Abstract
Assays that predict passive absorption of orally administered drugs have become increasingly important in the early drug discovery process. A molecule must have the potential to be orally absorbed for the compound to be considered a potential lead candidate. High-throughput assays are needed to meet the increased demand for screening of permeability characteristics. Automation of both noncell-based and cell-based permeability assays is discussed in this paper. The methods use a variety of multiwell filter plates in either 24- or 96-well format. UV/Vis or LC/MS analysis is used to generate data for the automated assays and results are compared to samples prepared manually. Two noncell-based assays: Parallel Artificial Membrane Permeation Assay (Kansey, M.; Senner, F.; Gubernator, K. Physiochemical high throughput screening: parallel artificial membrane permeation assay in the description of passive absorption process.
Introduction
The high cost of drug discovery research combined with the high failure rate of compounds in clinical trials has increased the demand for earlier detection of potentially negative properties. One of the common areas of failure in clinical trials is drug absorption, leading many labs to screen for ADME properties (absorption, distribution, metabolism, and excretion) earlier in the drug discovery process.
Absorption of drugs in the intestine can occur by the following methods: paracellular passive diffusion, transcellular passive diffusion, carrier-mediated transport, ATP-dependent efflux, and metabolic enzymes. 1 , 2 Cell-based drug transport assays, such as those using Caco-2 or MDCK cells, are common functional models for drug absorption. 3 - 5 Both Caco-2 and MDCK cells can be grown on either 24-well or 96-well filter plates. These plates can be used in drug transport assays to evaluate the absorption of drugs across the intestine. These assays are labor-intensive and expensive and are often pursued late in the drug discovery process due to the difficulty of running them in large scale.
Both the Parallel Artificial Membrane Permeation Assay (PAMPA) lipid and HDM Permeability assays are noncell-based in vitro techniques used to predict passive, transcellular membrane permeability only. Artificial membranes are created by adding phospholipids or lipid containing organic solvents to 96-well filter plates for the PAMPA assay. The HDM Permeability assay is performed using an artificial membrane created with hexadecane/hexane. 6 In both assays, a concentration gradient drives the passive diffusion of molecules, which is the principal mechanism of drug transport through biological barriers. These noncell-based permeability assays are fast, flexible, and cost-effective compared to alternative cell-based models.
Because both the cell-based and noncell-based assays use filter plates, the assays can easily be automated. The design of the filter plate with the ANSI & SBS dimensions (American National Standards Institute & Society for Biomolecular Sciences) and the rigid side walls make the plates compatible with most automation systems. The use of automation with these filter plates allows for more samples to be processed simultaneously and reduces the variability between samples.
Materials and methods
Robotic Workstations
Beckman Biomek FX Workstation (Beckman Coutler, Fullerton, CA) with 96 pipetting head and gripper mechanism (Fig. 1).
(A and B) The Biomek FX Workstation with a 96 head and gripper allows for total automation of noncell-based permeability assays. The design of the filter plates in the assay allows for easy gripping and handling by the gripper. This protocol was set up to process one plate at a time (A). MultiScreen filter plates and transport receiver plates used for permeability assays (B).
Tecan Freedom EVO Workstation and Tecan Genesis Workstation (Tecan Inc., Research Park, NC) configured with eight tips, 1000 or 2500 μL syringes (depending on application), RoMA, and on deck incubator (for cell-based assays) (Fig. 2).
