Abstract
A flexible and integrated flow-chemistry–synthesis–purification compound-generation and sample-management platform has been developed to accelerate the production of small-molecule organic-compound drug candidates in pharmaceutical research. Central to the integrated system is a Mitsubishi robot, which hands off samples throughout the process to the next station, including synthesis and purification, sample dispensing for purity and quantification analysis, dry-down, and aliquot generation.
Keywords
Introduction
Pharmaceutical research is under increased pressure to generate drug candidates in an ever more cost-effective and time-efficient manner. 1 In fact, competitiveness in follow-on drug research and development has been recently described as a race. 2
As part of our efforts to accelerate the cycle time for medicinal chemistry hit-to-lead and lead-optimization processes, we chose to develop a highly automated integrated compound library synthesis and purification platform based on segmented flow-chemistry technology. 3 We felt that automation could play a key role in this process by eliminating instrument face time on the part of the research scientist and allow for extended unattended operation time. Moreover, we mandated that any system developed would receive rapid acceptance by our medicinal chemistry community. 4
Small-molecule compound libraries have in recent years routinely been synthesized in parallel format; they are typically generated in a synthetic chemistry laboratory and transferred to another laboratory for purification, then on to a structural verification lab for analysis, and finally to a distribution lab to be sent off for bioassays. We describe herein an instrument station that combines many of the above functions, eliminating sample transfers and providing a platform that is efficient in terms of both time and laboratory staff.
System Design
The aim of an automated system was to accelerate the total compound-generation process as well as decrease the instrument face time by the operating scientist. To this goal, samples were handed from one system component to the next via a central robotics arm as shown in Figure 1 . Sample synthesis is achieved in a flow-chemistry process, the resultant solvent stream of which flows directly into a high-performance liquid-chromatography (HPLC) purification system. The purified fraction is aliquoted to generate samples for analysis as well as future biological assay. A parking lot is built in to hold samples requiring bulk dry-down for biological assay.
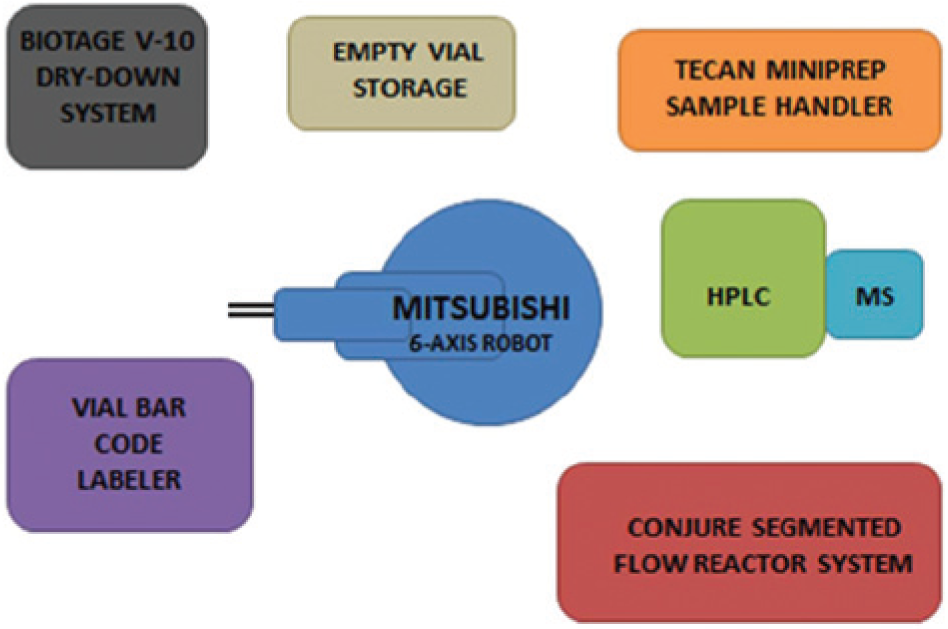
System outline.
Operation
Synthesis and Purification
Full details of the synthesis–purification system have previously been described; 3 the synthesis platform is built around an Accendo ConjureTM segmented flow-chemistry reactor, 5 seen in Figure 2 , to perform chemical synthesis. There are multiple advantages to a flow-chemistry platform, wherein reactions are carried out in stainless-steel coiled tubes, as opposed to the glass flask or vial typically used in laboratory-scale synthesis. Reactive intermediates can be generated and consumed in small quantities in situ, thus expanding the scope of reactions that can be safely conducted in a standard laboratory. Due to the ability to achieve higher temperatures in stainless steel, reactions can be run more quickly—typically, reaction times of 5 to 10 minutes can be achieved. Higher pressures can be achieved by allowing the ability to employ traditional solvents above their normal boiling point at atmospheric pressure. Reaction conditions can also be more carefully controlled, and temperatures rapidly increased or decreased, in a small-diameter stainless-steel reactor than in a glass vial with a large volume to surface area. Precise control of reaction conditions allows more reproducible reaction products in a flow-reaction system. Reactions are also inherently scalable in a flow-chemistry system from the laboratory all the way to the pilot plant, because the longer one allows reactant streams to flow, the more material is generated.

