Abstract
The CompacT SelecT is the latest generation automated cell culture system in the SelecT product line allowing incubation of up to 90 T-175 flasks and preparation of 210 assay-ready plates. We have successfully implemented the CompacT SelecT in support of a number of cell-based assays used in our Alzheimer's disease (AD) lead optimization programs. One of the distinguishing features of AD pathology is deposition of two neurotoxic forms of the beta-amyloid peptide (Aβ40 and Aβ42) in the brains of patients. It is thought that specifically lowering Aβ40 and Aβ42 in the brains of patients will halt the progression of the disease. The generation of Aβ requires sequential cleavage of the type-I integral membrane amyloid precursor protein (APP) by two proteases, β-secretase (BACE) then γ-secretase. In the specific examples presented here, we have transitioned two cell lines supporting drug discovery efforts for identifying β- and γ-secretase inhibitors (GSIs) from manual cell culture protocols to fully automation using CompacT SelecT. In Chinese hamster ovary (CHO) cells which over express wild-type APP (CHOAPP cells), robust secretion of Aβ40 was observed from cells cultured manually and with CompacT SelecT with signal:background ratios of 54–99 and 23–47, respectively. Despite the reduced signal:background observed with the CompacT SelecT cultured cells, the rank order of potency for a series of 18 BACE inhibitors in reducing Aβ40 secretion was identical when manually cultured cells were compared with CompacT SelecT cultured cells. The correlation coefficient when comparing the two sets of EC50 values was r 2 = 0.99. Similarly, CHO cells that over express a reporter construct for the Swedish mutant APP that generates high levels of Aβ peptide (CHOAPP-NL cells) cultured with CompacT SelecT exhibited a reduced secretion of Aβ40 and Aβ42 when compared with manually cultured cells; signal:background ratios for secretion of Aβ40 from CHOAPP-NL cells were 57–71 and 16–31, and for Aβ42 secretion 8–18 and 4–11 comparing manual and automated cell culture, respectively. Similar to the observations in CHOAPP cells, an identical rank order of potency for a series of GSIs was observed when CHOAPP-NL cells cultured manually and in CompacT SelecT were compared; r 2 = 0.99 and 0.98, respectively, for EC50 values for inhibition of Aβ40 and Aβ42 secretion. Taken together, the robust signal:background and identical pharmacological profile of inhibitors provide validation for the implementation of automated cell culture for CHOAPP and CHOAPP-NL cells in support of β- and γ-secretase-targeted drug discovery.
Introduction
Adoption of cell-based assays in modern drug discovery has increased dramatically in recent years. These assays have been implemented at all stages of the drug discovery process from hit identification to lead optimization and in compound safety profiling. A key driver for this transition has been the desire to work in a cellular context as early as possible to gain associated benefits such as a more physiological environment, ability to look at compound mechanism(s), and an early assessment of the ability of a compound to cross cellular membranes and gain access to intracellular targets. Membrane-associated proteins such as G-protein-coupled receptors and ion channels are ideally suited to cell-based assay approaches to take advantage of their signaling properties for configuration of high-fidelity assays of receptor function. This has also provided the ability to detect receptor activators, or agonists, positive allosteric modulators, and antagonists with the same assay format and often measured with sequential compound addition protocols in a single well.
Coincident with the increased adoption of cell-based assay approaches has been the increased demand placed on the cell culture facility to provide high-quality cells in ever-increasing numbers. Strategies to boost productivity of conventional cell culture have included brute force to generate large numbers of flasks or trays, use of suspension-adapted cells or adherent cells grown in flasks with multiple or stacked surfaces. A major innovation has been the development of fully automated cell culture platforms capable of maintaining, passing, and ultimately plating cells directly into cell-based assay-ready micro-titer plate formats. 1,2 The first such platform to be introduced was the SelecT, developed by The Automation Partnership in collaboration with pharmaceutical companies. Most recently, the CompacT SelecT with a reduced capacity compared with the original platform allowing for the incubation of up to 90 T-175 flasks, and preparation of 210 assay-ready plates has been introduced. We have successfully implemented the CompacT SelecT for automated cell culture with a diverse number of cell types. In this study, we describe the validation of automated cell culture supply of two CHO cell lines expressing amyloid precursor protein (APP) constructs in support of Alzheimer's disease (AD)-targeted drug discovery.
Materials and Methods
Manual Cell Culture
Cells stably expressing either wild-type APP (CHOAPP cells) or an APP reporter construct containing the Swedish KM/NL mutation (CHOAPP-NL cells) 3 were maintained in Dulbecco's Modified Eagle Medium (GIBCO Cat#: 11995) containing 1 × nonessential amino acids, 10% fetal bovine serum (maintenance media) supplemented with 1 × antibiotic/antimycotic, and 200 μg/mL of G418. The cells were passaged initially at a 1:100 dilution each week. For compound assay, CHOAPP and CHOAPP-NL cells were seeded at a density of 1 × 103 cells per well in 96-well plates and allowed to grow for an additional 3 days at 37 °C, 5% CO2, until 80–90% confluence was obtained.
