Abstract
Serum specimen pipetting is a labor-intensive process that can be a source of laboratory error, ergonomic stress, and a potential source of accidental exposure to infectious pathogens. Automated solutions for sample preparation reduce time spent on sample handling, minimize exposure to infectious pathogens, reduce laboratory error, reduce ergonomic stress, and save money. A fully automated method was developed for a custom-designed competitive Luminex immunoassay (cLIA) on a TECAN Genesis Workstation that measures antibodies to human papillomavirus (HPV) virus-like particles (VLPs). The automated program generated statistically similar data to assays performed manually. Overall, HPV 6, 11, 16, and 18 antibody titers obtained from samples prepared with the automated program were 3.2%, 6.6%, 3.6%, and 2.0% higher, respectively, than antibody titers from samples prepared manually. The agreement rates between methods for HPV positive and negative samples for HPV 6, 11, 16, and 18 were 100%, 96.8%, 98.4%, and 97.3%, respectively. An overview of the basic workflow of the automated HPV cLIA used to support our HPV vaccine Phase III clinical trials is described.
Introduction
Sample preparation comprises the most time-consuming and ergonomically stressful step in performing serological assays. 1 To test 192 samples, an individual spends from 30 min to over an hour opening individual sample vials, aliquotting, diluting, and transferring those samples from dilution tubes to microtiter plates. This process is not only time-consuming and tedious for the individual, but can also be a source of laboratory error. Numerous small repetitive motions also put the individual at risk for cumulative trauma disorders (CTDs). 2 Developing automated solutions for sample preparation reduces time spent on sample handling, minimizes exposure to infectious pathogens, decreases laboratory error, reduces ergonomic stress, and saves money.
The TECAN Genesis Workstation was chosen as an automation platform to develop a fully automated method to perform both pre-analytical serum sample preparation and reagent addition for a custom immunoassay to measure antibody responses to HPV L1 virus-like particles (VLPs). 3 This assay is being used to support epidemiological studies and vaccine clinical trials for the evaluation of HPV vaccines. 4 –7 The assay to be automated was a multiplexed HPV competitive Luminex immunoassay (cLIA) used to quantify antibodies to neutralizing epitopes on HPV types 6, 11, 16, and 18 VLPs. The HPV cLIA is a proprietary assay developed at Merck & Co., Inc (Wayne, PA). The basic principles of the assay were first described by Opalka et al., 3 while the optimization and validation of the assay have recently been described by Dias et al. 8 The assay measures HPV type-specific antibodies to known neutralizing epitopes on L1 VLPs in a competitive format, where known type-specific, neutralizing monoclonal antibodies compete with patient serum antibodies for binding to the neutralizing epitopes on the VLPs. 3 These four HPV VLPs are covalently conjugated to four separate Luminex micro-spheres that have distinct spectral addresses, which allows one to simultaneously measure antibodies to the four different VLPs.
In developing the Gemini script to automate the HPV cLIA, the TECAN had to perform the following: (i) transfer serum samples from 8-mL vials to a 96-well microtiter plate in duplicate; (ii) generate a duplicate, 12-point standard curve; iii) add high, low, negative, and antibody depleted human serum (ADHS) (Valley Biomedical, Winchester, VA) controls in duplicate; (iv) add fluorescent detection monoclonal antibodies (MAbs); and (v) add VLP-microspheres to all wells of the plate. This high-throughput serology assay has proven extremely valuable in performing epidemiology studies and evaluating the efficacy of prophylactic HPV vaccines.
Materials and methods
Instrumentation
The TECAN Genesis RWS200 (TECAN Inc., Research Triangle Park, NC) is a computer-controlled robotic system designed for the automation of sample preparation and analytical procedures. The system is equipped with a liquid handling arm, a deck, a personal computer that controls the instrument, a robotic movement arm, a plate reader, and a TECAN MultiChannel Pipetting Option (TeMO). A variety of carriers are used for the placement of microtiter plates, reagent troughs, sample tubes, and disposable tips on the TECAN deck. The instrument is controlled by a vendor-supplied proprietary software application, Gemini (TECAN, Schweiz ACT, Switzerland), which is built around a Microsoft Windows NT-based (Microsoft, Redmond, WA) graphical interface with pull-down menus and simple user prompts.
