Abstract
Liquid handling is a critical component of highly parallel processes such as high-throughput screening (HTS) and genomic analysis. Such processes require the use of smaller sample volumes and the ability to dispense without contact, driving the development of a variety of liquid-handling technologies to meet these needs. Such technologies have associated advantages and disadvantages, which makes choice of the right system application dependent. Syringe technology remains the most cost effective and versatile. It suffers, however, from the inability to dispense submicroliter volumes without contact.
We provide a brief overview of liquid-handling technology, and present a new method of sample transfer based on electrostatic forces (Jetha, N. N.; Marziali, A. Electrostatic device for active transfer of submicroliter samples from syringe pipettors. BioTechniques. 2006, 40, 148–151) that can be incorporated into liquid-handling systems, enabling highly accurate and repeatable non-contact dispensing of submicroliter volumes. (JALA 2006;11:278–80)
Liquid-handling systems are ubiquitous in molecular biology laboratories and are essential for highly parallelized processes such as high-throughput screening (HTS), DNA mapping, and sequencing. Their introduction has helped drive advances in genome and proteome research 1 and has enabled labs to reach throughputs in excess of 100,000 samples per day. 2 Liquid-handling technology is continuously evolving to meet the growing demands of high-throughput biology. It is the aim of this study to provide an overview of recent trends in the evolution of this technology and to review a new method of electrostatic sample transfer, 3 which may be incorporated into any liquid-handling system to substantially improve its performance in low-volume fluid transfers.
High-throughput molecular biology involves high-throughput reagent consumption, therefore it is imperative that reaction costs be minimized to maintain reasonable operating costs. All centers using high-throughput liquid-handling systems have made attempts to reduce sample volumes, driving manufacturers toward a concurrent decrease in minimum pipetting volumes of liquid handlers. To allow submicroliter accuracy in fluid dispensing, both with respect to fluid metering and fluid transfer into destination wells, liquid handlers have trended toward piezoelectric, ink-jet, or solenoid actuation 4 (http://www.deerac.com; http://www.biodot.com) and away from traditional syringe technology. These methods allow accurate dispensing of volumes down to 20 nL, and typically expel drops under pressure to ensure that they disengage from the dispenser and transfer to the destination well or surface. At the nanoliter scale, surface tension overwhelms gravitational effects, and drops metered by a syringe cannot be expected to reliably drop onto a destination plate.
Piezo, solenoid, and similar dispensing technologies, however, do not meter fluids in aspiration. These systems work best when dispensing fluid from a bulk reservoir and are, therefore, often used to dispense bulk reagents into destination plates, where many or all wells in the destination plate receive the same reagent, rather than source-to-destination fluid transfers where each well may contain a different sample. Some hybrid systems exist (BioJet Plus, Biodot Inc, Irvine, CA, USA) that use syringes to aspirate samples, and use a solenoid or similar device to expel drops at high accuracy. Though likely very effective, these systems require substantial hardware per dispensing channel, and can be very expensive if many channels are incorporated in the instrument. By comparison, a 96-channel syringe-based pipettor can be less than 1/10 of the cost of a 96-channel hybrid syringe/solenoid nanoliter dispenser (based on manufacturer quotations). Furthermore, the hybrid systems explored by the authors require substantial (i.e., ~5 μL) minimum volumes of aspiration to allow dispensing of 20 nL minimum volumes. For nanoliter-scale liquid transfers from a source to a destination plate, this would result in substantial dead volume and wasted reagents.
A departure from this limitation may exist in Labcyte's Echo 550 (Labcyte Inc, Sunnyvale, CA, USA), an acoustic transducer-based fluid transfer device that transfers volumes as low as 2 nL from a source to a destination plate, with no losses. Because of its unique and innovative transfer method, this instrument is currently limited to certain fluid types only, which currently excludes aqueous solutions.
Source-to-destination transfers have, therefore, typically been in the realm of syringe pipettors. Pipettors such as the Hydra Microdispenser (Art Robbins Instruments, Sunnyvale, CA, USA) have been shown to be capable of accurately metering volumes as low as 100 nL, but are left with the difficulty of transferring such small volumes from the end of the syringe needles to the destination plates. Touch-off methods (contact of the needles with the target well or fluid or direct contact between the needle and the plate during dispense) are therefore used to transfer metered volumes to the destination plate. These techniques, however, are sensitive to tip cleanliness, alignment, length, and flexibility, and thus the accuracy and repeatability of dispensing is compromised for submicroliter volumes. Furthermore, it is preferable to dispense without contact to reduce the possibility of contamination between sensitive assays.