Accendo ConjureTM segmented flow-chemistry reactor.
The commercially available Conjure system was modified to our specifications to accommodate different vial types, and it used a custom-fabricated piezoelectric (PZT) transducer module provided by Autochem Private Limited, 6 adjacent to the Conjure flow reactor, to accelerate the reaction rates for certain reaction types. In addition, the standard analytical HPLC interface of the Conjure system was modified through valve and syringe changes to allow direct injection of the entire reaction segment onto a preparative HPLC system for purification.
The in-house developed preparative HPLC–mass spectrometry (MS) system, using components from multiple vendors, was fully integrated with the flow–synthesis platform. A two-pump Gilson 305/306 preparative HPLC system, shown in
Figure 3
, using the At-Column Dilution configuration
7
was able to inject crude reaction segments directly from the HPLC interface module of the Conjure flow synthesizer onto the column (Phenomenx Luna C18[2], 5 µm, 21.2 × 50 mm). Separation was achieved through gradient methods using acetonitrile and either 0.1% aqueous trifluoroacetic acid or 10 mM ammonium acetate, as the mobile phase, at a total flow rate of 30 mL/min. An Agilent G1968 active splitter
8
and Gilson 306 makeup pump (1 mL/min flow) were used to deliver a 1000:1 split of the preparative stream to a Thermo MSQ-Plus mass spectrometer operating in APCI mode, with the main preparative stream being delivered via the Gilson 155 UV detector to the TecanTM fraction collector (
Figure 4
). The HPLC system was controlled through a custom software application, written in house, to control all components of the purification system. Synchronization with the Conjure system was achieved through digital I/O (input–output) lines, providing signals for

Preparative high-performance liquid-chromatography (HPLC)–mass spectrometry (MS) purification system.

Custom-modified TecanTM Miniprep 75.
A two-arm Tecan Miniprep, shown in Figure 4 , is driven via a custom-written library, allowing a separate thread to control each arm. What this allows is true simultaneous operation of both arms as well as independence between the two. The right arm is controlled by the software’s queuing system, and the left arm is controlled by a separate thread of the program, allowing the left arm to continue processing fractions if the main program encounters an error or is halted by the operator for any reason.
The heart of the system revolves around the Mitsubishi six-axis articulating arm as seen in Figure 5 .