Function of Key System Components
A picture of the TECAN Genesis System and its components is shown in Figure 1, and a schematic diagram of the deck is shown in Figure 2. The liquid handling arm (Fig. 1, item 1) has eight tip adapters linked to 1.0-mL syringes that are designed to fit disposable tips and is driven by fluidics. We exclusively used disposable 1000-, 200-, and 50-μL sterile filter tips from Molecular Bioproducts (San Diego, CA) to eliminate the possibility of carryover and cross-contamination of patient serum samples. 9 All disposable tips were discarded into the waste chute after use (Fig. 1, item 2), and all air gaps were restored in the liquid handling arm by flushing water through the tubing into the wash station (Fig. 1, item 3). The 1000- and 50-μL tips were stored on the deck of the TECAN (Fig. 1, items 4 and 5, respectively). One rack of 200-μL tips was stored on the TECAN deck (Fig. 1, item 6), while another two were stored within a hotel (Fig. 1, item 7). These tips were used to transfer fluid volumes between reagent troughs (Fig. 1, items 8-10), tubes (Fig. 1, items 11 and 12), a deepwell plate (Fig. 1, item 13), or microtiter plates (Fig. 1, items 14 and 15).

The TECAN Genesis System and its Components. (1) The liquid handling arm controls the liquid handling functions of the instrument using eight tips; (2) disposable tip waste slide; (3) liquid handling arm wash station; (4) 1000-μL disposable tips; (5) 50-μL disposable tips (five racks for six total plates); (6) 200-μL disposable tips; (7) 200-μL disposable tip storage; (8) 100-mL trough for ADHS diluent; (9) 25-mL trough for detection monoclonal antibody; (10) 25-mL trough for VLP microspheres; (11) 2-mL tube rack for standard and control tubes; (12) the 12 movable strip racks that can hold up to 192 sample vials; (13) deepwell used for standard curve dilution; (14) microtiter plate for detection antibody; (15) microtiter plate for VLP microspheres; (16) robotic movement arm; (17) TeMO; (18) four racks of 100-μL TeMO disposable tips; (19) disposable tip apron; (20) Microtiter Hotel.
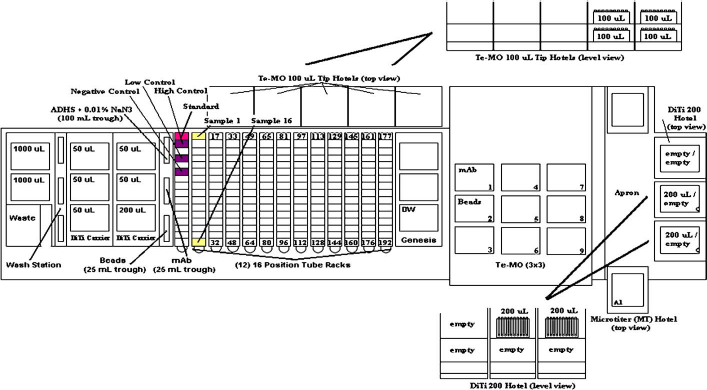
Top view of TECAN deck for the HPV cLIA. A schematic diagram detailing the placement of reagents and labware on the TECAN deck for the HPV cLIA.
The robotic movement arm (Fig. 1, item 16) is used to relocate microtiter plates as well as to replace disposable tips from one defined deck position to another. 9 The ability to replace disposable tips during a run allows the user to walk away from the system while the assay was being performed.
The 3 × 3 TECAN multichannel pipetting option (TeMO; Fig. 1, item 17) is a piston-driven 96-channel pipetter with nine positions (three slides with three positions each). We exclusively used 100-mL disposable tips (Phenix Research Products, Candler, NC) to eliminate the possibility of cross-contamination between assay plates. The 100-μL tips were stored in the rear of the deck of the TECAN within a modified deepwell hotel (Fig. 1, item 18). These tips were used for the addition of detection antibody and VLP-microspheres to the plates.
The disposable tip apron (Fig. 1, item 19) is a hotel deck extension to the right of the TECAN deck (robotic movement arm access only) that allows for the storage of more plates or tips. We used these hotels exclusively for storing more liquid handling arm disposable tips. Assay plates were stored in a hotel on the disposable tip apron (Fig. 1, item 20).