We have recently developed a new method to improve sample transfer from any liquid-handling system, with greatest advantage to syringe pipettors, by enabling them to dispense submicroliter sample volumes with high accuracy and without contact between the syringe needles and the microtiter plate. This is done through the application of an electrostatic potential between the pipettor needles and the destination plate upon dispensing of the sample. 3 The potential creates a non-uniform electric field between the pipettor needles and the target plate upon dispensing of the sample, resulting in droplet polarization and the generation of an electrostatic force that pulls the drops into the target well. Because the field is capacitively coupled to the drop, there is virtually no steady-state current (the steady-state field from leakage current is ~3 V/m in the drop), and consequently no risk of electrochemical damage to the DNA. The transient fields, which are comparable in magnitude to high electrophoretic fields, are quickly counteracted by the flow of other mobile ions in the drop. Total DNA motion under the expected transient 50 kV/m field (whose time constant is ~0.5 ms) is on the order of micrometers. This transient field should not compromise the integrity of the DNA as demonstrated in other work where DNA is subjected to fields in excess of 107 V/m and remains able to participate in enzymatic reactions. 5
The electric field is created by a device comprised of an adjustable high-voltage power supply and a microtiter plate charger (Fig. 1). The charger consists of an aluminum anode (on which the target plate rests) enclosed in an acrylic housing. To assess the effectiveness of the device, a series of wet-well dispensing (dispensing into the wells of a microtiter plate, which are prefilled with solution, 10 μL in this case) and dry-well dispensing (dispensing into empty wells) tests were performed. Sample solutions included distilled water, 95% ethanol, 25% polyethylene glycol (PEG), and BigDye Mix (Applied Biosystems, Foster City, CA, USA) mixed with fluorescent dye (fluorescein) for quantification of the dispensed volume. The liquid-handling system used for all experiments was a 96-channel Hydra Microdispenser (Art Robbins Instruments) fitted with 48 Teflon-coated stainless steel tips and 48 Teflon-coated Nitinol tips (a nickel titanium alloy). Results for non-contact dispensing of 100, 300, and 600 nL nominal sample volumes for distilled water with and without electrostatic assistance are presented in Table 1. The applied DC potential for electrostatic drop transfer (Table 1) was accompanied by a 20 kV pulse for a duration of 250 ms after dispensing of the sample by the Hydra Microdispenser was fully complete. (Experimental details, additional results, and data for 95% ethanol, 25% PEG, and BigDye Mix are available in Ref. 3.)