(A and B) The Tecan workstation with fixed tips, integrated LiHa, and incubator allows for total automation of drug transport assays. The protocol developed can process two MultiScreen 96-well Caco-2 filter plates (A) or two Millicell 24 filter plates simultaneously. Millicell 24 filter plate components (B).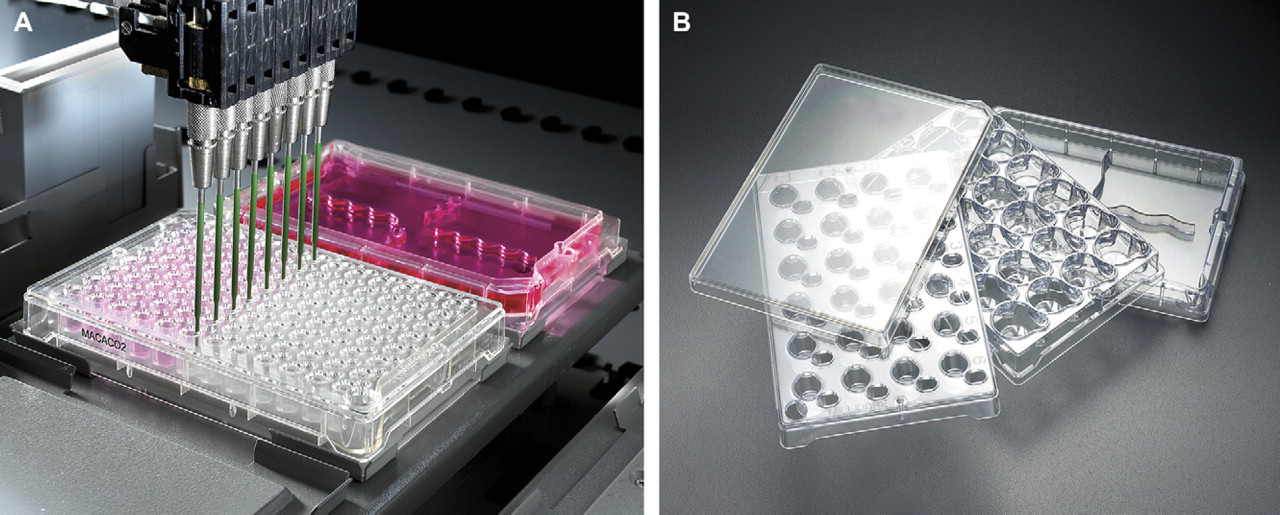
Hamilton MICROLAB STAR (Hamilton Company, Reno, NV) configured with 96-CO-RE pipetting head, eight-tip pipetting head, and iSWAP gripper mechanism.
Caliper Sciclone ALH 3000 Workstation (Caliper Life Science, Hopkinton, MA) configured with 96 pipetting head and gripper mechanism.
HDM Permeability Drug Transport Assay (Noncell-Based))
All drugs used in the HDM Permeability assay are commercially available and were purchased from Sigma–Aldrich (Milwaukee, WI) along with the reagents used. The filter plates (MAPBMN310 or MPC4NTR10) and acceptor plates (MATRNPS50 or MSSACCEPTOR) were purchased from Millipore (Billerica, MA). Each instrument was programmed with all steps of the HDM Permeability assay except for the final step (analysis using a UV/Vis spectrophotometer), which was done off-line using a Molecular Devices SprectraMax (Molecular Devices, Sunnyvale, CA). The automated steps of the permeability assay include the following:
Create membrane layer with hexadecane/hexane and incubate 60 min.
Create daughter plate from stock of drug compounds.
Add buffer to acceptor plate.
Add drugs (500 μM concentration) to filter plate and place it on top of the acceptor plate.
Incubate for 5 h.
Remove filter plate, analyze acceptor plate by UV/Vis spectrophotometer, and calculate the permeability rate.
Lipid PAMPA Drug Transport Assay (Noncell-Based)
As with the HDM Permeability assay above, all drugs and reagents used for lipid PAMPA were purchased from Sigma–Aldrich (Milwaukee, WI). The lipids were purchased from Avanti Polar Lipids, Inc. (Alabaster, AL). The filter plates (MAIPN4510) and acceptor plates (MATRNPS50 or MSSACCEPTOR) were purchased from Millipore (Billerica, MA). Each instrument was programmed with the same steps as the HDM Permeability assay. However, the membrane was created with the lipids after the buffer was added to the acceptor plate. The membrane does not need to be incubated before the addition of drug to the filter plate in the lipid PAMPA assay. The final incubation step lasts 16 h instead of 5 h as with the HDM Permeability.
Caco-2/MDCK Drug Transport Assays (Cell-Based)
Caco-2 (ATCC, Manassas, VA) and MDCK (ATCC, Manassas, VA) cells were grown for 3, 7, or 21 days depending on the cell line. The plates used were MultiScreen 96-well Caco-2 filter plate (MACAC02S5) and Millicell 24 filter plate (PSHT010R5) from Millipore (Billerica, MA). Nonradioactive drugs were purchased from Sigma–Aldrich (Milwaukee, WI), and radioactive drugs were purchased from PerkinElmer (Boston, MA). The Hanks buffered saline solution buffer (HBSS) was purchased from Fisher/Hyclone (Logan, UT) and Lucifer Yellow from Sigma–Aldrich (Milwaukee, WI). The final analysis was done either by LC/MS or using a Wallac 1450 Microbeta Trilux Plus Scintillation Counter (PerkinElmer, Boston, MA).
