Abstract
After a lively discussion at LabAutomation2005 in San Jose, we asked three major diagnostic companies to share their vision for the molecular diagnostics “rapid-growing-and-more-than-ever” market. Happily, all of them complied and we are pleased to present insight from Bayer Healthcare, Beckman Coulter, and Roche Molecular Systems. To complement these perspectives, we invited a heavy user of molecular diagnostic assays to comment on today's situation and share his hopes for the future of molecular medicine. It is with pleasure that we feature insight from Philippe Halfon of Alphabio Laboratoire.
Introduction: A New Era for Amplification-Based Assays
Within the rapidly expanding world of available tools for the genomics universe, in recent years we have seen an amazing evolution of real-time PCR-based assays and procedures. Interestingly, these have changed the way we identify and quantify genes and pathogens, and they have propelled molecular diagnostics to new dimensions of importance in the healthcare industry.
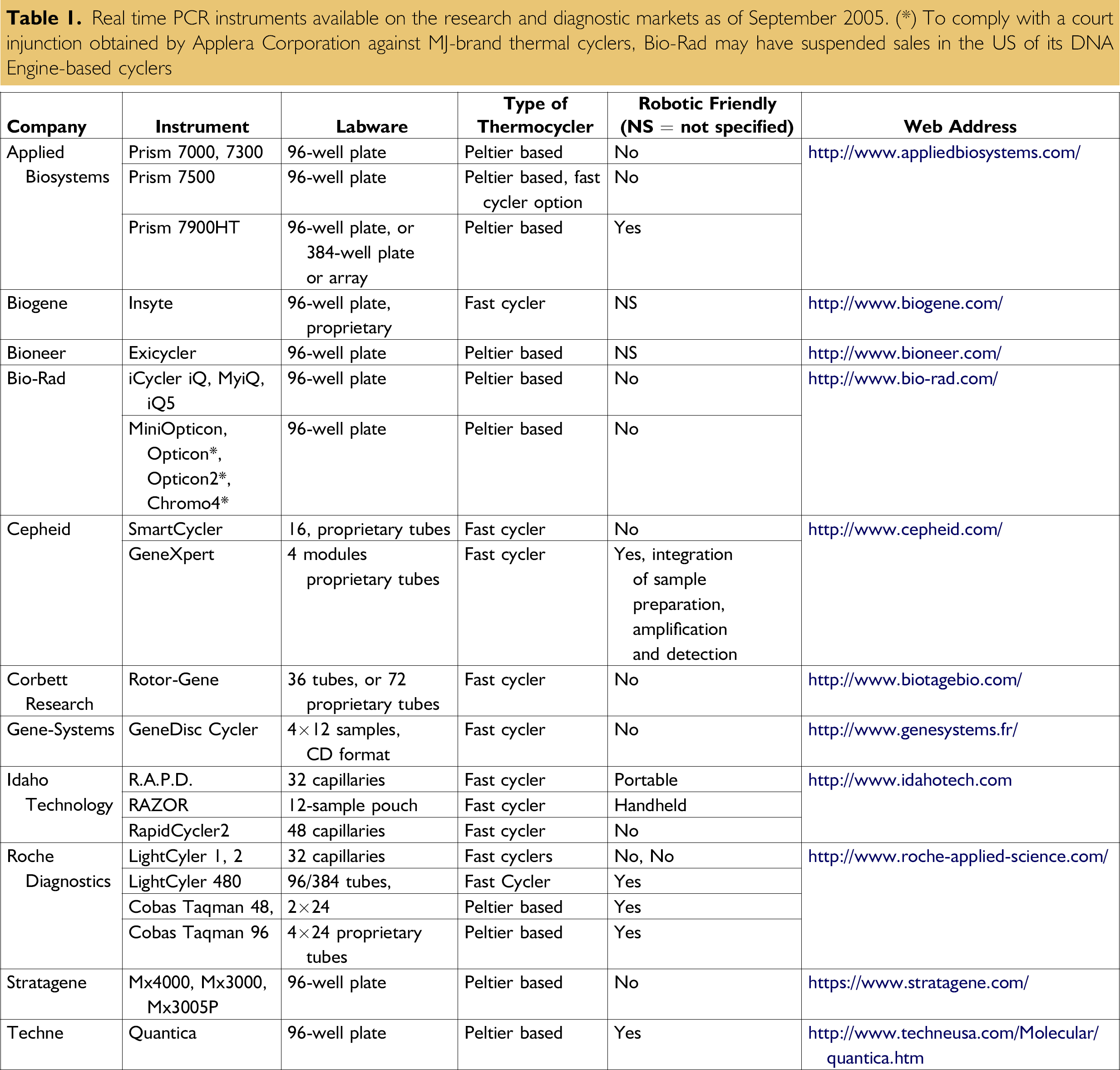
During the early 1990s, when Higuchi et al. 1 introduced the concept of PCR monitoring in realtime, only one instrument was available, the ABI Prism 7700 from Applied Biosystems, Foster City, CA, a precursor of today's new approaches. In 2005, more than 20 different instruments are available worldwide for real-time PCR in 96-well plate format or fast PCR-compatible labware (see Table 1). Not surprisingly, the molecular diagnostic assays that relied heavily on PCR plus electrophoresis or hybridization switched en masse to real-time PCR procedures. Molecular biology and medical journals are now reporting new diagnostic assays for mutation screening, pathogen detection and quantitation, and gene expression profiling using real-time PCR. This may not be the last technology that the biomedical testing continuum will benefit from, but we must admit that it has considerably changed our daily laboratory life, and more generally, our medical practice and healthcare vision. This is especially true when considering the huge impact that molecular testing will have on disease predisposition, drug metabolism, and drug resistance monitoring. It is defining a new era for personalized medicine.