Mitsubishi six-axis articulating arm surrounded by stations.
Scheduling of the system is handled via custom, multithreaded software as shown in Figure 6 , which orchestrates which sample is being processed at which station. Each process station has its own queue that requests a Mitsubishi transfer. Because this is a true parallel operation, careful scheduling of the Mitsubishi arm is critical to minimize idle time. As new fractions come off the HPLC and are dispensed via the auto-sampler’s left arm, the software receives fraction information from the HPLC. The program when launched dedicates one thread to the auto-sampler that runs independently of the queuing manager, allowing running of the front-end synthesis manually if the automation is down for maintenance.
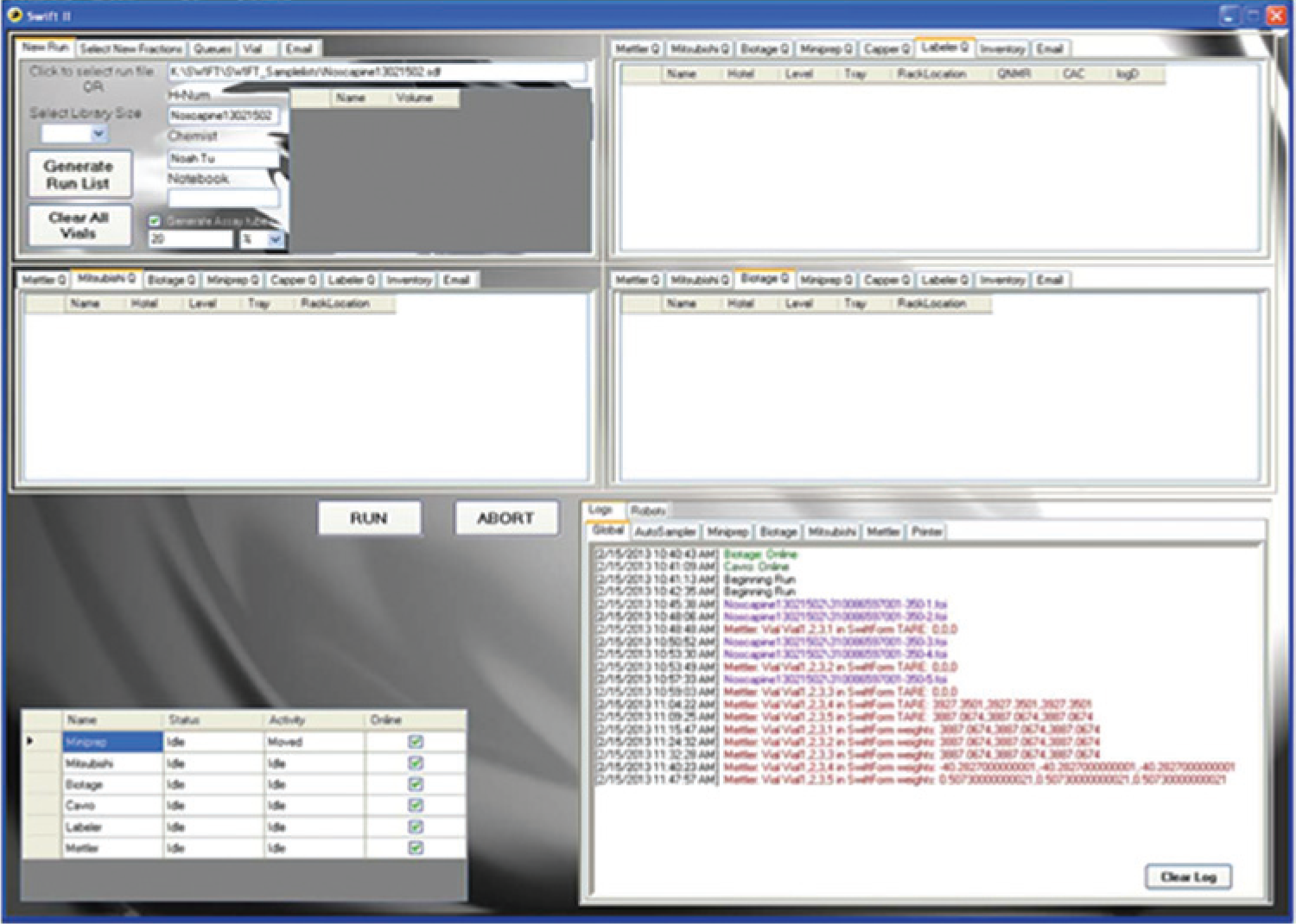
System-routing software.
The software also allows one or all of the systems to be taken offline during a run to allow hot fixes without interrupting the flow (e.g., if the labeler throws an error, the operator can take just that printer offline, fix the problem, and then place it back online, and the software will reincorporate the labeler into the queuing system). The operator can then manually apply the unapplied labels or have the system do it after the end of the current run. Similar offline capability is possible for the other systems as well. The only nonrecoverable failure is a Mitsubishi error. It cannot be placed into simulation during a run and have the run continue, or throw a fatal error and recover. The auto-sampler, however, will continue to index the fractions, allowing a run to be restarted at a later time.
At the time of fraction collection, the software selects an empty vial from the parking lot reserved for nuclear magnetic resonance (NMR) samples, as shown in Figure 7 . The Mitsubishi transfers the vial to the labeler, where a label is applied with the pertinent information. The arm then places the vial into a shuttle for the aspirating and dispensing of the purified compound for NMR analysis.

