Abstract
As the drug discovery process evolves and demands more challenging and relevant assays, we have experienced a recent and significant escalation in the number of cell-based high-throughput assays for both small molecule and target identification screens. This has resulted in an increased need for the reproducible production of high quality cells in large quantities. Historically, manual cell culture was the only option available for providing cells in sufficient numbers for small molecule ultra-high-throughput screens (uHTSs), representing a technology gap in our automated cell culture process.
Recently, we have applied an automated solution in the form of a novel 10-layer tissue culture flask, the HYPERFlask (Corning, Lowell, MA). This technology, when introduced as an upgrade to the SelecT (an automated cell culture system manufactured by The Automation Partnership, Ltd., Royston, England), provides a new approach to automating production of the high number of cells required for uHTS and consequently a highly desirable alternative to manual cell culture.
The HYPERFlask has a surface area of 1720 cm2 and can yield up to 3 × 108 cells after 3 days in culture. This can be compared to a typical yield of 2 × 107 cells from a T175 flask (with a surface area of 175 cm2), the current standard flask type for automated cell culture on the SelecT. Cells grown in both flask types are of comparable quality, as demonstrated by equivalent cell viability, yield per cm2, functional response, and pharmacology.
Introduction
Cell-based assays have become increasingly prevalent in many stages of drug discovery and target identification processes. 1,2 Traditional high-throughput screens used purified proteins or membrane preparations that were amenable to large-scale production, but limited to use in a minimal variety of assays. 3 –6 As the field of screening matured, new technologies made high-throughput cell-based assays a possibility, and the demand for large numbers of functional cells grew exponentially. 7 –15 Supplying cells in sufficient quantity and quality for ultra-high-throughput screens (uHTSs) presents a significant challenge for existing automated cell culture systems, primarily due to the extremely high numbers of cells required. This demand can range from hundreds of millions to billions of cells every day for 5–15 consecutive days depending on the format of the assay. Both system capacity and process time are limiting in these conditions, forcing the use of manual cell culture to support uHTS cell-based assays.
Automated cell culture using the SelecT 16 –18 offers several advantages over manual cell culture (Fig. 1). It removes many of the variables involved in growing cells. By maintaining cells with robotic protocols, greater consistency is achieved by alleviating variables when cells are transferred from one cell culture scientist to another. Automated cell culture also enables cell passaging and plating over weekends, allowing the delivery of plated cells for assay on Mondays. Importantly, when compared to manual cell culture, automated cell culture generates key parameters from the cell populations. These data are captured and can be applied to enhance the quality control of cell production. For example, percent viability, average cell size, and the presence of cell debris and aggregates can be compared to assay performance to identify the optimal cell conditions. Because cell counting is automated, 20 cell counts are routinely taken from each cell sample to generate a highly accurate number. Another key capability of the automated cell counter on the SelecT is that it produces and retains an image of the counted cells that can be critically important in trouble-shooting cell-based assays and avoids subjectivity in retrospective analysis of cells provided for assay.

SelecT. The SelecT, designed by The Automation Partnership, is the first automated tissue culture system. A consortium of six major pharmaceutical companies were involved in the system design. The SelecT automates cell seeding, harvesting, and passaging. It has a capacity of 182 T175 flasks and 420 plates.
The biggest disadvantage to using the SelecT for automated cell culture is the linear process of the system. The SelecT has only one arm, and is therefore only capable of handling one flask at a time, which can lead to long processing times. This was a significant driver for implementing the HYPERFlask. Another consideration is that the SelecT can only manipulate a 10-mL pipette, which has a maximum aspiration/dispense speed of 5 mL/s. Therefore, it can be difficult to dissociate a clumpy cell line into individual cells. These few disadvantages are outweighed by the quality control, consistency, and reliability obtained from using the SelecT.
We have used the SelecT to successfully support cell production for target identification and high content screening assays. Target identification and high content screens require moderate cell numbers, between 107–108 cells each assay day. 19 –24 However, for our process, automated production of cells in sufficient quantities to support cell-based uHTS screens (108–109 cells each assay day) has been limited by both speed and capacity of our current automation. Recent development of the HYPERFlask 25 in combination with a compatible upgrade of SelecT automation presented an opportunity to expand the capabilities of automated cell culture for uHTS applications.
The HYPERFlask is an innovative tissue culture flask that drastically increases the surface area of cell growth, while maintaining a footprint almost identical to that of a standard automation-compatible T175 flask (Fig. 2). The HYPERFlask consists of 10 essentially identical, interconnected “cassettes,” each containing a novel membrane that has been treated with CellBIND (Corning, Lowell, MA) for improved cell adherence. The membrane is gas permeable allowing exchange of oxygen and carbon dioxide through the base of the membrane. Gas enters the HYPERFlask through the sides and subsequently underneath each cell layer of the flask, allowing gas exposure to a large surface area within the flask (Fig. 3).