The Sunrise Reader (TECAN, Schweiz ACT, Switzerland) is an absorbance reader used in conjunction with CheckBox, a performance monitoring software application provided by TECAN (TECAN, Schweiz ACT, Switzerland). The software package offers predefined validated test procedures based on photometric methods, in addition to automated result evaluation and documentation. This software supports the automation of performance control and performance history of TECAN instruments and devices and is used to examine the precision of the Genesis liquid handling arm. The Gemini Software (Version 4.2) is used to control the TECAN Genesis Workstation.
Since the TECAN is a fluidics-based system, deionized, degassed water was used as the system liquid. Degassing the system liquid helps prevent bubbles within the tubing and is crucial in the liquid handling of the instrument.
The TECAN System is housed in a BioPROtect II, Class II A/B3 biological safety cabinet (Baker, Sanford, ME). The BioPROtect II is an enlarged clean-air and biocontainment enclosure built specifically to house high-volume, robotic liquid handling systems such as the TECAN Genesis. Encasing the TECAN within the BioPROtect II provides protection for the samples and personnel.
HPV cLIA Overview
A detailed understanding of the operating process of the manual assay was central to developing an automated program. The layout of the plate, specific volumes of serum, detection antibodies, and VLP-microspheres were a starting point for programming the TECAN. In the manual assay, a duplicate 12-point standard curve was added vertically into the first three columns of the plate in a final volume of 50 μL per well. Added to the remainder of the plate, columns 5 through 12, was 25 μl of ADHS. Both sera to be tested and the controls were diluted 1:2 in ADHS. Once the standard, serum samples, and high, low, negative, and ADHS controls were added to the plate, 25 μl of detection antibodies was added to all wells of the plate. Following the addition of the detection MAbs, 25 μL of VLP-microspheres was added for a final 1:4 dilution of the sera. Upon addition of the VLP- microspheres, the assay plate was covered with a foil seal and the binding reaction was allowed to reach equilibrium with an overnight incubation. Since this is a competitive assay, the monoclonal antibodies and serum were added first and the VLP-microspheres were added last so that there were no sample addition timing effects. Sample addition timing effects were observed when we tried to increase the sensitivity of the assay by adding the serum to the VLP-microspheres first, followed by the addition of the monoclonal antibodies. After the overnight incubation, the plate was filter-washed three times prior to being analyzed on a Bio-Plex instrument (Biorad, Hercules, CA). Additionally, stability studies have shown that the working concentration of the detection MAbs are stable for at least 3 months at 4 degrees.
Automation of the HPV cLIA
The automated method for the HPV cLIA was designed with the end user in mind. Since the deck was fully populated with plates, samples, and reagents, the potential for the operator to forget labware locations was a risk factor that was taken into consideration when designing the program. To eliminate the possibility of manually populating the deck incorrectly, a series of condition-controlled user prompts that queried the user at the beginning of the run were programmed within the HPV cLIA Gemini script. When running the program, the user was first asked how many samples were to be tested with a maximum allowable entry of 192 samples (6 plates). Based on the number of samples that were entered, variables and conditions within the Gemini script determined how much of the reagents were to be used during the process. For example, in an effort to conserve reagents, entering the number of samples to be tested controlled how many wells received the ADHS sample diluent, dictated the volume of stock quadriplex reference standard used to generate the duplicate standard curve, and determined the number of plates needed for the run. After entering in the number of samples, a series of prompts instructed the user to place the appropriate number of microtiter plates in specific locations.
Once the series of user prompts were completed, the program began with the dilution of the 12-point duplicate standard curve in a 2-mL 96-deepwell block. The liquid handling arm aspirated a specific volume of ADHS (based on the number of samples being run) and added it to the first three columns of the deepwell. After the ADHS had been added, the specified volume of the quadriplex reference standard was added to wells A1 and B1 and then mixed. An eleven, two-fold serial dilution of the reference standard was made in duplicate in C1, D1 through G3, H3. In order to eliminate carryover while generating the standard curve, new tips were used for each aspirate and dispense step. Next, ADHS was added to the sample wells (columns 5 through 12 on the plate); 50 μl of the standard curve from columns 1 through 3 of the deepwell plate were then transferred to the corresponding columns of the assay plate. After the addition of the standard curve, the controls were added to the assay plate in column 4 by adding 25 μl of ADHS, followed by 25 μl of controls. Lastly, the serum samples (25 μl) were added to the plate in duplicate. Running the samples in duplicate, rather than in replicate, was advantageous because there would be an obvious difference between the duplicate measurements if a sample was not added to the plate because the tip was clogged or was diluted incorrectly. Additionally, all serum samples were added to the plate such that duplicates ran down a column (i.e., A5, B5, etc.) on the plate rather than across the row (i.e., A5, A6, etc.; Fig. 3), since the Bio-Plex reads down a column from top to bottom, then left to right across a row.