Automated Cell Culture
Before automated cell culture, both CHOAPP and CHOAPP-NL cells were grown in bulk in a Corning 10-CELLSTACK culture chamber, harvested by trypsinization, and frozen in vials at a density of 5 × 106 cells per vial to maintain passage consistency for downstream assays. For each cell line, cells of a frozen vial were suspended to a T-175 flask with 30 mL of maintenance media containing 1 × Penicillin/Streptomycin, and the flask was supplied to the CompacT SelecT. Subsequently, a CompacT SelecT running protocol was performed for seeding 96-well plates. The protocol included cell counting using a built-in cell counter CEDEX, diluting the cell suspension to appropriate seeding concentrations, and plating in 100 μL final volume in 96-well assay plates. CHOAPP and CHOAPP-NL cells were diluted to 1.5 × 104 cells/mL and 3 × 105 cells/mL, respectively, before plating due to their different growth rates. The plates were then incubated in the CompacT SelecT plate incubator at 37 °C, 5% CO2 for an additional 3 days. This process of thawing a vial, suspending cells in a T-175 flask, and plating assay plates was repeated for each batch of plates produced.
Compound Assays
β-Secretase (BACE) inhibitors and γ-secretase inhibitors (GSIs) were prepared as 20 mM stock solutions in dimethylsulphoxide (DMSO). Medium was removed from cells growing in 96-well plates, cells were washed with UltraCULTURE media (Cat#: 12–725F, Lonza, Walkersville, MD) and fresh assay medium (UltraCULTURE media containing 25 mM HEPES and 2 mM l-GlutaMax) was added to cells. Compounds were serial diluted and then added to the assay medium. Cells were incubated with compounds for 22 h, the conditioned media (CM) was collected and assayed for Aβ levels as described below. Cell viability was tested using a CellTiter 96 Aqueous Non-Radioactive Cell Proliferation Assay (MTS: Promega, Madison, WI) according to manufacturer's instructions. Assay reagent was added to cells and incubated for 1 h. Plates were then read on a plate reader at 492 nm.
Aβ Measurement
Levels of Aβ40 and Aβ42 in CM were measured in an Aβ40/42 end-specific assay using a Meso-Scale Discovery (MSD) electro-chemiluminescent platform (MSD, Gaithersburg, MD). For CHOAPP cells, CM was analyzed undiluted. For CHOAPP-NL, CM was diluted 1:10 for Aβ40 measurement and 1:2 for Aβ42 measurement, and was mixed with biotinylated monoclonal 6E10 (Signet Labs, Dedham, MA), a rabbit anti-C-terminal specific antibody (Biosource International, Camarillo, CA), and a Ruthenium-conjugated anti-rabbit antibody (MSD, Gaithersburg, MD) for detection. This mixture was incubated at 4 °C over night in streptavidin-coated 96-well plates (MSD, Gaithersburg, MD). The next day, the plates were washed, a proprietary read buffer was added (MSD, Gaithersburg, MD) and the plates were read in an MSD SI 6000 plate reader. Cellular Aβ levels were determined using synthetic Aβ40/42 peptide (AnaSpec, San Jose, CA) standard curves.
Results
Absolute concentrations of Aβ40 and Aβ42 in CM from both CHOAPP-NL and CHOAPP cells were significantly lower when cells were plated using the CompacT SelecT method than when cells were prepared using manual methods. Media from CompacT SelecT-plated CHOAPP-NL cells contained 1.87 ± 0.64 ng/mL Aβ40 and 0.15 ± 0.01 ng/mL Aβ42: this is a 62% reduction of Aβ40 levels and 42% reduction of Aβ42 when compared to CM from manually prepared cells, which contained 4.96 ± 0.32 ng/mL Aβ40 and 0.26 ± 0.03 ng/mL Aβ42. Similarly, media from CompacT SelecT-plated CHOAPP cells contained less Aβ40 than CM from manually prepared cell cultures (0.59 ± 0.14 ng/mL for CompacT SelecT-plated cells vs. 1.18 ± 0.12 ng/mL) (Table 1).
Absolute concentrations of Aβ40 and Aβ42

As a consequence of reduced absolute levels of Aβ, the signal-to-background ratio, a key quality control parameter for Aβ detection assays was also reduced. The signal-to-background ratios for Aβ detection in CM from CHOAPP-NL cells were reduced from 58 for Aβ40 and 12 for Aβ42 for manually prepared CHOAPP-NL cells to 25 for Aβ40 and 6 for Aβ42 for CompacT SelecT prepared cells. The signal-to-background ratio for Aβ40 detection in CM from CHOAPP cells was similarly reduced (from 54–99 in range, 71 in average for manually prepared cells, to 23–47 in range, 35 in average for CompacT SelecT prepared cells) (Table 2).