HYPERFlask compared to standard T175 flask. The HYPERFlask (left) consists of 10 polystyrene growth surfaces each treated with CellBIND. The 10 layers have a combined surface area of 1720 cm2. Between each of the 10 interconnected growth surfaces is an air gap that allows passive gas exchange. The flask is barcoded and compatible with the SelecT. The standard T175 flask (right) has a growth surface area of 175 cm2 and gas exchange occurs through a vented cap.
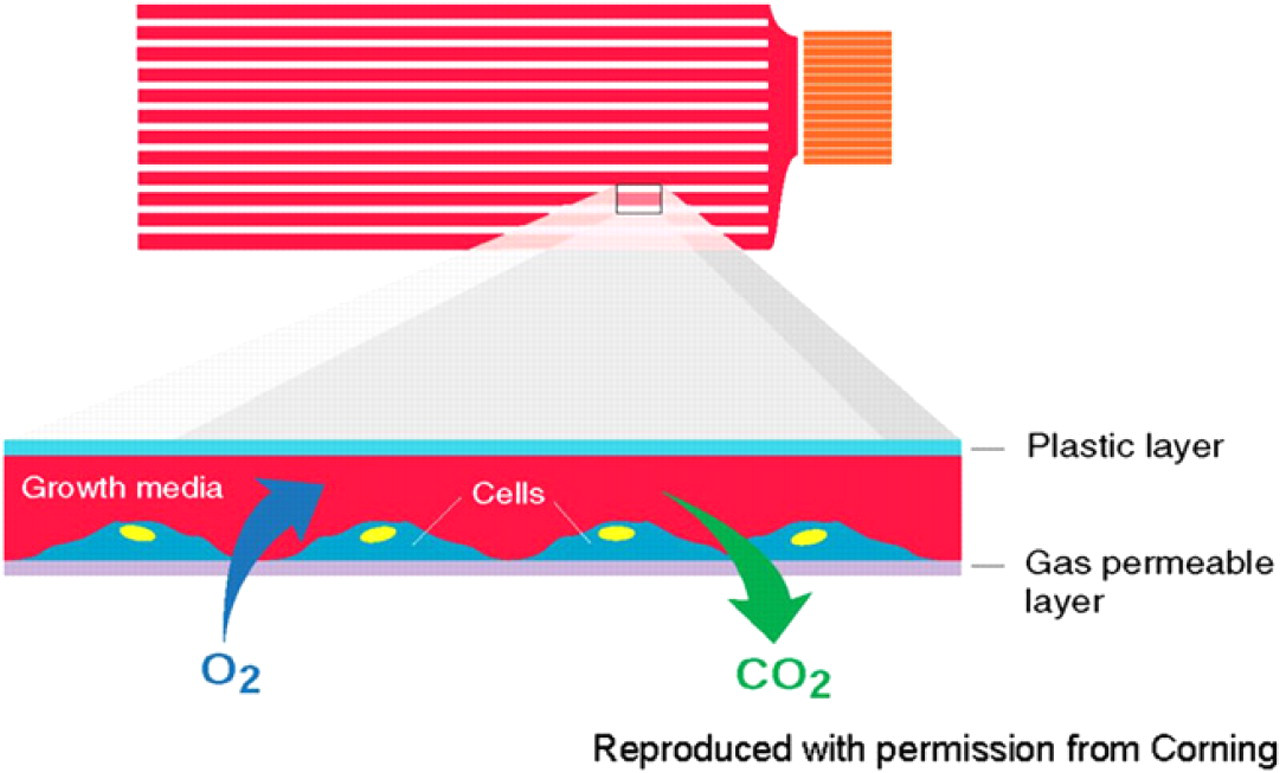
Diagram of gas exchange in a HYPERFlask. The cells grow on an ultra thin gas-permeable layer. Between each layer is an air space that allows gas exchange along the length of the flask. Each of the individual layers is known as a “cassette.”
Here, we demonstrate that the HYPERFlask is capable of producing approximately 10 times the number of cells when compared to a standard T175 flask. Furthermore, the quality of the cells grown in the HYPERFlask was equivalent to that of cells grown in a standard T175 flask when assessed by viability and assay performance in functional cell-based uHTS assays.
Materials and Methods
Cell Lines
Three recombinant Chinese hamster ovary (CHO) cell lines and a recombinant human embryonic kidney (HEK 293) cell line expressing various targets of interest were generated in house. A nonrecombinant HeLa S3 cell line was obtained from American Type Culture Collection (ATCC) (Manassas, VA).
Cell Culture Conditions
Manual validation of the HYPERFlask was performed using a recombinant CHO cell line that overexpressed an enzyme of interest (CHO #1). Cells were maintained in F12K, 10% fetal bovine serum (FBS), 1% penicillin/streptomycin, 0.5 mg/mL hygromycin, and 0.25 mg/mL Geneticin. A second recombinant CHO cell line, that overexpressed a target enzyme (CHO #2) was grown in Iscove's Dulbecco's modified eagle medium (DMEM), 10% FBS, 2 mM l-glutamine, 1 mg/mL Geneticin, and 1 × sodium hypoxanthine and thymidine (HT) supplement for automated validation. The functional validation was performed with CHO cells stably transfected with a nuclear receptor fused to a ProLabel tag (CHO #3). This cell line was maintained in F-12 Nutrient Mixture, 10% FBS, and 1% penicillin/streptomycin/l-glutamine. HeLa S3 cells were grown in DMEM supplemented with 10% FBS and 1% penicillin/streptomycin. HEK 293 cells were grown in DMEM, 10% FBS, and 1 mg/mL Geneticin.