Microtiter plate charger and power supply. (Top left) The microtiter plate charger loaded onto a Hydra-96 prior to dispensing of sample.
3

Test solution is distilled water (dH2O)
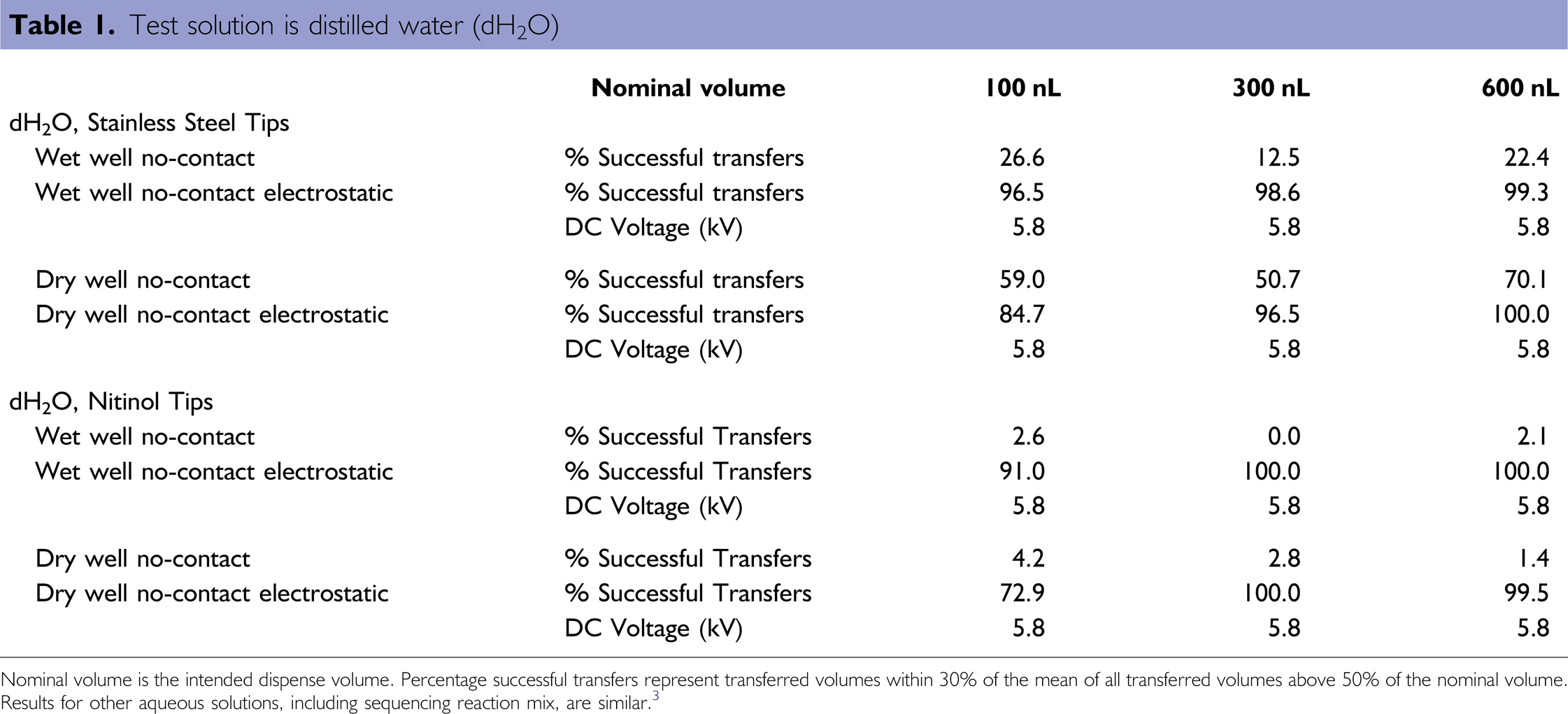
Nominal volume is the intended dispense volume. Percentage successful transfers represent transferred volumes within 30% of the mean of all transferred volumes above 50% of the nominal volume. Results for other aqueous solutions, including sequencing reaction mix, are similar. 3
Comparison of successful liquid transfers shows that non-contact wet- and dry-well dispensing improved tremendously for dilute aqueous solutions when using the electrostatic device. When dispensing BigDye sequencing reaction mix using Nitinol tips, transfer success increases from 10–50% for noncontact methods to 100% with electrostatic transfer for volumes of 300 nL and above. Results for other solutions including PEG and ethanol are presented in more detail elsewhere (supplementary material, Ref. 3). Thus, incorporating an electric field upon dispensing results in highly successful transfer of submicroliter volumes, enabling the use of syringe-based liquid-handling systems in applications where miniaturization of sample volumes is desirable. This technique of electrostatic drop transfer is a low-cost method that can be incorporated into any type of liquid-handling system to improve the accuracy and repeatability of dispensing in an extended range of volumes.
In summary, miniaturization of sample volumes and the ability to dispense samples without contact have become very important performance requirements for liquid handlers. Though many systems, using a variety of technologies, are available, which meet these demands at least in part, syringe pipettors are the most cost effective for high parallelization and have some unique abilities with respect to low dead volume transfers. Hitherto, they have suffered from difficulties dispensing submicroliter volumes reliably due to their reliance on contact or gravity for drop transfer from the pipettor needles to the destination. The method we have presented overcomes this obstacle, making syringe pipettors a viable choice for submicroliter volume transfers.
Footnotes
Acknowledgments
We would like to thank Jason Thompson, members of the UBC Applied Biophysics Lab, and the UBC Physics & Astronomy Machine Shop for their support. We would also like to thank Art Robbins Instruments and the BC Cancer Agency Genome Sciences Center for providing us with Hydra Microdispensers. This work was funded by GenomeBC. A PCT has been filed on the electrostatic drop transfer method.
