Abstract
IC50 analyses are typically sample, time, and labor intensive. They commonly require multiple dilution steps and consume significant amounts of sample compound. Aqueous intermediate dilutions of concentrated stock solutions can lead to rapid sample precipitation and the generation of false negatives (Spicer, T.; Fitzgerald, Y.; Burford, N.; Matson, S.; Chatterjee, M.; Gilchrist, M.; Myslik, J.; O'Connell, J. Pharmacological evaluation of different compound dilution and transfer paradigms on an enzyme assay in low volume 384-well format, Poster presented at Drug Discovery Technology, August 2005, Boston, MA). Hydrophobic compounds may stick to pipette tips or intermediate dilution vessels, reducing the concentration of the analyte in the dilution and also increasing the possibility of cross contamination. The requirement for multiple serial dilutions in common IC50 analyses causes significant accumulated error. Concentrations of dimethyl sulfoxide (DMSO), the typical solvent used to solubilize compound libraries, as low as 1% in the final assay solution can significantly affect the results of the experiment. Finally, the cost of pipette tips and intermediate dilution vessels, and the frequency of the DMSO washes of tips grows significantly as the number of compounds being analyzed is increased.
A system incorporating acoustic droplet ejection of compounds improves IC50 results by reducing the amount of sample used in the analysis to nanoliters, eliminating intermediate aqueous dilutions and accumulated pipetting error, lowering DMSO concentrations in the final assay to below 1%, and reducing costs of consumables (plastics, solvents, and their disposal). (JALA 2006;11:240–6)