Parking lot for 4-mL vials.
Sample labeling
Figures 8

Labeling station.

The Biotage V10 Evaporator fitted with an in-house, custom-designed head.

Mettler–Toledo resolvation station.

Computer-aided design (CAD) rendering of the labeling station.
Dry-Down and Evaporation
The Biotage V10 Evaporator, shown in Figure 9 , is where the sample is dried down and evaporated. Times vary from 4 to 6 min of total evaporation time for volumes of 0.5 to 1.0 mL. The head of the Biotage was custom modified in house to allow removal of the 4-mL vial via automation. While the evaporator is running, the assay vial of the same fraction is selected, labeled, and filled from the auto-sampler. If additional fractions have been generated by the HPLC while the evaporator is running, the vials are processed and queued for dry-down by the software.
Sample Aliquot for Analysis and Resolvation System
Because the Biotage would require as long as 120 minutes to dry down a single large fraction and because the NMR quantitation is performed offline anyway, the natural breakpoint between automated sample handling and manual sample handling in the current system design is at the sample dry-down stage. Due to the high cost of deuterated solvents, a fixed volume of deuterated solvent (160 µL) is then manually added to dried NMR samples. The removal of purification solvent (principally aqueous acetonitrile mixtures) for the remaining vials is accomplished via commercial batch-centrifugal evaporators.
As shown in
Figures 10
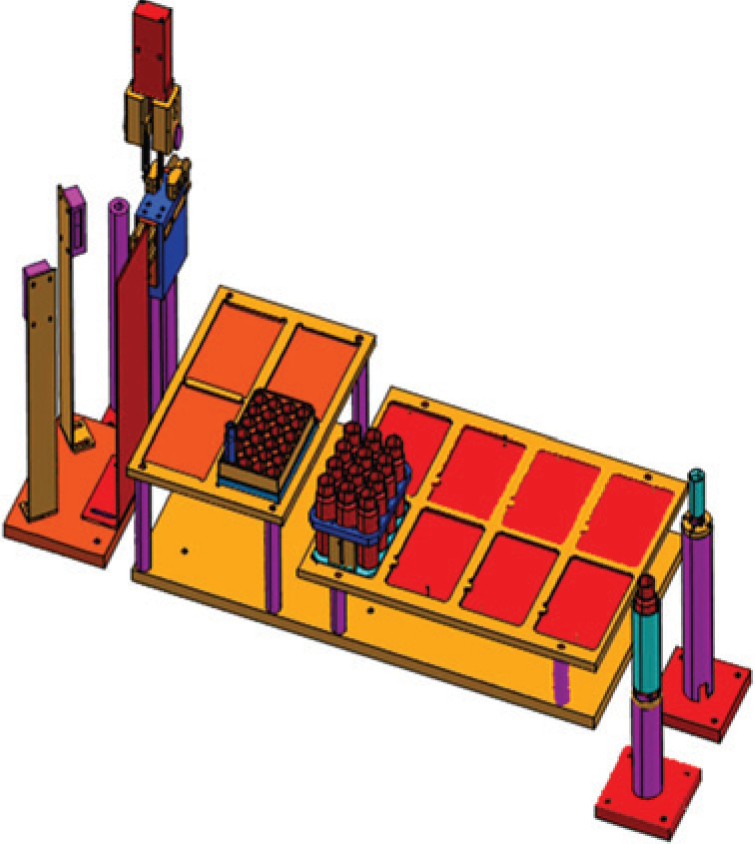
Computer-aided design (CAD) rendering of the resolvation station.
Structural evaluation and validation of samples are achieved by NMR spectroscopy of purified compounds. This process requires that samples be dissolved in a deuterated solvent such as d6-DMSO, rather than the acetonitrile–water mixture in which the samples elute from HPLC purification. An aliquot of the sample (5%) is used to create a sample for NMR validation. This is an automated process in the system, in which the requisite volume is calculated by software based on the HPLC flow rate and fraction-collection time (i.e., if the collected fraction elutes over 15 s of elapsed time, and the solvent flow rate is 40 ml/min, a 10-ml fraction will have been collected, and a 0.5-ml aliquot will be required for NMR). The fraction vial is auto-indexed on the auto-sampler system via the HPLC’s instructions. The auto-sampler is capable of dispensing 96 distinct purified fractions.
These automated validation and structural-determination processes were developed in house, 9 and they provide output data files that are archived and retrievable through in-place data-management systems.
Results and Conclusion