Only cell washing and drug transport steps are automated. The growing of the cells is not automated because our instruments are not in a sterile environment. The steps include:
Wash the cells three times with HBSS buffer.
Incubate plate at 37 °C for 30 min.
Remove the HBSS buffer.
Move filter plate to a clean receiver plate.
Add drugs to filter plate or receiver plate.
Incubate for 2 h at 37 °C.
Analyze both filter plate and receiver plate (done manually with LC/MS or using the Wallac).
Results
All the assays described are easily automated on various robotic platforms. In each assay, manual controls were run to show that there is no difference between automated and manually processed samples.
HDM Permeability Drug Transport Assay (Noncell-Based)
Eight drugs were run simultaneously on different days of the week using various robotic platforms (Biomek FX, Hamilton MICROLAB STAR, Tecan Freedom EVO, and Caliper Sciclone ALH workstations). The same eight drugs were not always used on each instrument and the assay was run on different days to look at day-to-day variability using automation. A manual control was run to compare automation to manual processing. The results (Fig. 3A–D) show no difference in rank ordering of compound permeability from day to day with any of the four robotic workstations. Similarly, there was no difference in rank order of compounds when comparing automation to manual. In most cases, the samples prepared with automation tended to have less variation (lower standard deviation) than the manual samples, indicating that automation may reduce variability in the process.
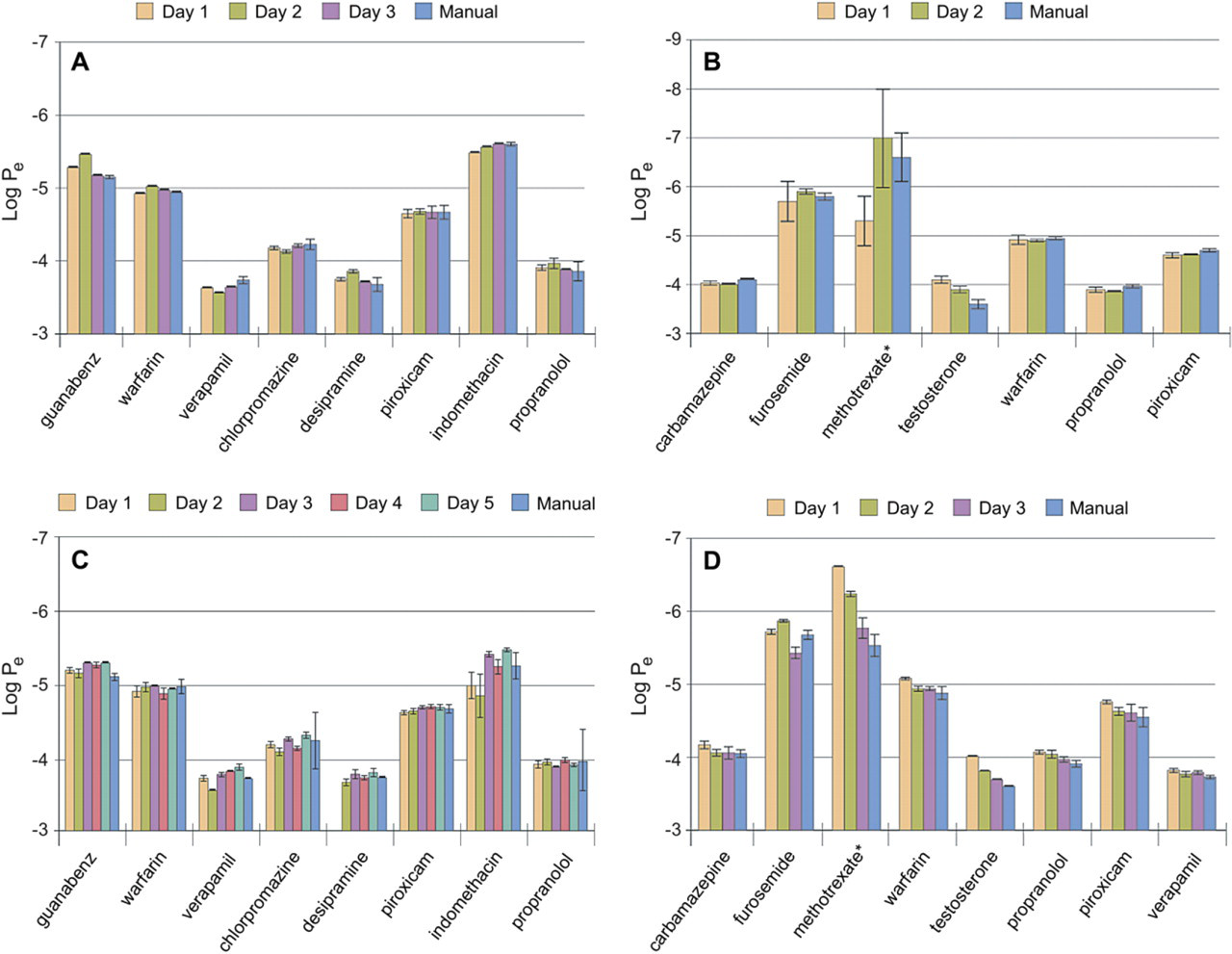
(A–D) HDM Permeability assay results. Each value is an average log
