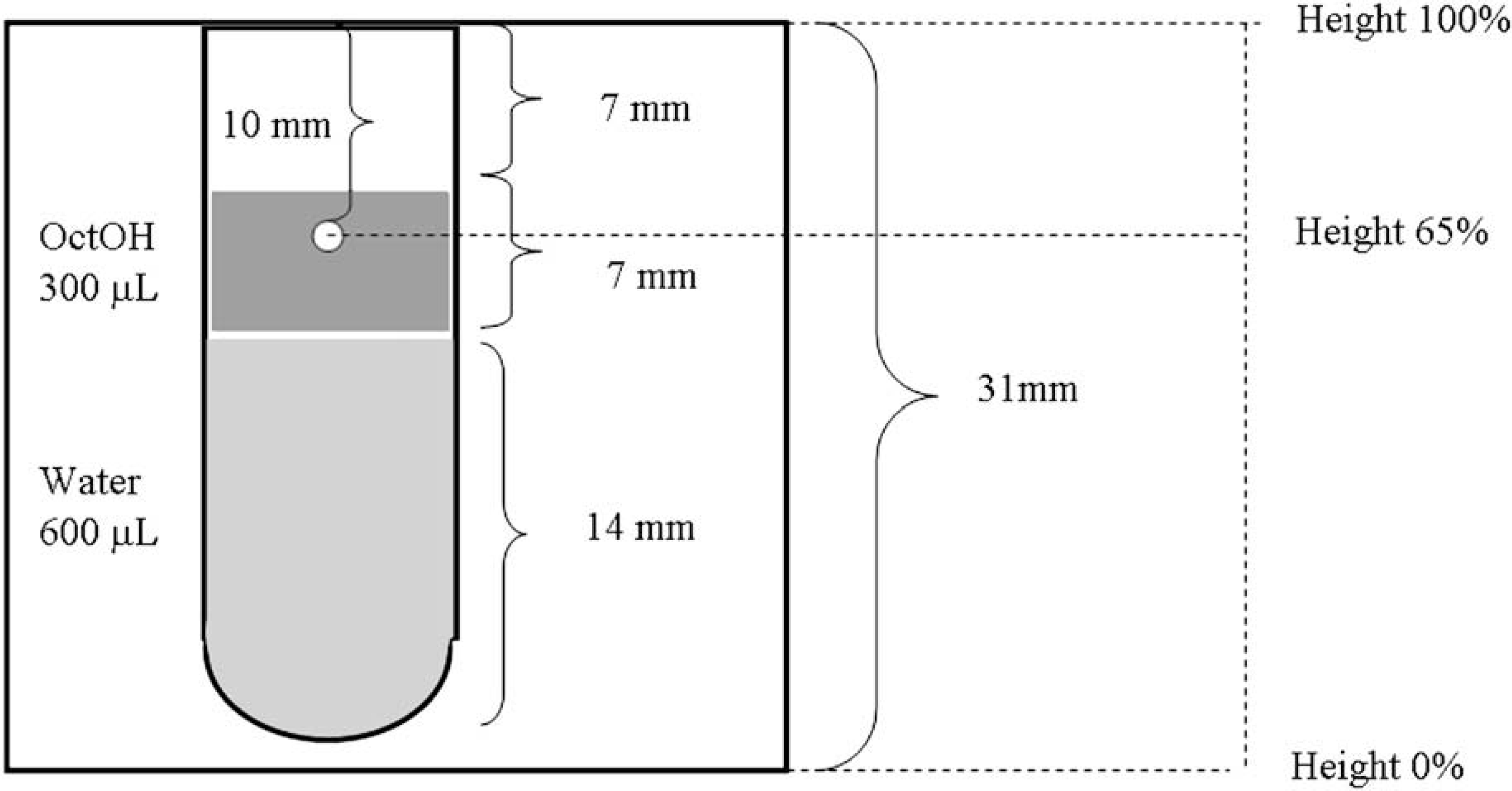
Cross-section of a 96-well plate with 300 μL of octanol and 600 μL of water. At the 10-μL octanol sampling point, the height at 65% was input into the automated liquid handler.
To validate this dilution method, we examined the relation between automatic and manual dilution. Table 1 shows the LC–MS area of chlorpromazine. Automation involved sampling of 10 μL octanol followed by 2500-fold dilution controlled by the workstation of the automated liquid handler; the 2500-fold diluted octanol sample was analyzed by LC–MS. Manual involved manually sampling 10 μL of octanol by eight-channel pipette (Biohit eLine, Helsinki, Finland) with low-binding polymer tip (Nippon Genetise) followed by a manual 2500-fold dilution; the 2500-fold diluted octanol sample was analyzed by LC–MS. The results of automatic and manual dilution were similar; therefore, the automatic dilution process was adopted to increase throughput.
LC–MS area of chloropromazine (n = 8)

Automation involved sampling of 10 μL of octanol that was diluted 2500-fold in the workstation of the automated liquid handler; the 2500-fold diluted octanol sample was analyzed by LC–MS. Manual involved the manual sampling of 10 μL of octanol using an eight-channel pipette (Biohit eLine, Helsinki, Finland) with a low-binding polymer tip (Nippon Genetise) followed by manual 2500-fold dilution; the 2500-fold diluted octanol sample was analyzed by LC–MS. RSD, relative standard deviation.
