Abstract
Molecular diagnostics presents challenges to clinical laboratories that are under pressure to consolidate and automate. There is a need to evaluate molecular automation for process efficiency and suitability for high-throughput environments in core laboratories.
A fully automated molecular instrument platform (the BD Viper System with XTR Technology in extracted mode [BD Viper System with XTR]), was evaluated for automation efficiency, labor requirements, and system robustness. System productivity was predicted using time and motion studies as well as process simulation.
Preanalytical steps required 15 min of skilled operator time. The BD Viper System with XTR fully automated DNA extraction, amplification, and analysis of 368 specimens (736 results for Chlamydia and Gonorrhea). Time and motion studies estimated that the total hands-on full time employee (FTE) burden was approximately 35 min per run, of which 41% was high complexity, 29% medium complexity and 29% relatively unskilled labor. A skilled operator can easily operate four instruments for 8.5 h and generate data on 2944 results (four runs on each instrument for a total of 1472 clinical specimens) for
We determined that fully automated molecular analysis of GC and CT is possible in a core laboratory facility with significant throughput and minimal impact on labor demand using a fully automated and robust molecular diagnostics platform (such as the BD Viper System with XTR).
Introduction
Molecular diagnostics has enabled genetic testing of inheritable diseases, as well as specific identification of microorganisms that cause infection. Because of the ability to exponentially amplify both ribonucleic acids (RNA) and deoxyribonucleic acids (DNA) using specific polymerases, molecular methods can be both highly selective and provide almost single molecule sensitivity.
1
The increasing demand for molecular testing coupled with financial pressures to reduce the cost of performing these tests has resulted in the need for automation, especially for sexually-transmitted disease (STD)-targeted assays which routinely have high sample volumes and can be processed in batches. Until recently, execution of these tests has required extensive training in molecular biology, and an educational background equivalent to a masters or Ph.D. level scientist. Automation can standardize the performance of molecular testing methods, simplify the process of analyzing numerous specimens, and increase the throughput of specimen testing, while reducing labor costs traditionally linked to manual testing modes. Laboratories that have adopted automated molecular systems have been able to move
Automation, which combines process management software and laboratory robotics, has been increasingly used to reduce the need for human labor in clinical laboratories. 2 Robotic workstations are defined as automated platforms that perform selected laboratory tasks 2,3 including various aspects of molecular biology. 4 –6 For example, amplified DNA or RNA is monitored in a closed tube system using real-time fluorescence to measure product yield as a means to determine the presence of infectious agents in a clinical specimen and potentially to further characterize or genotype microorganisms by conducting DNA melt curve analysis 7 for greater clinical impact. The latest generations of analytical systems that will enable molecular diagnostic integration into the core laboratory integrate sample extraction, nucleic acid amplification, and detection 8,9 either on one instrumented platform or in a multi-instrument modular format.
Several partially or fully automated molecular diagnostic systems are currently available, such as the Roche COBAS AMPLICOR System (Roche Diagnostics, Indianapolis, IN), 10,11 the Abbott m2000 (Abbott Diagnostics, Abbott Park, IL), 9 the GenProbe Tigris DTS (GenProbe, San Diego, CA), 9 and the BD Viper System with XTR (BD Diagnostic Systems, Sparks, MD). 9 Because limited information is available regarding the process efficiency, skill level, and task intensity necessary to operate these instruments, we undertook to analyze the BD Viper System with XTR, which is designed for unattended extraction, amplification, and detection of CT and GC in urine or swabs of vaginal and urethral mucosa. Our goal was to evaluate labor requirements for the BD Viper System with XTR operation in a core clinical laboratory.
More than 90 million cases of STDs are diagnosed each year 12 ; however, up to 90% of infections may remain asymptomatic. 13 Thus, there is an increasing demand for testing STDs such as CT and GC. In addition, with a growing use of liquid-based cytology (cervical smears placed in liquid media), it is anticipated that molecular testing for CT in high-risk subjects will increase. 14 This is anticipated to place a burden on the molecular diagnostics laboratory to perform high-throughput analysis of aliquots from such cytology specimens. Furthermore, application of molecular assays is expected to increase for all microbiological testing 15 and even more dramatically because of discovery of single nucleotide polymorphisms for the prospective diagnosis of chronic diseases. 16 Automated molecular diagnostic platforms will be required for nonspecialty and high-throughput laboratories to perform these tests.