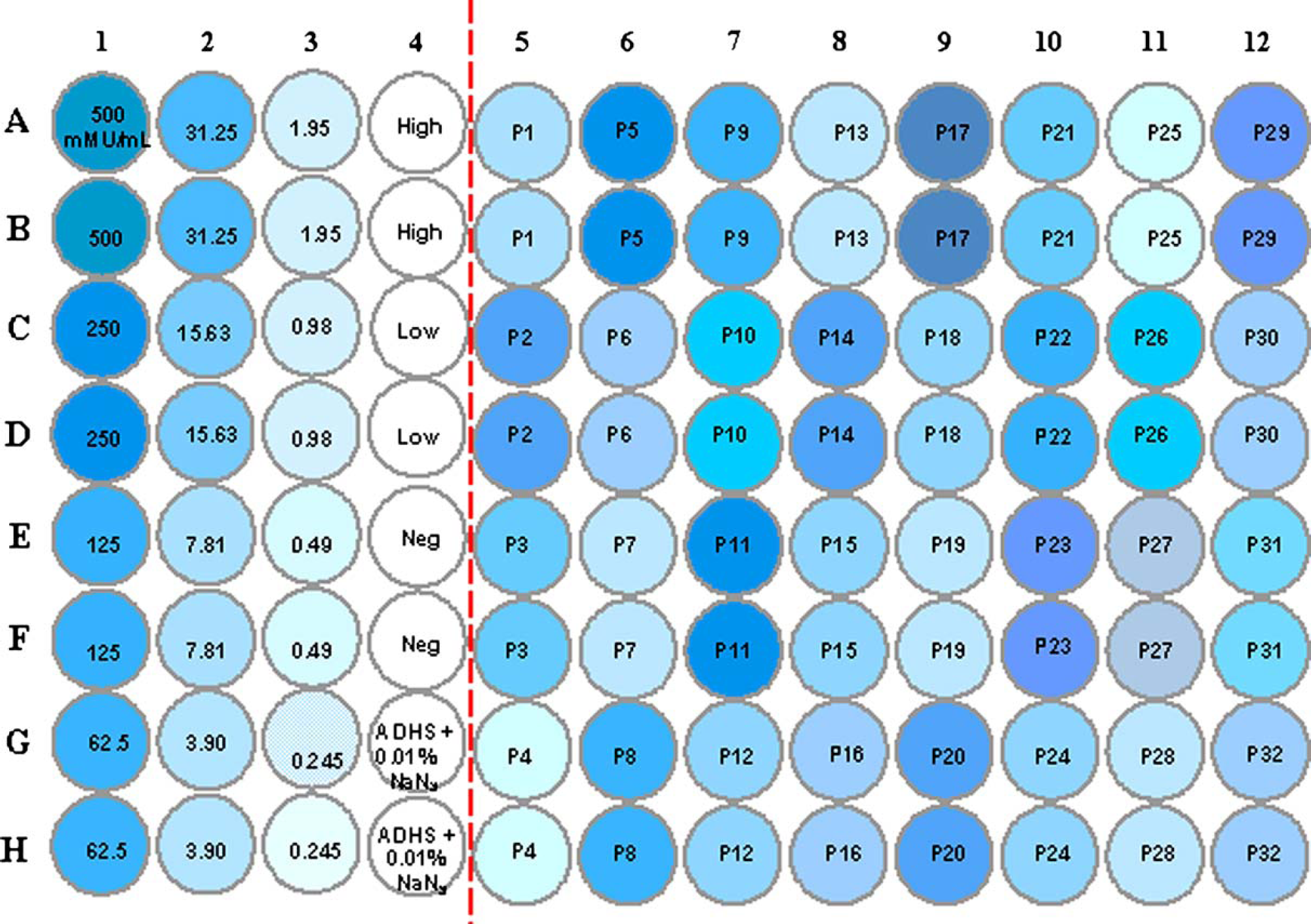
HPV competitive luminex immunoassay (cLIA) plate layout. A 12-point standard curve is plated in duplicate in wells A1–H3, duplicate high and low controls are placed in A4, B4, C4, and D4, respectively. A duplicate negative control is placed in E4 and F4 and straight ADHS + 0.01% NaN3 is placed in G4 and H4. Thirty-two patient samples are tested, in duplicate, in wells A5–H12. Volumes per sample well for the 1:4 dilution were as follows: ADHS + 0.01% NaN3 = 25 μL, heat inactivated serum sample = 25 μL, detection mAb cocktail = 25 μL (H6.M48, K11.B2, H16.V5 and H18.J4), and VLP microspheres = 25 μL. Total volume per well = 100 μL.