Keywords
Traditional IC50 Process
The traditional method of preparing IC50 samples requires multiple serial dilutions as shown in Figures 1 and 2. Serial dilutions are time consuming and each successive dilution increases the accumulated error.
Method 1. Five serial dilutions are made to cover a 12-point, half-log concentration curve. Although the illustration shows dilutions in microcentrifuge tubes, the dilutions can also be done with an intermediate plate as shown in Figure 2. Method 2. A microplate can be used for the intermediate dilutions rather than microcentrifuge tubes. The numerous serial dilutions involved will increase the compounded error. Samples must be mixed, possibly by aspiration or sonication, at each step to ensure complete mixing. The potential for inadvertent transfer of high-concentration fluid is high and requires significant washing of tips or cannulae at each step.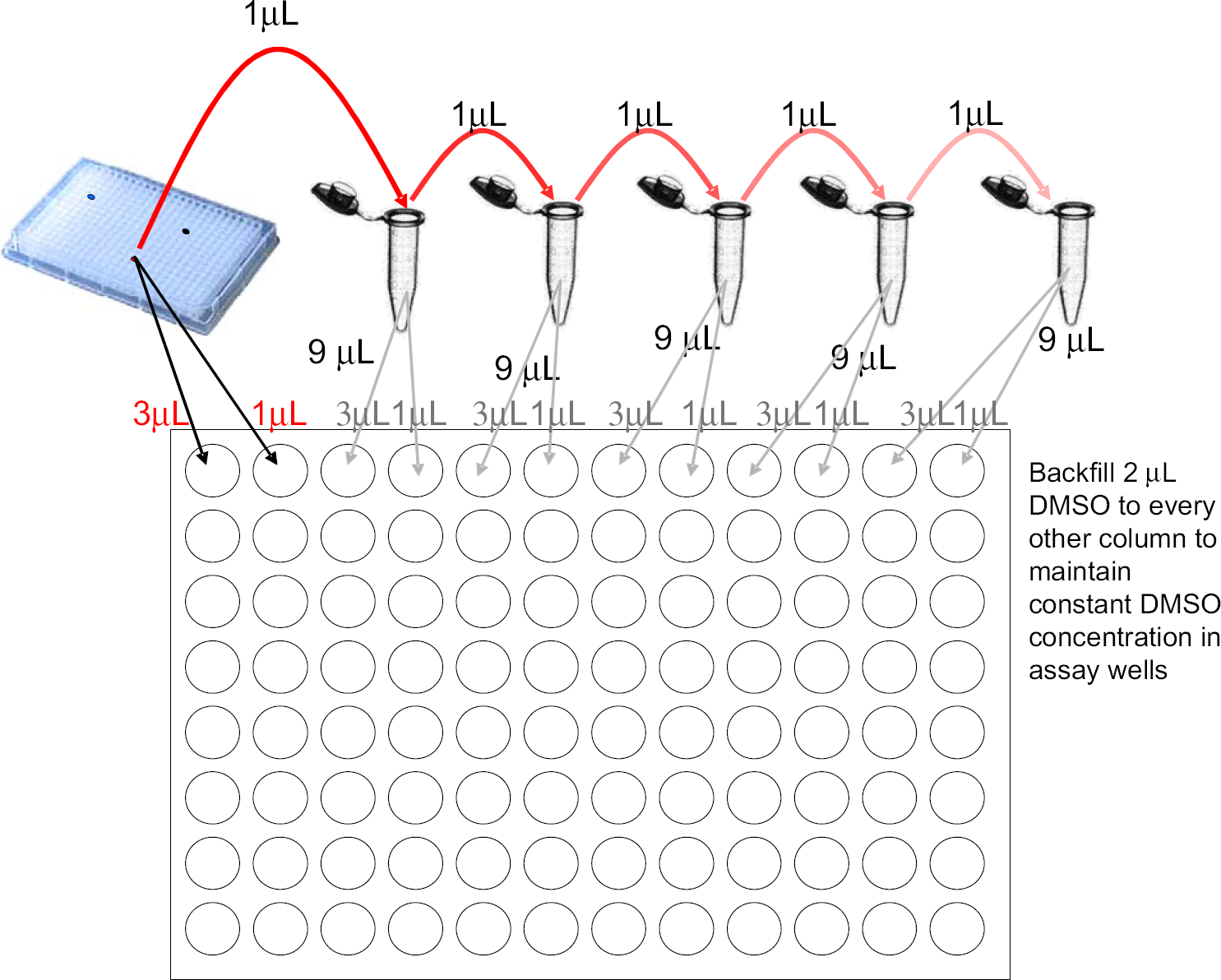
Significant amounts of sample are used in the manual method shown. Method 1, as shown, uses 5 μL of sample for one 12-point curve and 13 μL if run in triplicate. It also uses 57 μL of dimethyl sulfoxide (DMSO) for a single analysis and 90 μL when run in triplicate. Using Method 1, the final amount of DMSO in each assay well is 3 μL. To keep the final concentration of DMSO below 1%, Method 1 requires an assay volume of 300 μL. Method 2 presents a more automation-friendly process that has the added benefit of reducing both the amount of sample used and the volume of DMSO required for dilution. Table 2 shows a comparison of the amount of sample and DMSO required for these two methods as well as for the acoustic droplet ejection (ADE) method.
