Abstract
A unique high-performance liquid chromatographic (HPLC) workflow specifically designed for the rigors of process development has been developed. A key feature of the workflow is the creation of an HPLC software–hardware platform designed to automatically and systematically screen samples using a matrix of columns and eluents to aggressively search for impurities. The workflow platform was assembled from commercial hardware components and both custom and commercial HPLC software. The platform can be used to challenge existing HPLC methods or to develop new methods. The screening conditions are complementary to each other, and are useful to assess the complexity of a sample and to chromatographically resolve impurities that may coelute using any single method. The workflow has been designed to support several different modes of HPLC, and can be used with absorption detection, photodiode array spectrometers, evaporative light scattering (ELS) devices, and mass spectrometric (MS) detection. The custom software interface contains a data-viewing feature to simplify analysis of results. The platform is designed to be used by process scientists, and the same simple user-interface is used to control analytical HPLC, LC–MS, and preparative HPLC. Three real world examples are provided to illustrate the utility of the platform to rigorously assess the complexity of samples and to develop new and improved HPLC methods. (JALA 2005;10:381–93)
Keywords
Introduction
Scientists involved in pharmaceutical process research and development (PR&D) rely heavily on analytical data obtained using chromatographic techniques such as high-performance liquid chromatography (HPLC) or gas chromatography (GC). Through critical evaluation of these data, scientists assess the progress of processes, formation of reaction byproducts, and the quality of synthesized materials. HPLC, in particular, is widely accepted in pharmaceutical laboratories for the following reasons: sample preparation time is usually short, qualitative and quantitative data can be recorded for samples of widely varying polarity in a single run, various forms of detection can increase the specificity and information content of assays, and regulatory agencies have accepted HPLC as a mature and reliable technique. Within the past 15 years, HPLC has very likely impacted the development of every drug candidate within the pharmaceutical industry.
Several years ago many pharmaceutical companies were faced with a number of challenges, which included a several-fold increase in the number of projects in the development pipeline, shortened development timelines, and the need to improve upon the understanding of processes being developed. It was also recognized that process understanding could be greatly enhanced by developing a systematic means to aggressively search for side products in process streams and impurities in starting materials, intermediates, and active pharmaceutical ingredients (APIs). These challenges prompted us to reflect upon how HPLC had previously been used within PR&D and how it could be used more effectively in the future. Specifically, the opportunity existed to greatly improve the sophistication of our HPLC capabilities by (1) creating expert-based HPLC systems designed to improve the HPLC capabilities of scientists with little chromatography experience, (2) integrating analytical HPLC analyses with other workflows such as impurity isolation and identification (preparative HPLC and LC–MS), and (3) improving the use of HPLC data with respect to reduction, reprocessing, and visualization and to support parallel experimentation. Developing such capabilities also presented the opportunity to standardize around a single software–hardware platform that could be deployed to all Bristol-Myers (BMS) Squibb Process R&D laboratories, thus simplifying administration and better enabling data sharing and future application development.
This paper describes the integration of HPLC, along with its associated infrastructure, into a broader workflow. Some critical elements that are emphasized include HPLC hardware, attributes of the custom software to support an automated multidimensional screening protocol, and the design of expert-based HPLC systems for several different modes of HPLC. Three examples are provided to illustrate the utility of the workflow and how it has been used to advantage within the PR&D environment.
The MeDuSA concept
At the very outset of process development work, it is important to develop a “quality-indicating” HPLC method to assess quality of intermediates and APIs being produced. It is equally important to recognize that an HPLC method must evolve in parallel with the synthetic process and the change in impurity profiles. The latter would suggest that methods be checked or challenged on a fairly frequent basis. However, this work is often overlooked because process scientists frequently lack the time and chromatographic knowledge to systematically evaluate different HPLC conditions.
HPLC method development can be a time-consuming process if it is done in a linear, one column or one eluent at a time, trial-and-error manner based on experience and intuition. It can also be an inefficient process if method development efforts are terminated too early based on a single promising result. An approach we have taken is to screen samples using a defined set of columns of various selectivity in combination with mobile phases of differing pH and solvent composition. The screening conditions are complementary to each other and can reveal HPLC conditions that provide improved resolution, selectivity, peak shape, and run times. Data obtained from screening are useful to assess the complexity of a sample, to address purity or impurity issues, or for first-time development of a method. Depending upon the screening results, additional method optimization may be required, but ultimately a high level of confidence in a method's ability to separate all components in a sample is gained. Within BMS, this screening protocol has been named MeDuSA (an acronym for
The first MeDuSA system was designed for reversed-phase HPLC to aid the many users of this technique within PR&D. A standard set of 10 columns, 4 eluents, and defined run conditions was selected to be applicable for a wide range of analytes and to allow scientists to systematically screen HPLC conditions without the need for extensive chromatography experience. The rationale for many of the conditions selected for MeDuSA can be found in the cited references, all of which blend theory with practical application, and serves as a useful guide for HPLC method development.4–6 The use of scouting gradients for samples of unknown complexity (i.e., early route scouting samples) is also described. 7 Our experiences with HPLC, particularly those involving isolation and identification of impurities from drug substances and products, influenced the columns, eluents, gradient conditions, and modes of detection that were selected. The columns were chosen to provide a diverse range of selectivity, but column ruggedness and reproducibility were important considerations. A description of the columns used for reversed-phase MeDuSA, along with method conditions and eluent compositions, is listed in Table 1. A schematic diagram of a MeDuSA Super-8 HPLC (four eluent pairs) is illustrated in Figure 1. In this diagram, two high-pressure pumps are used to deliver low- and high-organic solvent strength eluents of pH 2, 4, 7, and 9 that are paired using solvent lines A1B1, A2B2, A3B3, and A4B4, respectively.
Schematic of MeDuSA Super-8 HPLC.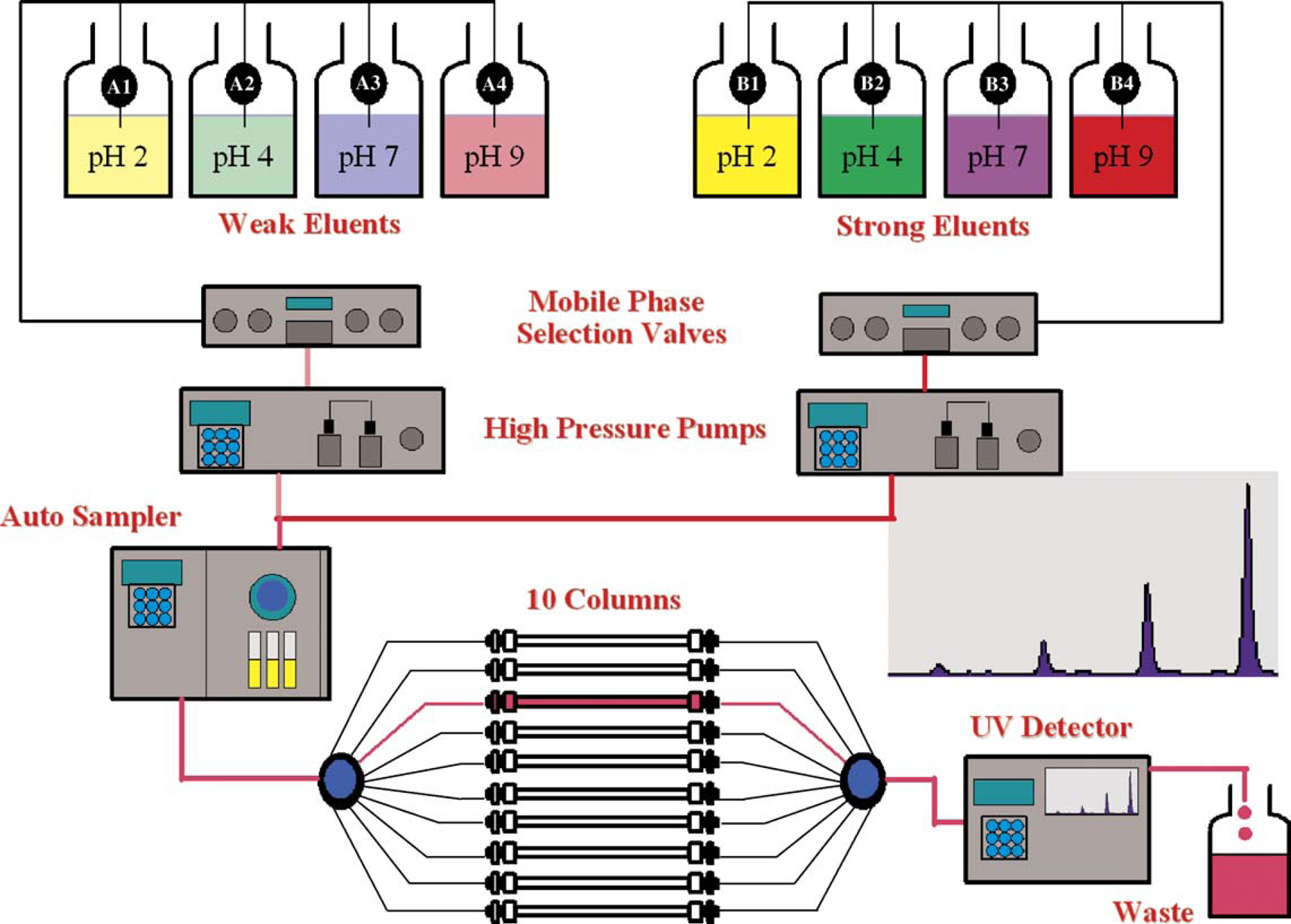
HPLC conditions for reversed-phase MeDuSA

Compatible with pH 9 eluent.