Real time PCR instruments available on the research and diagnostic markets as of September 2005. (*) To comply with a court injunction obtained by Applera Corporation against MJ-brand thermal cyclers, Bio-Rad may have suspended sales in the US of its DNA Engine-based cyclers
The success of real-time PCR in the molecular diagnostic community is obvious. Real-time PCR has simplified, automated, and replaced most of the procedures used after a regular PCR reaction such as gel electrophoresis or reverse-hybridization in a microtiter plate. Ease of use, short time-to-result, and low cost have always been of importance to laboratories. Now, real-time PCR offers new opportunities to increase efficiencies: extract DNA or RNA, set up the real-time PCR reactions, run a real-time PCR instrument method, and you are done. Molecular diagnostic procedures have been shrunk to just a few steps, including sample preparation, reaction setup, and real-time PCR. Of course, many of these assays still are not real-time PCR compatible, but more than 75% of molecular tests presumably are amenable to this technology.
Not only has the real-time PCR revolution had a clear impact on the lives of its users, but it also has stimulated an entirely new molecular biology market, which provides new reagents and chemistries for real-time procedures. The Taq-Man procedure (Roche Diagnostics, Basel, Switzerland), hybridization probes, Molecular Beacons (PHRI, Newark, NJ), Scorpion Primers (DxS Ltd., Manchester, U.K.), and Lux Primers (Invitrogen, Carlsbad, CA) are just a few of the new chemistries now available for rapid assays based on real–time PCR. Real-time NASBA (Nucleic Acid Based Amplification, Biomérieux, Marcy l'Etoile, France) and soon, real-time TMA (Transcription Mediated Amplification Gen-Probe, San Diego, CA) are two other major “chemistry upgrades” that will certainly boost this market.
Presumably, the major impact of real-time PCR technology has been the rapid development of automated platforms, directly linked to thermocyclers of course, but also linked to the front-end of PCR assays, along with the special case of nucleic acid extraction. The concept of integrating two instruments for PCR assays is not new, but with real-time PCR as the detection step, it makes the concept simpler than ever.