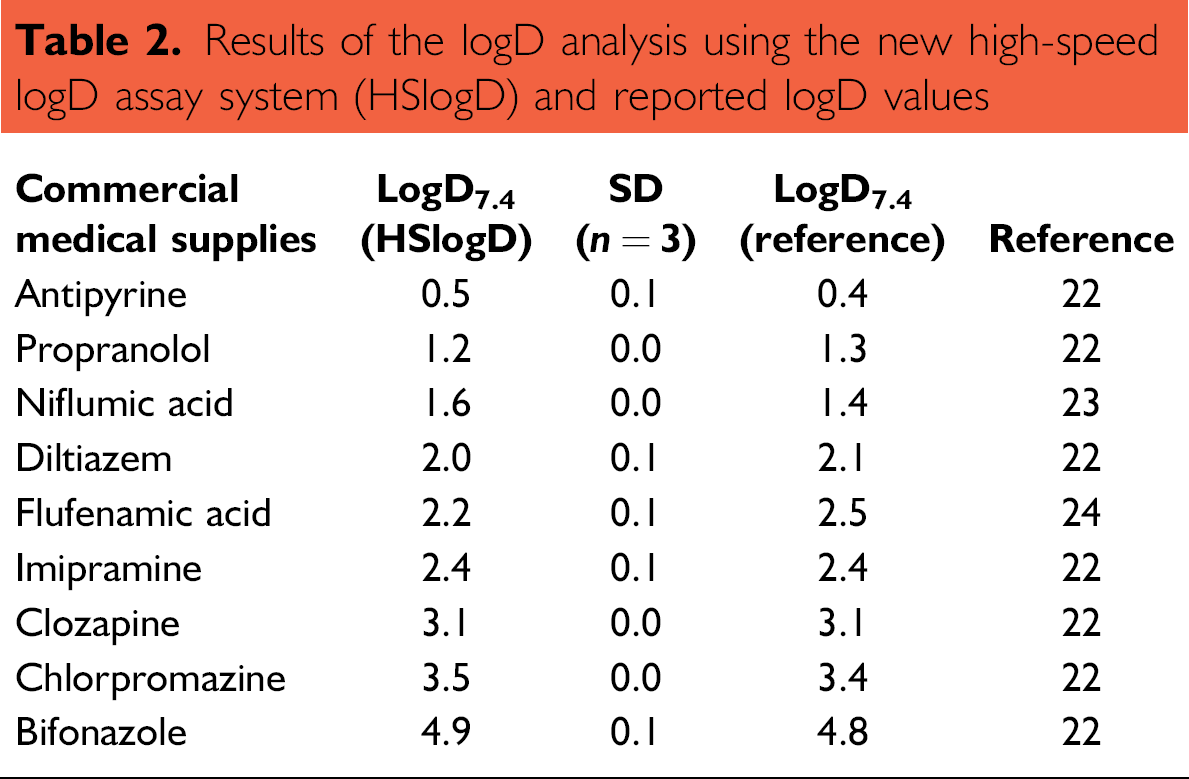
Several generic drugs were measured using the HSlogD system with the introduced automated liquid handler (Biomek2000) and were compared with reported logD values (Table 2). 22 –24 There was good agreement between the HSlogD results and reported logD values over a wide dynamic range (logD from 0 to 5).
Results of the logD analysis using the new high-speed logD assay system (HSlogD) and reported logD values
Throughput of the HSlogD System
Figure 4 shows the sample preparation scheme of the conventional shake-flask method and the new HSlogD method. For the HSlogD method, 10-mM DMSO stock solution was used instead of solid sample in the shake-flask method. Using 10-mM DMSO stock solution enabled removal of the time-consuming powder weighing process. In the HSlogD system, 1.1% of DMSO was included in 300 μL of octanol/600 μL of water, but did not influence the value of logD, as previously reported. 20 For the HSlogD system, a water-plug aspiration/injection method was used to analyze the water phase automatically instead of using a manual separation process as in the shake-flask method. It is time-consuming to separate the octanol phase from the water phase because octanol has high viscosity. The water-phase aspiration/injection method replaced the manual separation process, which increased throughput. However, this method still involves time-consuming processes. In this report, we introduce the automated liquid handler to dilute the octanol phase to the LC–MS analytical concentration instead using manual dilution as in the shake-flask method. Introduction of the automated dilution system enabled elimination of the manual dilution process, and dramatically increased throughput. In the HSlogD system, LC–MS analysis was performed instead of LC–UV analysis. Generally, most compounds that are synthesized in the early stages of the drug-discovery process are not perfectly pure. To obtain good chromatographic separation, a longer gradient time was required in the LC–UV system compared to the LC–MS system. By introducing SIM detection, the influence of impurities was reduced, shortening analysis time. To increase the measurement of logD values, the conventional shake-flask method was improved by using a 10-mM DMSO stock sample, the water-plug aspiration/injection method, LC–MS detection, and an automated liquid handler. As a result, the throughput was dramatically increased, allowing the analysis of 400 compounds per week compared with 10 compounds per week for the conventional shake-flask method.