The choice and composition of the MeDuSA eluents are advantageous from the standpoint of enhancing chromatographic selectivity, but they also enable integration of evaporative light scattering (ELS) detection, LC–MS analyses, and preparative HPLC into the common work practice. The volatile nature of the eluents together with the screening protocol affords an easy approach to convert methods that commonly employ phosphate buffer to LC–MS compatible conditions. This is important to enable molecular weight confirmation of components, but it can also remediate material balance issues that are common when phosphate buffers are used. Such issues can occur if highly retained compounds do not elute from the column. This is an established limitation because the high proportion of organic solvent required to elute strongly retained compounds can also cause precipitation of phosphate salts within the HPLC system. The composition of the MeDuSA eluents is also important from a preparative HPLC standpoint because all components can be readily removed from collected effluent fractions by rotary evaporation followed by lyophilization to render salt-free impurity isolates.
Following the success of the first MeDuSA system, several other MeDuSA variants were developed to extend the screening concept to other modes of HPLC. A mini-MeDuSA system was developed to support parallel experimentation. The mini-MeDuSA system is similar to the original MeDuSA system in that it incorporates the same four pairs of eluents and columns of the same phase type. The difference is that shorter columns are employed that can be used at higher flow rates. With appropriate adjustments to the standard gradient, the mini-MeDuSA has delivered efficient separations with short run times (5 min or less). It has been used extensively to support analyses of parallel reactions during early route scouting and in other situations where speed and sample turnaround time are critical. Chiral reversed and normal phase MeDuSA systems have also been developed to support processes involving chiral intermediates and APIs.
An hplc workflow for pr&d
A diagrammatic representation of how MeDuSA was incorporated into an HPLC work practice for PR&D is illustrated in Figure 2. The workflow centers around the use of MeDuSA (encased within the centralized beige oval) to either challenge existing methods or as a first step to develop a suitable method if one does not exist. When MeDuSA is used to challenge an existing method, a sample is screened, and the results are compared against a chromatogram recorded using the existing method. If a method does not exist, the same screening protocol is used, and the most promising conditions are chosen from the screening results. In either scenario, the most promising leads from the MeDuSA screen may be further developed to optimize resolution of all components. The possible outcome of the workflow is to either find the current method acceptable or develop a new or improved method.
Flow diagram of PR&D's HPLC workflow.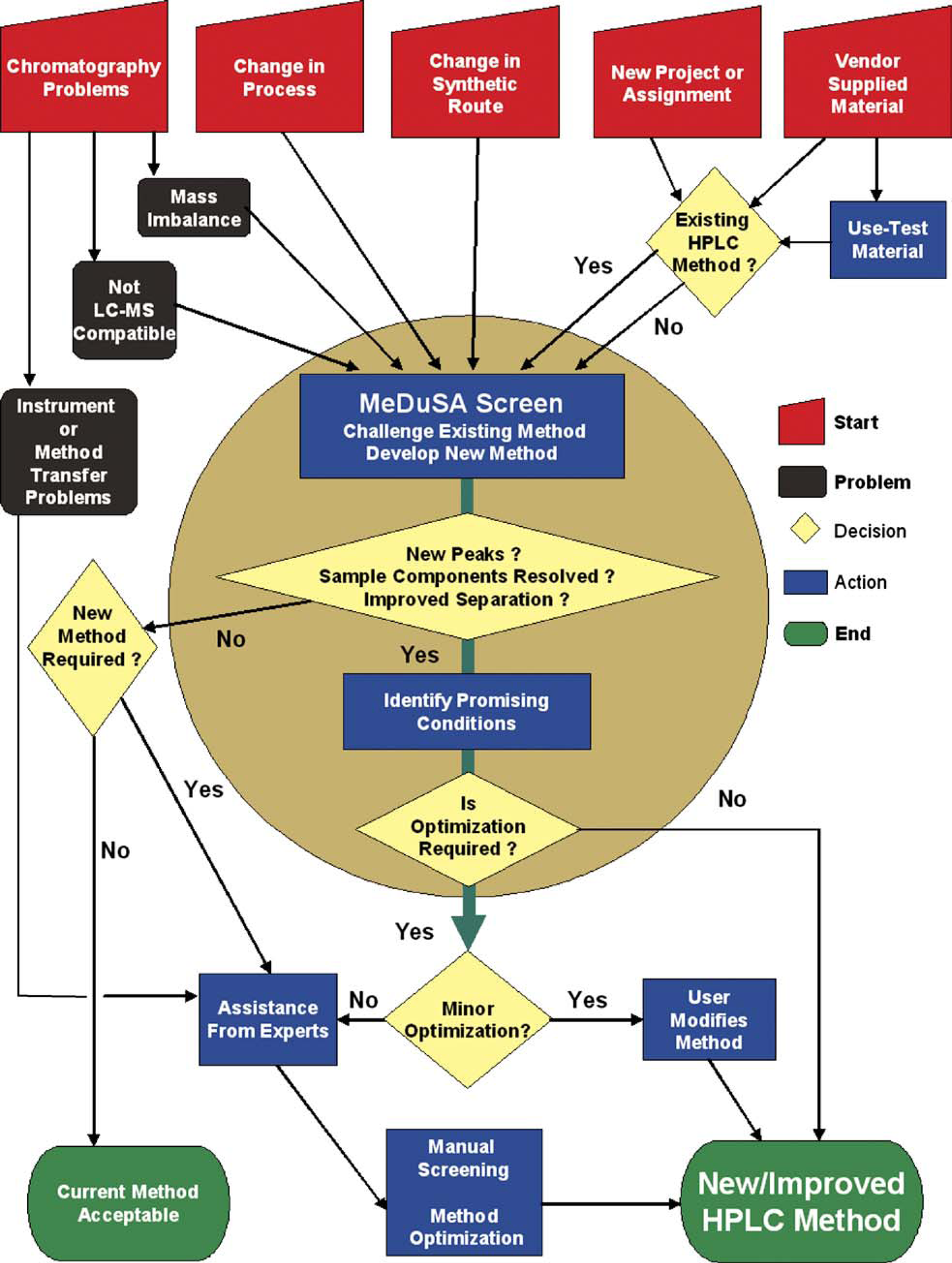
Based upon MeDuSA, a simple workflow was created for scientists to follow in response to five common situations, illustrated by the five red trapezoids at the top of the workflow. These five trapezoids represent entry points to the HPLC workflow and can be viewed as potential opportunities for impurities to be introduced into the process development continuum and/or remain undetected. The situations include chromatographic problems, changes in the process, changes in the synthesis, new project assignments, and vendor-sourced materials. Whereas the same screening protocol and decision tree can be used to effectively deal with the five possible sources of impurities, it is incumbent upon each process scientist to remain attentive to the five impurity entry points and to use MeDuSA in a timely and appropriate manner. It is also important that the scientist analyzes samples that are appropriate for each situation. For example, when assessing a method used for API prepared via a new synthetic route, it is common to analyze a system suitability cocktail containing known and expected impurities. However, this can be misleading because the existence of some impurities can be difficult to predict. A mother liquor or crude batch may be more appropriate for screening because potential impurities are more likely to be present in these samples.