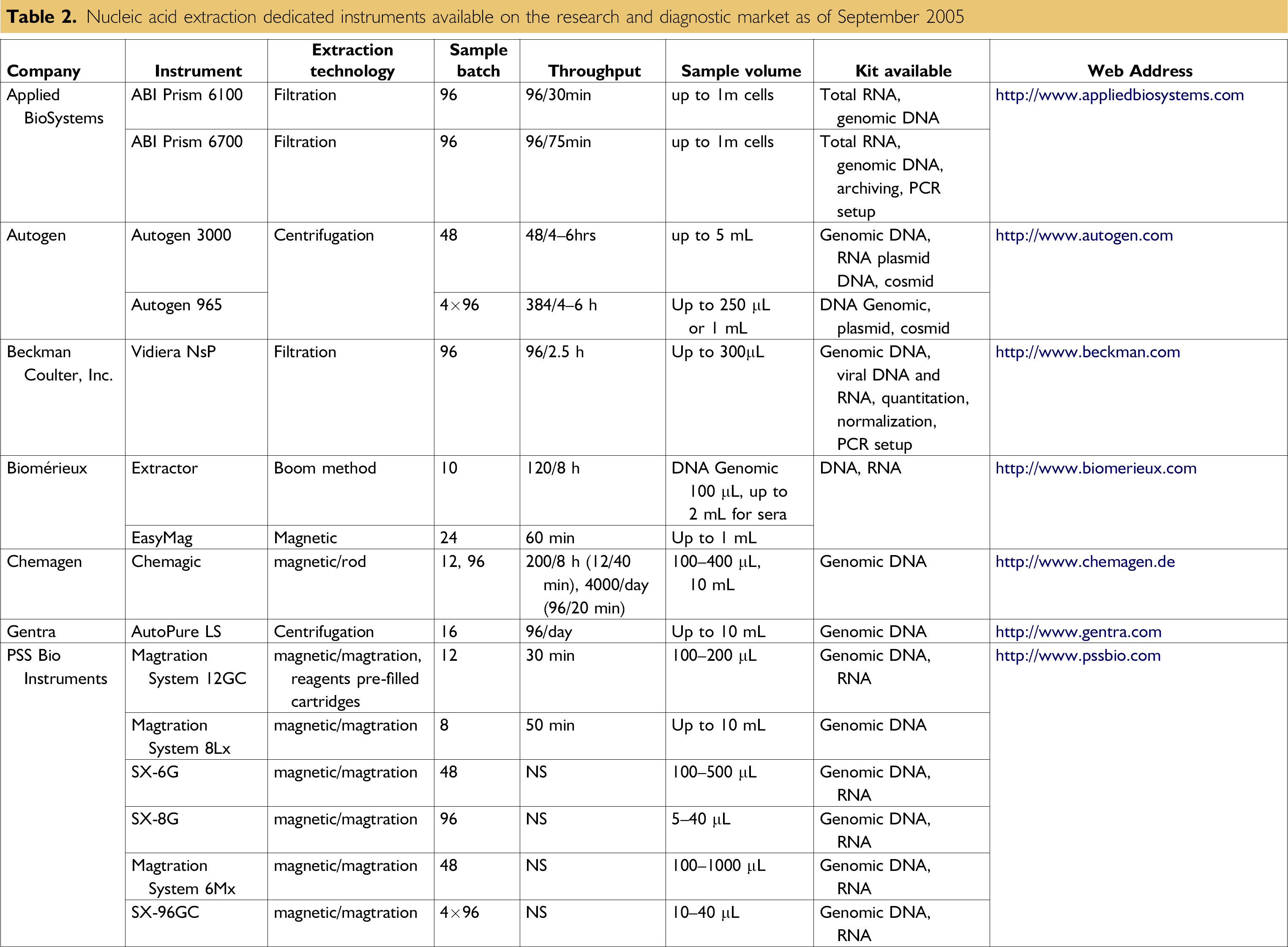
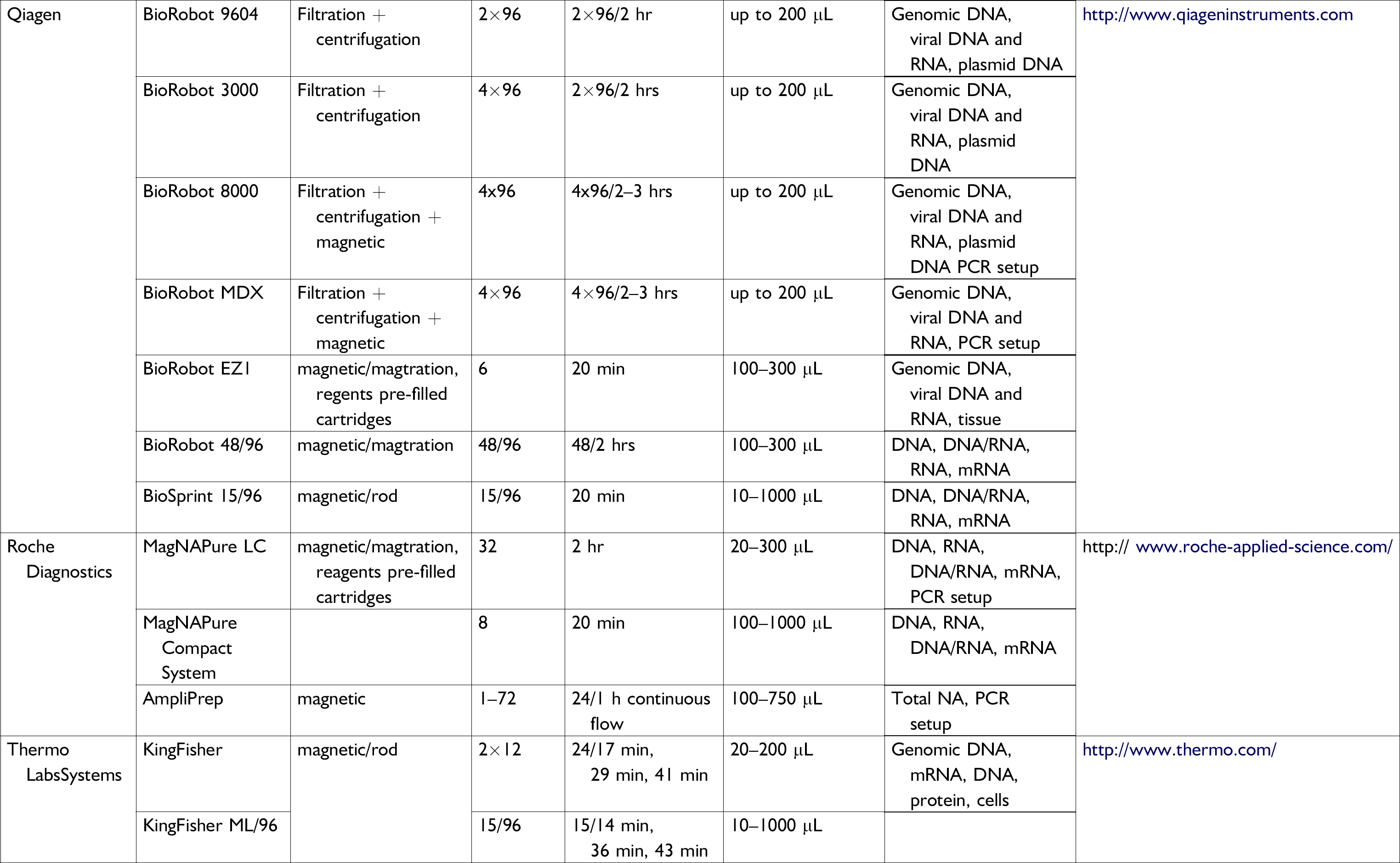
Not only is the market for real-time PCR instruments a fast-growing one, the market for nucleic acid extraction platforms has shown a constant evolution in the last 5 years, with the development of more than 20 instruments (see Table 2). From centrifugation-based instruments to magnetic-beads-based platforms, the market is growing steadily from R&D-based instruments to diagnostic labeled configurations.
Nucleic acid extraction dedicated instruments available on the research and diagnostic market as of September 2005
Filtration-based procedures have been driving the nucleic acid extraction market during the past 10 years. Platforms that automate such procedures are more common and have been the first to enter the diagnostic market. Not surprisingly, many molecular diagnostic assays have been validated with these reagents and platforms. Now that simplicity and shorter time-to-result are two major requirements, not to mention lower cost, the fast magnetic-beads-based procedures have become favorites. Various technologies for magnetic beads are available on numerous platforms, with some of them already in vitro diagnostics directive 98/79/EC compliant. No doubt, they will be the next generation of diagnostic instruments.
In this context, diagnostic companies have participated in this new evolution of molecular biology. Many small and major companies are now providing real-time amplification assay kits and reagents. Automated solutions for nucleic acid extraction packaged with reagents are becoming available from major diagnostic companies, and numerous commercial options are available elsewhere. Providing reagents with dedicated automated instruments is a major advancement for the molecular diagnostics business. These are available from a few of the players in this business, Roche Diagnostics being the initiator of this strategy. From a business perspective, molecular diagnostics is just beginning to enter the world of clinical diagnostics.
Bayer healthcare LLC: AUTOMATION FOR GENOMICS