The time needed to process one plate does not vary much between different automated systems. The protocol on each system took between 1.2 and 1.5 h to run, depending on the workstation. This time does not include the 5-h incubation step at the end, but it does include setting up an equilibrium/standards plate used for calculating the permeability rate (log
Lipid PAMPA Drug Transport Assay (Noncell-Based)
Eight drugs were run simultaneously on different days using two robotic platforms (Caliper Sciclone and Tecan Freedom EVO) (Fig. 4A, B). The same eight drugs were not always run on both instruments. Variability from day to day was examined by comparing the same drugs run using the same automated protocol on different days. A manual assay was run at the same time. The results show no difference in rank ordering of compound permeability from day to day with either of the two robotic workstations. There was no difference in rank ordering when comparing automation to manual processing. In most cases, the automation samples tended to have less variation (lower standard deviation) than the manual samples, indicating that automation may reduce variability in the process.
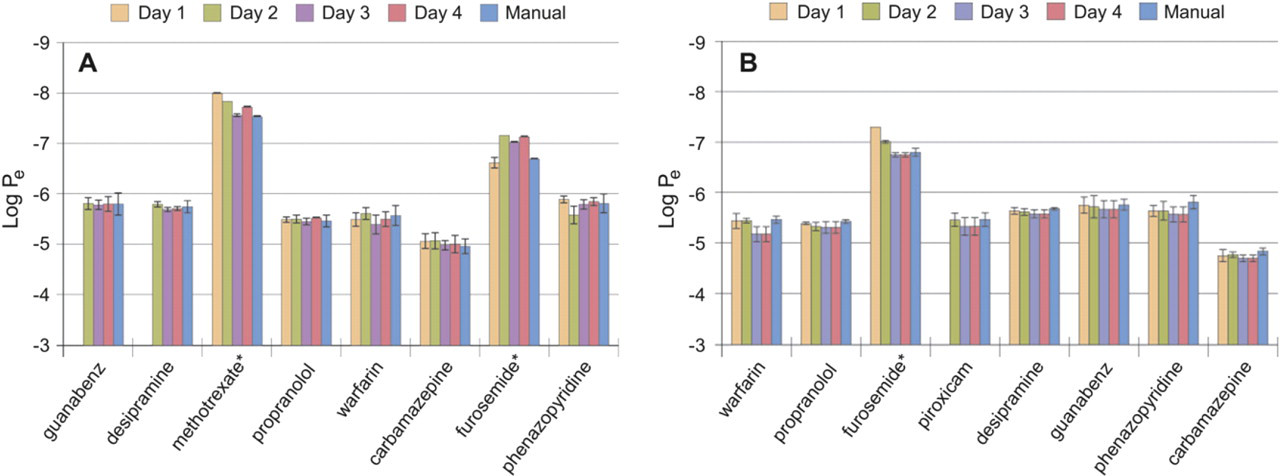
(A & B) Lipid PAMPA assay results. Each value is an average log
Caco-2/MDCK Drug Transport Assays (Cell-Based)
The cell-based assays are easily automated using either the Millicell 24-well filter plate or the 96-well format (Multi-Screen Caco-2 filter plate). Table 1 shows data for the Millicell 24 filter plate on the Tecan Freedom EVO system. There was no significant difference between the plates processed using automation and plates processed manually for both cell lines. Automation tended to have lower variability between replicates (lower standard deviation) than samples handled manually. Table 2 shows data from 96-well filter plates (Multiscreen Caco-2 filter plate) that had been processed using a Tecan Genesis. As with the 24-well filter plate, there was no difference between the manual and automated data.