There are two types of chromatography problems that MeDuSA is useful in remediating. The first involves the inability of a method to detect all components present in a sample (mass balance issue). This can occur if compounds in the sample coelute, are retained on the HPLC column, or are invisible to a particular mode of detection. MeDuSA is ideally positioned to deal with these situations because it screens a matrix of chromatographic conditions (columns, mobile phase composition, and pH) to increase the probability that all components in a sample are separated and detected (UV–vis, ELS, MS). The pH of the mobile phase is maintained with a highly organic soluble, volatile component (see Table 1) enabling very high organic content in the gradient, and thereby ensuring that strongly retained compounds are eluted from a column. The second type of chromatography problem involves conversion of phosphate-based methods to LC–MS compatible conditions. HPLC methods often employ phosphate buffers to control mobile phase pH, yet the presence of phosphate prevents direct application of the method to mass spectrometric detection. In this case, the method needs to be redeveloped using a volatile buffer, and MeDuSA provides a direct means to convert phosphate-based methods to LC–MS compatible conditions. A third type of chromatography problem noted in Figure 1 is that involving instrument or method transfer of an HPLC method and is outside the scope of MeDuSA. Examples include methods transferred between low- and high-pressure mixing systems, methods transferred between HPLCs with different delay volumes, and the appropriate adjustment of gradients when switching between premixed and neat solvents.
Changes to the process or synthetic route may create new impurities that may go undetected even on using an established HPLC method. Samples generated after changes have been made in a process or synthesis should be screened in order to challenge the existing method. A new project or assignment may come with or without an HPLC method(s). If there is an existing method, one of the first steps the scientist should undertake is to screen critical samples using MeDuSA to challenge the quality of the information provided by the method. At this stage, LC–MS is highly effective at revealing components that may have weak chromophores, to confirm retention times of known compounds, and to look for isobaric compounds such as regioisomers. If a new project or assignment does not involve the transfer of an existing HPLC method, then early route scouting samples should be screened and the most promising HPLC conditions identified.
Vendor-supplied starting materials represent the final entry point to the workflow. Typically, these starting materials are obtained from multiple sources and may be synthesized using different routes. The synthetic routes may be proprietary and therefore unknown to BMS. The routes may also undergo frequent developmental changes, which result in different impurity profiles. By routine screening, MeDuSA provides an effective means to scrutinize/characterize impurities in vendor-sourced materials.
After the MeDuSA data have been evaluated, either the current method is determined to provide sufficient separation of all sample components or more promising conditions are identified. If the initial MeDuSA method requires optimization, either the PR&D scientist or an HPLC expert can undertake these studies. MeDuSA was designed for both experts and novice HPLC users, and the expert may choose to use MeDuSA to program more complex methods or to use nonstandard columns and eluents to further optimize a separation. Arrival at either endpoint (represented by the green boxes at the bottom of Fig. 2) provides better insight into the true complexity of a sample while leading to a high level of assurance that all components in a sample have been chromatographically resolved. Overall, the application of MeDuSA as part of a systematic workflow helps ensure that the best possible HPLC methods are used throughout development, while providing assurance that impurity issues are resolved without adversely affecting development timelines.
Develop, deploy, and maintain the infrastructure
Establishment of a formal HPLC infrastructure was necessary to support the workflow envisioned for a large deployment of MeDuSA-capable HPLC systems. Principal requirements included standardization of HPLC hardware and software to simplify administrative and support efforts, a mechanism to facilitate data sharing and archiving, and the dedication of a core group to oversee maintenance and support functions. The following discussion elucidates how the HPLC infrastructure was developed, how it has been deployed to over 200 process scientists, and how it is being maintained.
The software required to support our workflow needed to be user friendly, simple to learn, and able to perform many routine HPLC tasks in the background (i.e., column flush and column equilibration) to ensure high quality and reproducible chromatographic data. It was also necessary that the software be tailored for MeDuSA screening as well as for routine analysis once a method is chosen. A program developed within BMS Drug Discovery met many of our software requirements and offered customization that commercial chromatography software packages lacked. The program had been initiated by Dr. H. Weller, who partnered with Shimadzu Scientific Instruments Inc. (Columbia, MD) to codevelop an automated system for HPLC.8, 9 The software package featured an intuitive and simple user-interface that could be used for analytical HPLC, preparative HPLC, and LC–MS. Sample queuing was quick and effortless, custom report printouts were generated automatically, and features incorporated within a password-protected administrative layer would ensure that many of the mundane tasks associated with good chromatographic practice (e.g., pump prime at start up, autosampler flush, column flush and equilibration, baseline stability check before injection, sleeping conditions during the day when no samples are queued, and shutdown at a designated hour in the evening) were performed in the background. HPLC data files containing complete HPLC method information (column attributes, gradient table, eluent composition, detection parameters, etc.) were combined with synthesis information and mass spectrometric results for a sample, and the data packet was then automatically archived. The software also logged HPLC errors and e-mailed them to a core service group to ensure rapid resolution of any problems. Shimadzu eventually marketed a variant of this product under the name Discovery VP. The name serves to reaffirm connection with large Pharma's Drug Discovery departments and acknowledges the underlying Class-VP software through the validation and productivity (VP) insignia.
Whereas Discovery VP offered many attractive features, additional software enhancements were required to support the HPLC workflow envisioned for PR&D. Specifically, use of more sophisticated methods and automated HPLC method screening was needed to increase the probability of resolving all components in a sample (≥0.1% level). An automated procedure to collate and stack the chromatograms into a stack-plot report was developed to simplify viewing of results and to more easily assess the complexity of samples. This feature generates a stacked plot report consisting of method information, stacked chromatograms, and a list of the eluents and columns used for screening. This report can be copied and pasted into an electronic notebook. The stacked plot reports help to assess the most promising column–eluent combination to resolve all sample components. The enhancements also include a new architecture for naming data files and for creating user folders within instrument folders to support global sharing and reprocessing of data. The enhanced software interface developed was given the name Process-VP and is now marketed by Shimadzu under the Discovery VP logo.
