Abstract
Surface-based assays have been used extensively for the functional and structural analysis of biomolecules such as DNA or proteins. These experiments are established by the analysis of binding between acceptor molecules and immobilized receptors on a platform. Site-specific printing of receptor molecules on gold, glass, or polycarbonate (PC) surfaces is conventionally performed by the chemical derivatization of a surface, priming it to covalently bind to subsequently deposited receptor molecules. Unlike conventional methods, we have developed a new fabrication method for bioactive PC surfaces by directly molding PC granules doped with receptor molecules. PC-based receptor molecules were synthesized and commercially available PC granules were doped with these synthesized molecules. In our proof-of-concept study, PC doped with dye 1 (Fig. 1) was used as the receptor molecule. Using an aluminum mold and a hot press machine, PC-based objects were manufactured through compression molding using doped PC granules. Affinity analysis was evaluated by monitoring the localization of a monoclonal antibody elicited against dye 1 to the surface of the molded platforms by fluorescence microscopy. The results illustrated effective binding of an anti-dye 1 monoclonal antibody to the surface, substantiating successful display of assemblies of molecular receptors on the surface through compression molding. Although conventional surface functionalization methods impose limited applications and alter desired opto-mechanical properties of the polymer, our investigation provides a versatile means for the fabrication of bioactive PC-based platforms. It can also be used for engineering and imbedding receptor arrays within three-dimensional objects with applications to the production of opto-medical devices or biosensors.

Introduction
Surface-based assays (binding of acceptor molecules and immobilized receptors on a platform) have been used extensively for the functional and structural analysis of various biomolecules. The development of novel platforms has gained in importance due to the recent shift toward genomic, proteomic, and metabolomic studies. In this domain, gold, glass, or plastic surfaces are commonly used. 1 The existing related art in the production of biosensors is based on chemical micropatterning of these surfaces for site-specific immobilization of receptor molecules. This is usually performed by chemical derivatization of gold, glass, or polycarbonate (PC), preparing it for covalent attachment of the ascribed receptors. Subsequently, the platform can be used in various surface-based assays in which binding of acceptor molecules to the immobilized receptors are studied.
Among the aforementioned materials, PC has been used enormously in recent years for production of various laboratory and clinical devices including biosensors as it posses outstanding mechanical and optical properties. 2 Specifically, PC provides better optical properties than many types of glasses, high impact (Hardness-Rockwell M70) resistance, and high working temperature (115–130 °C). Moreover, it is moldable at a commercial scale for high-throughput production of PC-based components. 3
Currently practiced approaches in the execution of polymer-based surface assays are generally based on production of polymer-based objects and subsequent printing of receptors to the surface through surface functionalization methods. 2 These methods mostly have limited applications and alter the desired opto-mechanical properties of the polymer, as method such as inkjet printing leave regions with uneven surfaces and therefore require the generation of larger surfaces from the printing of thousands of droplets. 4 However, we have provided a novel method for production of chemically active PC-based platforms by directly molding PC granules doped with receptor molecules. 5 This discovery suggests a versatile method for the fabrication of various biosensors.
Method
Our production method is based on manufacturing biosensors from moldable PC granules doped with receptor molecules. For our proof-of-concept experiment, we decided to dope PC granules with a fluorescence coumarin dye-based receptor. Efficient doping would be possible by synthesis of the receptor molecule that can attach efficiently to regular PC granules. Different synthesis strategies were studied and the molecular design shown in Figure 1 was chosen. 5 As shown in compound 1, a fluorescence coumarin dye-based receptor (shaded in green) has been attached to a PC tail (shaded in blue) through a polyethylene glycol linker (shaded in red).

