Abstract
PatchXpress, an automated 16-channel parallel patch clamp system, was used to determine inhibition of human ether-a-go-go related gene (hERG) potassium channels by known blockers. A monoclonal cell line stably expressing hERG potassium channels was generated in CHO-KI cells. Results were compared to conventional patch clamp experiments using similar voltage protocols and solutions. Success rates were evaluated for cell recordings under a variety of conditions, including Accumax versus trypsin treatment to harvest cells, single versus double compound additions, and polystyrene versus glass-coated compound plates. We found that the average success rates rose from 27% with trypsin treatment to 38% with Accumax treatment, which improved to 55–65% following long-term culturing using only Accumax to harvest cells. Two drug additions (spaced 1 min apart with suction off) were also found to produce data that more closely matched conventional experiments. Finally, polystyrene versus glass-coated compound plates were evaluated, and we found that for some compounds (but not all), preparation of compound samples in glass-coated plates resulted in inhibition that more closely matched data obtained by conventional experiments. Therefore, we have established an assay to evaluate the ability of compounds to inhibit hERG potassium channels, which closely matches data produced using conventional methods but with much greater throughput.
Introduction
The human ether-a-go-go related gene (hERG) potassium channel has been implicated as a potential mechanism underlying acquired long QT syndrome (aLQTS); therefore, potential candidate compounds are routinely screened against hERG channels expressed in recombinant cell lines. 1 –3 However, the standard patch clamp technique is low throughput, labor intensive, and requires highly skilled experimenters, factors that greatly limit the number of compounds that can be tested in this assay (generally one compound/day). Using the PatchX-press 7000A (Molecular Devices Corporation, Sunnyvale, CA), we sought to develop assay conditions that would produce data as similar as possible to conventional experiments and that would increase throughput approximately tenfold over that of conventional experiments.
PatchXpress 7000A is an automated patch clamp instrument that can record up to 16 simultaneous and parallel recordings at one time. 4,5 The instrument uses the Sea/chip planar electrode (AVIVA Biosciences, San Diego, CA) instead of traditional patch clamp pipette electrodes. 6 Each Sea/chip consists of 16 planar patch clamp recording apertures of ∼ 1–2 μm in diameter, and high-resistance (gigaohm) seals are achieved once cells are seated. The PatchXpress can perform both ligand-gated and voltage-gated experiments, and multiple drug concentrations can be applied to a single cell. As a result, concentration-response curves can be generated from individual cells to produce IC50 values for compounds of interest. Overall, once assay conditions are optimized, the automated parallel instrument can increase assay throughput and reduce the personnel required to perform experiments while maintaining high data quality and accuracy.
Experimental Details
Cell Culture
A CHO-K1 cell line stably expressing hERG potassium channels was maintained in media containing a mixture of 0.5X Dulbecco's Minimum Essential Media and 0.5X F-12 Nutrient Mixture (GIBCO/Invitrogen, Carlsbad, CA), supplemented with 10% heat-inactivate fetal bovine serum (Summitt Biotechnologies, Fort Collins, CO), 0.5 mg/mL genticin (GIBCO), and 1X penicillin/streptomycin (GIBCO). Cells were passaged every second day, split using a 1:10 or 1:20 ratio, and used between passages 20 and 55 for all experiments.
Briefly, cells were washed with Accumax (Innovative Cell Technologies Inc., San Diego, CA), and treated again for 10–15 min at 42°C. Cells were centrifuged at room temperature for approximately 1–2 min at 250xg. The supernatant was aspirated and cells resuspended in 10 mL total growth media, and then diluted into fresh media.
Cell Preparation
For PatchXpress experiments, cells were harvested as previously described, but cells were resuspended in 150 μL of extracellular solution (see composition below) instead of media. The cells were then incubated for approximately 5 min at room temperature and loaded onto PatchXpress at a final concentration of 1 × 106 cells/mL.
For conventional experiments, cells were harvested as previously described, then resuspended in media at a target density of 2 × 104 cells/mL and plated onto poly-D-lysinecoated glass coverslips (Becton Dickinson, Bedford, MA) 1–2 days prior to the experiment.
Solutions
The following solutions were used to isolate potassium currents in cultured cell lines. (1) Extracellular (in mM): NaCl (150), KCl (4), HEPES (10), CaCl2 (1.2), MgCl2 (1), adjusted to pH 7.4 with NaOH. (2) Intracellular (in mM): KCl (140), HEPES (10), EGTA (5), MgCl2 (6), ATP-Na2 (5), adjusted to pH 7.2 with KOH.