Aβ40 and Aβ42 signal-to-background ratios of CHOAPP-NL and CHOAPP cells
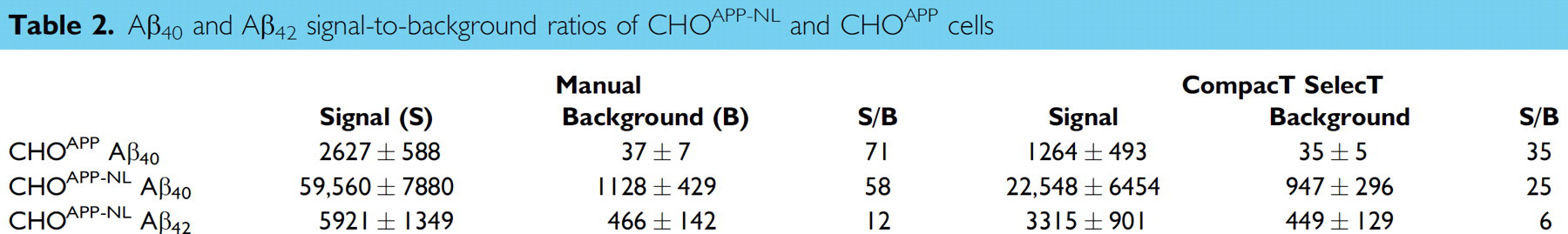
Irrespective of the reduced Aβ generation, signal-to-background ratios remained well within assay criteria (γ-secretase Aβ40 ≥ 25; Aβ42 ≥ 5; and BACE Aβ40 ≥ 35), and EC50 values for both BACE and GSIs were closely correlated regardless of method of cell preparation. When 17 BACE inhibitor compounds were tested in assays on CHOAPP cells, the correlation coefficient for the EC50 values for Aβ40 reduction was 0.9993 (Fig. 1) when assay results from CompacT SelecT and manually prepared cells were compared. Similarly, when 16 GSIs were tested on CHOAPP-NL cells the correlation coefficient for EC50 values for Aβ40 reduction was 0.9958 (Fig. 2), and correlation coefficient for the EC50 values for Aβ42 reduction was 0.9835 (Fig. 3). Furthermore, rank ordering of compound potency was almost always retained regardless of the method of cell preparation (Tables 3 –5).
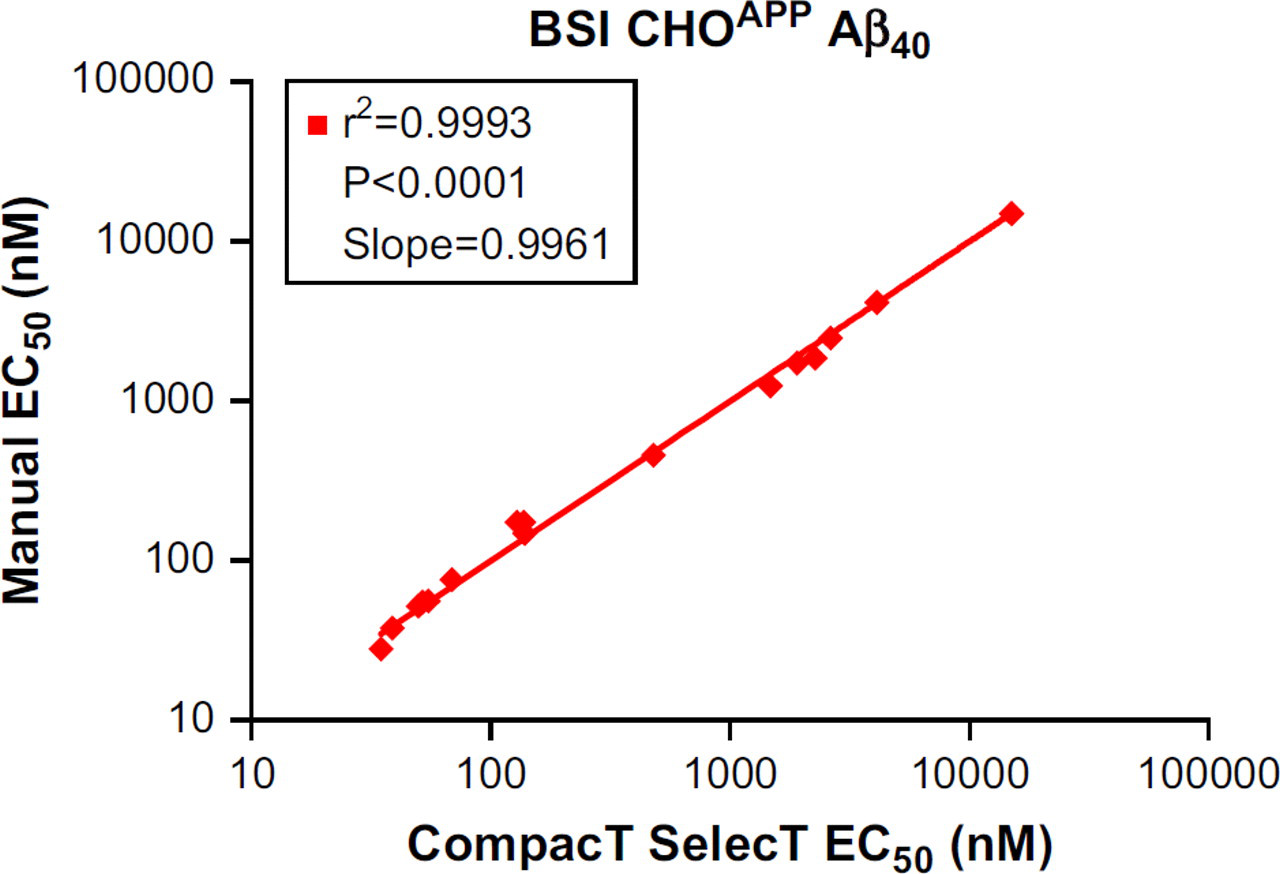
Correlation of EC50 values of β-secretase inhibitors (BSI) for reduction in Aβ40 levels in CHOAPP cells: CompacT SelecT versus manual.