All cell lines were grown at 37 °C in a humidified environment containing 5% CO2. All incubation steps contained in protocols below were performed in this environment. The following products were purchased from Invitrogen Corporation (Carlsbad, CA): FBS (catalog #10082), penicillin/streptomycin (catalog #15140), hygromycin (catalog #10687), Geneticin (catalog #10131), Iscove's DMEM (catalog #12440), L-glutamine (catalog #25030), HT supplement (catalog #11067), F-12 Nutrient Mixture (catalog #11765), penicillin/streptomycin/l-glutamine (catalog #10378), and DMEM (catalog #11965). F12K was purchased from ATCC (catalog #30–2004). For dissociation, Accutase was purchased from Innovative Cell Technologies (San Diego, CA) (catalog #AT-104). Trypsin (catalog #25300) and Trypsin LE (catalog #12605) were both purchased from Invitrogen Corporation.
Manual Protocols
Automated Protocols
Functional Validation
Recombinant CHO cells stably expressing a human nuclear receptor fused to a ProLabel tag (CHO #3) were grown in a HYPERFlask and in T175 flasks using identical seeding densities to compare the functionality of the cells.
In 3456 well assays, compounds were preplated in 5 nL drops before the addition of cells and agonist. This was followed by a 3-h incubation at 37 °C in a humidified atmosphere containing 5% CO2. Detection reagent was added and plates were incubated 1 h before reading on the ViewLux (PerkinElmer Life and Analytical Sciences, Inc., Waltham, MA) for a 90-s exposure.
Results
Manual Validation of HYPERFlask
The recombinant CHO cell line (CHO #1) was selected for these experiments as an example of a robustly adherent cell line with a doubling time representative of a typical cell line used in uHTS assays.