Voltage Pulse Protocol
Voltage-dependent potassium currents were recorded from single cells using the whole-cell configuration of the patch clamp technique. 7 Each cell was held at a negative holding potential of −80 mV. During voltage steps, cells were depolarized from −80 mV to −40 mV for 100 ms to measure and subtract the leak current, depolarized to +20 mV for 1000 ms, and finally repolarized to −40 mV for 500 ms. At the end of the repolarizing pulse to −40 mV, the cell was returned to a holding potential of −80 mV. Data points were collected at a frequency of 0.1 Hz (one stimulus every 10 s). Drugs were applied once a stable baseline had been recorded for 5 min. For PatchXpress experiments, 0.1% DMSO (v/v) in extracellular solution was added as a negative control before drug application. Drugs were then applied either once for 5 min, or twice with the first addition followed by the second addition one min later, and data recorded for 5 min following the second addition. For conventional experiments, drugs were continuously bath-applied and data collected for 5–10 min until steady-state inhibition was achieved and cumulative concentration response curves were generated. The peak amplitudes of outward currents at the end of the 500 ms repolarizing pulse to −40 mV were measured using Clampfit software (Molecular Devices Corporation). Once the peak amplitudes were measured, the effects of the compound were reported as a percentage change compared to the peak amplitudes of the baseline peak amplitude. pIC50s were calculated using the nonlinear curve-fitting function of Prism software (GraphPad Software, San Diego, CA).
Results
Figure 1A,B illustrates the difference between the conventional patch clamp assay and the planar configuration used in the PatchXpress 700A instrument. Figure 1C shows the voltage pulse protocol that is used for experiments, both conventional patch clamp and PatchXpress methods.
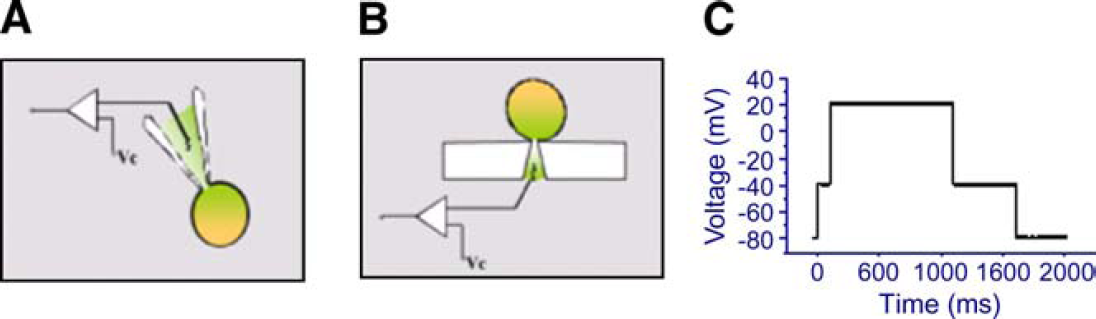
Methods used in conventional patch clamp and PatchXpress: (A) conventional patch clamp used with glass pipette electrode, (B) PatchXpress with planar electrode, and (C) pulse protocol used in both conventional patch clamp and PatchXpress.
Figure 2A illustrates a representative trace obtained on PatchXpress following application of 30 μM olanzapine, a maximally inhibitory concentration. A representative time course of a PatchXpress experiment, where increasing concentrations of olanzapine are applied to the cell and outward current measured every 10 s. The pooled data were used to generate concentration response curves, shown in Figure 2C. Data generated using PatchXpress were very similar to data generated by conventional methods for olanzapine at all concentrations tested (Fig. 2D).