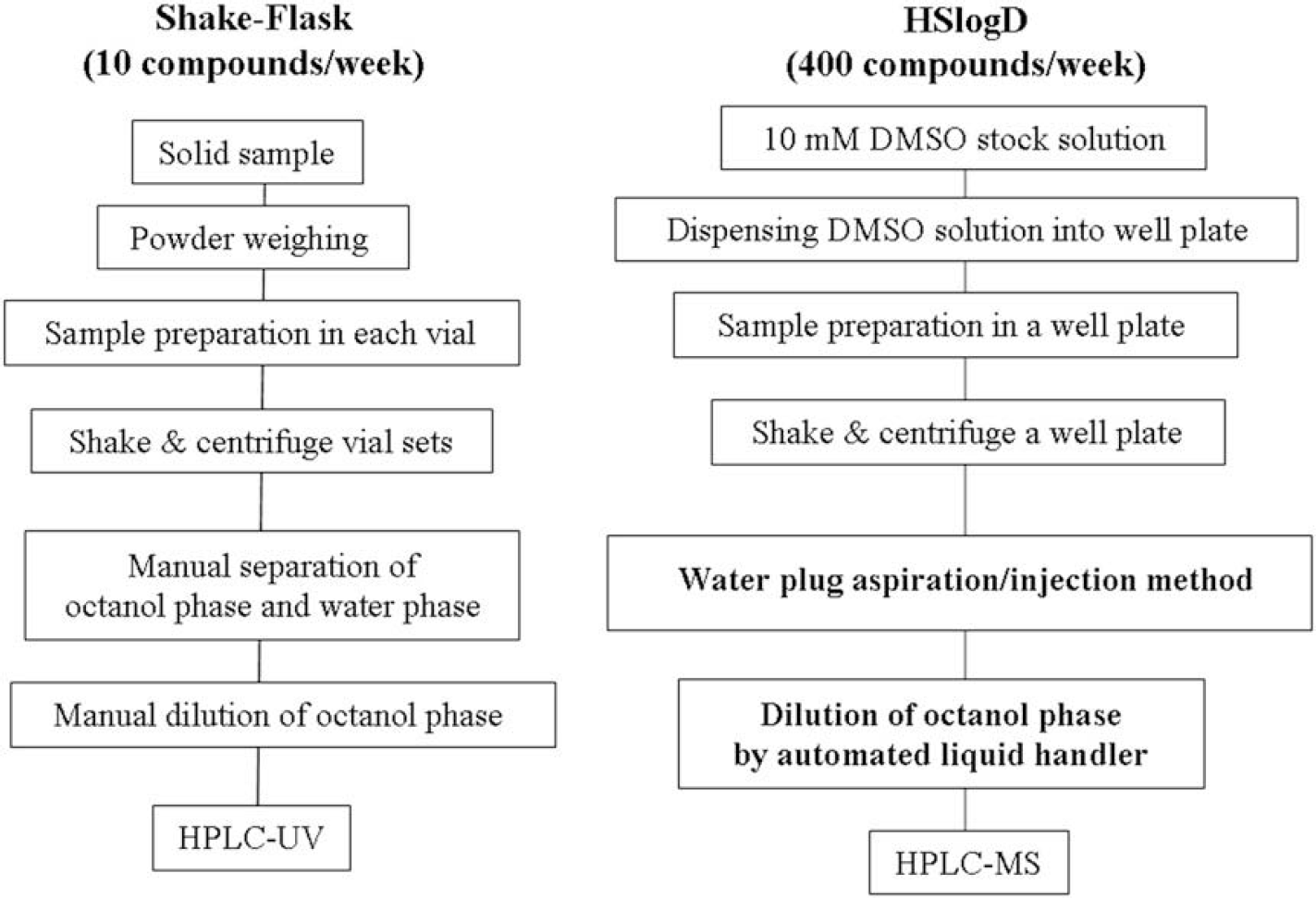
Sample preparation schemes of the conventional shake-flask method and the new HSlogD method.
Conclusions
We previously developed a 96-well HSlogD using a water-plug aspiration/injection method for auto liquid sampling (HTS-PAL) to analyze a water phase without contamination of the upper octanol phase. 20 In a previously reported HSlogD system, the octanol phase was diluted manually to the concentration required for the LC–MS assay. In this report, we introduce an automated liquid handler (Biomek2000) to dilute the octanol phase automatically. As a result, the arduous octanol dilution process was eliminated, and the throughput was dramatically increased, permitting the analysis of 400 compounds per week with a range of 0 < logD < 5.
Footnotes
Acknowledgments
We thank the staff members of the analytical chemistry group at Tsukuba Research Institute (Banyu Co., Ibaraki, Japan) for assistance and Michiyo Nagano and Kazue Ogino (Banyu Co.) for supporting analyses. We also thank Satoshi Ozaki and Hirohide Nambu (Banyu Co.) for supplying the automated liquid handler (Biomek2000).