The “Genomics Revolution” is fueled by anticipation that knowing the relationship between a genetic profile and disease will revolutionize the way medicine is practiced. Patient genotyping will be used to predict risk for developing a disease, diagnosing a current illness, predicting disease progression, and selecting and predicting therapeutic response. Simultaneously, the genomic revolution is advancing a therapeutic revolution that is resulting in an explosion of new drugs to treat or control diseases by specifically targeting the molecular pathways of the disease. Genomics provides the information for new drug discovery and the tailoring of these drugs to enhance efficacy and minimize toxicity. A key driver for the genomics revolution has been the development of new technologies and the automation of procedures for sample handling, genome interrogation, and data analysis.
Cancer and cardiovascular disease involve a complex interaction of multiple genes and environmental factors. A gene variation has the potential to alter the disease course and the response to therapy. To improve knowledge of complex diseases and to understand the relationship between a genetic profile and disease, scientists have developed technologies to evaluate individual base changes within genes and how these changes may affect cellular processes.
Bayer Diagnostics recognizes the need for molecular tools and assays to complement the genomics-based medical breakthroughs that will lead to individualized treatment. Automation and more complex data analysis will become a mainstay in clinical diagnostics laboratories. Building upon Bayer's technology and platform base in detection and quantification of viral nucleic acids, Bayer has invested in the automation of analytical procedures including sample handling and high throughput nucleic acid signal and target amplification technologies. Additionally, Bayer, through its acquisition of Visible Genetics, Inc., in 2002, now has a sequencing platform and viral resistance products. More recently, Bayer entered into a collaboration with Amersham for joint development of assays and sequencing instrumentation in a high throughput setting. These relationships give Bayer access to sequencing platforms suitable for both the viral and human genomics markets.