Types of source plates for ADE

Source plates for ADE must be acoustically compatible for droplet ejection. Five plates are commercially available with a variety of formats and working ranges. ADE requires a flat-bottomed plate to efficiently transfer acoustic energy into the well. Droplet ejection can only occur when fluid covers the bottom of the well. In most cases, the minimum working volume is set by the minimum fluid required to sufficiently coat the bottom of the well.
Comparison of the amount of sample (blue) and the amount of DMSO (yellow) required for analysis

ADE IC50 Process
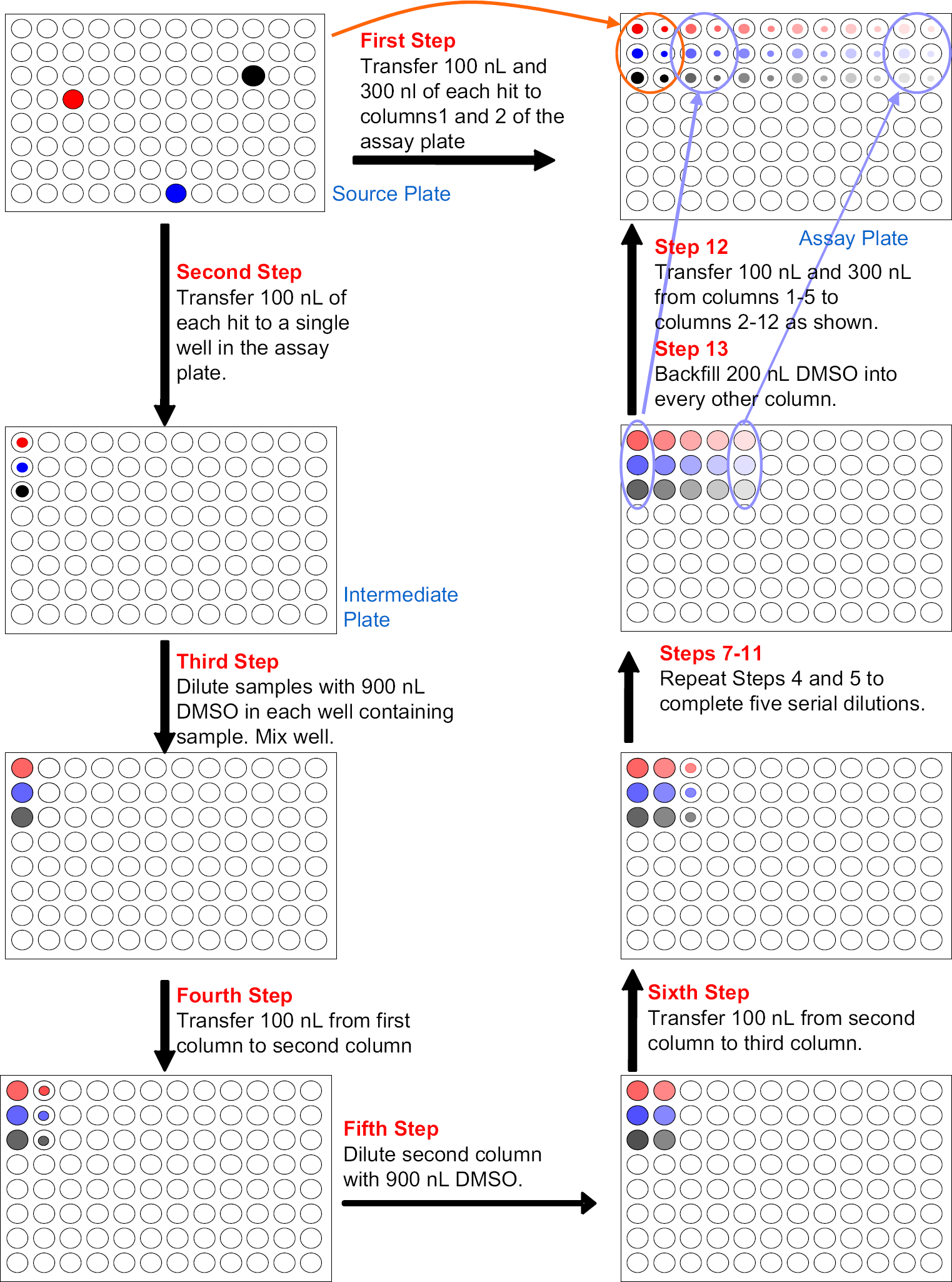
The ADE method, as illustrated in Figure 3, is slightly more complex, but brings significant benefits.
The source plate shown at the upper left has three compounds, which the researcher wants to further characterize with the determination of IC50 values. Fluid is acoustically transferred from the source plate to the first four columns of the assay plate. Well A1 receives 75 nL of the red compound; A2, 25 nL; A3, 7.5 nL; and A4, 2.5 nL. The other chosen compounds are moved in a similar manner to rows B and C. The second step transfers 250 nL of the red compound to well A1 and 2.5 nL to well A2. After all the chosen compounds are moved to the intermediate plate, the third step uses a bulk filler to add 25 μL pure DMSO to each well. Column 1 of the intermediate