Structures of PC-PEG-DYE construct containing a coumarin dye-based receptor (shaded in green), a polyethylene glycol linker (shaded in red), and a polycarbonate (PC) solublizing anchor (shaded in blue). The figure depicts a molecule of the PC-PEG-DYE construct interchelated in between two molecules of PC as it would appear on the surface of a molded object. The addition of the PEG linker serves as a spacer to extend the dye molecule away from the PC surface and to provide an environment favorable for protein binding. The dye used was 7-dimethylamino-4-coumarinacetacetic acid, which we have shown can be readily coupled to form amide linkages. 6
Doping studies were conducted on commercially available Makrolon CD2005 PC milled to 150 ± 50 μm and doped by shaking 100 mg of resin in 1 mL of compound 1 at 50 nM in ethanol (as determined by the concentration of the dye, Fig. 1) for 2 h. After washing with water or phosphate buffered saline (PBS) buffer, doped PC was produced. 5 Our assay strategy was based on the implementation of a dual-color fluorescence-based sandwich assay using primary and secondary antibodies. This is shown schematically in Figure 2. Murine monoclonal antibody (mAb) XRI-TF35 7 and FITC-labeled anti-mouse immunoglobulin G(IgG) were chosen as primary and secondary antibodies, respectively.
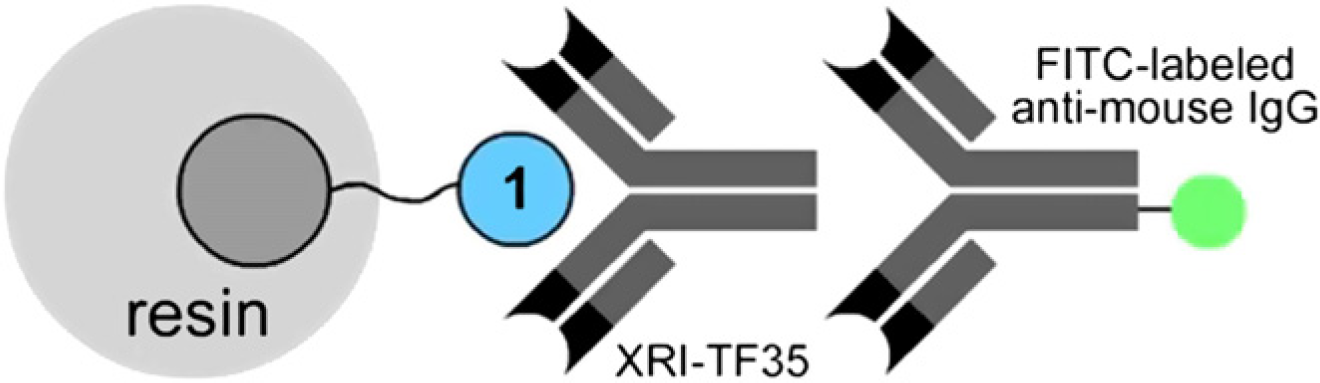
Illustration of dual-color fluorescent analysis on doped PC resins depicting the binding of a primary XRI-TF35 murine monoclonal antibody (mAb) to the receptor in blue. The affinity event was visualized by examining the secondary FITC-labeled anti-mouse IgG mAb in green.
With respect to production of PC-based object, objects were pressed by means of compression molding. A hot press machine as shown in Figure 3a was used. The machine consists of two stainless steel plates, the temperature of which plates can be electronically controlled in a closed loop manner. Set points for the temperature of the plates can be assigned. The temperature of the plates is measured using a bipolar thermostat and fed back to the electronic controller, which controls the electrical heating source. The lower plate can be moved up/down using a hydraulic cylinder. Maximum pressure as much as 1500 psi can be applied.