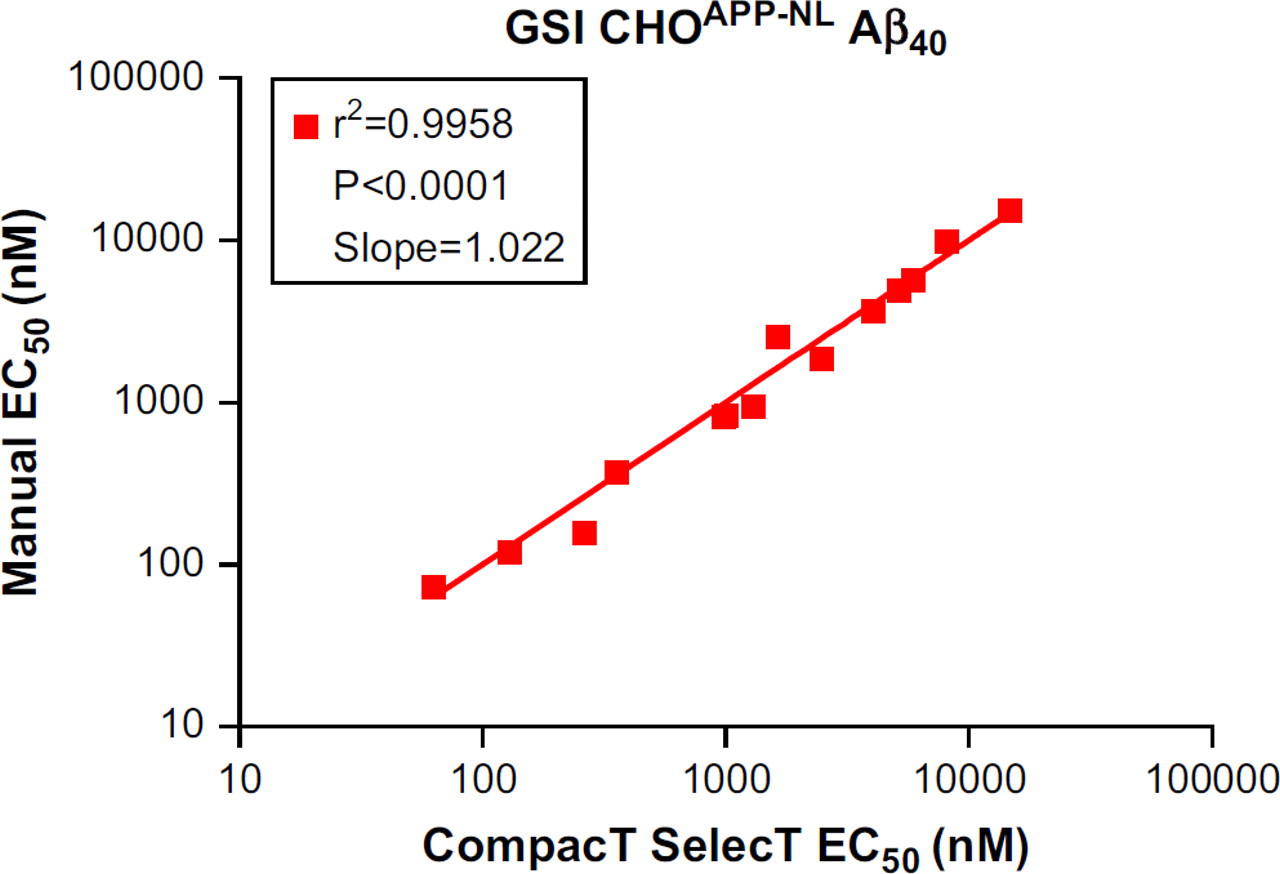
Correlation of EC50 values of γ-secretase inhibitors (GSIs) for reduction in Aβ40 levels in CHOAPP cells: CompacT SelecT versus manual.
