Abstract
Hydrotropic solubilization is a technique that can be used to improve the solubility of drugs that are poorly soluble. This technique involves adding a large amount of a second solute, known as a hydrotrope, which increases the aqueous solubility of the poorly soluble drug. Hydrotropes such as sodium citrate, sodium benzoate, and urea have been shown to be effective in enhancing the solubility of poorly soluble drugs. This technique has several advantages over other solubility enhancement techniques, including its cost-effectiveness, eco-friendliness, and the fact that it does not require chemical modification of hydrophobic drugs or the use of organic solvents. Hydrotropic agents are now being used to develop various dosage forms, including solid dispersions, mouth-dissolving tablets, and injections, to improve poorly water-soluble drugs' therapeutic effectiveness and bioavailability. This review paper will provide an overview of hydrotropic solubilization techniques.
Introduction
The solubilization of pharmaceuticals with low solubility is a common obstacle in screening new chemical compounds and in the design and development of therapeutic formulations.1,2 The Indian and US Pharmacopoeias contain a significant number of drugs that are poorly soluble or insoluble in water. This presents a major challenge for drug development, as poor solubility can result in poor bioavailability. Solubility is one of the most important physicochemical properties for drug development. 3 Many newly developed drugs have poor solubility. Organic solvents such as methanol, chloroform, dimethyl formamide, and acetonitrile are often used to solubilize these drugs, but these solvents have drawbacks such as high cost, volatility, pollution, and toxicity. Hydrotropic agents offer a safe, eco-friendly, and cost-effective alternative for solubilizing poorly soluble drugs. 4 Hydrotropy was first reported by Neuberg in 1916. 5 It refers to the phenomenon where the addition of a large amount of a second solute increases the aqueous solubility of another solute. Examples of hydrotropic agents include concentrated aqueous solutions of sodium benzoate, sodium salicylate, urea, nicotinamide, sodium citrate, and sodium acetate. 6 Hydrotropic agents are typically anionic organic salts with two essential parts: an anionic group and a hydrotropic aromatic ring or ring system. The high aqueous solubility is due to the anionic group, which may have a minor effect on the phenomenon. 7
Solubility
Solubility can be described both quantitatively and qualitatively. Quantitatively, it is the concentration of the solute in a saturated solution at a specific temperature. Qualitatively, it refers to the tendency of a substance to dissolve spontaneously. Solubility is the process by which two or more components combine to create a uniform molecular mixture. A saturated solution is one in which the solute and solvent are in equilibrium. In pharmaceutical sciences, solubility can be expressed in various ways when quantitative data is available. Some of the common units used to express solubility include parts, molarity, normality, formality, mole fraction, percent solution, volume fraction, and molality. Drug solubility refers to the maximum concentration of drug solute that can dissolve in a given solvent under specific conditions such as temperature, pH, and pressure. The solubility of a medication in a saturated solution is considered a static feature, while the dissolution rate of the drug is regarded as a dynamic property that is more closely related to the rate of bioavailability. 8
Need of Solubility
The absorption of drugs from the gastrointestinal (GI) tract can be hindered by several factors, 9 with the most prominent being the drug molecule’s inadequate water solubility and membrane permeability. For an active drug to be effectively delivered orally, it must dissolve in gastric and/or intestinal fluids before permeating the gastrointestinal walls and entering systemic circulation. Therefore, two specific areas within pharmaceutical research prioritize enhancing the oral bioavailability of active agents by improving their solubility and dissolution rate. The Biopharmaceutics Classification System (BCS) is a scientific framework used to categorize therapeutic substances based on their water solubility and intestinal permeability. For BCS class II and IV drugs, the rate-limiting step is the release of the drug from the dosage form and its solubility in stomach fluid, rather than the absorption process. Thus, enhancing the solubility of these drugs can lead to an increase in their bioavailability.10,11
Hydrotropic Solubilization
Around 70% of newly discovered drug candidates have low water solubility, presenting a challenge for the pharmaceutical industry to find techniques to make drugs more water-soluble. 12 The therapeutic efficacy of a medication depends on its bioavailability, which is largely related to the solubility of the drug moiety. 13 There are now various formulation techniques that can enhance solubility and dissolution to increase oral bioavailability. 14 In addition to these advancements, hydrotropy is a recognized technique for resolving solubility-related issues. This study aims to construct several hypothetical systems, examine them, and discover the geometric characteristics of hydrotropic substances to aid researchers in studying hydrotropy for the development of drug delivery. 15 Within the context of the biopharmaceutics classification system, a number of corticosteroid drugs are classified as belonging to class II (BCS). These drugs have low solubility in the body’s fluids, resulting in poor oral bioavailability. 16
Hydrotropy