Once standard, controls, and diluted serum samples were added to the assay plates, the program paused, prompting the user to add the detection MAbs and VLP-microspheres to 25-mL troughs on the TECAN deck. Both reagents are light-sensitive, so these reagents were not added to the deck at the beginning of the program, and the lights of the biosafety cabinet were turned off. In addition, settling of the VLP-microspheres was avoided by adding this reagent to the deck at this time point. The detection MAb reagent solution contained 1% Triton X-100, and care was taken to avoid having bubbles in the trough. After these reagents were added to the deck, the liquid handling arm pipetted the detection MAbs and VLP-microspheres into separate assay plates located on the TeMO. Once this step was completed, the assay plates were transferred to the TeMO for the addition of 25 μL per well of detection MAbs, followed by the addition of 25 μL per well of VLP-microspheres. Upon completion of the program, the operator was prompted a final time to recover any remaining detection MAbs and VLP-microspheres within the reagent plates. To recover any remaining detection MAbs within these plates, the liquid handling arm aspirated 200 μL from the first four columns of the plates and dispensed the volume recovered within the tips into the appropriate reagent trough. This was repeated for the remaining columns of the microtiter plate. The VLP-microspheres were recovered in the same fashion. After the liquid handling arm recovered both the detection MAbs and VLP-microspheres, the operator removed the remaining detection MAbs and VLP-microspheres in the trough, pouring the reagents back into separate amber bottles upon completion of the program to be used for subsequent assay runs.
The total time the detection MAbs and VLP-microsphere were on the deck was less than 15 min. The assay plates from the TECAN were removed, covered with a foil seal, and placed in a Thermomixer (Brinkmann, Westbury, NY) for an overnight incubation of 15-25 h at a setting of 600-800 rpms (set point) with the temperature off. The overnight incubation allowed the reaction between the VLP-micro-spheres and antibodies to reach equilibrium, ensuring that there would not be a timing effect that would impact the assay.
After the overnight incubation, the contents of the assay plate were transferred to a 96-well, 1.2-mm filter plate (Millipore, Billerica, MA), using a stand-alone TeMO prior to being analyzed on the Bio-Plex system. A 96-well 2-mL deepwell plate was filled with 925 μL of wash buffer (phosphate buffer saline, 0.10% Triton X-100, 0.05% NaN3) and placed on the TeMO (Fig. 1, item 17). The wash buffer was aspirated from a deepwell rather than a trough to avoid any cross-contamination. During the filter-wash step, the program paused and instructed the operator to manually vacuum the filter plate. Once the filter plate was placed back in the appropriate position on the TeMO, the program resumed after the operator clicked “OK”. The plate was filter-washed three times with a wash volume of 175 μL per wash, and added a final volume of 125 μL to the filter plate to reconstitute the VLP-microspheres. All filter plates were placed in separate Thermomixers for approximately 5 min, at a setting of 900 rpm with the temperature off, before being analyzed on a Bio-Plex. Lids were not placed on the filter plates in the Thermomixer because this caused the wells of the filter plate to leak.