The ability to integrate components and automate sample handling has provided an efficient compound-generation system. The average cycle time to synthesize, purify, validate, and distribute samples from a 48-member library of 10-mg compounds is 3 days, nearly one-fourth of the cycle time for typical nonintegrated processes.
Although the time needed for a compound to pass through the synthesis, purification, distribution, and dry-down steps is about 30 min, the ability of the system to simultaneously handle compounds at multiple production stages shortens the overall production time for the library. On average, the system produces a new compound every 12 min. Thus, for a typical library size of 30 or fewer members, the entire process can be completed within 6 h with dry quantitative NMR (QNMR) samples (5% aliquot) ready to be dissolved in deuterated solvent. This enables acquiring of QNMR data overnight. If an additional set of samples is aliquoted for biological assays, the samples can be placed in batch-centrifugal evaporators overnight and be ready for submission to biologists in the morning. The turn-around time for this type of library is down to 24 h.
The process is further important in its impact on staff efficiency. The typical processes to achieve what the system accomplished in total would require a technician to intervene and transfer samples from laboratory to laboratory. Setting up a typical library-production run on the system required the operator to load common laboratory consumables (chemicals, empty vials, solvents, etc.) and reaction parameters exported from an electronic lab notebook. Once the integrated production process starts, it requires minimal to zero intervention from the technician.
A practical example of this has been published by Tu et al. to demonstrate accelerated click-chemistry library production. 10 Other libraries have been produced using a diverse array of chemistries, including amide bond formation from amines or carboxylic acids, reductive animation reactions, nucleophilic displacement reactions, sulfonylation reactions, and urea formation. Library sizes have ranged from 10 to 48 members with an average size of 20.
Footnotes
Acknowledgements
The authors wish to thank Rick Yarbrough, David Whittern, and Dr. Stephen Spanton for assistance with NMR results and David Blanchard, David Dingle, and Jonathan Trumbull for providing valuable input to process and instrument design. All contributors are AbbVie employees or are retired from AbbVie.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JDS, NPT, TAN, PAS, SWD, and JYP are all employees of Abbvie. JEH is a retired employee of Abbvie.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was sponsored by AbbVie. AbbVie contributed to the study design, research, and interpretation of data, reviewing and approving the manuscript.