Tecan Freedom EVO Millicell 24 filter plate
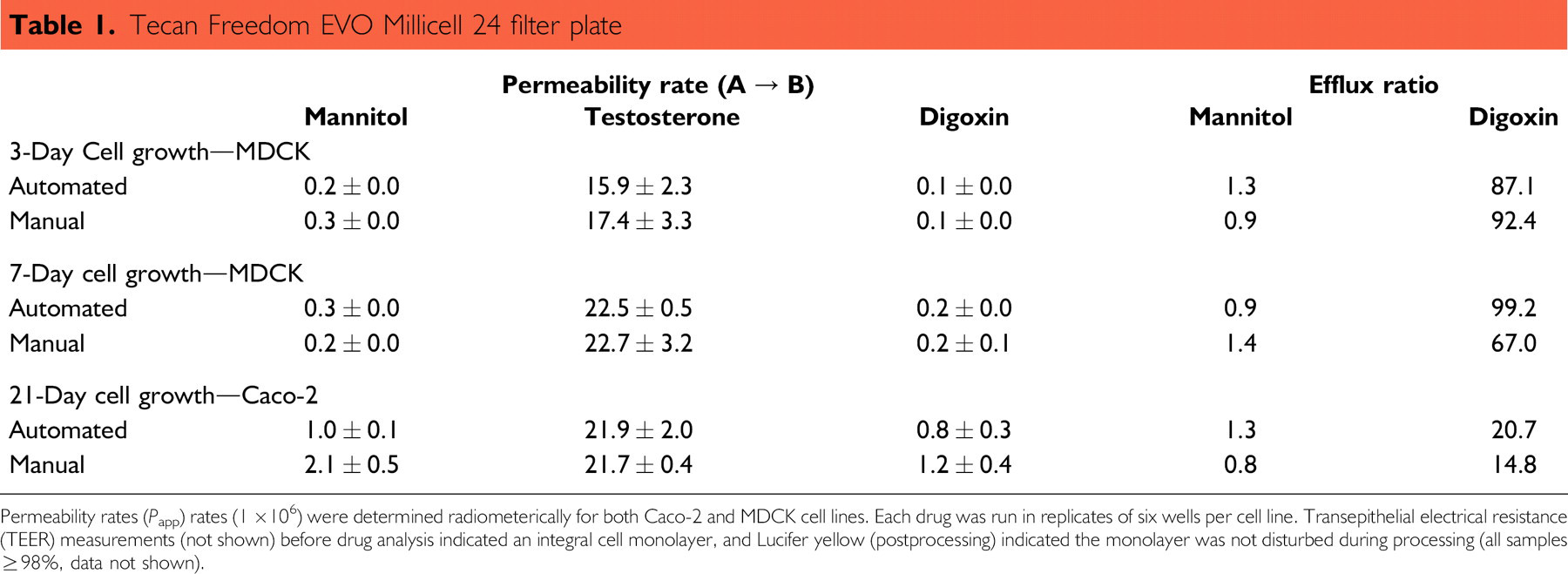
Permeability rates (
Tecan Genesis MultiScreen Caco-2 filter plate

Permeability rates (
Conclusion
Permeability assays (both cell- and noncell-based) are critical for the drug discovery process. Automation of these assays is an important step in the realization of high-throughput processing of permeability properties and is easily achieved by using filter plates compatible with robotic systems. The use of automation expands the potential for early screening of compounds to reduce attrition rates due to ADME failures. The results achieved using automation and manual processing are comparable and in fact, the use of automation reduces variability between replicates. Both noncell-based and cell-based drug transport assays have been successfully automated on a variety of common liquid handlers. These applications do not require custom accessories and can be easily programmed as walk away methods. The overall time it takes to complete an assay may not be improved over manual processing, but freeing the user to do other tasks is an important benefit.
Acknowledgments
We would like to thank Andrew Arena and Greg Kazan for their help in generating the data for this paper.