Methods
System Analytical Performance
The analytical performance of the BD Viper System with XTR has been reported elsewhere. 9,17,18 These studies are summarized in the discussion.
Process Evaluation
A two-day process evaluation and optimization was performed using time-stamped videotape and process analysis for each labor step (see Fig. 1; Table 1). We assumed that one technologist equivalent (up to a maximum of 8.5 h of labor) was used to perform all labor activity for analyzing 92 specimens and four control samples for each run. The work shift spanned the time from 8:00 AM to 4:30 PM allowing for multiple runs. We also assumed that pre-bar coded specimens would be delivered to the analytical area as is common in most commercial level clinical laboratories.
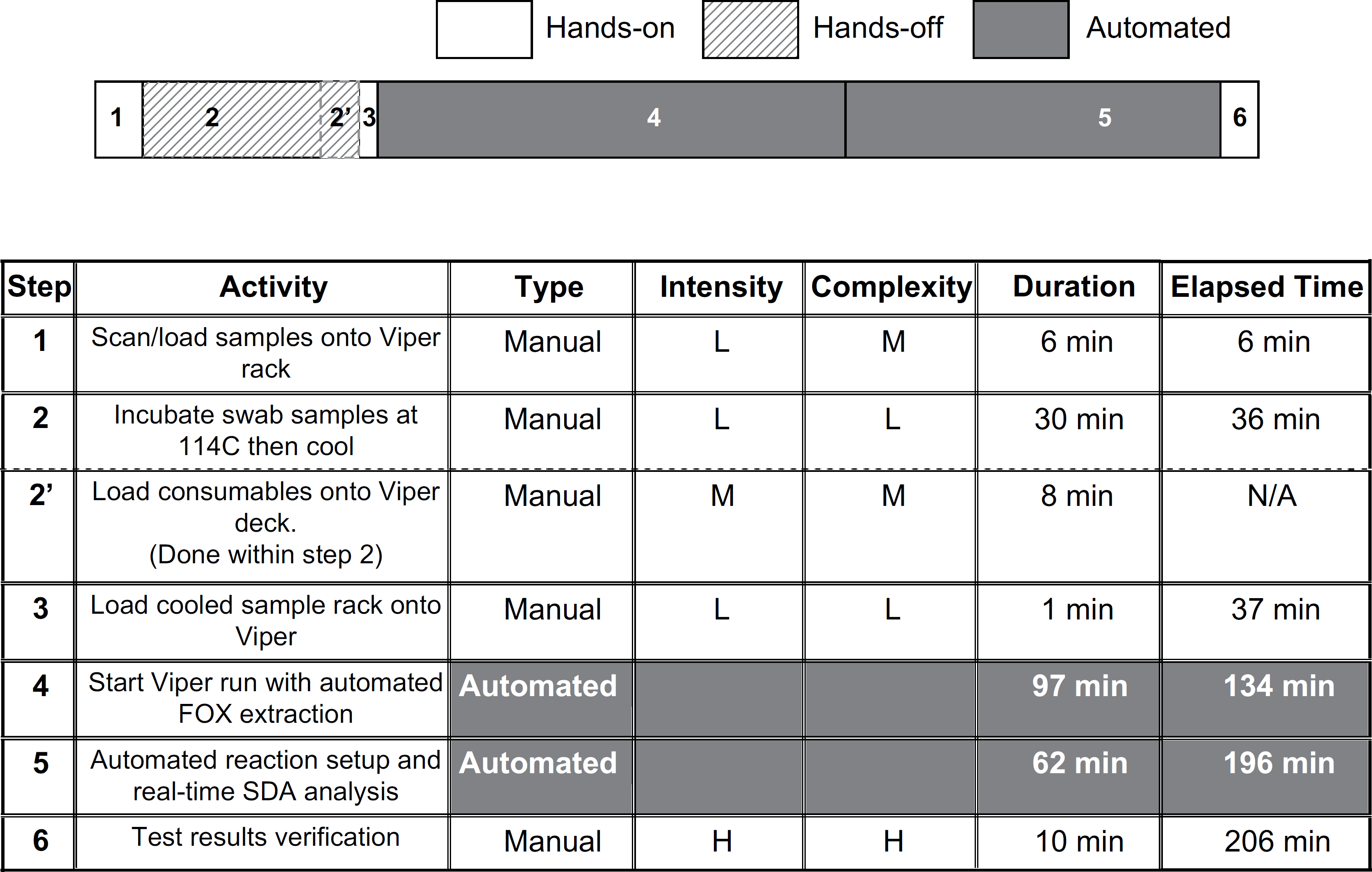
Sequential flow of manual and automated steps and the time required for each step in the analysis of
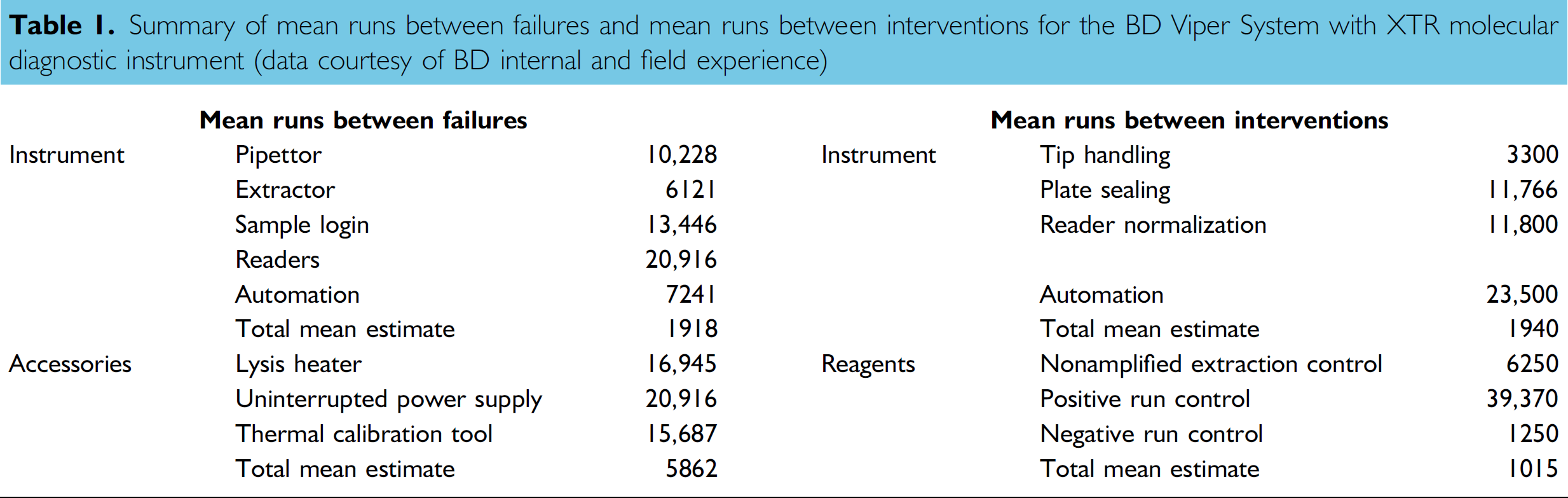
Summary of mean runs between failures and mean runs between interventions for the BD Viper System with XTR molecular diagnostic instrument (data courtesy of BD internal and field experience)
The various steps required to run a batch of clinical specimens on the BD Viper System with XTR was assessed with respect to task intensity and complexity, which provides additional valuable information to determine the skill level of the operator that may be required to successfully complete each task. In Figures 1 and 2, shading was used to indicate which operations require technologist manual hands-on time versus other operations that where either hands-off or fully automated by the BD Viper System with XTR. Skill level was divided into three levels: low complexity (may be performed by a clinical laboratory associate, or an entry-level clerk), moderate complexity (may be performed by a clinical laboratory scientist or a trained medical technologist with at least 1 year laboratory experience, and several months experience with the BD Viper System with XTR), or high complexity (may be performed by a clinical molecular biologist or a technologist with at least 6 months experience using the instrument on a weekly basis). Task intensity was scored as low intensity (such as loading of reagents, wiping down surfaces, or other simple task with no significant time pressures), moderate intensity (routine pipetting, loading of consumables, or other moderately complex task with some opportunities for errors), and high intensity (organizing hundreds of specimens, or operating software under a time pressure where training is essential).