Many of the features of Process-VP are accessed through the administrative layer, and these can be used to customize an HPLC instrument for a specific application or to tailor it for a particular mode of chromatography. A description of the normal phase chiral MeDuSA serves to illustrate some of these features. Chiral columns can be very expensive (ca. $1600 each) and require a higher level of care to ensure that they are used with the intended/appropriate mobile phases. The Process-VP welcome page for a normal phase chiral MeDuSA HPLC is illustrated in Figure 3 and provides the status of the instrument, complete descriptions and details of the columns and eluents being used, and access to the administrative layer (“Preferences” and “Hardware Config” buttons). From the administrative layer, columns can be configured to use only certain eluents, and columns can be placed into groups. These settings can be verified from the “Details” tab associated with each column. The normal phase chiral MeDuSA HPLC is configured with two identical banks of five columns each. One bank is dedicated for use with eluents containing trifluoroacetic acid (TFA) and the other for eluents containing diethyl amine (DEA). Each modifier is known to cause column memory effects, and it is advantageous to protect against the possibility of introducing the TFA eluent onto any column that has been used with DEA and vice versa. On-column mixing of these eluents may also cause detrimental chromatographic effects due to DEA–TFA salt formation.
Process-VP welcome page showing chiral normal phase MeDuSA columns and eluents.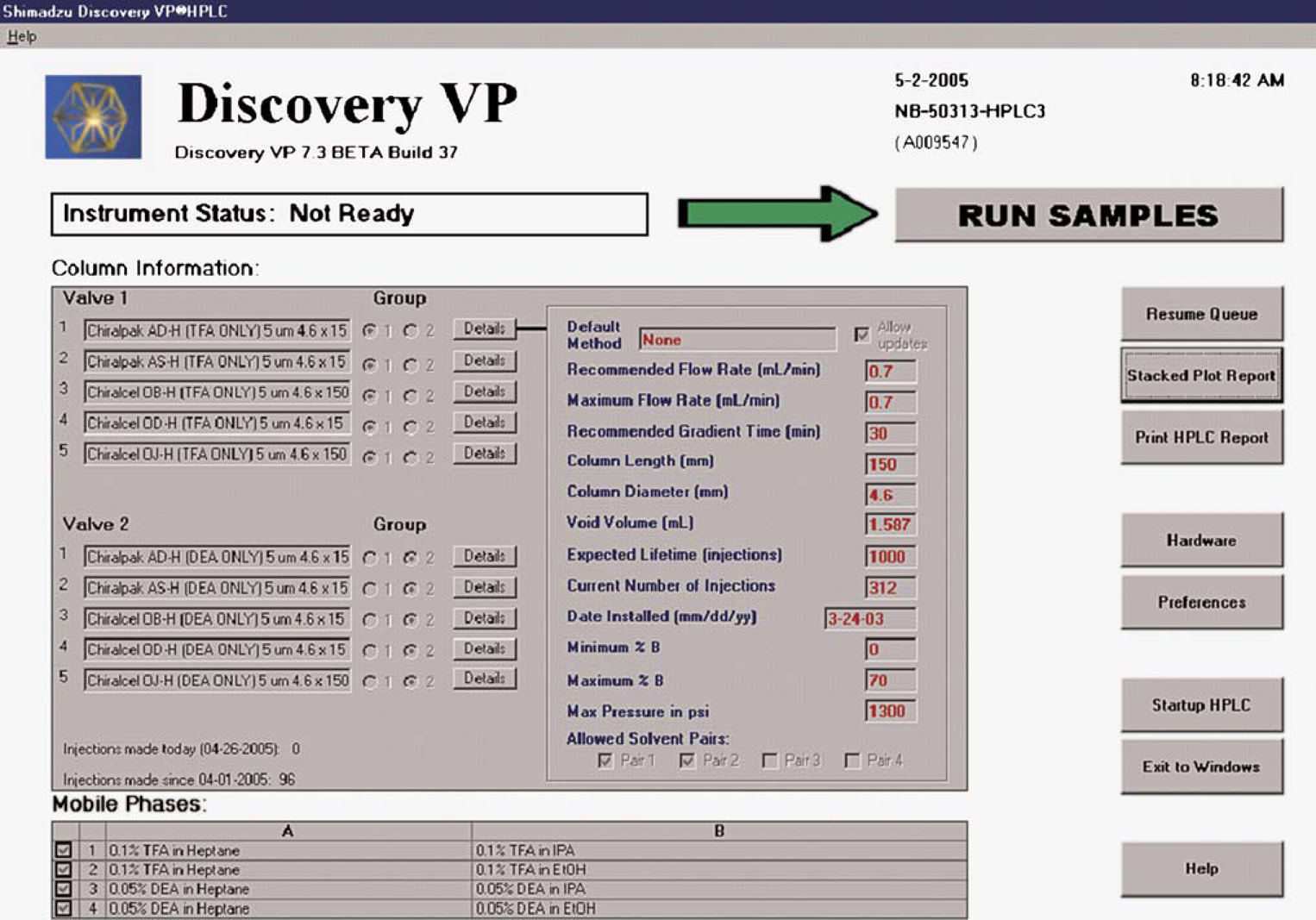
Another important feature of Process-VP is that it can control Shimadzu's dual wavelength absorption and photodiode array (PDA) detectors, Sedex's ELS detectors, and Shimadzu's single quadrupole mass spectrometer (MS). 10 Within Process-VP, these detectors can be configured and readily controlled through the user-interface. The ability to record multidimensional data for samples can be helpful, especially in cases where an impurity is revealed by only one mode of detection, when molecular weight information is needed, or in situations where analytes lack chromophores or have quite different absorptivities. Providing process scientists with access to a variety of HPLC detectors enables characterization of impurities, which results in a better overall understanding of processes, and allows scientists to make better decisions in a timely manner without the direct support of HPLC experts. A screen capture of an instrument utilizing a dual wavelength absorption detector in series with PDA and ELS detectors is illustrated in Figure 4. The bottom panel shows the running queue and provides access to the method builder page, which is used to queue samples and define run conditions.