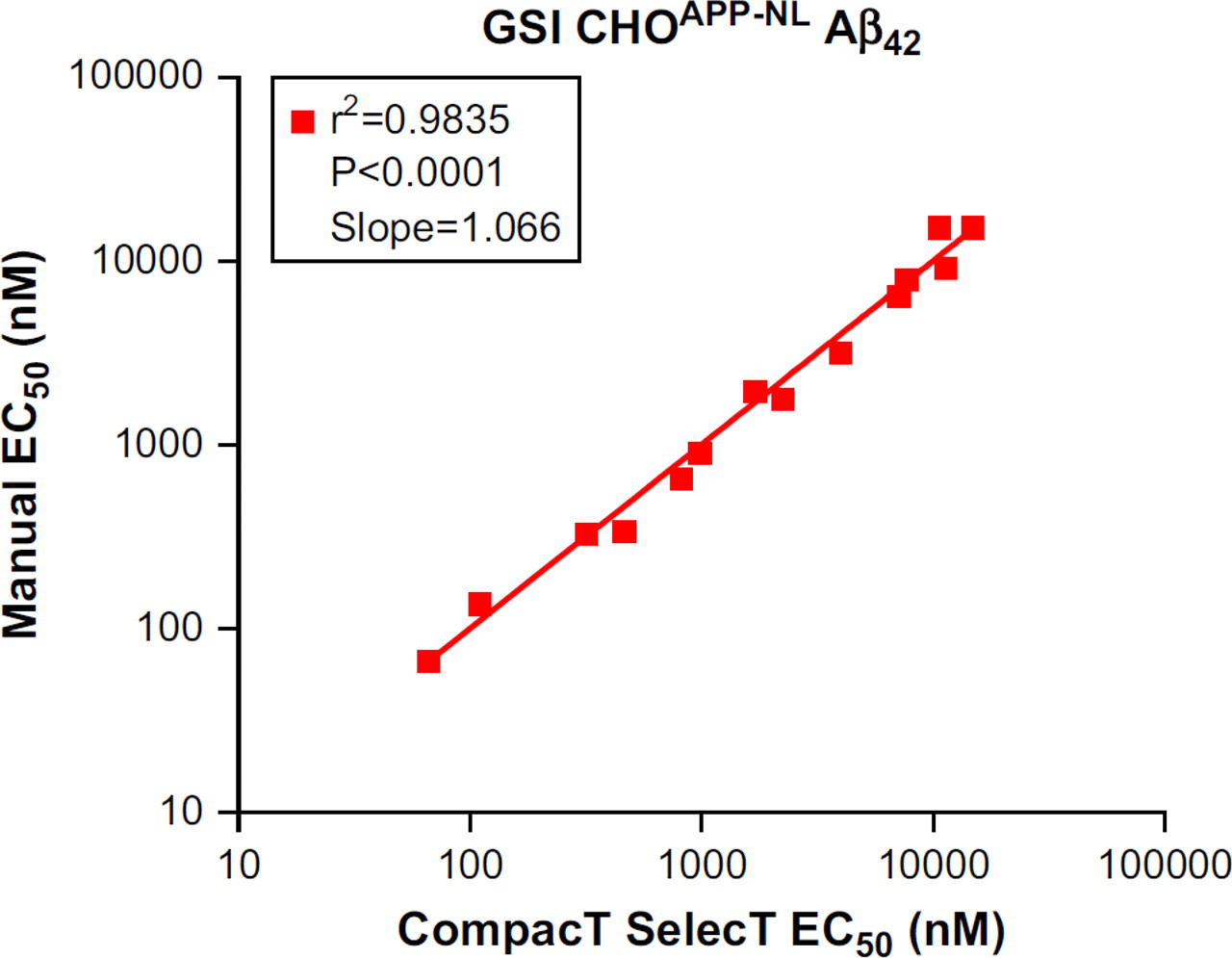
Correlation of EC50 values of γ-secretase inhibitors (GSIs) for reduction in Aβ42 levels in CHOAPP cells: CompacT SelecT versus manual.