plate now holds a solution of each compound that has been diluted 100:1, and Column 2 holds solutions that are diluted 10,000:1. The fourth step acoustically transfers four separate volumes of the 100:1 dilution (75, 25, 7.5, and 2.5 nL) to columns 5–8 of the assay plate followed by four volumes of the 10,000:1 dilution to columns 9–12. The assay plate now has compound in each well at concentrations over six orders of magnitude.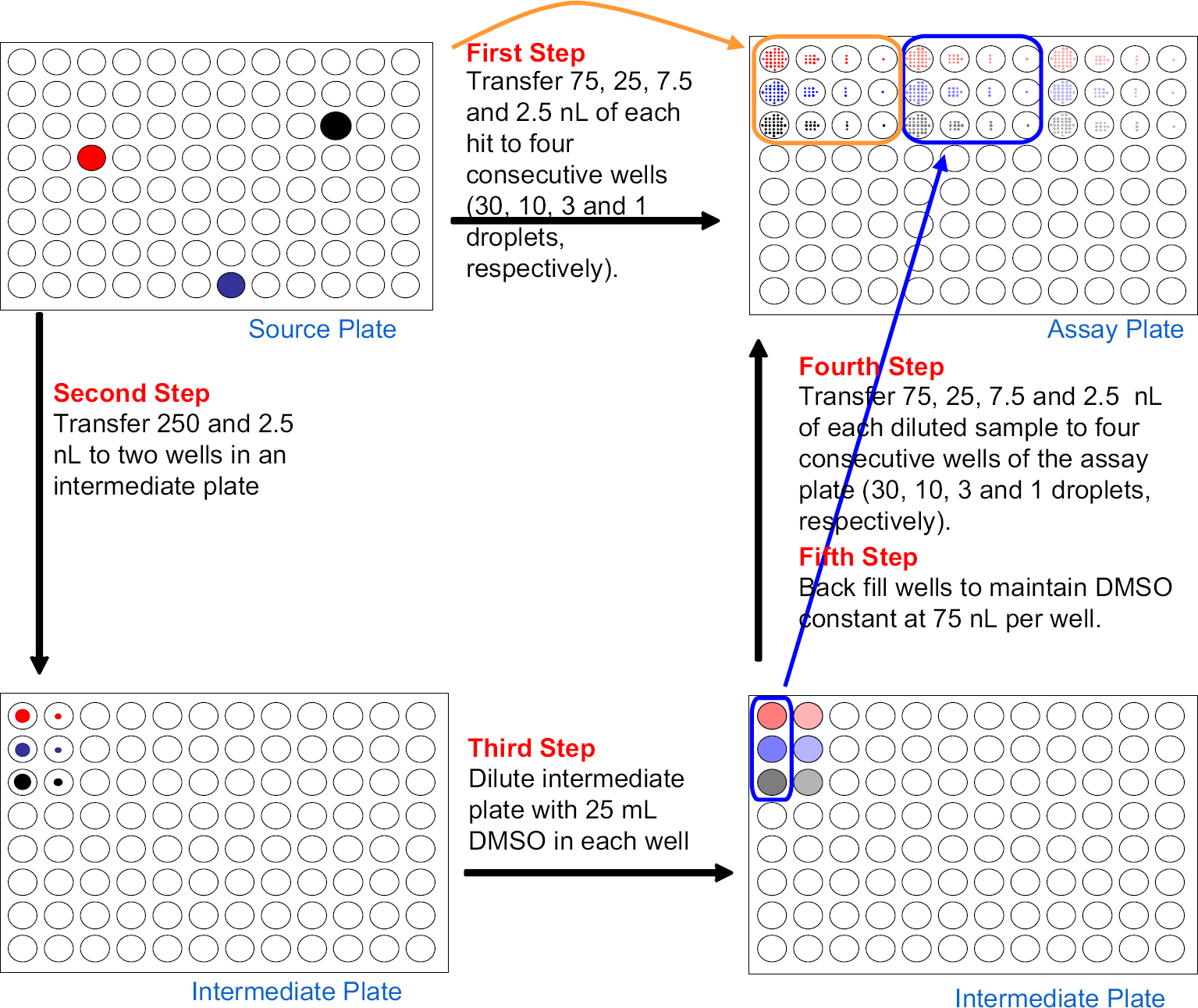
ADE uses sound to move liquids. 2 Unlike other liquid transfer techniques, no solid implement, pipette tip, or pin tool, ever touches the fluid being transferred. Fluid is ejected directly from a source plate to a destination plate. The Echo 550 and Echo 555 liquid-handling systems (Labcyte Inc., Sunnyvale, CA) use ADE to transfer droplets of 2.5 nL. Increased volumes are obtained with multidroplet transfers where the droplets may be ejected from a well in the source plate as rapidly as 500 times a second. Figure 3 illustrates how liquid is moved from a source plate to a destination plate. Table 1 shows the types of source plates that can be used for ADE on an Echo system. The assay plate obtained from the process outlined in Figure 3 provides samples at 12 concentrations over six orders of magnitude while using smaller amounts of compound and DMSO (see Tables 2 and 3).
Comparison of the amount of DMSO in the final assay

We have been reminded that the dynamic range of ADE provides the capability to run an abbreviated dose–response curve without requiring an intermediate dilution plate as shown in Figure 5.