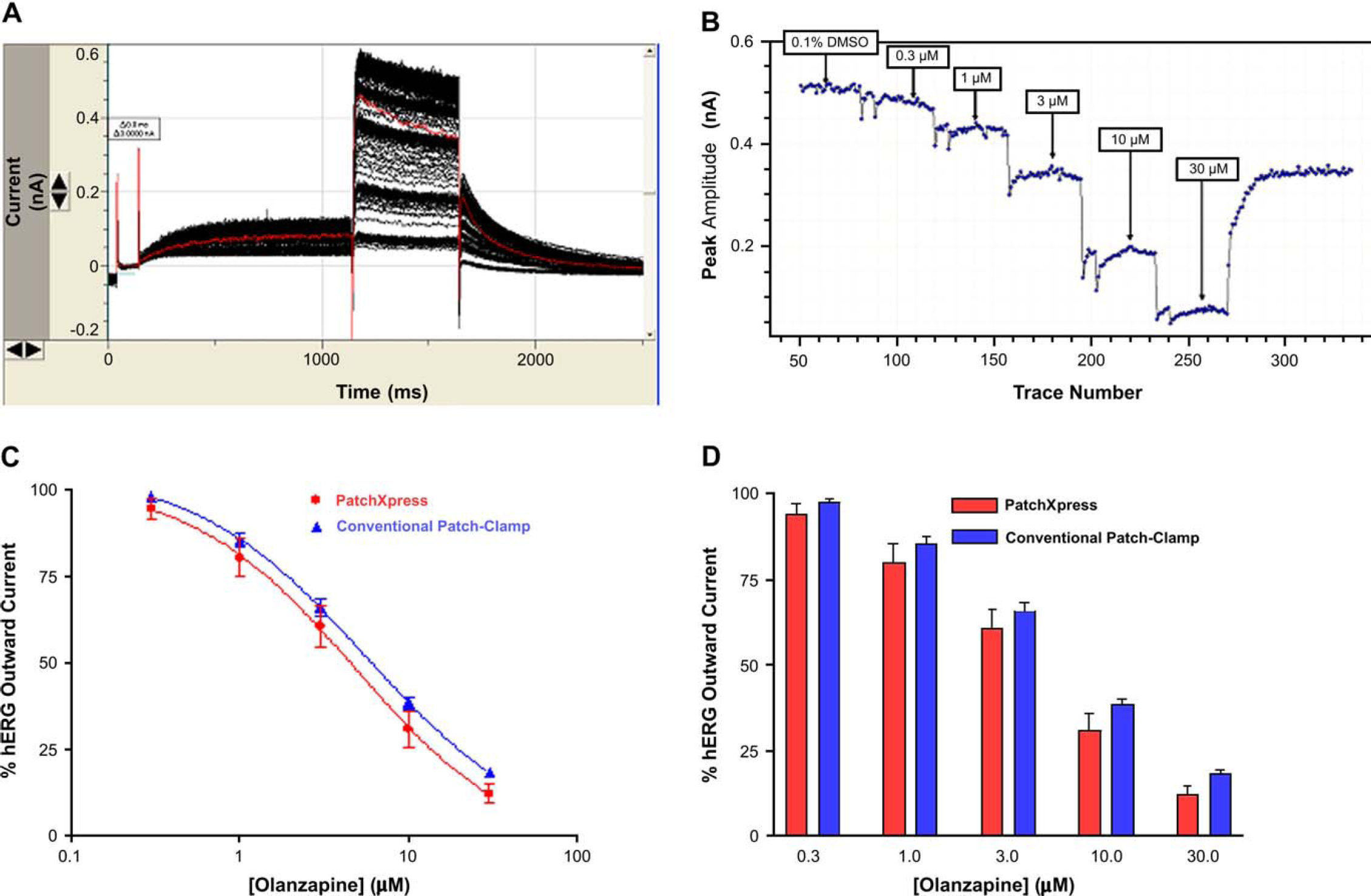
Effects of olanzapine on hERG. (A) Whole-cell hERG currents from PatchXpress were elicited by a 1-s-depolarizing pulse to +20 mV from a holding potential of −80 mV. The cell was then returned to −40 mV to generate large outward currents. (B) The effects of 0.3, 1, 3, 10, and 30 μM olanzapine from PatchXpress are shown. (C) Dose-response relationship for olanzapine block of peak currents at −40 mV. The measured IC50 values were 4.5 μM for PatchXpress and 5.0 μM for conventional methods. (D) The effects of 0.3, 1, 3, 10, and 30 mM olanzapine from PatchXpress and conventional patch clamp methods.
Initially compounds were applied to PatchXpress once per concentration; however, following a single drug addition, compared to conventional methods, compound potency generally decreased when measured by PatchXpress (Fig. 3A), and the correlation between pIC50s generated on PatchXpress and conventional experiments was not robust (r2 = 0.75; Fig. 3B). Because residual bath solution was left in the chamber during PatchXpress experiments, the experiments were repeated using two drug additions so that the final concentration in each chamber would be close to the expected concentration (Fig. 4A). Additionally, more compounds that had been previously tested using conventional methods were tested on the PatchXpress, and overall the correlation between pIC50s generated on PatchXpress and conventional experiments was greatly improved (r2 = 0.97; Fig. 4B).