View of online instrument monitoring UV1, UV2, PDA, and ELS signals.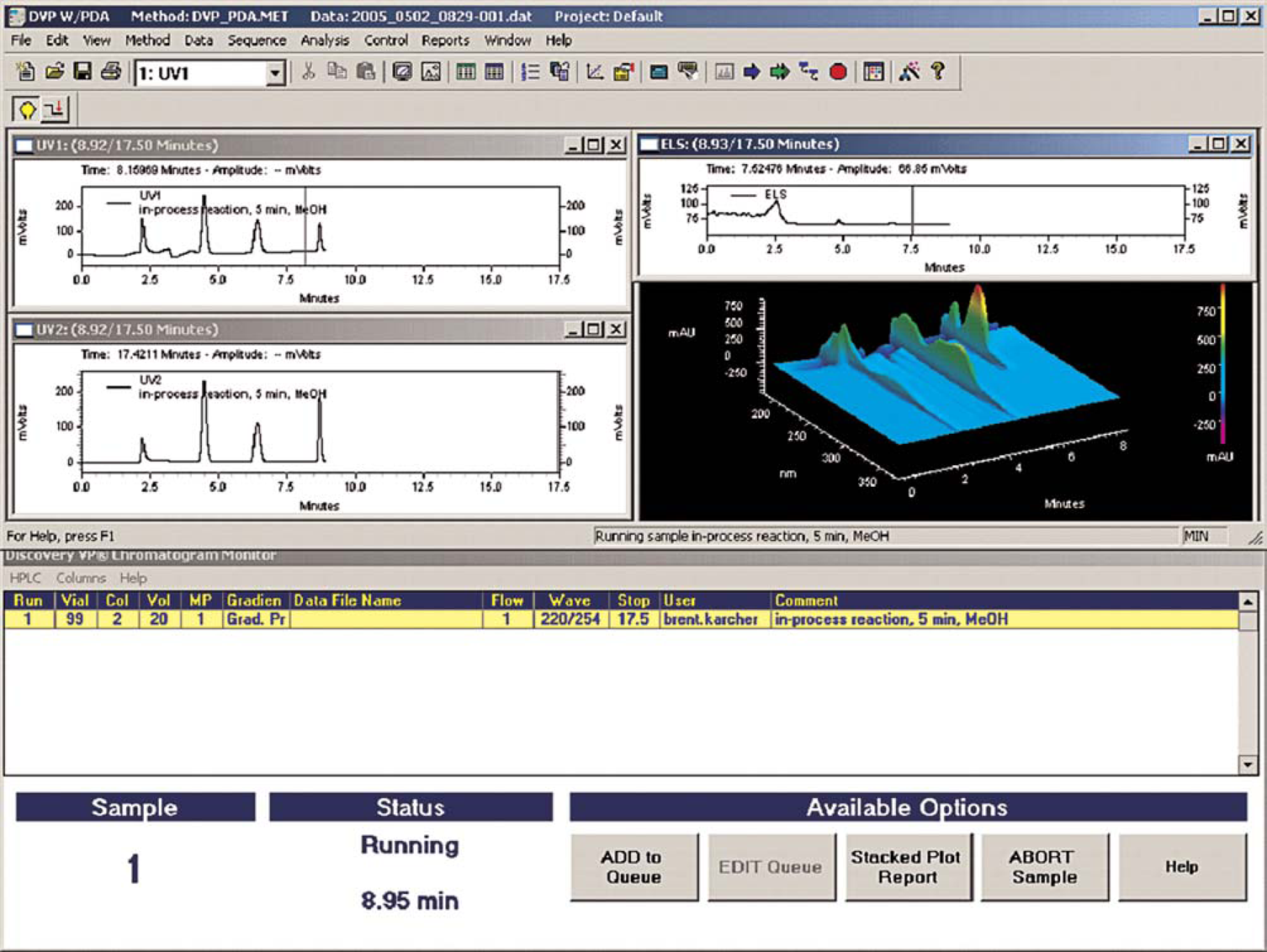
The method builder page represents the primary user-interface and can also be accessed from the welcome page by clicking the “Run Samples” button. A method builder page for a reversed-phase MeDuSA HPLC equipped with dual wavelength absorption, PDA, and ELS detectors is illustrated in Figure 5. From this page, the columns, eluents, modes of detection, and methods used for screening are selected. The columns for this HPLC have been grouped based upon whether they can tolerate the pH 9 (NH4OH) eluent. If a column is selected with a mouse click, the allowed eluents for that column appear in the “Solvents” box located underneath the column groups. Grouped columns can be selected together by clicking the “All” button. Eluents can also be selected as a group by clicking the “All” button that is associated with the “Solvents” box. The “All” buttons simplify queuing of MeDuSA runs. Multistep gradients can be programmed by entering sector times with corresponding values of the strong eluent (%
Process-VP method builder page.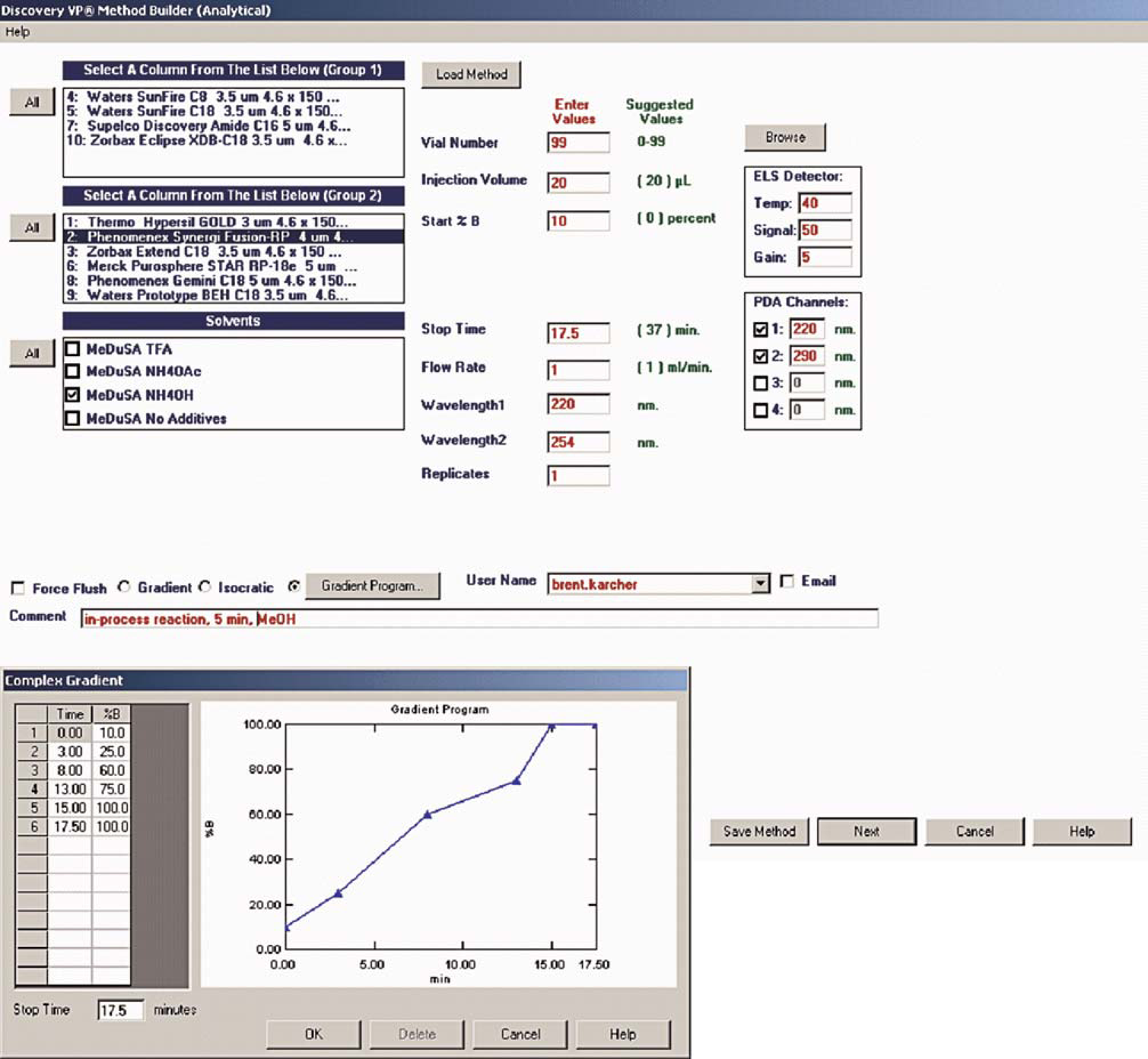
Process-VP is currently deployed on over 200 HPLCs across four BMS PR&D sites to support our scientists. To administer and maintain this infrastructure efficiently, hardware has been standardized. Each HPLC system can use 10 columns, either 2 or 4 pairs of eluents, and is equipped with a dual wavelength absorption detector. Complete descriptions for all columns and eluents are entered via the password-protected administrative layer prior to deployment of an HPLC instrument. This facilitates the capture of complete and accurate method information into a standardized custom report designed specifically for PR&D. Capturing this information is important to ensure that scientists working on the same project can share accurate methods and results. An illustration of the custom report is shown in Figure 6. The report includes method information, full-scale and magnified views of chromatograms, and a report table containing peak area and area percent data. A standard method file with sensitive integration settings is used to ensure that low-level components are recognized for inclusion in area percent calculations, and the magnified chromatogram provides visual confirmation of these components. Each HPLC that is deployed is set up to monitor two wavelengths simultaneously, and a custom report is generated for each wavelength monitored.