Two HYPERFlasks were each seeded with 1.7 × 107 cells at a concentration of 9.9 × 103 cells/cm2. The cell suspension was harvested from the HYPERFlask and counted using the Cedex counter on the SelecT. Yields of 3.5 × 108 and 3.8 × 108 cells were obtained with 99.2% viability in both cases, based on Trypan Blue exclusion. This yield was compared to the same cell line, seeded at equivalent density, grown in a T175 flask and harvested on the SelecT (Fig. 4). The T175 yielded 3.0 × 107 cells. In this experiment, the HYPERflask gave a 12-fold increase in yield when compared to the T175 flask.
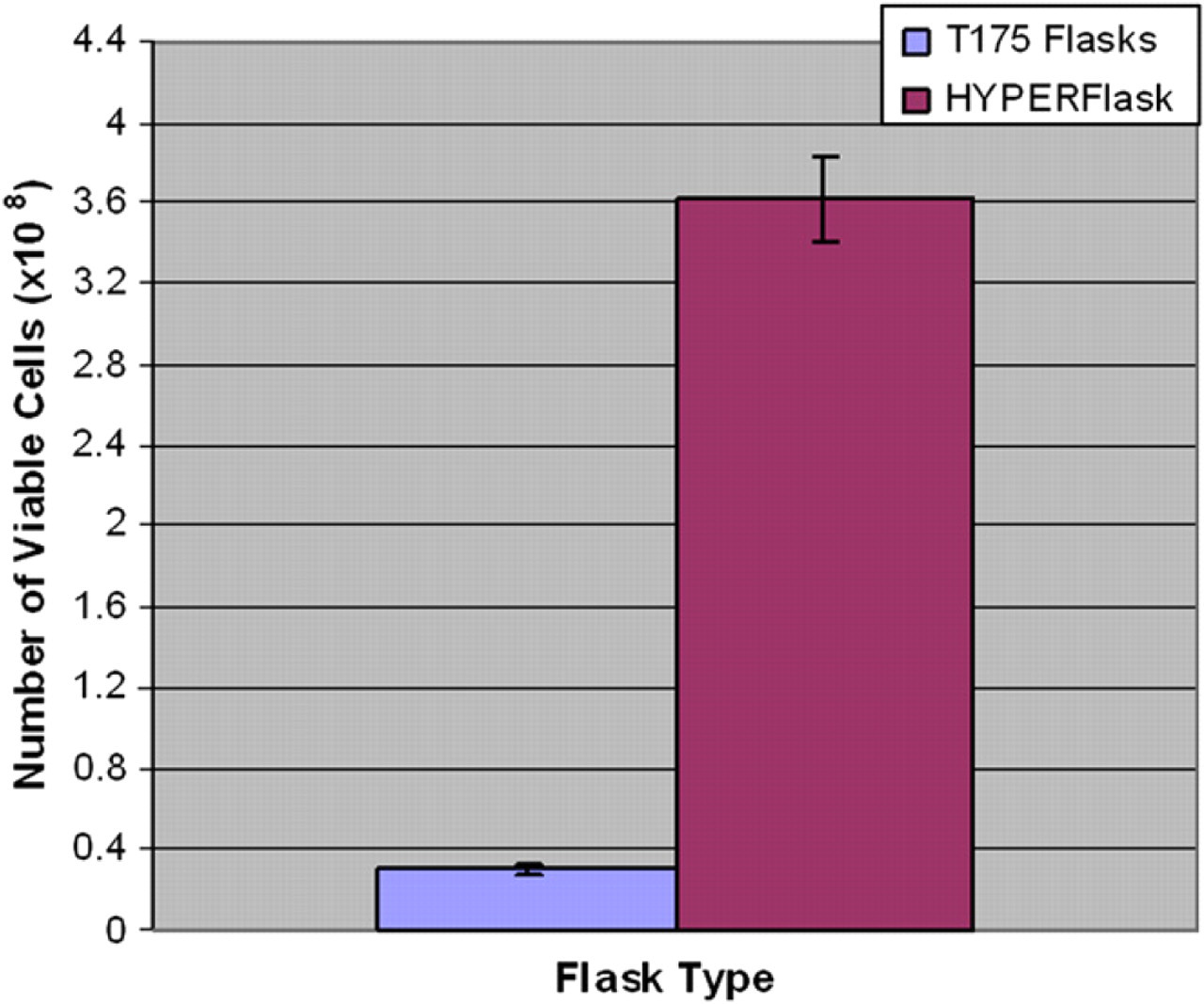
In manual protocols, cells harvested from HYPERFlask show greater than 10-fold cell yield when compared to a T175 flask. Yields of viable recombinant Chinese hamster ovary (CHO) cells (CHO #1) harvested from a HYPERFlask (
Automated Validation of HYPERFlask
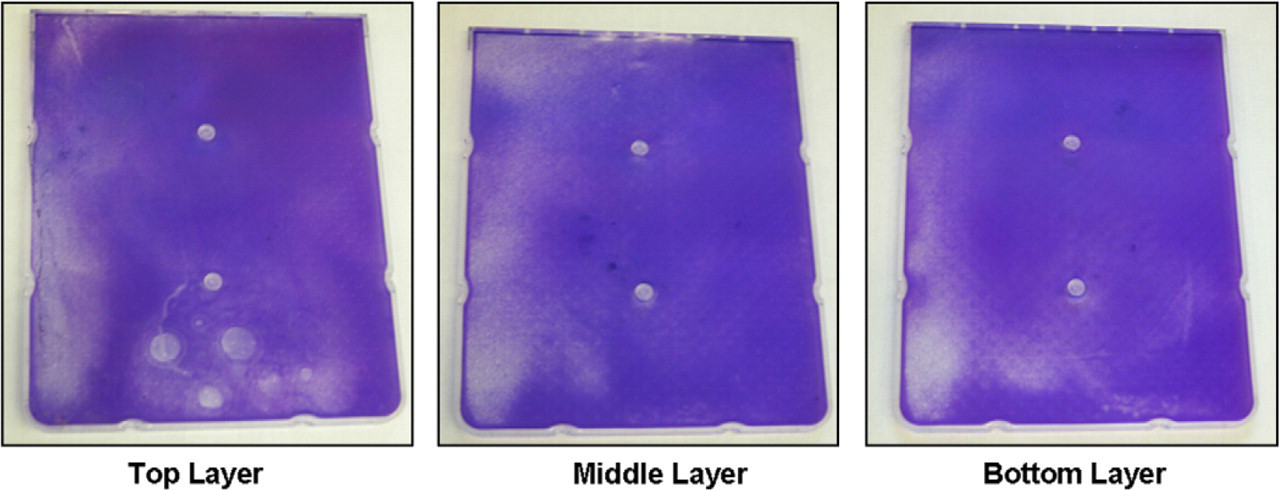
Homogeneous distribution of cells is illustrated by Crystal Violet Staining of HYPERFlask cassettes. HeLa cells were seeded at 25,000 cells/cm and incubated for 3 days in a HYPERFlask. The cells were stained using Crystal Violet, then cassettes were separated at the manifold and examined visually and under 10× magnification. Crystal Violet staining demonstrates that cells were evenly seeded using SelecT.

Cell lysing captured during cell count of Chinese hamster ovary cells on SelecT using Cedex. This image illustrates that cell lysing was occurring when cells from a HYPERFlask were dissociated with TrypLE. Circle A shows healthy cells. Circle B highlights genomic DNA resulting from cell lysis.
When cell lines are not robust under dissociation with TrypLE, we have previously used Accutase successfully. Accutase was therefore tested in automated HYPERFlask protocols and cell lysing was no longer observed.