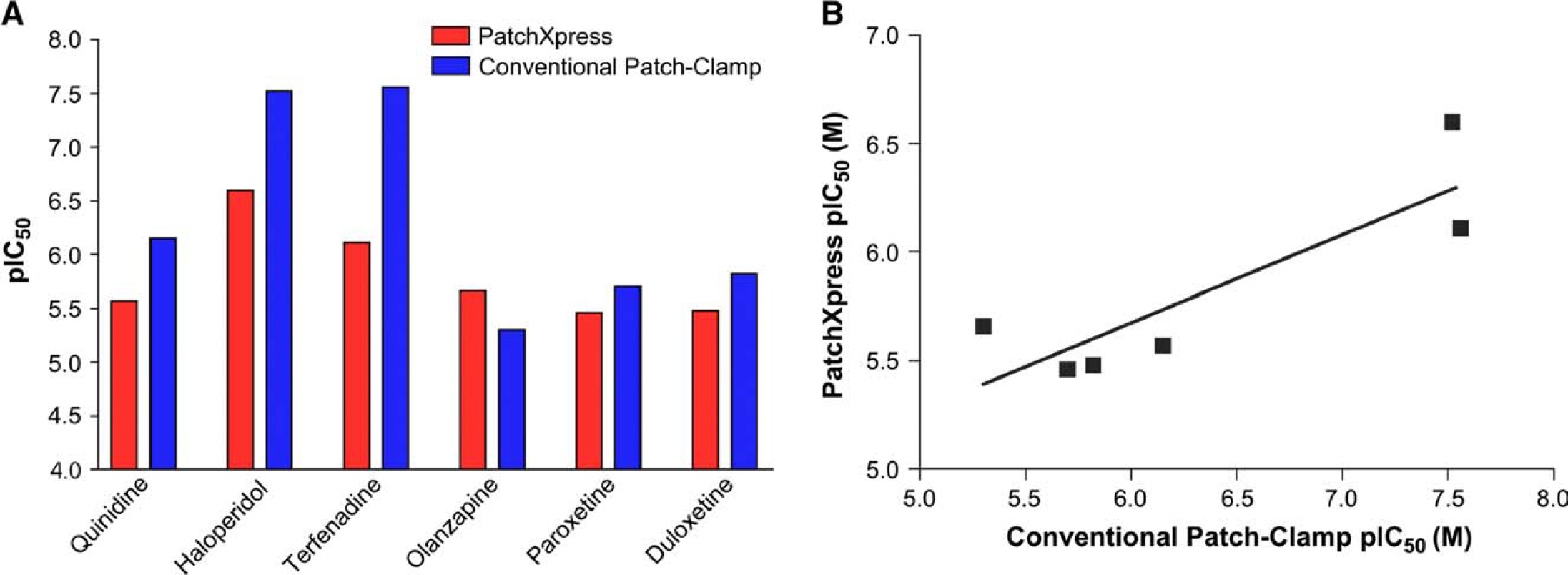
Effects of various compounds on hERG following single compound addition. (A) The potencies of various compounds were determined by measuring the pIC50. The pIC50s obtained using PatchXpress and conventional methods. (B) The correlation between PatchXpress following single compound addition and conventional patch clamp.