When genomic variations are spread over large distances of the genome, sequencing may not be the favored approach for identification of single nucleotide polymorphisms or mutations. Other multiplex technologies such as liquid- or solid-based arrays are more suitable. Toward this end, Bayer has gained access to the Luminex multiplex liquid bead array platform and xMAP® technology that can currently detect and quantify up to 100 different targets simultaneously. This technology allows for interrogation of DNA, RNA, and protein -based targets. Bayer is also evaluating solid-based arrays that could detect and quantify larger numbers of concurrent targets and could serve both the laboratory and point-of-care markets.
Advances in technology, coupled with improved understanding of the human genome and/or the relationship of pathogen to disease, will lead to better diagnostic products and personalized management of complex diseases. Bayer Diagnostics is investing in platforms and assays that will help revolutionize the way medicine is practiced in the coming years.
Beckman coulter, Inc.
Beckman Coulter has been serving research genetics with automated solutions through its Biomek family of products since 1987. These laboratory automation workstations have enjoyed a significant market share because of their performance and flexibility in research genetic analysis. They have been applied to sample isolation and purification, as well as pre-PCR reaction setup and post-PCR applications. Beckman Coulter is also the leading supplier of automated diagnostic lab systems that connect hematology, homostasis, general chemistry, and immunodiagnostics instruments into a single command console. The objective in a routine diagnostics laboratory is to go beyond automating individual disciplines of testing processes by automating sample movement throughout a laboratory of many testing disciplines. This objective is in contrast to the molecular diagnostics testing marketplace, which we believe is still in an early stage of automating the various test processes.
Labor shortages and productivity needs drive the economics of automating a moderate-complexity clinical lab, while the metrics that drive automation in high-complexity molecular labs are more plentiful. Labor shortage is also prevalent in these high-complexity labs, but lack of standardization of the testing process across all assay types is a challenge for developing an automated and integrated platform. The molecular-based infectious disease market has been driving automation of molecular diagnostics with standardization of assays. However, the economics of purchasing an automated integrated system mainly for virology testing is not attractive in many labs, unless this capital purchase can be leveraged to automate additional assays.
The preanalytical step of sample preparation in molecular testing represents one area that can be automated independent of the type of assay. This will enable the laboratory to leverage their capital outlay over molecular virology and pathology assays and makes the economics more attractive for the broader base of labs performing, or wishing to perform, molecular testing for patient management. Many molecular diagnostic labs have told us they are planning to consolidate as many assays as possible to the fewest number of detection platforms. PCR and real-time PCR have been adopted for many clinical molecular testing techniques to meet this desire or need. However, these methods are not as practical for assays that require multiple marker identification or ones that require separation technologies such as capillary electrophoresis. Beckman Coulter believes that multiple modes of detection will be required eventually for the various molecular assays required to fulfill unmet clinical needs.
Beckman Coulter's initial plan for automation in molecular diagnostics is to leverage its experience in research genetics through the Vidiera™ family of products. The Vidiera products will simplify and automate the molecular testing process in high-complexity molecular labs. One of our initial platforms, Vidiera NsP (Fig. 1), is designed to offer complete walkaway automation of the preanalytical step. The platform will prepare the sample from a primary tube, optionally quantify and normalize the extracted nucleic acid, and set up the reaction for amplification. It is versatile enough to set up reactions for most downstream amplification/hybridization platforms that labs are using today.

The new Vidiera™ NsP nucleic sample preparation system from Beckman Coulter provides flexible automated nucleic acid isolation, quantitation and normalization, and reaction setup.
We are also introducing a separation and detection platform, Vidiera NsD, which is based on the separation technology of capillary electrophoresis. This open platform will enable labs to consolidate homebrew assays postamplification, which are not well suited for real-time PCR.
Beckman Coulter continues to offer the research market automated solutions to speed discovery and to aid clinical research. An enhancement to our microarray detection platform, SNPStream™ Genotyping System, is the addition of chemistries that enable up to 48 reactions per well in a 384-well microtiter plate. These chemistries are also scalable to lower-density platforms. We have the ability to offer scalable technologies using the same supporting chemistries across multiple platforms. We have the solutions to automate the reaction setup for these platforms, and we have in development an automated sample preparation solution, ArrayPlex SA, that is the result of a joint venture with Affymetrix, Santa Clara, CA, to automate processing of the Affymetrix chip.