Results
After developing and beta testing the automated HPV cLIA, we first sought to establish whether antibody titers determined manually were equivalent to antibody titers generated from the assay performed on the TECAN. The manual versus TECAN HPV cLIA titers for each HPV type are shown in Figure 4. The solid line represents perfect concordance, which happens when both methods produce the same antibody titers. The dashed lines show the actual concordance results from the manual versus TECAN evaluation. When concordance is high (concordance slope = 1, % difference = 0%), the two lines (dashed and solid) in each of the graphs in Figure 4 overlap. As shown in Figure 4, the two methods produced nearly equivalent antibody titers. In addition to evaluating the concordance between the two methods, we also determined the agreement rate between the two methods with respect to the number of samples that fall above or below the serostatus cutoff. The serostatus cutoff is the titer value that determines a positive score in an assay when an individual has antibodies to an HPV type. For HPV 6, 11, 16, and 18, the serostatus cutoffs are 20, 16, 20, and 24 mMU/mL, respectively. The concordance slope, average percent difference, and percent agreement for serostatus are shown in Table 1. To determine the overall concordance of the two methods, a concordance analysis was performed using the method described by Tan et al. 11 The overall concordance slope was estimated to be 0.99, 1.00, 0.97, and 0.96, meaning that on average, for every 2-fold increase in titer obtained from samples diluted manually, there was a 2.0-, 2.0-, 2.0-, and 1.9-fold increase in titer for samples diluted with the TECAN for types 6, 11, 16, and 18, respectively. Titers obtained from samples diluted with the TECAN were, on average, 3.2% higher [90% CI = (+1.8%, +4.5%)], 6.6% higher [90% CI = (+2.8%, +10.6%)], 3.6% higher [90% CI = (-3.0%, +10.6%)], and 2.0% higher [90% CI = (-1.8%, +5.8%)] than those obtained from samples diluted manually for types 6, 11, 16, and 18, respectively. The serostatus agreement rate between the two methods was 100%, 96.8%, 98.4%, and 97.3% for HPV 6, 11, 16 and 18, respectively. These results demonstrate that processing samples on the TECAN produce antibody titers that are equivalent to those obtained when processing samples manually.
Performance characteristics of the HPV cLIA performed on the TECAN versus manually

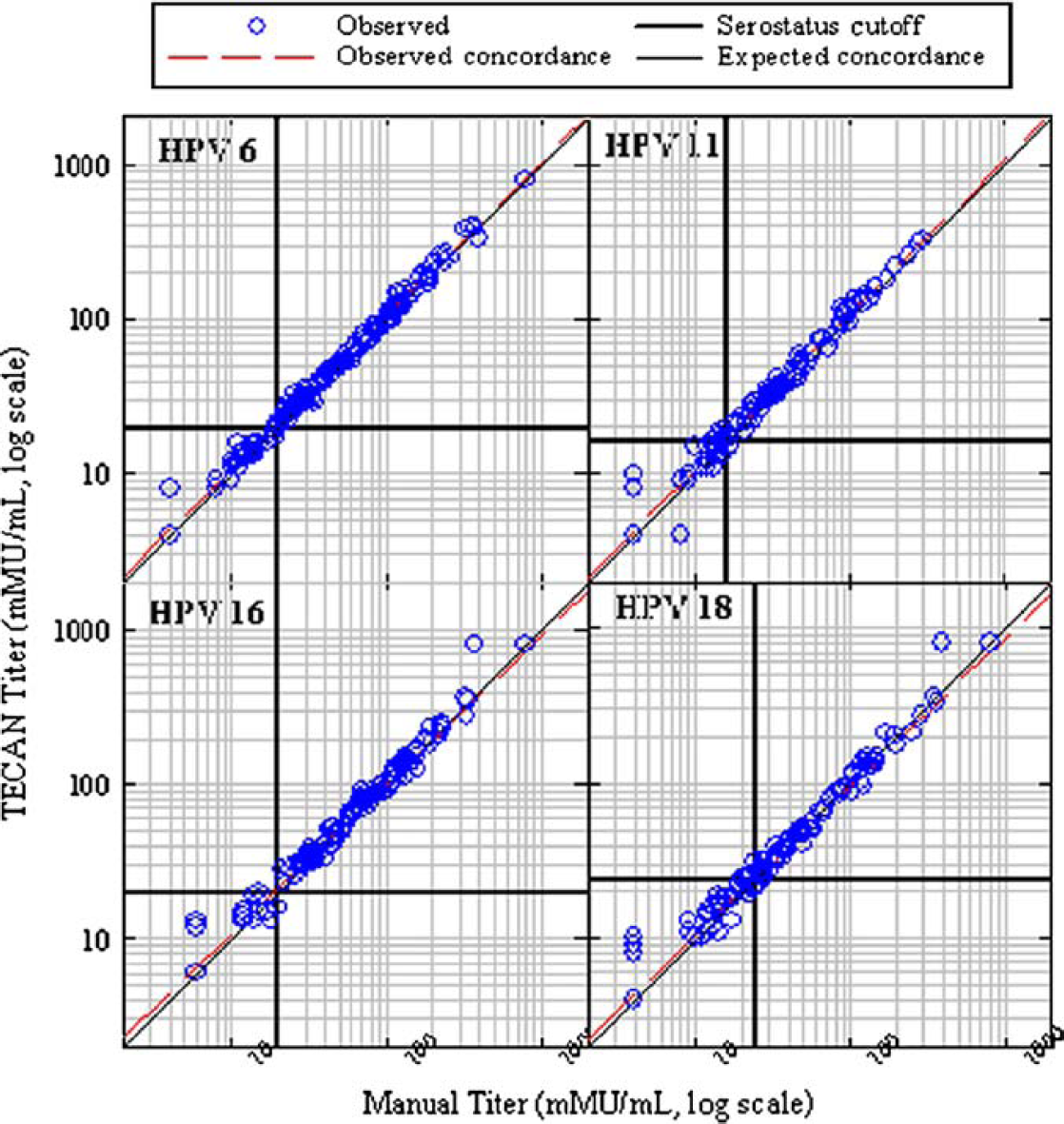