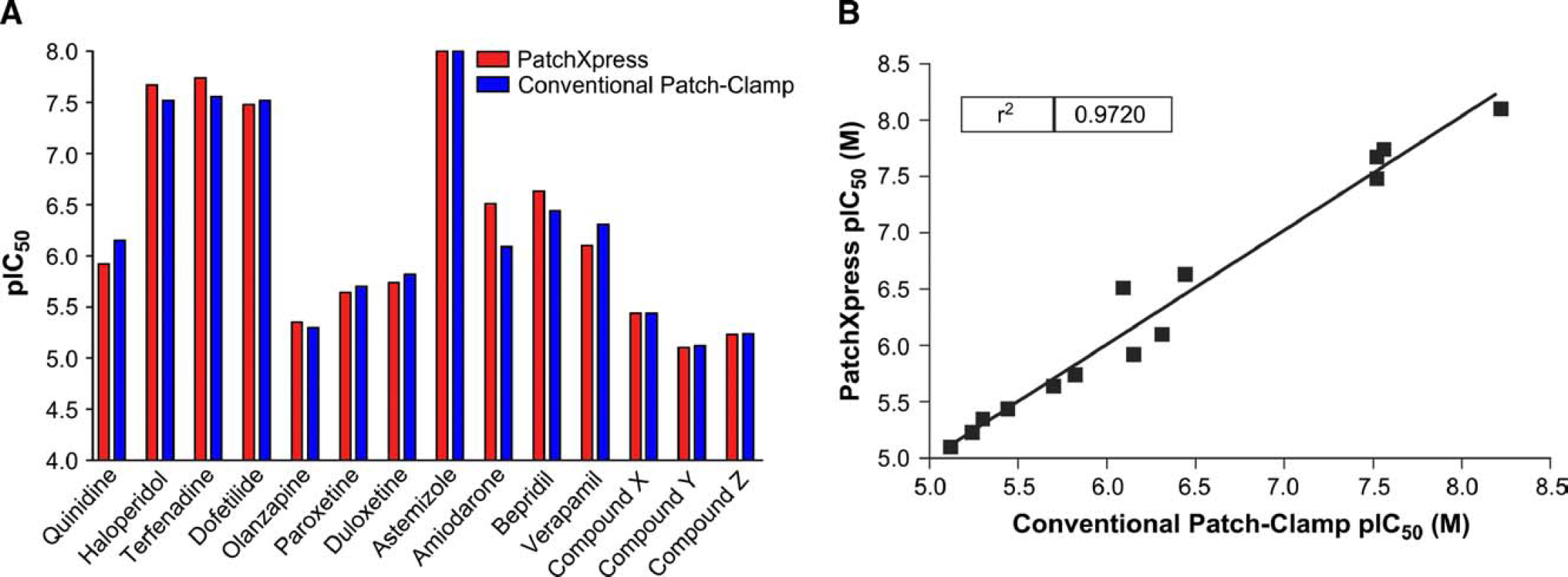
Effects of various compounds on hERG channels following double compound addition. (A) The potencies of various compounds were determined by measuring the pIC50. The pIC50s obtained from PatchXpress and conventional patch clamp experiments. (B) The correlation between PatchXpress following double compound addition and conventional methods.
Finally, because compounds are often screened at a single concentration so that larger numbers of compounds can be tested and prioritized for further experiments, the PatchX-press assay was used to screen at a single concentration and data compared to conventional experiments (Table 1). Compounds had been screened on PatchXpress using polypropylene compound plates. However, when novel compounds were screened, the ability of some compounds to inhibit hERG channels was decreased and not well correlated with conventional data (r2 = 0.65; Fig. 5A), presumably due to compound adsorption to the plate. To address this potential issue, glass compound plates were used and experiments repeated. By using glass-coated compound plates, screening compounds at a single concentration produced a robust correlation between conventional and PatchXpress data (r2 = 0.88; Fig. 5B).
Percent hERG outward current (baseline = 100%; mean ± SEM) a
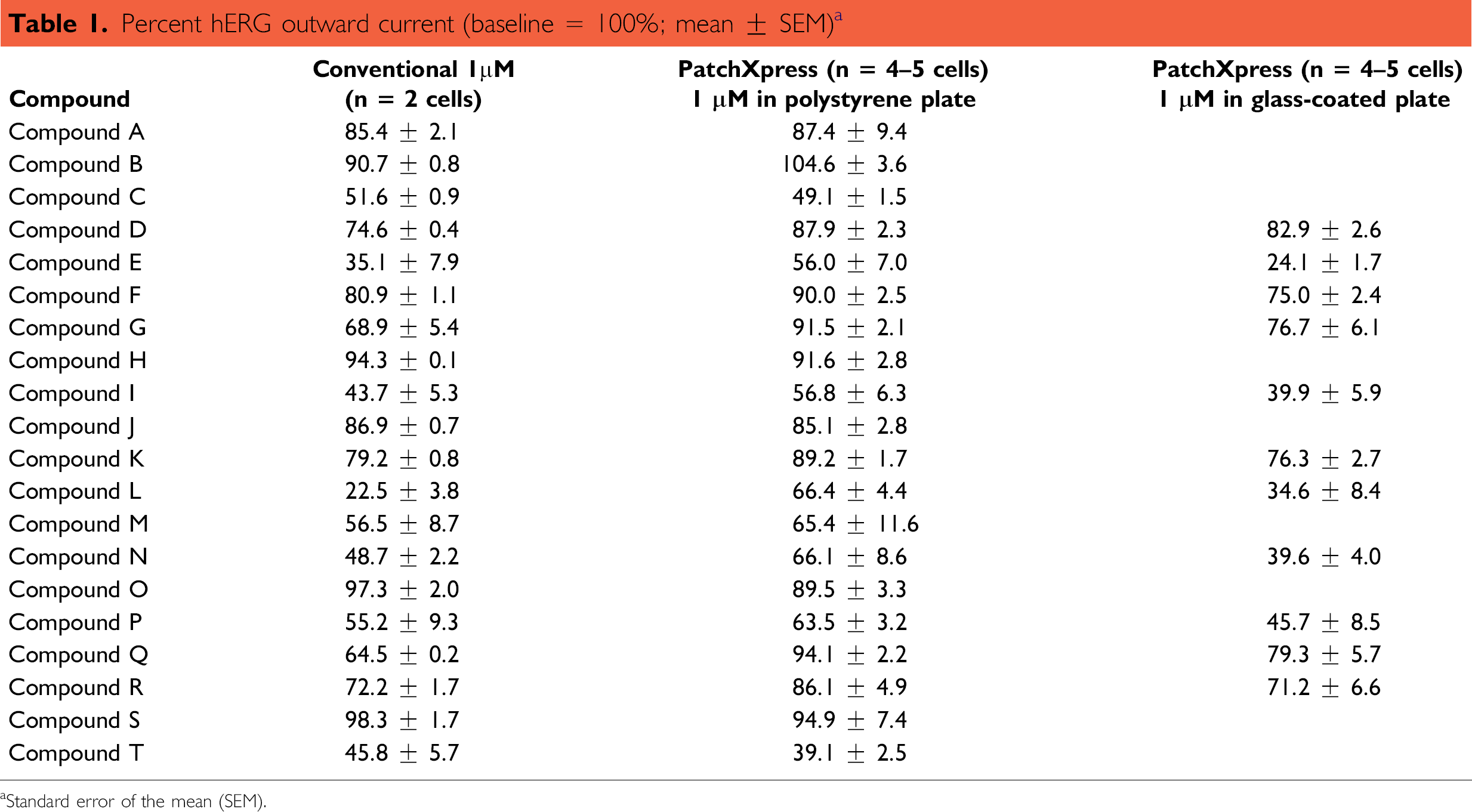
Standard error of the mean (SEM).
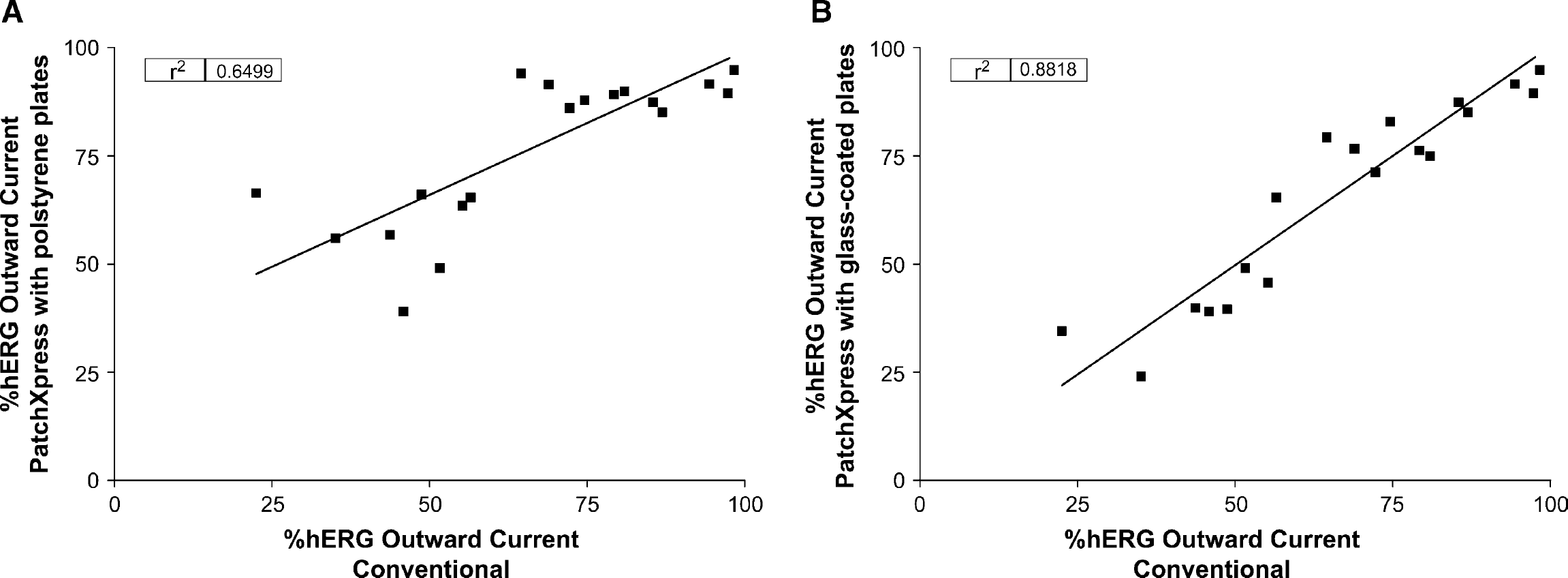
The correlation between conventional patch clamp and PatchXpress at a single testing concentration. (A) The correlation between conventional patch clamp and PatchXpress experiments using polystyrene plates as compound plates.(B) The correlation between conventional patch clamp and PatchXpress experiments using glass-coated plates as compound plates
Discussion