In the hydrotropy solubilization process, a significant amount of a second solute is added to increase the water solubility of a third solute. This process is known as solubilization. Complexation, which involves weak interactions between hydrotropic substances such as sodium alginate, sodium acetate, sodium benzoate, and urea, and weakly soluble drugs, is used to increase solubility. This process is more closely related to complexification. It is caused by the presence of many salts containing large anions or cations that are themselves highly soluble in water. This occurrence is known as “salting in.” Hydrotropic solutions are not considered colloids because they are non-colloidal and have a weak link between the hydrotropic agent and the solute. 17
Mechanism of Action of Hydrotrope
The mechanism of action of hydrotropes must be clarified since this issue is still being debated and needs to be settled. Numerous researchers have speculated on the likely operation of a hydrotrope. The three ideas proposed to explain hydrotropic activity are as follows: a) the formation of a complex between the solute and the hydrotrope; b) the breakdown or fragmentation of a water-based tetrahedral complex; c) the spontaneous creation of hydrotrope connections.
18
The Spontaneous Creation of Hydrotrope Connections
This theory posits that the molecules of hydrotropes will self-aggregate to form organized clusters in the hydrophilic fluid. This will facilitate the establishment of structured clusters. Moreover, it was hypothesized that the enhanced hydrophilicity of the solute might be ascribed to the trapping of solute molecules inside the structured clusters.19,20 The Minimum Hydrotropic Concentration (MHC) is the concentration at which hydrotropes start to aggregate. This concentration is designated as MHC. 21 It was hypothesized that the molecular structure of hydrotropes comprises planar aromatic rings that are layered atop one another to create aggregates. Similar to the micellization process, the aggregation phenomenon happens simultaneously in the aqueous solvent as the solubilization of the hydrophobic chemical commences. It has been found that the capacity of hydrotropes to self-aggregate is essential for various applications in many domains, such as the solubilization of medicines. The enthalpy, entropy, and free energy of a system will all suffer alterations due to the aggregation of hydrotropes.22,23
Complex Formation Between the Solute and Hydrotrope
It's possible that the medication and the complexing molecules won't have a direct affinity for one another while the process of solubilizing the treatment is going on. However, they still need to communicate with one another to reduce the amount of time order to reduce the amount of time that the medication and the complexing molecules spend in contact with water. Stacking may take place either between molecules of the same species, known as self-association, or between molecules of different species, known as hetero-association (co-association). Most of the stacking process takes place between molecules with a planar shape, which enables those molecules to have the least amount of surface area accessible to water. A simple 1:1 complex has two components, one molecule of each medication and one molecule of a complexing agent. The complex is made up of two molecules total. It is possible to produce a sandwich complex with a ratio of 1:2 when the complexing agent surrounds the core molecule on both sides. This occurs when the sandwich complex is produced. 24
The Water Molecules Form a Tetrahedral Complex that May Dissolve or Fracture
Hydrotropes may improve the solubility of weakly or sparingly soluble solute ions via their interaction with hydrotropes. This is because hydrotropes reduce water’s surface tension, destabilizing solid forms such as ice crystals. Consequently, a reduction in surface tension breaks down the barrier between solutes and solvents, enhancing their dispersibility and solubility in water. This is because the barrier between the two no longer exists.19,20
Advantages of Hydrotropy and Mixed Hydrotropy
1. This solvent is less hazardous than another organic solvent. 2. Using hydrotropes for solubilization is a straightforward and inexpensive method because it only requires mixing the drug substance and specific hydrotropes with the solvent. This can provide answers to solubility problems in a wide variety of commercial and pharmaceutical applications. 3. Hydrotropes have a common application in today’s increasingly intricate formulas. 4. One of the main positive aspects of the hydrotropic technique are that it does not alter the chemical activity of the drug active ingredients. 5. Hydrotropes have broad formulation, compatibility, and effectiveness, are inexpensive, and are considered a greener solvent. 6. Because of their high selectivity over other solubilization techniques, hydrotropy is suggested as being superior as a point of safe, cheap, simple, accurate, and precise method. 7. Mixed hydrotropy precludes the use of an organic solvent to prevent problems. 8. For optimum criteria, mixed hydrotropy is one of the best approaches. 9. In the field of science, hydrotropy has a role in extraction and separation. 10. Certain properties of hydrotropes, such as high selectivity, solvent character being independent of pH, and absence of emulsification are beneficial. 11. The solute can be easily recovered from hydrotrope solutions by simple dilution. 12. The main benefits of mixed hydrotropy are that a combination of agents can lower the individual concentration that should be effective as well as less toxic. 13. For solubility of poorly water-soluble drugs, mixed hydrotropy has a synergistic effect on the solubility of the substance.