Process-VP custom report.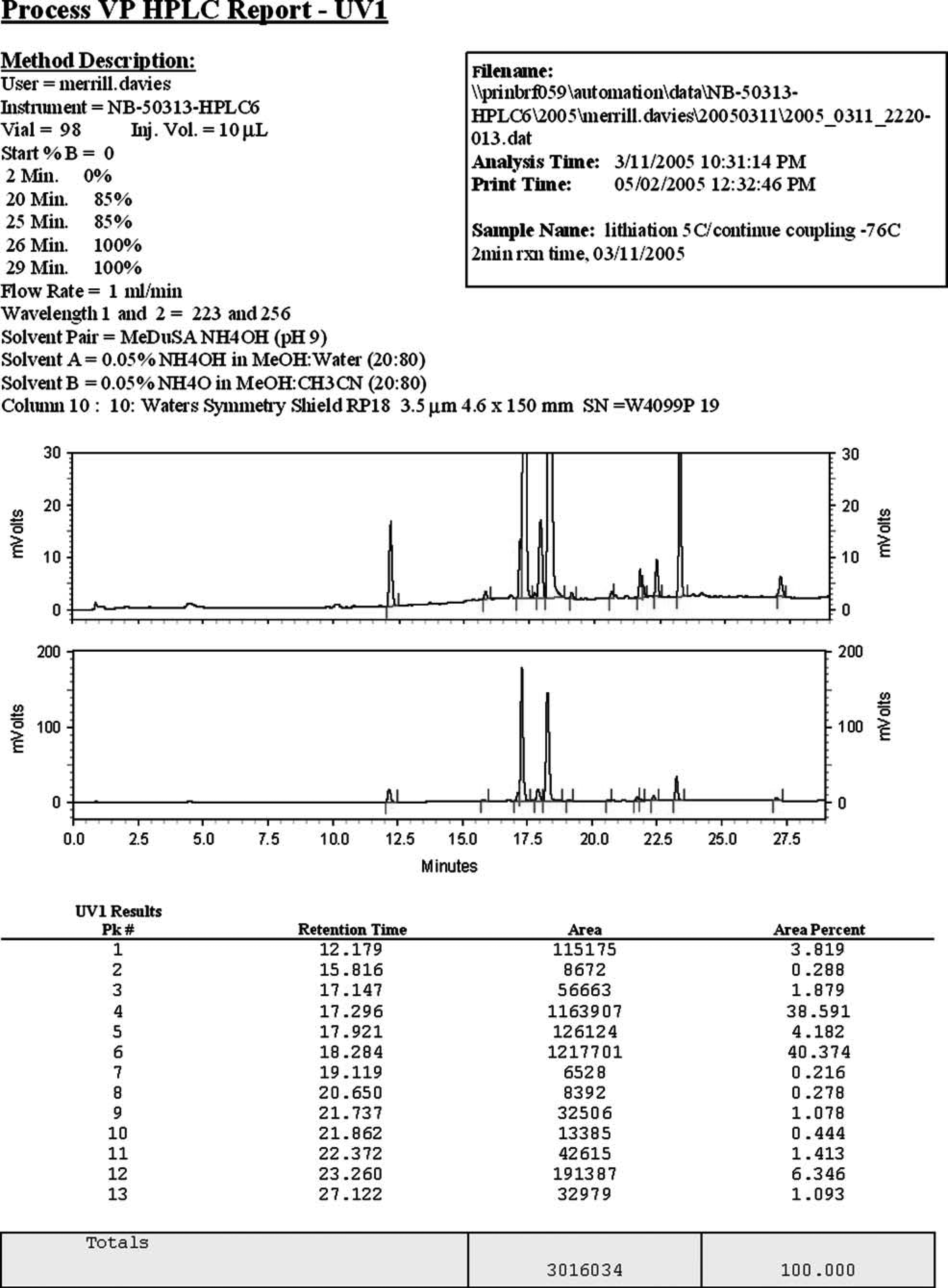
The automated features and intuitive user-interface simplify training. Training is normally administered at the time an HPLC is deployed to a new user and includes hardware and software aspects, as well as desktop reprocessing of data. A training session normally takes 2 h, and because the same user-interface is used to control LC–MS and preparative HPLC systems, training serves as a stepping-stone to enable “walk-up” access to molecular weight determinations and preparative HPLC isolation of intermediates and impurities.
All chromatographic data are stored on a protected server, which is accessible to desktop computers for reprocessing. Mass spectrometric files are reprocessed using the native MS acquisition software, and chromatographic data are reprocessed using Class-VP. Class-VP can be used to export results to Excel for further manipulation or to access charting or graphical functions, as is often necessary with large data banks from parallel reactions. Data are kept live for 3 years to allow project team members to share data (often across sites). After 3 years, the data are moved to an archive media where it can be easily mined and retrieved for display or additional reprocessing. To provide a consistent data management architecture, each HPLC instrument is given a descriptive name based on its location. Data file paths include the HPLC name, the year in which the file was recorded, a “firstname.lastname” folder of the person who queued the sample, a numeric folder to represent the year–month–day that the sample was run, and finally the time–date stamped data files. The architecture is logical so that staff can easily locate data files, and Class-VP provides file finding capability according to sample ID, acquisition date, and/or file modification date. Windows explorer also provides additional file searching capability.
Process-VP automatically handles many of the tedious tasks associated with good HPLC technique, and thereby avoids may fundamental chromatographic problems. In addition, the use of the standard MeDuSA premixed eluents (Table 1) eliminates the possibility of microbial contamination in the mobile phases. These eluents work equally well with HPLCs that use high- or low-pressure mixing, and contribute to improved method transfers from one HPLC instrument type to another. Having standard eluent recipes, which are prepared and delivered by a Lab Services group within BMS, has also helped to improve the quality and reproducibility of HPLC analyses. Standardization of equipment has simplified our ability to maintain the HPLC infrastructure, and the number of service calls has decreased with the implementation of the systematic HPLC workflow. Finally, software and hardware standardization has benefited the BMS scientists who travel between PR&D sites by providing a common HPLC instrument that can be used in a walk-up mode.
Real world implications and applications
The different types of MeDuSA systems developed have been used to support a diverse range of projects, chemistries, and sample types. The following three examples illustrate how the workflow can be used to solve a variety of chromatographic issues to keep projects on track. Although there are many examples to choose from (100+), these three were selected to illustrate the specific attributes and design elements of the standard reversed-phase MeDuSA, the mini-MeDuSA, and the normal phase chiral MeDuSA systems.
The first example involves a sample corresponding to the forced degradation of a drug candidate using dilute base (0.1 N NaOH). The sample had been analyzed using an existing analytical HPLC method, and a large shoulder was observed on the peak corresponding to the API. This shoulder indicated the presence of a closely eluting degradant, but the method could not provide meaningful quantitative information. This sample was evaluated using the standard reversed-phase MeDuSA screen, and a stacked plot report summary of some of the screening data is shown in Figure 7. In this figure, the results for the TFA (upper panel) and NH4OAc (lower panel) eluents for 3 of the 10 columns have been manually combined onto a one-page report. Each chromatogram has been magnified to show the significant retention time region between 25 and 42 min. The chromatogram in the foreground of the upper panel is similar to the separation that was achieved using the existing method and shows a small shoulder on the main component peak. The two chromatograms that are stacked above reveal the main component peak to be resolved into two components. The only significant difference between the analytical method and the conditions used to record the middle chromatogram (labeled 2) is that the eluent of the analytical method did not contain methanol. The ability to resolve this peak into two components is attributed to the different selectivity afforded by using methanol in the MeDuSA mobile phases. In the lower panel, the results for the ammonium acetate eluent for the same three columns are shown. These chromatograms show a much improved separation over the TFA results and provide a better appreciation for the actual complexity of the sample. Using peak area percent data for quantification, the main component was found to be approximately 40% degraded at the 10-day time point. LC–MS analysis of the sample using the MeDuSA ammonium acetate eluent provided molecular weight data for the degradant peaks and enabled tentative structures to be proposed.