Yields and Viability from HYPERFlask
Comparison of cells grown in T175 flasks, TripleFlasks, and HYPERFlasks

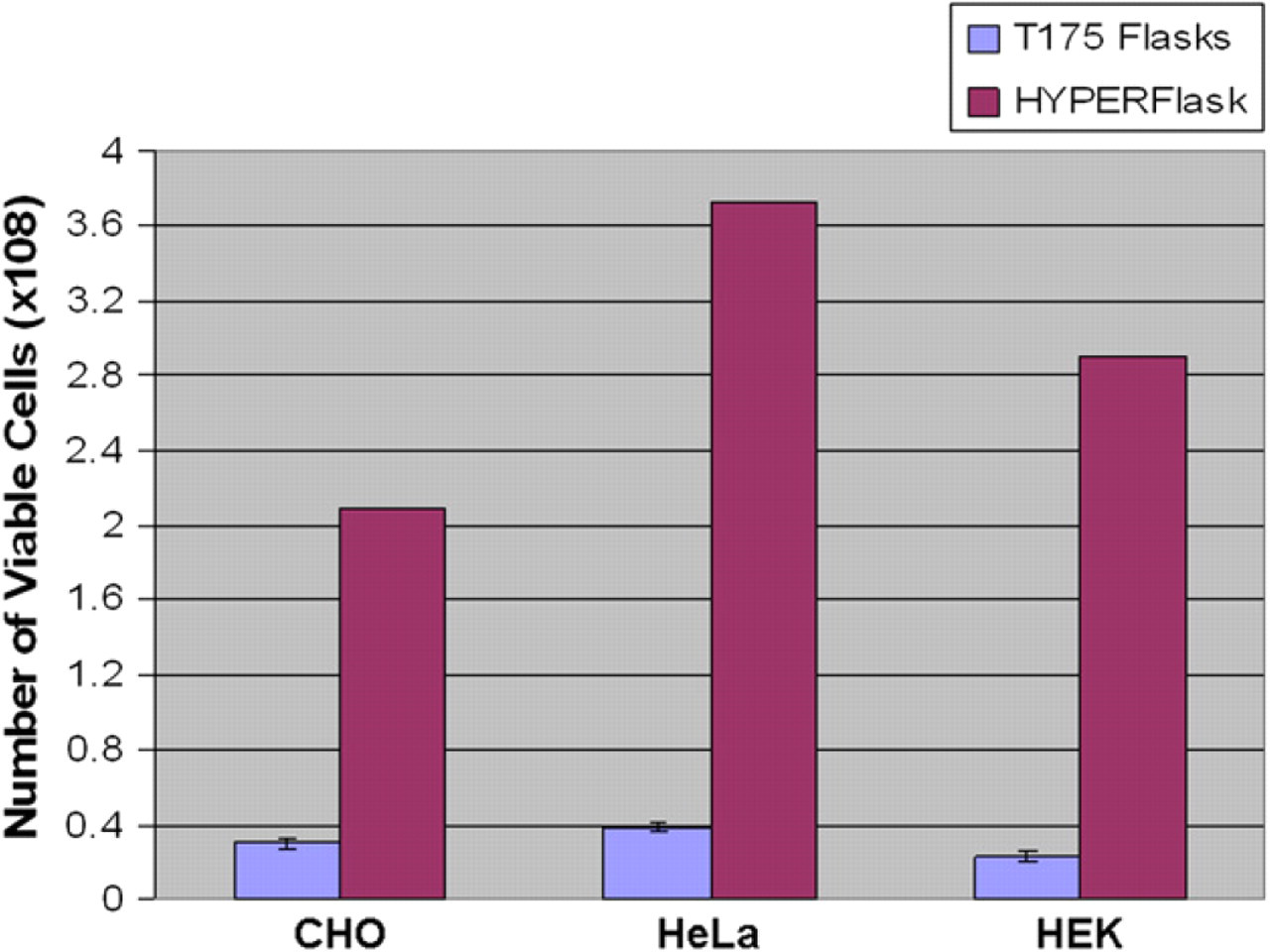
Comparison of cell yields from a range of cell lines grown in HYPERFlasks or T175 flasks. A variety of cell lines were harvested from HYPERFlasks and T175 flasks. In all cases, a significant increase in yield was observed when cells were grown in HYPERFlask.
The Nunclon TripleFlask (Nalge Nunc International, Rochester, NY) was also evaluated in this comparison, because this flask was the original solution for high density automated cell culture on SelecT. The Nunclon TripleFlask is a 500-cm2, three-layered flask with similar footprint to a T175. Using automated protocols, two recombinant CHO cell lines were grown and harvested from HYPERFlasks and yields compared with those obtained from Nunclon TripleFlasks. Yields from HYPERFlask averaged threefold higher than those obtained from Nunclon Triple-Flasks, with comparable high viability of harvested cells (Table 1).