Rank order of EC50 values for Aβ40 reduction by BACE inhibitor (BSI) compounds using CHO cells
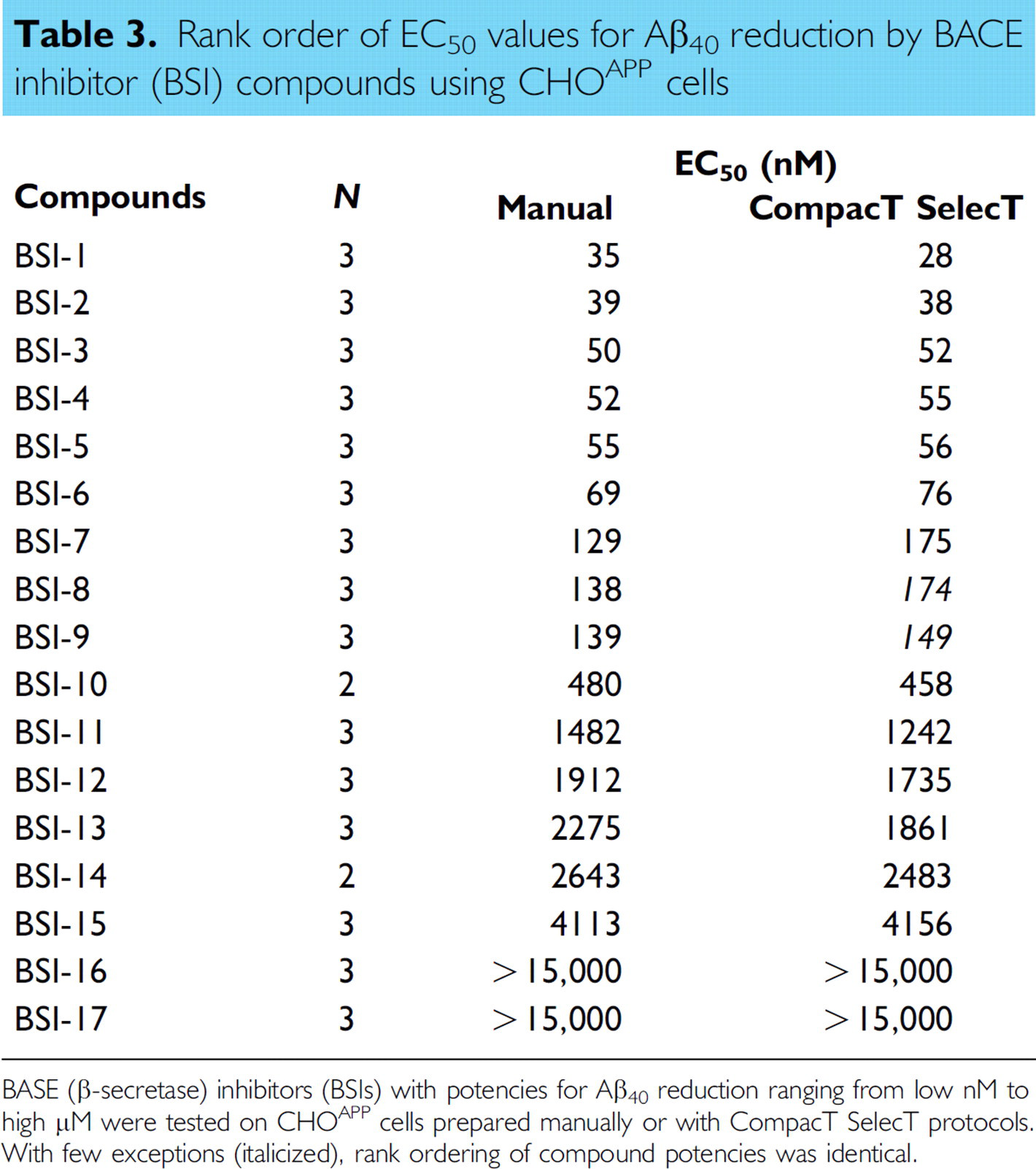
BASE (β-secretase) inhibitors (BSIs) with potencies for Aβ40 reduction ranging from low nM to high μM were tested on CHO cells prepared manually or with CompacT SelecT protocols. With few exceptions (italicized), rank ordering of compound potencies was identical.
Rank order of EC50 values for Aβ40 reduction by γ-secretase inhibitor (GSI) compounds using CHOAPP-NL cells
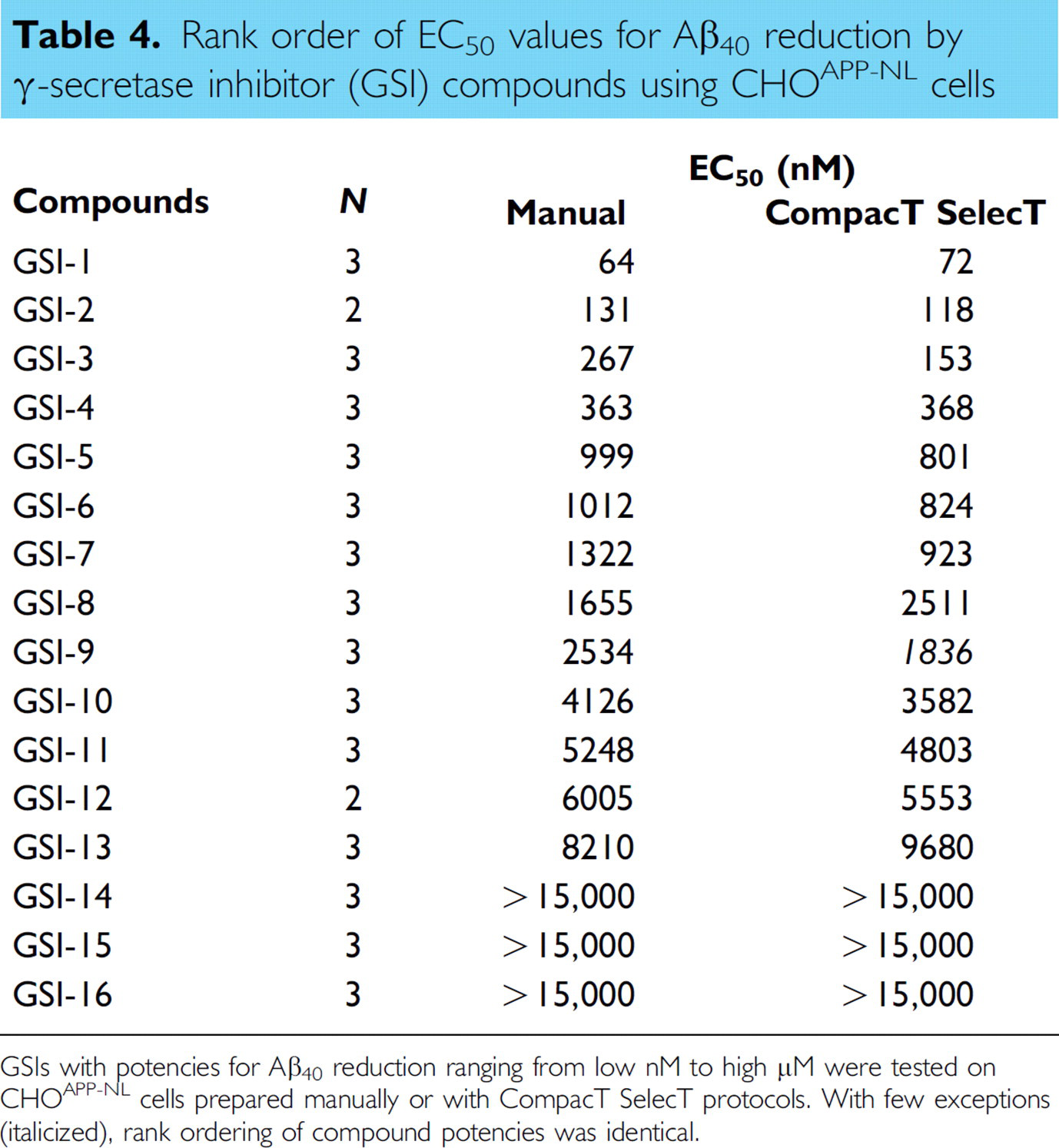