Disadvantages of Hydrotropy and Mixed Hydrotropy
1. There are chances of weak interaction between the hydrotropic agent and drugs. As water is used as a solvent, complete removal of water cannot be achieved. 2. The use of hydrotropes is limited for some hydrotropic agents because of toxicity.
These are the limits of hydrotropic agents: • Sodium benzoate: Oral medications may contain up to .5% of benzoic acid, and the same percentage is allowed in medicines administered parenterally.
25
• Nicotinamide (NAD): It has been noted in a study that supplementation with nicotinamide mononucleotide, which boosts blood NAD concentrations, is deemed safe and well-tolerated when orally administered up to 900 mg daily. The clinical efficacy, as indicated by blood NAD concentration and physical performance, appears to peak at a daily oral intake of 600 mg.
26
• Potassium citrate: The goal of the treatment is to bring urinary citrate levels back to normal (above 320 mg/day and as close as possible to the average normal value of 640 mg/day), and to raise the urinary pH to between 6.0 and 7.0. It is recommended not to exceed a daily dose of 100 mEq of Urocit®-K, as doses higher than this have not been studied.
27
• Sodium Citrate: Commonly used in the medical field to prevent the formation of kidney stones. The recommended dosage for adults typically ranges from 10-30 mL, which should be diluted in up to 6 oz of water or juice and taken orally after meals and at bedtime as needed.
28
• Sodium acetate: Sodium Acetate Injection, USP (2 mEq/mL) is used. It is a sterile, nonpyrogenic, concentrated solution of sodium acetate in water for injection. The solution is administered after dilution for safety purposes.
29
• Caffeine: Regarding the safe consumption of caffeine for humans, it is generally accepted that up to 400 milligrams (mg) per day is safe for most healthy adults.
30
• Urea: The typical range for urea concentration in human blood is between 6 to 20 mg/dL (2.1 to 7.1 mmol/L). Ideally, the urea level should be within the range of 12 to 20 grams over a 24-hour period in urine. The normal range for urea creatinine typically lies between .59 mg/dl and 1.35 mg/dl.
31
Approach
BCS Classification of Drug.