Concordance of antibody titers obtained from samples diluted at 1:4 with the TECAN Genesis Workstation to those produced manually for HPV 6, 11, 16, and 18 using the HPV cLIA. The lower limit of quantitation for HPV 6, 11, 16, and 18 was 8, 8, 12, and 8 mMU/mL, respectively. For graphical purposes, values less than the LLOQ were assigned values of 4 for HPV 6, 11, and 18, and 6 for HPV 16. The upper limit of quantitation for the four HPV types was 400 mMU/mL. Values greater than 400 mMU/mL were assigned values of 800 mMU/mL for graphical purposes.
The timing of the various steps in the HPV cLIA, manually or on the TECAN, are listed in Table 2. Diluting the standard curve and the addition of the standard curve, ADHS, and controls to one plate took more time on the TECAN, while additional samples, detection MAbs, and VLP-microspheres took less time when performed on the TECAN. The total time to complete one plate manually was 15 min, 49 s, while the total time for the TECAN to complete one plate was 13 min, 24 s. When comparing the total time to prepare 6 plates (the maximum number of plates that can be processed in one TECAN run) manually versus on the TECAN, it took 1 h, 40 min, 2 s manually, whereas it took 1 h, 27 min, 49 s on the TECAN. For the second day of the assay in which the contents of the assay plate were transferred to a filter plate and washed, it took 12 min, 35 s manually, whereas the TECAN took 4 min, 33 s. These results demonstrate that performing this assay on the TECAN took less time than performing the assay manually.
Timing comparison of various steps of the HPV cLIA performed manually and on the TECAN

Total time for TECAN to complete 6 plates is 1h 34min 15s when all robotic movements are accounted for.
Discussion
We describe here the workflow of a TECAN Genesis Workstation robotics program used to test up to 192 samples (6 assay plates) in the HPV cLIA. The development and implementation of this automated method for sample preparation was not an easy task and was met with many challenges. The first challenge to automating the HPV cLIA was logistics. Even with a two meter TECAN, the deck space quickly dwindled once the appropriate number of disposable tips and plate carriers were added to the deck. Because this assay is used to determine previous HPV exposure, a decision was made to use disposable tips with filters to reduce the risk of sample contamination or crossover. The number of disposable tips needed to complete various portions of the program (i.e., dilution of the standard curve, addition of controls, and addition of samples) dictated how much space remained on the deck for other TECAN racks (such as tube racks).