GSIs with potencies for Aβ40 reduction ranging from low nM to high μM were tested on CHOAPP-NL cells prepared manually or with CompacT SelecT protocols. With few exceptions (italicized), rank ordering of compound potencies was identical.
Rank order of EC50 values for Aβ42 reduction by γ-secretase inhibitor (GSI) compounds using CHOAPP-NL cells
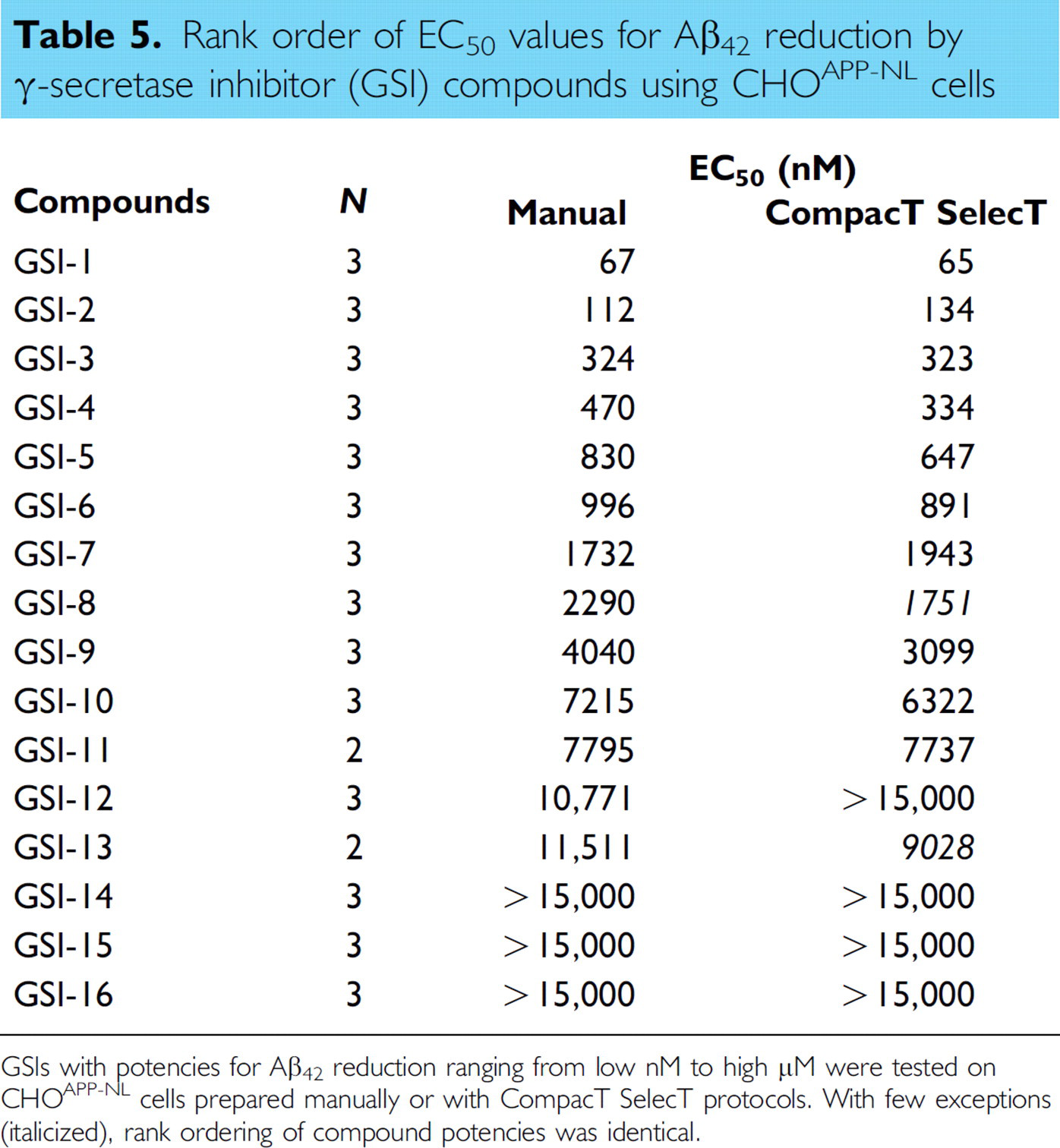
GSIs with potencies for Aβ42 reduction ranging from low nM to high μM were tested on CHOAPP-NL cells prepared manually or with CompacT SelecT protocols. With few exceptions (italicized), rank ordering of compound potencies was identical.