An acoustic transducer is coupled to a flat-bottomed source plate by a droplet of water. The source plate is held stationary, whereas the transducer and destination plate are free to travel. When centered under a source well, the transducer automatically changes position in the vertical axis to maintain acoustic focus at the fluid meniscus in the source well. A single droplet has a volume of 2.5 nL. The rate of droplet ejection depends upon the instrument, the source plate type, percent hydration of the DMSO, and fluid volume. Droplets are ejected at 200 Hz (on the Echo 550) to 500 Hz (on the Echo 555). Because the destination plate is independent of the source plate, ADE offers any-well-to-any-well capability and lets the researcher choose the total volume transferred to any well. ADE also allows the user to place two different samples into the same well. Any water on the bottom of the plate after transfer is removed automatically. A 384→384 transfer of 5 nL in a “picture perfect” format takes approximately 185 s with the Echo 550 and 92 s with the Echo 555. ADE can be used without an intermediate plate to provide a two-decade dose–response curve ready plate.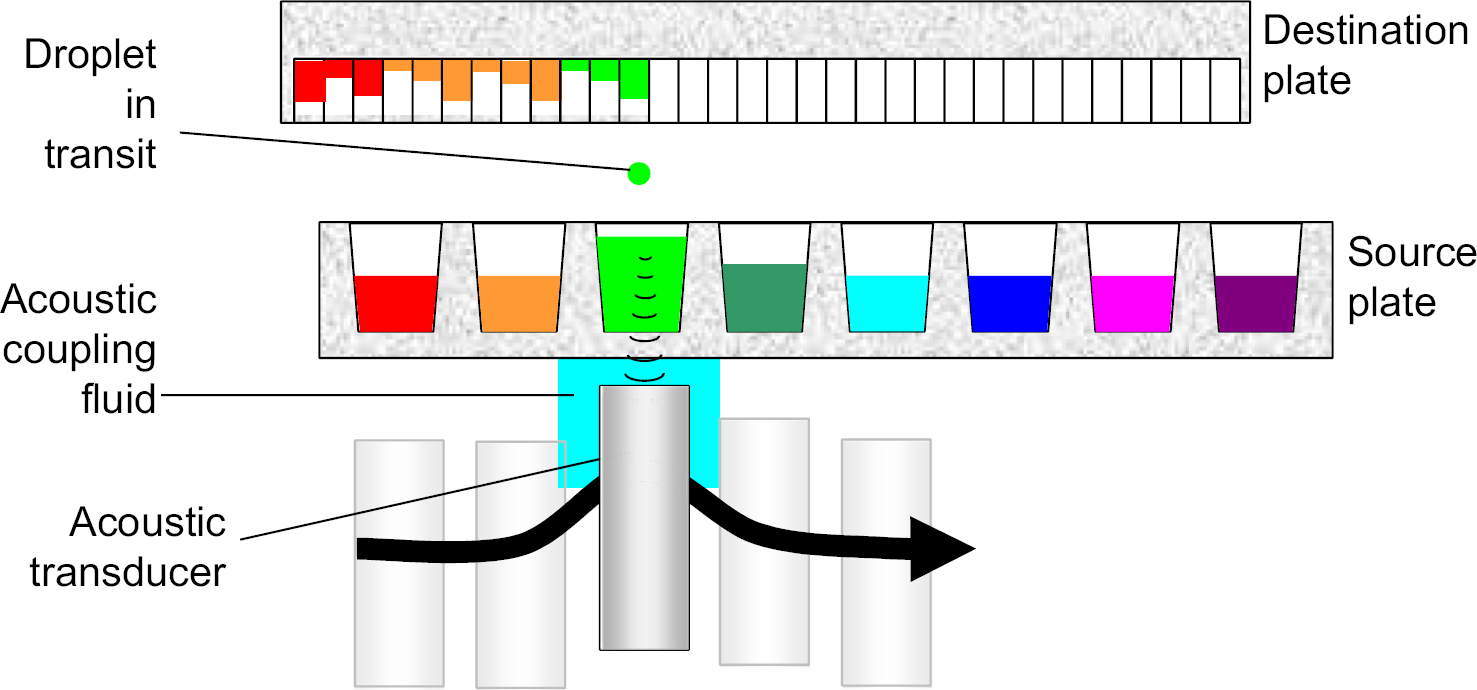

DMSO Dilution vs. Aqueous Dilution—Elimination of False Negatives
Some laboratories make use of an aqueous solution rather than DMSO for serial dilutions. This is done for two reasons—first, to reduce the amount of DMSO used in the development of hundreds or thousands of analyses and, second, to reduce the concentration of DMSO in the assay. Unfortunately, many compounds have a very limited solubility in aqueous solutions and the sample may rapidly precipitate or “crash out” of solution.