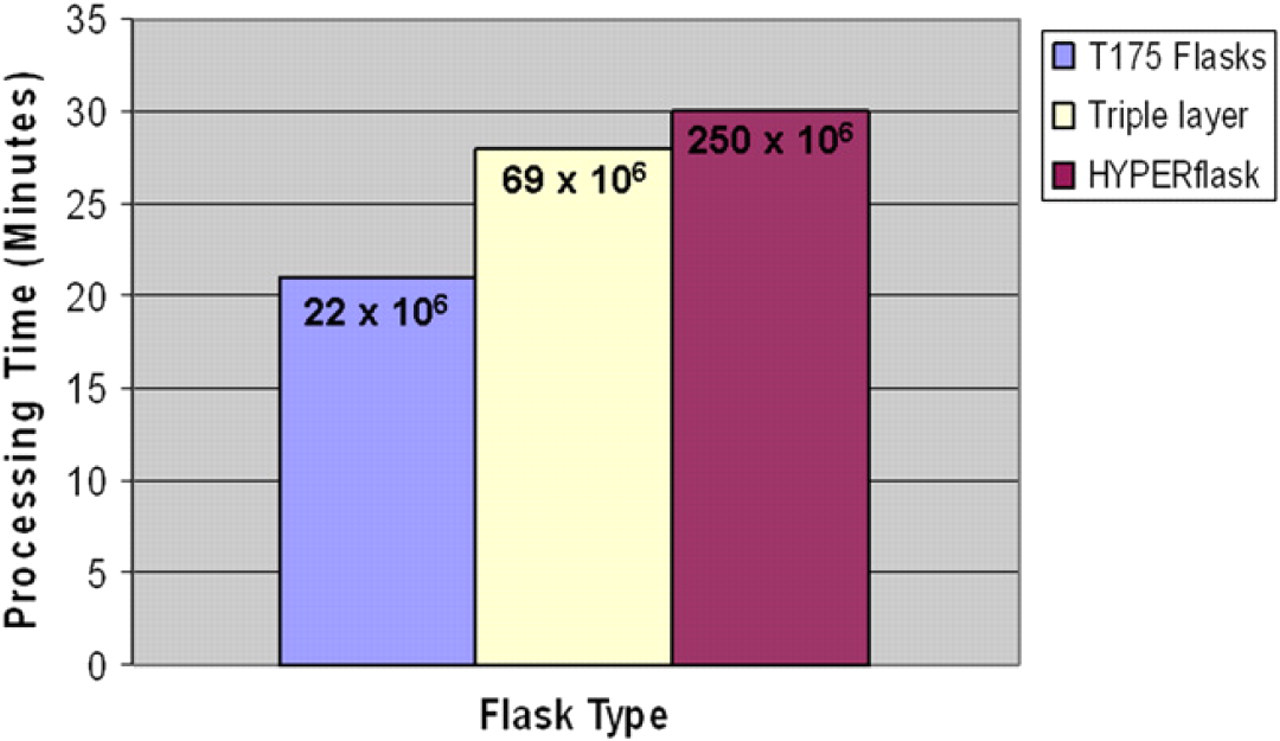
Comparison of harvesting times and cell yields. Chinese hamster ovary #3 cells were seeded at 30,000 cells/cm into three flask types and incubated for 3 days. Flasks were harvested on SelecT, and harvesting time and cell yield compared. A T175 flask takes the shortest amount of time to harvest, at 22 min, however the HYPERFlask takes just 8 min longer with a ∼ 10-fold increase in cell yield.
Functional Validation
To compare the function of cells grown in HYPERFlasks with those grown in T175s in automated cell culture, we implemented a functional translocation assay using the recombinant CHO cell line (CHO #3). Each population of cells was characterized in terms of signal-to-background ratio of control agonist. EC50 values of control and reference agonists were also compared. In addition, we monitored the consistency of these parameters over time as cells were stirred by a stir bar in a collection flask (stir flask) at room temperature before assay.
During uHTS, it is considered advantageous that the cells are stable in their response for 3–4 h in a stir flask after dissociation and before plating for assay. To test functional stability, cells were plated and assayed immediately after dissociation (