We envision some forms of these multiple technologies will migrate into clinical research, and eventually the assays of clinical relevance will transfer to diagnostics. We believe that the integration of these technologies into a single automated platform is possible, and Beckman Coulter is defining that path by leveraging the internal strengths across our company. In the future, we see the discipline of “molecular diagnostics” as just another discipline in the moderate-complexity clinical lab.
Roche molecular diagnostics: evolution of Nucleic Acid Testing (NAT) automation d enabling transformation of molecular technology from research to IVD
The molecular diagnostics market can still be considered to be in its infancy. Compared to areas like clinical chemistry, only about 7% of all diagnostics laboratories are using molecular methods. This is mainly a result of the highly complex nature of the technology and the limited number of In Vitro Diagnostic (IVD) tests that are commercially available. Both factors have led to centralization of testing because larger laboratories have been better equiped to handle the procedural complexity as well as the higher cost structure. Larger testing volumes in these sites have led to consolidation of the tests into batches to ensure efficient processing.
On the other hand, early adopters in hospitals and universities have expanded the number of molecular tests through laboratory-developed tests (homebrew) beyond the portfolio currently offered throughout the industry. This market segment most recently adopted open real-time PCR platforms such as the LightCycler instrument and the ABI Prism 7700 as their testing platforms. These early adopters also spearheaded the use of automated sample preparation platforms like the MagNA Pure LC instrument or the BioRobot, increasing test volumes while at the same time reducing the complexity of the technology.
The development of IVD platforms always trailed behind the availability of such open platforms in the “homebrew” segment. With the introduction of the COBAS AMPLICOR analyzer in 1995 in Europe, Roche Diagnostics launched for the first time an instrument developed specifically for the molecular diagnostics market. Integration of amplification and detection in a single instrument substantially reduced both hands-on time and total turnaround time. In 2001 this was followed by the launch of COBAS AmpliPrep instrument in Europe, the first sample preparation instrument focusing on the needs of routine diagnostic laboratories and designed to work with the COBAS AMPLICOR analyzer as a backend instrument.
In 1999, Roche launched the MagNA Pure LC sample preparation instrument for the research market that uses generic magnetic-particle-based capture. In the same market segment, the LightCycler instrument enabled researchers to convert their tests to real-time PCR. Real-time instruments substantially decreased the turnaround time and in addition reduced the risk for contamination through the use of completely sealed amplification vessels.
Introduced in 2003, the COBAS TaqMan 48 analyzer was the first real-time instrument in the industry specifically targeting routine diagnostic customers. In the meantime, Roche had changed sample preparation technology on the COBAS AmpliPrep instrument to generic capture as well and adapted it to fit the TaqMan technology. The launch of the COBAS AmpliPrep instrument with the COBAS TaqMan analyzer in 2005 is now allowing customers to convert their testing for most virology assays to real-time technology in combination with fully automated sample preparation. The offering of two differently sized TaqMan instruments combined with a sample preparation instrument accommodates a variety of throughput and workflow requirements. The larger Taq-Man analyzer can be directly connected to the sample preparation instrument, thus allowing walkaway automation for the first time.
What will be the next steps in the automation of molecular diagnostic assays? Integration of amplification and detection has been a major advance in increasing the ease of use and reducing hands-on time for operators. Obviously, the integration of the sample preparation step is the next logical extension. Glass magnetic particles have turned out to be easy to automate and can be adapted to most applications (Agen-court Bioscience, Beverly MA; Agowa, Berlin, Germany; Dynal Biotech, Oslo, Norway; Seradyn, Indianapolis, IN; etc.). However, more development of this technology and its automation will be required in order to offer a broader collection of sample materials such as tissue.
Size and cost of current equipment can still become a barrier for adoption of PCR for smaller laboratories. Integration of process steps as well as miniaturization of components will help eliminate this barrier. Decreasing reaction volumes will also help in reducing the overall turnaround time of PCR assays, which will be critical for the development of new assays with shorter turnaround time requirements such as those products targeted for emergency room and hospital use.
With the introduction of the FDA-cleared AMPLICHIP CYP450 test, Roche has entered into a new area in the diagnostics of highly complex targets. Whereas the current microarray is making use of existing automation, future platforms will require a high degree of integration of the different process steps. Connectivity with existing lab equipment for sample preparation and amplification will reduce the number of new platforms required and decrease the investment hurdle. While microarrays will be indispensable for highly complex assays, multiplexing of real-time assays may allow targets with lower complexity to be addressed most efficiently. It is conceivable that both types of tests will converge at the lower end of the complexity scale.
Figure 2 outlines the launch of Roche's instrument platforms for molecular diagnostics, research and diagnostic homebrew, and the decreasing turnaround time for results.