3
If the sample precipitates or is absorbed by microplates or pipette tips due to its high degree of hydrophobicity, the amount of material in the final assay well may be much lower than anticipated (Fig. 6).
IC50 values were obtained for more than 1000 compounds by each of two methods. The values obtained from a traditional serial dilution technique using aqueous buffers for dilutions were plotted along the horizontal axis. The values obtained by using ADE and eliminating all aqueous dilutions are plotted along the vertical axis. All compounds in the green quadrant are active (i.e., IC50 value less than 20 μM) by both techniques, and all compounds in the gray area are inactive by both techniques. The compounds found in the bright yellow quadrant are inactive by the traditional aqueous serial dilution technique, but active when determined via the ADE technique. More than 100 active compounds were missed by the aqueous serial dilution method in this study. Graph modified from reference 1.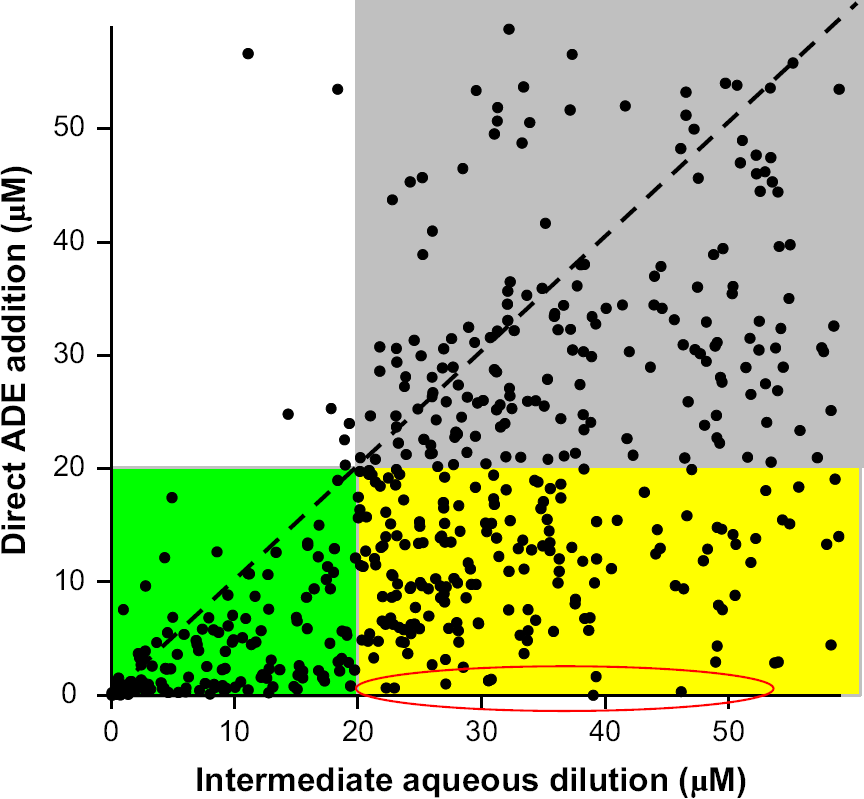
Spicer et al. 1 compared the IC50 values obtained from over 1000 compounds via a process that included aqueous serial dilutions, and compared these values to those obtained from an ADE-based process. They showed that 110 compounds that were judged active when kept in DMSO were misidentified as inactive when the process contained aqueous serial dilutions. Some of the compounds with the lowest IC50 values (i.e., most active) showed little activity when an aqueous dilution is used and would have been missed. Figure 3, modified from Spicer et al., compares the IC50 values of more than 1000 compounds obtained via direct addition of sample in DMSO by ADE vs. IC50 values obtained using aqueous serial dilutions.
This pattern of false negatives holds even when the IC50 cut-off value is changed. For example, 44 compounds that have IC50 values < 10 μM by the ADE technique have measured values < 20 and > 10 μM by the aqueous technique. Thirty-one compounds with IC50 values < 2 μM by ADE were judged to have IC50 values > 10 μM by the aqueous dilution technique.