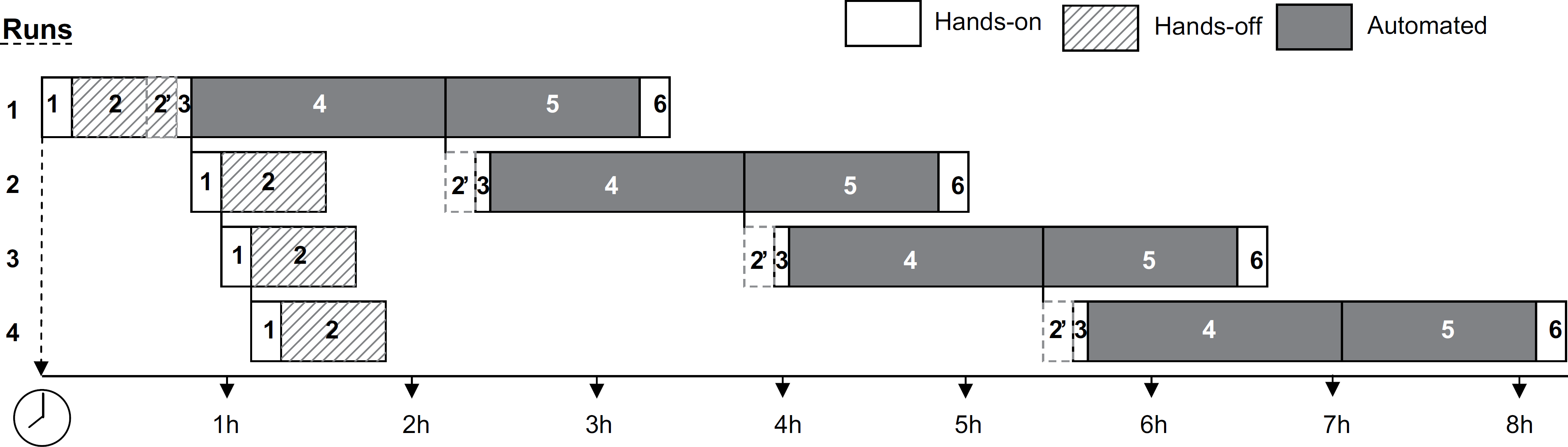
Felder–Foster plot for maximizing the number of BD Viper System with XTR runs in a given day using one instrument. This figure depicts how a single operator can plan an 8.5-h shift for maximal throughput of clinical specimens (in this case, swabs) on a single BD Viper System with XTR instrument. Automated routines are shaded in dark gray, whereas hands-on and hands-off tasks are not shaded or shaded with gray hatch marks. When these multiple runs are performed, please note that the operator can expedite Steps 1 and 2 and prepare these clinical specimens well in advance to frontload their workday with a subsequent delay until Step 2 can be completed (loading the deck with consumables when the Viper robotic arm is at rest and the door can be opened). Note that for the samples comprising the four runs, the nonshaded tasks do not overlap and Step 2 contains overlap between the runs but this comprises a hands-off heat incubation and cooling step. It is notable that with this workflow, at least three of four of the workdays is heavily automated allowing the operator to complete other tasks in the laboratory.