Stacked plot results (from example #1).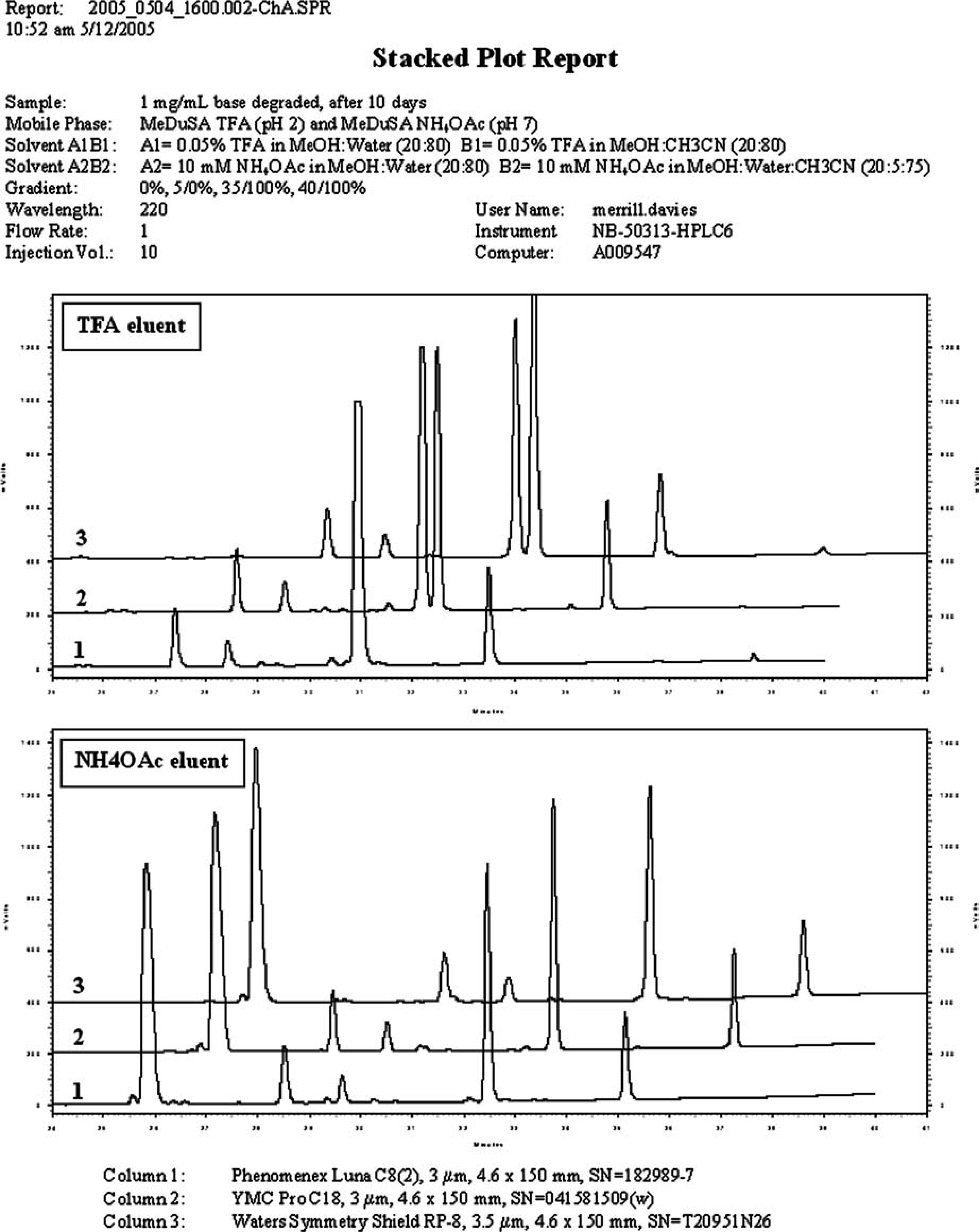
The second example demonstrates the power of mini-MeDuSA to combine efficient separations with short run times. An early development project had experienced chromatography problems, and a sample was screened to challenge the existing in-process method. The existing in-process method used both pH 2 (TFA) and pH 7 (NH4OAc) eluents. A mini-MeDuSA screen was undertaken, and the results were available within 3 h. Chromatograms from the pH 2, pH 7, and pH 9 eluents on the YMC Pro C18 RS column are presented in Figure 8. The pH 9 eluent clearly showed the best separation and the appearance of a small, previously unseen peak. The peak was not observed with either the pH 2 or 7 MeDuSA eluent. Mass spectrometric analysis using the pH 9 mini-MeDuSA conditions revealed the small peak to be an in-situ intermediate. The appearance of this intermediate and the ability to monitor its level enabled scientists to gain a higher level of understanding and control of the reaction.
HPLC screening results (from example #2).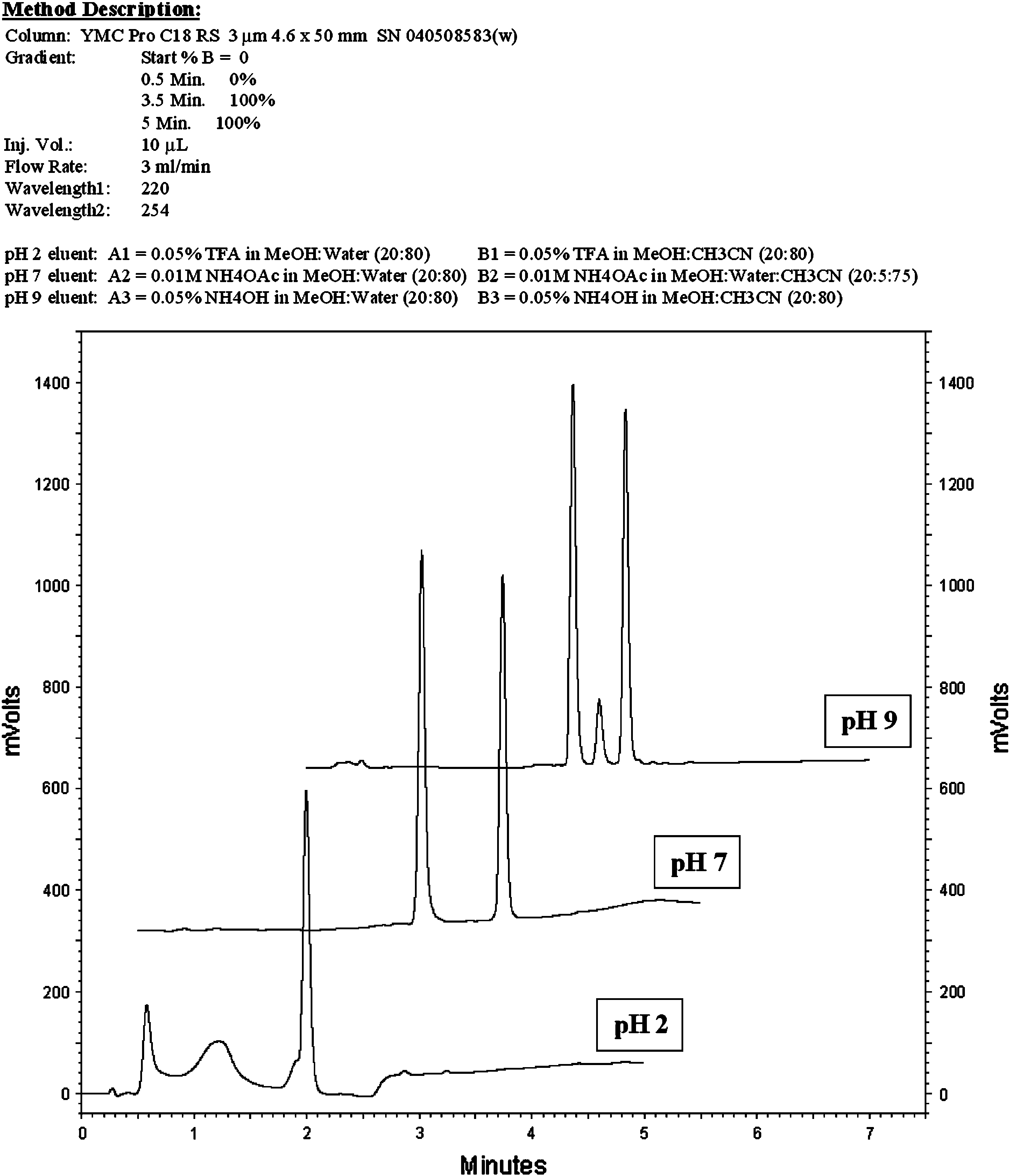
The final example involves a route scouting program to support a project in the early stages of development. In this instance, a chiral assay for protected amino acids was needed. Standards of l-alanine, d-alanine, and an l-alanine–l-alanine dimer, each protected with a carbobenzyloxy group (CBz), were used to prepare a cocktail for screening. Although normal phase HPLC may not be an obvious first choice to separate small, highly polar molecules, the MeDuSA data provided unanticipated noteworthy results. The stacked plot report featured in Figure 9 illustrates the results for protected amino acid samples chromatographed using an eluent comprising 0.1% TFA in heptane:isopropanol (80:20). The chromatogram illustrated in the foreground shows an efficient separation of all three components, with a run time of approximately 12 min. This chiral separation was developed within a day of receiving the sample, and illustrates the value of performing a broad screen rather than focusing on HPLC conditions that may seem to be appropriate based upon prior chromatographic experience or knowledge.