Machine and molds. (a) Hot press machine; (b) pieces of cylindrical mold; and (c) compression molding in action.
Production of small cylindrical lenses using a simple three-piece closed mold was implemented. The mold consists of three parts: (1) an aluminum cylinder (outer diameter (OD) = 0.8 in., inner diameter (ID) = 0.40 –0.005 in., height = 0.8 in.); (2) an aluminum disk (OD = 0.405+0.0025 –0.0025 in., height = 0.2 in.); and (3) a Teflon plunger (OD = 0.405+0.0025 –0.0025 in., height = 0.6 in.) (Fig. 3b). The disk is inserted into the cavity of aluminum cylinder from one end. Then, the cavity of the cylinder is filled with the material and the assembly is placed on the lower plate of the hot press machine. After the material is melted (melting and softening temperatures of the material are 220–230 °C and 150–160 °C, respectively), 3 the Teflon plunger is inserted into the cylinder and pressed down by moving the lower plate of the hot press machine (Fig. 3c). Teflon material has been chosen to avoid clogging of the plunger into the cylinder of the mold by the molten PC. The manufacturing tolerances of parts as mentioned above would allow enough tightness for forming a cylindrical object out of the molten material inside the mold. Moreover, the aluminum disk can be taken out easily from the cylinder after the molding process. PC-based cylinders with OD ∼ 0.4 in. and various heights depending on the amount of material poured into the mold can be formed through compression molding using the mentioned closed-mold design.
Experimental Results
A sandwich assay based on the concept shown in Figure 2 was implemented before molding doped PC granules, and was analyzed through dual-color fluorescence analysis. 5 A concentration-dependent binding event was observed between the primary antibody and the receptor, as well as between the secondary antibody and the primary antibody. Because the best binding effect was observed with primary and secondary antibody at 3 μM, this concentration was used to perform the assay on the surface of molded object.
After inserting the aluminum disk into the cavity of aluminum cylinder, the cavity was first filled with PC resin, and samples of resin doped with 1 were sprinkled randomly over the top of the mold (∼ 99.9% PC resin vs 0.1% doped resin by mass). The mold was then placed on the lower plate of the hot press machine. Plate temperature was increased until the resin inside the mold melted, then the Teflon plunger was inserted and pressure of approximately 20–50 psi was applied. The best molding results were obtained by quick water cooling of the mold assembly after compression. Figure 4 shows the result of compression molding with doped resin. Figure 5 shows images taken from the surface of the molded lens using a fluorescence microscope configured for imaging the receptor molecule (the 7-dimethylamino-4-coumarinacetacetic acid moiety depicted in Fig. 1 is excited at 377 ± 50 nm and emits at 447 ± 60 nm). Figure 5a and 5b taken from the object with UV light illumination shows that dye molecules are locally stabilized after compression molding, whereas Figure 5c shows that the object is transparent to visible light.

Transparent molded lens using PC resin and doped PC granules.

Images depicting the surface of compression-molded PC lenses. (a, b) Blue fluorescent images denoting regions of PC that contain dye molecules after compression molding. These images confirm that the dye molecule is stable to the molding process. (c) Image of the molded surface taken with white light illumination. Fluorescence in images (a) and (b) was captured with excitation at 377 ± 50 nm and emission at 447 ± 60 nm.
To verify the existence of the receptor molecules on the surface of the molded object ready to bind with the antibody, the same sandwich assay performed on the doped resin (Fig. 2) was performed on the molded surface as illustrated in Figure 6. The surface was first treated with the primary antibody and then with the secondary antibody. Figure 7 shows the result of dual-color fluorescence analysis performed on the surface of molded object. Regions that fluoresce in blue (Fig. 7a) contain the receptor molecule. Regions that fluoresce in green (Fig. 7b) are the result of binding of FITC-labeled antibody complex to the receptor regions on the surface. The experiment demonstrates that regions that appeared on the surface of the lens (g1 in Fig. 7) were able to bind to the green fluorescent antibody, whereas regions inside the molded lens (g2) did not take up the antibody complex. 5 We chose to show the results of our proof-of-concept experiments by producing a lens-shaped object to illustrate the possibility of producing optical components with bioactive surfaces. Optical-based diagnostic devices would definitely benefit from this type of disposable bioactive optical component.

Illustration of dual-color sandwich assay performed on the molded surface.

(a) Blue fluorescence from regions containing the receptor; (b) green fluorescence from regions after treatment of the surface with 3 μM XRI-TF35 mouse monoclonal IgG antibody and 3 μM FITC-labeled anti-mouse IgG antibody. Blue fluorescence was captured with excitation at 377 ± 50 nm and emission at 447 ± 60 nm. Green fluorescence was captured with excitation at 500 ± 24 nm and emission at 542 ± 27 nm.
Conclusion and Future Work
Our investigation provides the capability to produce chemically active objects in any geometric form using PC granules doped with synthesized PC-based receptor molecules. This method as illustrated here by molding a lens and performing an antibody affinity sandwich assay can be used to manufacture optical devices containing patterns of fluorescent materials or receptors for subsequent analyses. The production method is consistent with high-throughput manufacturing process of PC objects by injection modeling or compression molding; thus it provides a great potential for low-cost high-throughput fabrication of devices useful for performing various home-based or environmental assays. For instance, compact disk-based biochip production and analysis could benefit extremely from this technique by directly manufacturing of compact disks with bioactive PC surfaces. Further studies are underway to engineer the optimal method to apply this approach for molecular systems with reactive labels and biologically relevant ligands.
Acknowledgment
This work was supported by the NSF grant BES-0520868. We thank Bayer Material Science for the sample of PC used herein. The XRI-TF35 mAb was obtained as a gift from the Xenobe Research Institute, San Diego, CA.