Accumulated Error vs. Single-Dilution ADE Technique
The use of serial dilutions has its drawbacks. Dilution error for each transfer accumulates increasing the uncertainty of compound concentration at each dilution. Cesarek and Nie 4 suggested a sixfold increase in the coefficient of variation (CV) over a serial dilution used in their laboratory. The dispensing errors of the Echo 550 and Echo 555 liquid handlers remain below 8% CV from high to low volume transfers 5 and can be less than 2%. 6 Designing a 12-point, six-log IC50 curve with acoustic transfer requires only a single-dilution step.
Reduction of DMSO in Assay
Tjernberg et al.7, 8 recently showed that even low concentrations of DMSO in the final assay can cause a variety of negative effects. First, DMSO at concentrations below 1% may lead to denaturation of the protein, as determined by the loss of alpha-helical content measured by circular dichroism and the change in enthalpy of the protein. This denaturation leads to a subsequent loss of activity. DMSO may also increase the amount of protein aggregation as measured by dynamic light scattering, mass spectroscopy, and ultracentrifugation, again leading to a loss in measured activity. It is possible to adjust for the effects of DMSO on the target molecule with the judicious use of controls.
Walsh et al. 9 showed that low levels of DMSO could hide the evidence of genotoxicity in cell-based assays, and they suggested that other solvent systems be investigated to eliminate or reduce DMSO.
DMSO can reduce the binding of proteins and ligands by 90% or more. Reduced binding of a drug-like compound to the target protein will result in reduced apparent effect, agonistic or antagonistic, of the compound on the protein. It is much more difficult to use controls to adjust for the effects of ligand:protein dissociation, because the manner of binding may differ among test compounds. In the examples described above, DMSO concentration can be substantial when performing serial dilutions with DMSO. Although aqueous dilutions will reduce the concentration of DMSO, they are prone to other problems as discussed.
Cost of Consumables
Methods 1 and 2 require five intermediate dilutions. The typical price of a 0.6-mL microcentrifuge tube used in Method 1 is about $0.03. The typical cost of a 384-well intermediate dilution plate is $2.60. Methods 1 and 2 require six pipette tips (at ~$0.025) per sample (regardless of replicate number), one for the original source plate and one for each of the dilution tubes or wells. The number of tips can be reduced by stringently washing them before reuse, but the potential for contamination remains great. Using a microplate for the dilutions substitutes the cost of a microplate for the microcentrifuge tubes.