Preanalytical Activity
Low complexity preanalytical steps included scanning the bar code on the tube and racking specimen tubes in a special instrument rack that detects and registers the location of each tube and prevents inadvertent specimen lifting during the sample piercing process. The BD Viper System with XTR accepts urine and swab specimens. The swab specimens were preincubated at 114°C for 15 min to promote dissolution of muciod content which if present, can interfere with subsequent pipetting operations. Our simulation was based on the analysis of swab specimens being present in each run. The racked specimens were then cooled down for a 15-min period either directly on the system (when the first or only run was being performed) or off-deck if the BD Viper System with XTR was already processing another batch of clinical specimens. The system was prepared by loading DNA extraction reagents troughs, eight racks of 96 disposable tips equipped with barrier filters, extraction tubes, and four microplates containing dried down strand displacement amplification (SDA) reagents. The instrument then performed all of the remaining tasks consisting of an automated deck system layout check followed by sample aspiration, DNA extraction, reaction setup, and real-time amplification and analysis.
DNA extraction was fully automated using magnetic ferric oxide (FOX) particles and height-adjustable magnets. Extraction consisted of the stepwise addition of lysis buffer, binding acid, wash buffer, and elution buffer. The DNA-bound magnetic particles were captured and released during the washing and elution steps. Neutralization buffer was added to each eluted sample to adjust the pH and ionic strength to ensure compatibility with the downstream SDA reactions and the DNA eluates were automatically transferred to the priming microwells occupying the analytical station for subsequent target amplification. The quality of DNA obtained by this method is sufficient to support molecular amplification methods using either SDA or PCR where analytical detection limits below 100 copies/mL are required for robust clinical sensitivity for a variety of human clinical specimen types (data not shown).
The BD Viper System with XTR features a safety shield that is used to protect the operator from the moving robotic arm. It may be opened after the amplification samples have been transferred to the fluorescent readers thereby allowing spent consumables from the current run to be removed from the deck followed by immediate loading of consumables for the planned next run. If the operator decides to equip the deck for a next run, the real-time amplification reactions in process are uninterrupted.
DNA amplification and detection were performed simultaneously using SDA technology 19,20 incubated in a fluorescent reader integrated onto the instrument deck to accomplish real-time monitoring of the amplification reactions. Only two prepackaged plates and a transfer step were required to complete reaction setup for SDA. Use of these dried down single unit of use amplification microwells increased the efficiency of reaction setup and eliminated any wasted reagent costs associated with amplification master mixes and bulk reagents that are used by other automated systems. Adhesive plate seals were automatically secured on each amplification plate providing a tight barrier to prevent evaporation of reagents during elevated temperature incubation and to prevent the liberation of contaminating amplified nucleic acids into the environment.
The SDA technology is an isothermal reaction that is ideally suited for automation because a simple single temperature incubator is required. Furthermore, accumulation of reaction products can be monitored in real time resulting in rapid quantification of results.
Postanalytical Activity
At the end of the automated run, the results were verified by a trained technologist and the tightly sealed amplification microwells (96-well format in 8-well strips) as well as any remaining spent consumables were manually removed and discarded. At the end of the shift, the operator cleaned the BD Viper System with XTR with the recommended cleaning solution to prepare the instrument for a subsequent shift.
BD Viper System with XTR Productivity Simulation Over an 8-hour Shift
The BD Viper System with XTR can be run in a multitasking mode to maximize its productivity (Fig. 1). When the amplification reactions were automatically placed in the fluorescent reader, the safety shield was automatically unlocked so that the deck could be configured for another subsequent run. It is also notable that during the FOX extraction step, the operator can prepare another batch of clinical specimens for the next run by gathering and configuring the next batch of sample tubes to be loaded onto the sample rack. Given this multitasking requirement to leverage maximum clinical sample throughput within a shift, we increased the index of complexity and intensity for managing such multiple runs in concert.
Simulating Maximum Productivity for One FTE Running Multiple BD Viper System with XTR
We simulated the number of BD Viper System with XTR that can be run in one day by a single full time employee (FTE). Felder—Foster plots (process depiction and complexity burden) were prepared to determine the optimal number of instruments and operator availability for performing the various required duties.
BD Viper System with XTR Reliability
The overall system mean runs between failures (MRBF) metric was assessed as hardware or software failures that required replacement and/or repair by a BD field service representative (Table 1). In addition, mean runs between interventions (MRBI) were assessed as instrument failures that may have required the operator to restart a run but did not require a field service visit. Similar data were also examined for MRBF and MRBI for individual system components. The data for these evaluations were obtained from the manufacturer who obtained supplier failure rate specifications, historical data from system testing, empirical accelerated life testing, and failure modes and effects analyses. Data were compiled over a total of 741 analytical runs on nine BD Viper Systems with XTR over a 12-month period.