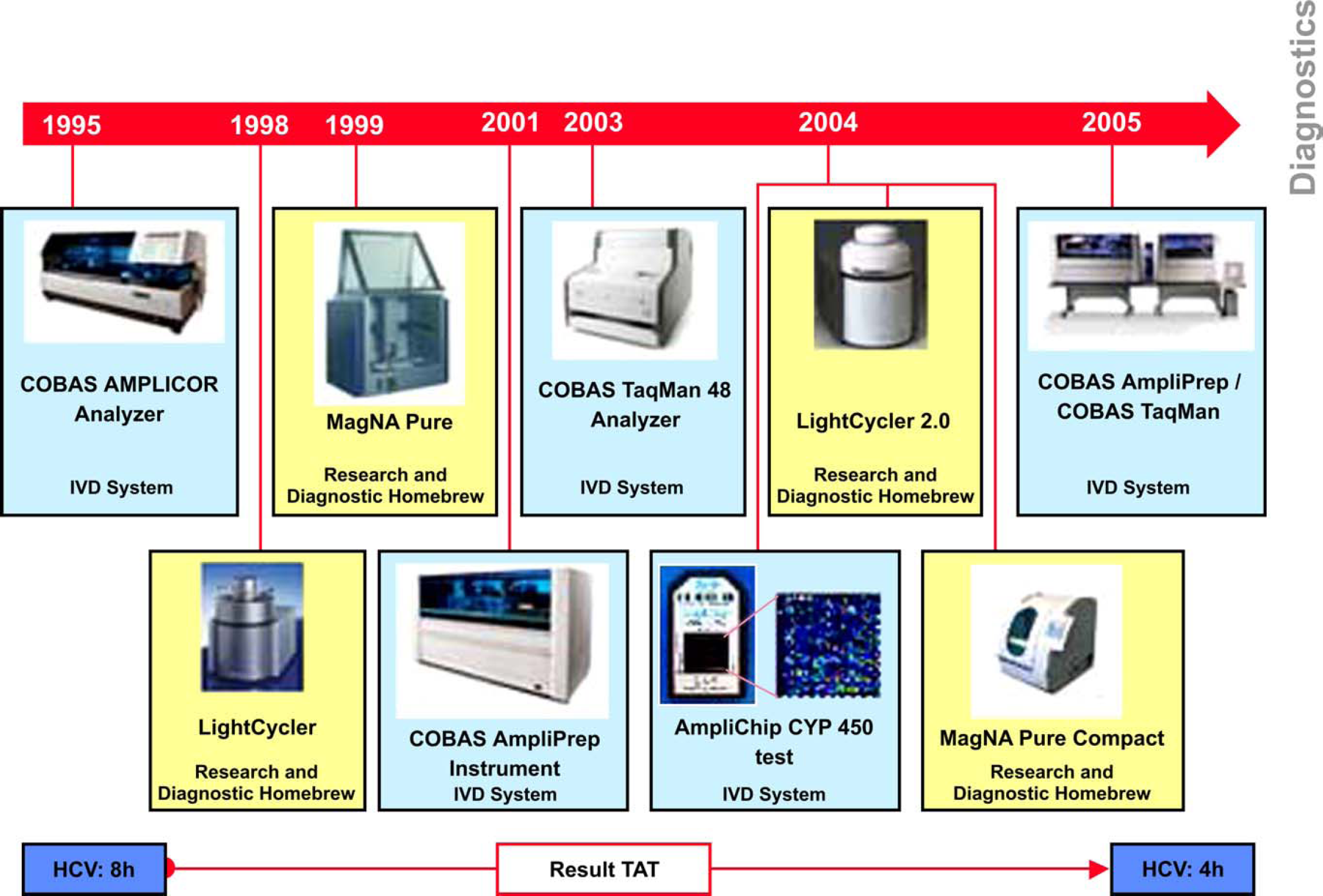
Evolution of Roche platforms for molecular diagnostics, research, and diagnostic homebrew.
In summary, we believe that the future of molecular diagnostic automation will be characterized by further integration of process steps and standardization. In addition, adaptation to a broader variety of targets and sample materials will be required. It is not too far-fetched to assume that molecular diagnostics will ultimately develop in a manner similar to other diagnostic disciplines such as immunochemistry. Thus, in the future we may even see platforms for decentralized testing sites such as the intensive care unit or even point-of-care settings.