The second challenge was liquid handling. Many factors come into play when aspirating and dispensing fluid. These include the viscosity, volume, and conductivity of the samples and reagents, aspirate and dispense speeds, and the air gaps that separate the sample from the system fluid. Air gaps separating different liquids in the pipetting tips are called separation air gaps, while air gaps preventing droplets at the end of the tips, while the liquid handling arm moves over the worktable, are called trailing air gaps. Air gaps, viscosity, and other factors ultimately can affect the accuracy and precision of liquid handling. The TECAN Gemini software application incorporates a unique tool, a liquid class database, that helps to manage these factors. The TECAN default serum liquid class was used for the HPV cLIA. The only change made to the default serum liquid class was that the clot detection feature was enabled. Similar to using the default serum liquid class, the default water liquid class was used as the liquid class for the detection MAbs and VLP-microsphere liquid class. These predefined liquid classes not only saved us time when developing this application, but it has also allowed us to apply these liquid classes to other serological assays.
Another feature of the TECAN is liquid detection, which enables the instrument to aspirate and dispense liquid with minimal carryover of liquid on the outer surface of the tips. Liquid detection works by measuring the capacitance between the tips and the bed of the machine. The values obtained are dependent on the conductivity of the media that the tip is traveling through (i.e., air, gas, or sample). Thus, when the tips of the liquid handling arm travel down to the surface of the liquid, the software polls the instrument for a change in capacitance signal. This capacitance change dictates the height at which the instrument will start to aspirate (or dispense) the liquid. Just as the instrument looks for a change in capacitance when the tips enter the liquid, the software polls the instrument for a change in capacitance as the tips exit the liquid. If a capacitance change is not detected within a defined distance of leaving the liquid, a clot detection error message occurs, indicating there is a clot hanging from the tips and bridging what should be an air gap. Enabling this clot detection feature prevents pipetting errors due to very viscous samples, or samples with clots. For the HPV cLIA, the clot detection feature was enabled since serum, not plasma, samples were tested. If the clot detection error occurred while running the HPV cLIA on the TECAN, the operator manually removed any solid material hanging from the tips and allowed the instrument to retry the aspiration step. Enabling the clot detection feature significantly helped decrease the number of sample retests.
Another challenge we faced was bubbles on the surface of the liquid in the reagent troughs. If the disposable tip did not break bubbles on the first detection, it was possible that the TECAN would start aspirating the liquid at the height of the bubble. This would cause air to be aspirated instead of the liquid, affecting both the accuracy and precision of the results. To avoid this potential error, each operator visually inspected the reagents in the troughs to ensure there were no bubbles.
Overall, the time associated with processing six plates (192 samples) was slightly faster on the TECAN (1:27:49 vs. 1:40:02). Similarly, the time associated with the transfer/filter wash step on the second day of the assay was almost three times as fast when performed on the TECAN as that performed manually (4:33 vs. 12:35). Although not a major reason for automating this assay as compared to the ergonomic and safety issues, automating the HPV cLIA did provide time for the analysts to work on other tasks.
The antibody titers produced from assays performed on the TECAN were statistically similar to the antibody titers from assays performed manually. These results demonstrate the ability of the TECAN workstation to produce reproducible antibody titers that are comparable to antibody titers from assays performed manually.
Faced with a shortage of laboratory personnel and increasing numbers of samples to be tested, there are clear advantages to automating routine laboratory assays. Many of the bottlenecks laboratory personnel face are produced by manual sample processing, which can reduce productivity, cost efficiency, and assay throughput. The implementation of an automated system to perform these otherwise manual and time-consuming routine procedures vastly improved productivity and increased job satisfaction. With many time-bound projects and strict deadlines, our laboratory personnel rely on these automated liquid handlers for precision, accuracy, efficiency, cost savings, and safety; our lab has advanced by improving the quality and speed in which the data are produced, and by reducing turnaround times and decreasing repetitive stress injuries.
The use of a robotic liquid handler to pipette serum and reagents for this multiplexed serological assay has reduced the time spent on sample preparation, increased the number of samples analyzed per day, and reduced the risk of accidental blood-borne pathogen exposure. In addition, automated programs have reduced manual sample handling, which can be a source of laboratory error. The TECAN pipetting program described here represents a platform for pipetting serum for multi-plexed serology assays and supports our epidemiology studies and phase III vaccine clinical trials.