Results
Process Evaluation
Using our Felder–Foster plots, we summarized the results of the BD Viper System with XTR (Fig. 1). The time to the first 184 CT and GC results (from 92 clinical specimens) was 196 min (3 h, 16 min). If the technologist was required to verify the results before release, then the elapsed time of the entire process would be 206 min (3 h and 26 min) allowing an additional 10 min to survey the results. Using one instrument, our measured data from one run allowed us to predict using simulation that a laboratory can complete four full analytical runs (736 results for CT and GC on 368 clinical specimens) by 4:30 PM after starting at 8:00 AM (Fig. 2). This model allows for over an hour (1 h and 15 min) for system maintenance; however, the actual requirement is 17 min to thoroughly clean the instrument according to our videotaped observations. The manual labor utilization for this system was only 35 min of direct interaction with the BD Viper System with XTR for each run, or a total of 140 (2 h 20 min) (27.5% of the workday) labor requirement for four runs. The incorporation of automated extraction on the BD Viper System with XTR significantly reduces the amount of operator manual time that may be calculated to be 22.8 s per specimen or 11.4 s per CT or GC result.
We simulated the maximum capacity for a single technologist to operate the BD Viper System with XTR and calculated that 32 analytical runs could be completed in an 8.5-h work shift using eight instruments that would generate 5888 results (processing 2944 specimens). This level of capacity would be challenging until a technologist had gained sufficient experience in operating the system and kept a regimented time schedule moving between the eight instruments. Given this consideration, we estimate that one technologist could more reasonably run six BD Viper Systems with XTR during an 8.5-h shift that would generate 4416 CT and GC results from 2208 clinical specimens after initial training.
The BD Viper System with XTR demonstrated an estimated MRBF of 1918 runs that translates to 352,912 reported CT and GC patient results from 176,456 clinical specimens (Table 1). The table also contains individual subsystem component performance estimated from data supplied from the manufacturers in-house and field generated testing data. The MRBI was estimated to be 1000 runs corresponding to approximately 184,000 specimen results. From these data, one can estimate that for an average of four runs per day, the BD Viper System with XTR will operate for 250 days (about 8 months and 6 days) without a failed run for system-related issues.
Discussion
The BD Viper System with XTR consists of a single integrated analytical platform employing a variety of technological innovations to enable efficient DNA extraction, amplification, and analysis of GC and CT from urine and swab clinical specimens that minimizes operator time and technical skill necessary to complete complex molecular testing. Analytical performance of this system was comparable with other systems on the market. 9 Previous studies have demonstrated relatively good sensitivity for GC and CT detection over a broad range of specimen transport conditions. 17 Furthermore, overall sensitivity was 98% for CT and 98.6% for GC in male urine, urethral swabs, and urine transport preservative, 18 and greater than 95% in random urine specimens. 9
Various technological features were engineered into the BD Viper System with XTR to facilitate specimen collection, to prevent the potential for specimen cross contamination, and to eliminate errors from unskilled operators. Once specimens are placed in the specimen collection and transport device they are not opened again by human operators. The robotic system can access the contents by using barrier tips (pipette tips incorporating filter material) to pierce a unique dual-layer foil cap that provides an airlock preventing pressurized specimens from forming an aerosol that could lead to system and testing laboratory contamination. A locking specimen rack prevents specimen lifting during the piercing process and maintains a fixed array. Adhesive seals are used on top of the amplification microplates to prevent amplified nucleic acid contamination. Finally, a highly reliable, high-precision industrial grade select compliance articulating robot arm (SCARA) robotic arm was used for automated pre-programmed movements to assure long-term system reliability.