Molecular diagnostics from reagents to platforms requirements: a french reference laboratory wish list
Guided by government rules, competitive environments, quality assurance programs, and economic pressures, the routine diagnostics for infectious disease laboratories reached the laboratory with complete automation. For the past two decades, PCR technology has allowed laboratories to quantify and identify genes and pathogens and has brought clinical diagnostics to a new dimension. Molecular diagnostics companies have solved problems found 50 years ago with routine glucose analysis, and brought to the market assays like complex human immunodeficiency virus (HIV) genotyping with the same level of precision and confidence. How did these companies win their technological wager? Was it by passing over the nucleic acid barrier and considering it as a simple protein or a lipid fraction? And how did these companies make the step to DNA or RNA extraction, real-time PCR reaction setup, and real-time PCR instrumentation? They had to consider the constraints met in a laboratory.
A central laboratory involved in molecular diagnostics will need:
reliable workflow in compliance with a central facility,
a quality assurance program to be included at each step of the process,
a guarantee for a contamination-free process,
tests with high sensitivity, specificity, and large dynamic range,
adherence of results to standard International Units,
participation in worldwide programs for pathogen variability monitoring.
The use of sensitive and quantitative PCR-based assays has changed the management of viral infections. For HIV, Hepatitis C virus (HCV) and Hepatitis B virus (HBV) testing, the challenge of molecular diagnostics has been driven by the following key points:
Correlation between the assays has not been addressed at this time despite the use of International Units. Presumably, the assay's internal standards are different among diagnostics companies, and for this reason, it is recommended that a single methodology be used to monitor a patient's viral load.
Quantification uniformity among subtypes is a crucial point for customers' acceptance. It requires similar sensitivity and dynamic range among the various subtypes of a single pathogen.
Availability of assays for fluids other than plasma or sera (cells, peripheral blood mononuclear cells, bone marrow cells, cerebrospinal fluid, sperm, sputum, urine, liver … etc.).
Development of new genotyping technologies to monitor disease progression and treatment—this point is being achieved. However, an assay's accuracy is still dependent on interpretation algorithms from various sources, with various rules, from simple to complicated interpretation.
In this context, what are the commercial options for viral disease management?
Both small and large companies have participated in this new evolution of molecular biology. But today, only a few players are able to offer a complete product line to clinical laboratories, providing reagents and dedicated automated instruments. It is indeed a very new era, with new options for laboratories.
Roche Diagnostics is one of the companies that offer an all-in-one solution strategy by providing its own reagents and automated solutions for nucleic acid extraction (MagNA Pure LC, COBAS AmpliPrep), and amplification plus detection (COBAS AMPLICOR, LightCycler, COBAS TaqMan 48 and 96).
Abbott Molecular Diagnostics, Abbott Park, IL, also participates in this market, but chose a different approach by organizing each step with various combinations, like extraction reagents and instrumentation (Promega Corp., Madison, WI, and Tecan AG. Mannerdorf, Switzerland), or real-time reagents and instrumentation (Artus Biotech, Hamburg, Germany; Abbott Diagnostics, Celera Diagnostics, Rockville, MD).
Gen-Probe is actually a unique example of molecular diagnostics integration, with the molecular screening of infectious diseases implemented in a unique and single instrument, the TIGRIS.
Others like Bayer HealthCare Diagnostics, Tarrytown, NY, with Chiron, Emeryville, CA, Biomerieux, DiGene, Gaithersburg, MD, have developed tools for this market, but they have not yet developed a final integrated solution.
Maybe the most significant advancement brought by these companies' strategies lies in sample preparation steps, with a special mention of viral RNA extraction. Regular sample preparation protocols are time consuming, labor intensive, susceptible to contamination, and limited to low-volume samples. A fully automated, high-throughput nucleic acid purification system was needed for the sensitive detection of HCV and HIV RNA. We are experimenting with HCV, HBV, and HIV viral load testing using the two most advanced systems available in this category.
The Abbott m1000 is a fully automated sample preparation system. It processes samples with volumes up to 1 mL using magnetic-bead procedures. It brings higher throughput and larger volumes handling to viral assays with 48-sample batch processing in about 2 h, starting with primary bar-coded tubes. However, with this open-platform concept, some concerns remain about sample carryover.
The Roche COBAS AmpliPrep instrument purifies RNA or DNA targets in a continuous flow that includes loading samples in closed tubes on racks, lysis, capture, washing, elution, PCR setup, and archiving back in closed tubes. Sample batch is from 1 to 72 samples but unfortunately does not allow primary tube input. The whole process is however contamination free (closed cabinet, uracyl DNA gycosylase-based chemistry for PCR) and sample carryover free with closed tubes during process.
In our hands these two systems have enhanced our workflow reliability, quality assurance programs, assay sensitivity, and dynamic range. The complete investigation continues. The major remaining challenge is to find the best balance between performance, cost, and automation capacity. Right now, it has clearly enhanced our laboratory organization while bringing new perspectives to patient monitoring and treatment, and more generally to molecular medicine.