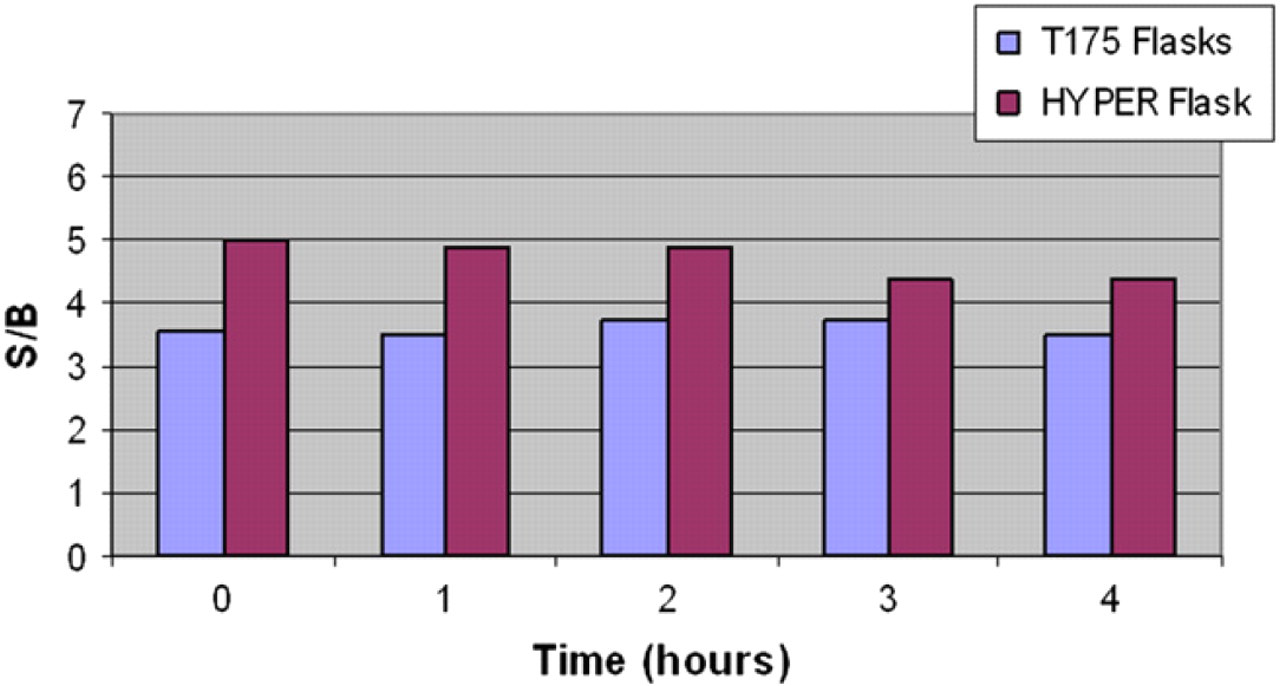
Stability test with cells cultured in T175 flasks (blue) or HYPERFlask (red). Cells from a T175 flask and a HYPERFlask were tested for functional stability over a 4-h period. Cells were plated in 3456 well plates and assayed at hourly intervals using the Path-Hunter assay according to Methods. The signal-to-basal ratio is plotted as a function of time (h).
In addition to evaluating the function of cells grown in the HYPERFlask and T175 with a single concentration of agonist, we performed dose–response titrations of several reference compounds. Figure 10 illustrates that we observed comparable potencies and efficacies for all compounds tested.
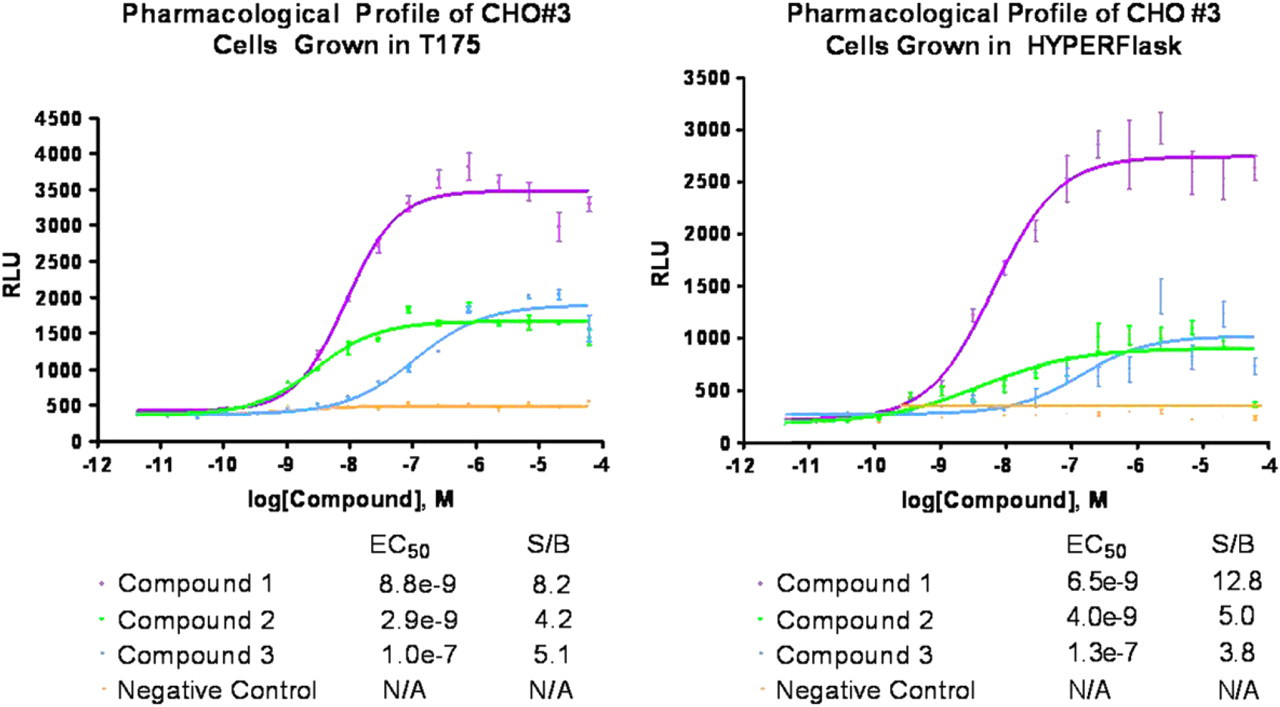
Dose–response curves of control compounds (agonist, antagonist, and partial agonist) using cells from T175 (left) and HYPERFlask (right). A 16-point dose titration for each compound was performed in a white 3456 well plate using the PicoRaptr (Aurora Discovery). Cells and PathHunter reagents were added according to Methods. Luminescence was read using the ViewLux.