Discussion
To improve the speed, accuracy of plating, and consistency in cell culture for downstream assays necessary in a dynamic and productive research environment, we implemented the use of a CompacT SelecT to fully automate manual cell culture techniques. As cell-based assays have become increasingly more popular in drug discovery efforts, it became necessary to ensure a consistent supply of cells of sufficient quality and quantity. In this article, we present a comparison of the functional assessment of cells grown manually to cells grown in a CompacT SelecT for use in our AD lead optimization program.
AD is a progressive neurodegenerative disorder characterized by Aβ amyloidosis of the brain parenchyma and vasculature. 4,5 The Aβ peptide is derived from the APP by the sequential proteolytic activities of BACE and γ-secretase. Because Aβ pathology is characteristic of AD, and because Aβ is reported to be a neurotoxic peptide, 6 the reduction of Aβ levels by secretase inhibition is a widely accepted therapeutic strategy for AD. 7
We have developed assays to measure effects of γ-secretase and BACE inhibitors on the generation of Aβ in vitro. These assays use cells expressing different APP constructs. For GSIs, we use cells expressing a reporter construct that encodes the Swedish Mutant (KM/NL) APP-751 isoform (CHOAPP-NL). Because γ-secretase is responsible for the C-terminal heterogeneity of Aβ, we measure the effects of GSIs on the generation of both the most abundant Aβ isoform, Aβ40 and the minor Aβ isoform, Aβ42. The mutation in the CHOAPP-NL construct results in the generation of higher levels of total Aβ enabling detection of the minor Aβ42 isoform. However, the Swedish mutation is adjacent to the BACE cleavage site, and the relative potencies of BACE inhibitor compounds might differ based upon enzyme substrate. Therefore, for BACE inhibitors, we use cells expressing a construct that encodes the full-length wild-type sequence of the APP-695 isoform (CHOAPP) and measure the effects of compounds on the generation of the Aβ40 isoform.
These assays are performed weekly and require the plating of fourteen 96-well plates that are both labor intensive and tedious. When we compared manual cell culture with automated cell culture, we found that the absolute concentrations of Aβ40 and Aβ42 in CM from both CHO cell lines were lower when using the CompacT SelecT and, as a consequence of reduced absolute levels of Aβ, the signal-to-background ratio, a key quality control parameter for Aβ detection assays, was also reduced. Despite this overall reduction in Aβ levels and concomitant reduction in the signal-to-background ratio of the assay, the assay was still within acceptable tolerability limits. Furthermore, the EC50 values for both BACE and GSIs were closely correlated regardless of cell culture method and the rank ordering of compound potency was accurately maintained. The automated cell culture method was therefore deemed acceptable for CHO cell production in our Alzheimer's program.
There are several differences between manual and automated cell culture that may have resulted in the differences observed in Aβ production. For example, CompacT SelecT culture was accomplished from frozen vials of cells, which may have needed more time in culture to fully recover Aβ production. Also, CompacT SelecT culture was performed without G418 in the media to minimize pump usage, and maximize the number of cell lines that could be maintained with a common media. Finally, cell seeding densities into 96-well plates were different between manual and automated procedures, which may have resulted in slightly different final cell numbers per well.
Despite these lower Aβ yields in the automated cell culture procedure, the benefits of the automated system were enough to convince us to implement the system. CompacT SelecT enabled us to reduce the time researchers spent passing cells, ensure consistent quality of cells, maintain the same cell passage for downstream assays, increase flexibility in batch size and numbers of cell plates produced, increase flexibility of downstream processing by enabling continuous unattended cell maintenance/plating operation, and provided more accurate cell counts and viability measurements. Full integration of the CompacT SelecT has enabled us to increase production of a diverse number of cell types and to support a greater number of critical drug discovery programs.