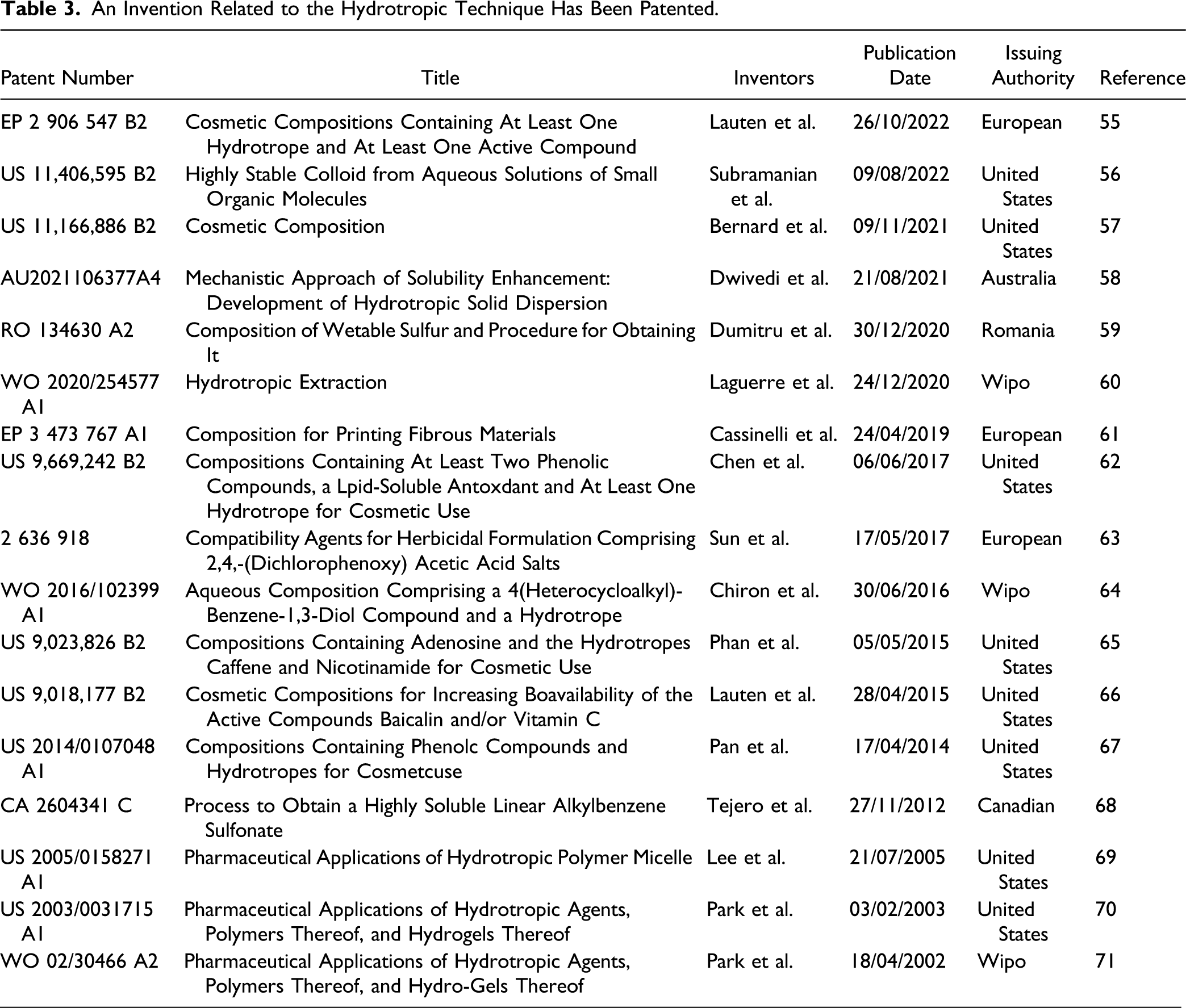
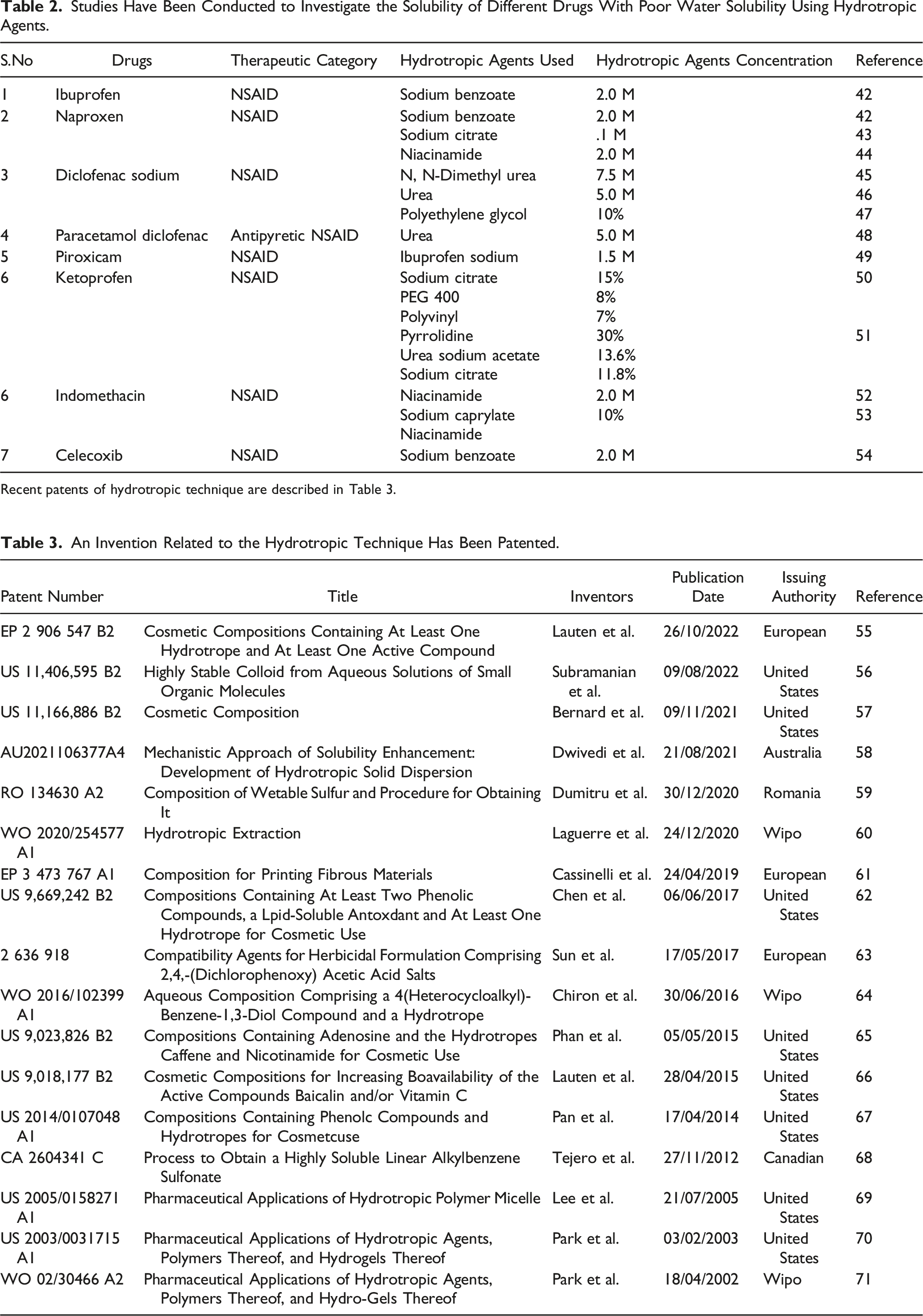
Studies Have Been Conducted to Investigate the Solubility of Different Drugs With Poor Water Solubility Using Hydrotropic Agents.
Pharmaceutical Application
Hydrotropes have been employed in the realm of pharmaceutical science for the purpose of drug formulation and stabilization. Hydrotropes have been utilized in various applications, including the production of parenteral, oral, and topical medications, as well as in the development of novel drug delivery systems. It is primarily utilized for pharmaceutical substances that exhibit challenges in solubility.