Discussion
We have demonstrated that automation of the HYPERFlask is a viable solution to satisfying the requirements of uHTS for the supply of large quantities of cells without compromising cell quality. In addition, for some cell types, the flask exceeded the 10-fold increase in cell yield predicted by the approximately 10-fold increase in surface area of HYPERFlask when compared to a T175 flask. One possible explanation for this increased yield may lie in the novel architecture of the flask. Gas exchange occurs below the entire growth surface of each cassette of the HYPERFlask. This is likely to allow highly efficient gas exchange, supporting high cell numbers. In comparison, gas exchange in a T175 flask is via five small holes on the vented cap. An additional feature of the HYPERFlask is a CellBIND treatment on cell culture surfaces that increases the flask's wettability. 25 This may also contribute to increased yield of cells by maintaining cell contact and adherence during culture.
In validating a range of cell types and morphologies, we addressed the variety of cell lines commonly used in uHTS screens. CHO K1, HeLa S3, and HEK 293 cells have all been grown and harvested successfully. Some optimization was necessary for these cell types, particularly in the cell dissociation step. Although the cell lines described here were not amenable to dissociation with TrypLE, this cannot be assumed for all cell types and we would consider TrypLE an appropriate reagent for dissociating cell lines that may have a tendency to clump, or are extremely adherent and require long incubation protocols.
At higher cell densities, clumping of harvested cells emerged as an issue that was subsequently resolved by seeding and harvesting cells at lower cell density. Further optimization of dissociation conditions or off-line resuspension of harvested cells may also address this issue. One observed limitation of the SelecT is that trituration protocols are restricted to the use of a 10-mL pipette, at a relatively slow pipetting speed of 5 mL/s. Because HYPERFlask harvest protocols generate large cell volumes that may contain clumps, this on-line trituration method may not adequately break up clumps for some cell types.
This does raise the question of applications of these flasks for cell lines with more challenging morphologies, particularly cell types such as the hepatocellular carcinoma cell line HepG2 that are prone to clumping using standard tissue culture techniques. In the cell types investigated so far, this has not been an issue. Although decreased cell density in the HYPERFlask may compromise yield, transition of a cell line into HYPERFlask can still be considered, because savings will still be realized in improved SelecT incubator capacity and process times.
Another important advantage driving application of the HYPERFlask to uHTS is the rapid preparation time for harvesting. In cell-based assays, cells are continuously stirred in plating protocols and this is a key parameter used in designing cell delivery schedules for screening. Minimizing the time between cell dissociation and plating is desirable and is predicted to result in assays requiring fewer batches of cells. Benefits to cell culture workflow are immediate—fewer cell deliveries per assay day will be required. Decreasing variables in assay protocols is always desirable, thus increased assay stability should also be an important downstream improvement.
Considerations in choosing cell lines that would benefit from culture in the HYPERFlask center around acceptable yields. Assuming a 10-fold increase in yield, protocols should require seeding from three or fewer flasks. Once this threshold is reached, multiple T175 flasks would need to be maintained on the SelecT for seeding of HYPERFlasks. This would lead to a substantial occupation of incubator space for scale-up and would not be an efficient use of the HYPERFlask. Similarly, compromises in yield should be limited—a sevenfold increase when compared to a T175 flask is acceptable, in most cases. Anything less than this would indicate moving cell culture into Nunclon TripleFlasks as an alternative approach.
The advantages and applications of high density cell culture in HYPERFlask are numerous. The implementation of HYPERFlask in combination with cell culture automation using the SelecT has allowed us to achieve the standard of quality control desirable for uHTS. In addition to live cell assays, we plan to use automated high density cell culture to generate large batches of cells for preparation of frozen vials to support frozen cell assays. This will minimize the number of batches of frozen cells in assay protocols. Similarly, high volumes of cells required for membrane preparations are appropriately supported by high-density cell culture, releasing significant incubator capacity for other processes and allowing more efficient use of our automated cell culture system.
It can be noted that, to date, the SelecT is the only automated cell culture system capable of handling the HYPERFlask. There is one commercially available alternative to SelecT, the Cellerity, an automated cell culture system manufactured by Tecan (Mannedorf/Zurich, Switzerland). 27 The Cellerity automation is limited to the use of RoboFlasks for cell culture. 25 RoboFlasks have dimensions that conform to Society for Biomolecular Screening format; therefore, the Cellerity is incapable of using the HYPERFlask.
In general, any process requiring large cell numbers that are sensitive to process time or batch-to-batch variability would benefit from the enhanced quality control of automated cell culture and would be a candidate for implementation of HYPERFlask automation on the SelecT.
Acknowledgments
We are grateful to Matt Golding and Stephen Guy (The Automation Partnership), Todd Upton (Corning), and Julie Kerby and Mathew Leveridge (Stem Cell Sciences), for their contributions to the protocols described in this manuscript.