Stacked plot results (from example #3).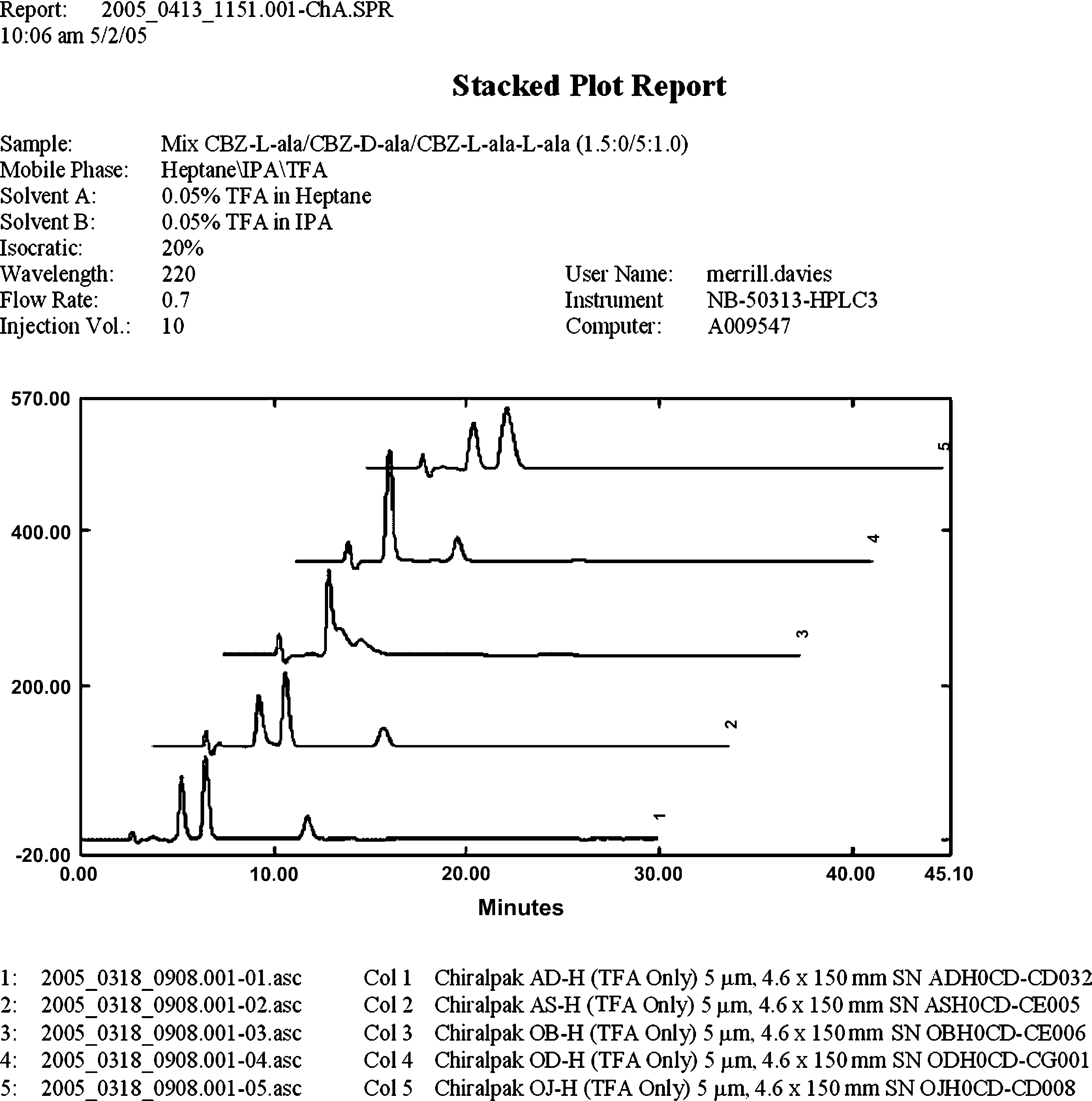
Outcome and conclusions
The development and implementation of a systematic HPLC workflow has provided process scientists at BMS with a means to aggressively challenge analytical methods and to aggressively look for impurities throughout process development. At the heart of the workflow are a variety of expert-based MeDuSA HPLC systems that facilitate the automated analysis of samples using complementary chromatographic conditions. Process scientists have used the MeDuSA HPLCs to uncover impurities that were previously unseen with existing methods, and the systems have been used routinely to develop new or improved HPLC methods. The normal phase chiral MeDuSA HPLC has achieved significant inroads in the automated development of selective methods for chiral intermediates and APIs. The capability of Process-VP software to control a variety of detectors has enabled the MeDuSA HPLCs to record multidimensional data for HPLC peaks. With the same simple user-interface on analytical HPLC, LC–MS, and preparative HPLC systems, process scientists are empowered to challenge existing chromatography methods and assumptions of sample purity, obtain molecular weight information on a new impurity, and isolate those impurities for full characterization. By enabling scientists with this information earlier in development, a better overall understanding of processes is achieved, and a higher level of confidence is gained in the analytical methods that guide process decisions. Ultimately, better decisions can be made in a timely manner, and late stage impurity issues can be avoided.
From a broader perspective, the workflow developed serves as an example of how analytical and informatics technologies can be integrated to enhance capabilities for the user community, while increasing productivity and reducing administrative costs through standardization. It has also set the foundation to enable PR&D's further evolution as additional applications are developed around this core infrastructure.
Footnotes
Acknowledgments
We thank Harold N. Weller for his continued support and whose Discovery VP application provided the foundation of our custom software. We are indebted to Patrick Armstrong, Andy Sasaki, Larry Beasley, Kerry Hill, Curtis Campbell, Susan Steinike, Terry Adams, Thomas Moran, and Dana Loyd of Shimadzu Scientific Instruments, Inc. for their significant contributions to the development of Process-VP.