Classic process evaluation techniques have been used in clinical laboratories when determining return on investment for analytical systems. 21,22 However, little work has been published on quantifying molecular diagnostics automation systems from a labor efficiency perspective. Therefore, we used time-and-motion studies and objective review of task intensity and complexity to develop new measures of automation efficiency. These data were then used to map an optimal process flow using the example of the BD Viper System with XTR. Task complexity peaks were observed at the initiation of an analytical run. After system initiation, the need for highly trained laboratory professionals was minimized. Thus, for laboratories performing a limited number of runs, a skilled technologist would only be required for brief predictable periods thereby allowing for multitasking with other instrumented platforms.
Our time-and-motion, skill level, and task intensity Felder–Foster plots revealed that one full time technologist operating one BD Viper System with XTR run manually works only 6.9% of the workday (35 min, which may be calculated to be 22.8 s per specimen or 11.4 s per result) We calculated that one technologist could complete 32 runs generating 5888 results (2944 specimens for both GC and TC analysis in each tube) using eight Viper XTR instruments. However, the repetitive nature of handling 5888 tubes in an 8-h shift would require advanced skills to establish a much regimented predictable schedule needed to attend several instruments. Our analysis also indicates that the skill level demands on the operator changed during a run. We found that the most skill demanding tasks were associated with instrument setup, specimen accessioning, and result validation. Subsequent steps could be performed using less skilled labor.
In addition to optimal use of appropriately skilled operators, our studies revealed opportunities for the multitasking of various processes to optimize system throughput. We found that labor could be used optimally if operators placed consumables on the instrument during the time that the clinical specimens were preincubating at 114°C to eliminate mucoid content. This efficient use of labor concentrated all the moderately skilled tasks into one 15-min up-front period. The Felder–Foster plot could serve as a guide regarding staffing appropriately skilled individuals at selected times of the day to maximize the use of available human resources, particularly when labor utilization is performed simultaneously or in an asynchronous fashion. Simulation studies revealed that as instruments are increased from one to four, then one can still use one operator optimally for preparing specimens and setting up the instrument if the runs are performed asynchronously (shown in Fig. 2), but the skill level of this operator would likely have to be higher because of the complexity of moving between instruments and coordinating the runs.
The BD Viper System with XTR has a high throughput (92 specimens [184 total GC and CT results]) for each run of 3 h and 12 min. The total capacity per instrument is 368 clinical specimens (736 total GC and CT results) per 8.5-h shift as compared with the Gen-Probe Tigris DTS performing GC and CT analysis that provides 492 results per similar shift (246 specimens + 4 controls per day), the Abbott m2000 system provides 270 CT and GC results per 8.9-hr shift (135 specimens + 9 controls per day) and involves a manual transfer between the sample processing and target amplification modules of the system. The COBAS TaqMan 96 automation solution processes 72 samples (up to four different tests) for 288 specimens per day, and 576 GC and CT results per 8-h shift). 9
System reliability plays an important role in maintaining system throughput. Additional labor savings can result from using robust automation technology to eliminate repairs and reduce preventative maintenance visits. The BD Viper System with XTR uses robust engineered components to reduce system downtime. For example, the SCARA was selected for high precision movements (repeatability = ±0.10 mm), high speed, and a minimum of failures. The unique approach to delegate most of the liquid and consumable handling steps to a programmable robotic arm as opposed to moving tubes and plates across the instrument deck provides a high degree of system reliability, while maintaining flexibility for system expansion. The robot is equipped with a pipette head that uses a positive displacement syringe pumping mechanism for each channel of the pipette so that no tubing or vacuum manifolds are used to transmit aspiration or dispensing forces. The BD Viper System with XTR demonstrated robust MRBF and MRBI metrics. Data on these parameters were not readily available for other similar automated molecular diagnostic systems on the market.
We conclude that the high degree of reliability demonstrated by the BD Viper System with XTR is ideally suited for core clinical laboratories running molecular assays such as STDs where large numbers of clinical specimens are batched and rapid sample throughput is required with consistent results.
Footnotes
Acknowledgments
We thank the engineering team at BD for their helpful input into the data-gathering phase of this research and Lawrence Mertz for his review of the manuscript. Robin A. Felder and Maria L. Foster were provided a consulting fee to cover their time conducting these studies. Mike J. Lizzi, Brent R. Pohl, Dustin M. Diemert, and Bryan G. Towns are employees of BD Diagnostic Systems.