Chemically and mechanistically diverse compounds were used to assess the ability of the PatchXpress instrument to produce data that is both similar to conventional experiments and of high quality. A number of parameters were explored to optimize the assay for PatchXpress and improve the correlation between PatchXpress and conventional data. These parameters required optimization due to slight differences in methodology between automated and conventional experiments (planar versus operator-controlled pipette patch clamp and noncontinuous, small-volume drug delivery via plates versus continuous bath application via solution reservoirs).
Using a cell line in which a high percentage of the cells express current, and optimizing the cell culturing conditions (i.e. using Accumax to harvest cells), improved overall success rates tremendously, particularly for PatchXpress but also for conventional experiments. Cell conditions were more critical due to the random nature by which cells “land” on the aperture using planar technology, as compared to conventional experiments where an operator chooses cells based on morphology.
Secondly, double drug additions were required to ensure adequate mixing of fluids and to minimize drug dilution due to the residual dead volume in each Sea/chip chamber. Compounds are delivered continuously via bath application in conventional experiments and allowed to reach steady-state equilibrium, so drug dilution is not an issue.
Finally, glass plates were found to reduce compound adsorption and improve the correlation between conventional and PatchXpress experiments. Compounds for both conventional and PatchXpress experiments are solubilized in DMSO, then diluted in extracellular buffer and stored in 20-mL-glass vials the same day of the experiment. However, the entire 20 mL of solution is added to large reservoirs immediately before bath application for conventional experiments and allowed to continuously flow through tubing until steady-state concentrations are reached within the bath chamber. For PatchXpress experiments, only 300 μL of solution is added