Further cost is incurred for pipette tips or pipette tip washing at each step, in addition to the cost of the dilution vessels. The ADE technique illustrated in Figure 3 requires only a single 384-well Echo qualified flat-bottom plate as a consumable (Table 4). Not only does ADE save the cost of plastic for the researcher, but it is also environmentally sound because less solid and liquid waste is generated in the process.
Cost of consumables
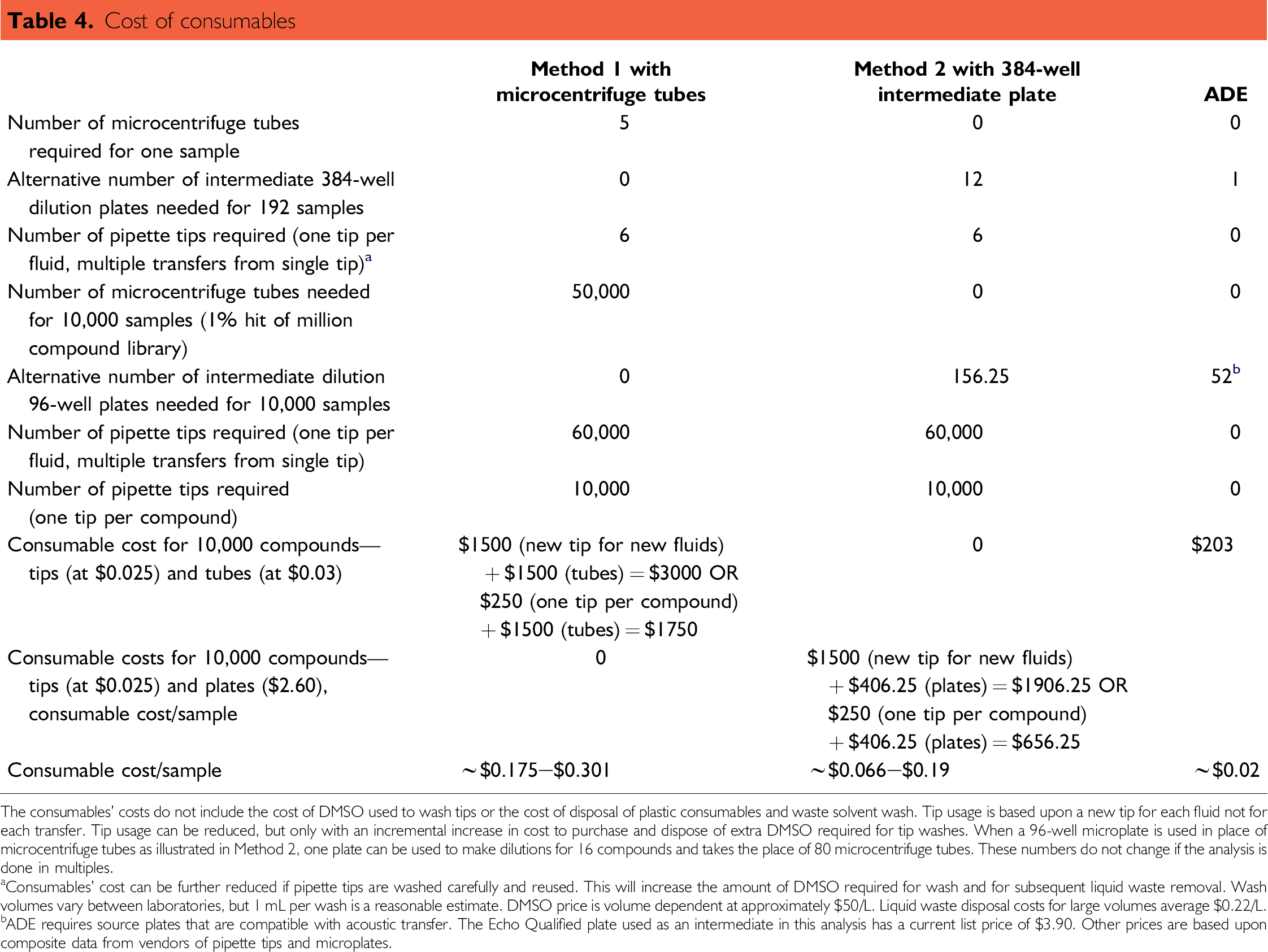
The consumables’ costs do not include the cost of DMSO used to wash tips or the cost of disposal of plastic consumables and waste solvent wash. Tip usage is based upon a new tip for each fluid not for each transfer. Tip usage can be reduced, but only with an incremental increase in cost to purchase and dispose of extra DMSO required for tip washes. When a 96-well microplate is used in place of microcentrifuge tubes as illustrated in Method 2, one plate can be used to make dilutions for 16 compounds and takes the place of 80 microcentrifuge tubes. These numbers do not change if the analysis is done in multiples.
Consumables’ cost can be further reduced if pipette tips are washed carefully and reused. This will increase the amount of DMSO required for wash and for subsequent liquid waste removal. Wash volumes vary between laboratories, but 1 mL per wash is a reasonable estimate. DMSO price is volume dependent at approximately $50/L. Liquid waste disposal costs for large volumes average $0.22/L.
ADE requires source plates that are compatible with acoustic transfer. The Echo Qualified plate used as an intermediate in this analysis has a current list price of $3.90. Other prices are based upon composite data from vendors of pipette tips and microplates.
Conclusions
ADE improves IC50 analyses in a number of ways. First, it requires less precious source material. Second, the elimination of serial aqueous dilutions provides improved IC50 values by reducing precipitation or adsorption of sample during aqueous dilutions. The elimination of aqueous dilutions eliminates the incidence of falsely high IC50 values. Third, ADE has high precision (%CV generally less than 5% at 2.5 nL). The elimination of multiple serial dilutions eliminates their accumulated error. Fourth, the potential for cross contamination of assay wells is eliminated, because ADE has no physical contact with the solution being moved. Fifth, the small amounts of DMSO transferred by ADE allow lower concentrations of DMSO in the assay, especially as the volume of assays decreases. This may also provide improved IC50 values. Finally, the cost of the assays drops with ADE. The reduction comes from three areas—the elimination of pipette tips and most dilution plastics (tubes or plates), the reduction in the amount of waste generated, and the reduced volume of reagent used (ADE allows smaller transfers and smaller assay volumes 10 ).