to each well in a 96-well plate and may be stored in the plate for up to 2 h prior to addition to the chamber. Therefore, compound adsorption to polystyrene may be a greater issue when using small volume plates as compared to large solution reservoirs that are used in conventional experiments. We reproduced the compound storage conditions used in conventional experiments by using glass-coated plates (rather than less-costly polypropylene plates typically used in screening) to store compound solutions prior to application for PatchXpress experiments.
Overall, data obtained using optimized assay conditions on PatchXpress were found to be robust, reproducible, and correlated well with conventional data. The optimized assay can be used as an initial compound screen to assess hERG channel inhibition and prioritize compounds to be tested further in other assays. Additionally, PatchXpress can be used for more detailed compound characterization, as potency against the channel was accurately determined by the automated instrument for a number of known hERG blockers as well as previously tested internal compounds.
In addition to the accuracy achieved using the PatchX-press, the overall capacity of the instrument increases the throughput of compound testing approximately eight- to tenfold in our application. Not only does the instrument increase the throughput and the number of compounds that can be tested, the need for highly skilled electrophysiologists was reduced since the PatchXpress can be used following minimal training. By implementing a PatchXpress assay to screen and characterize compounds against the hERG potassium channel, the number of compounds tested has increased, and the cost for testing has decreased because less personnel is required to operate an instrument, while data quality and accuracy are maintained. Many more compounds can be evaluated to assess hERG channel inhibition without sacrificing data quality.