Analytical Application
This technique is employed in analytical chemistry to mitigate the utilization of organic solvents and improve the solubility of the sample in an aqueous medium. Hydrotropes have been employed in a variety of methods, There are three commonly used analytical techniques in the field of chemistry: titrimetric analysis, spectrophotometric analysis, and thin-layer chromatography. These methods are widely employed for the quantitative and qualitative analysis of various substances.33,34
Chemistry Application
Hydrotropes are utilized in order to facilitate the separation of compounds, enhance solubility, and carry out extraction processes. Hydrotropes are commonly employed in green synthesis due to their eco-friendly nature. An illustration of the application of the Gibbs phase rule in the determination of the fundamental principles of statistical thermodynamics of hydrotropes is presented. 35 Hydrotropes are employed to investigate the behavior within microemulsions. 36
Pharmacognosy Application
In pharmacognosy, a variety of extraction processes are used. Hydrotropes can be used as an assisting agent to increase the percentage of active constituents obtained. For example, when a hydrotropic agent is added to the solvent during the extraction of reserpine from the roots of Rauwolfia vomitoria, the yield is comparable to that of conventional extraction methods. Sodium-cumene sulfonate (Na-CS), sodium-p-toluenesulfonate (Na-PTS), and sodium-n-benzylbenzenesulfonate (Na-NBBS) are three representative hydrotropes that were used for the extraction of reserpine at equivalent concentrations. 37
Hydrotropic salts such as sodium salicylate, sodium cumene sulfonate, sodium phenol sulfate, and sodium xylene sulfonate can be used as modified hydrotropes in the process of extracting lignin from Birch wood. Two treatments were conducted: a conventional process and a modified process. 38
Computational Application
Software is utilized to forecast the mechanism through which hydrotropes enhance solubility by employing computer simulations, predominantly relying on the classical molecular dynamics simulation technique. 39
Biochemistry Application
Sodium xylene sulfonate (SXS) and formic acid are both hydrotropes commonly employed in pretreatment processes due to their ability to enhance enzymatic activity. For instance, when subjected to these hydrotropic agents, reed exhibits enhanced digestibility. 40 In Asia, rice straws are commonly chosen as the primary feedstock for the production of fuel ethanol. Prior to the ethanol production process, these rice straws undergo pretreatment using hydrotropic agents such as sodium cumene sulfonate (NaCS) and sodium cumene sulfonate (NaCS). Hydrotropes are considered to be a more environmentally friendly solvent option, offering numerous advantages. Notably, they present themselves as an appealing substitute for biomass refineries. Additional benefits associated with hydrotrope solutions include the expeditious recuperation of the solute through straightforward dilution with water, as well as the ability to reuse the hydrotrope solution following its reconcentration. The study examined the effects of biomass loading, time, temperature ranging from 40 to 80°C, and hydrotrope concentrations of 10% and 30%. 41
Current and Future Prospects
Current Prospects of Hydrotropic Solubilization
One potential benefit of hydrotropic solubilization is its ability to enhance medication delivery efficacy through the augmentation of drug solubility in aqueous formulations. The enhanced solubility frequently results in improved drug absorption and distribution within the body, hence augmenting its therapeutic effectiveness. Hydrotropic agents exhibit the ability to be employed in a diverse range of pharmaceutical dosage forms, encompassing oral solutions, suspensions, and injectable formulations. The flexibility inherent in this approach enables pharmaceutical companies to create medications that are more aligned with the specific needs and preferences of patients. Enhancing drug solubility through hydrotropic solubilization has the potential to expedite the drug development process, resulting in reduced development time and costs. Researchers have the potential to circumvent the necessity of creating wholly novel pharmacological molecules, a procedure that is both time-consuming and costly, by employing pre-existing APIs (active pharmaceutical ingredients) in more soluble forms. The utilization of hydrotropic solubilization frequently incorporates the employment of hydrotropes that are biodegradable and biocompatible, rendering them a more ecologically sustainable alternative in contrast to other solubilizing methodologies that rely on hazardous solvents.
Future Prospects of Hydrotropic Solubilization
With the acquisition of a more profound understanding of the fundamental mechanisms and interactions involved in hydrotropic solubilization, researchers have the potential to discover novel and improved hydrotropic agents. This could result in further enhancements in the solubility and delivery of drugs. Combination therapies involve integrating hydrotropic solubilization with other drug delivery methods, such as nanoparticles or liposomes, to develop novel and targeted drug delivery systems. The integration of this combined approach has the potential to significantly transform pharmacological therapy and create new opportunities for treatment. With the accumulation of additional research and favorable outcomes, it is likely that pharmaceutical companies will incorporate hydrotropic solubilization into their drug development pipelines. The potential outcome of increased adoption rates is the advancement of medication commercialization, resulting in the production of a greater number of pharmaceuticals characterized by enhanced solubility and bioavailability. The future viability of hydrotropic solubilization is contingent upon the acceptance and evaluation of the safety and effectiveness of hydrotropic formulations by regulatory agencies. The provision of explicit guidelines and comprehensive support by these bodies has the potential to foster increased utilization of such formulations within the pharmaceutical sector.
Conclusion
This paper presents a comprehensive analysis of the prospective applications of hydrotropic solubilization in the pharmaceutical sector. It discusses various major discoveries and draws relevant conclusions. Hydrotropic agents possess the capacity to substantially enhance the solubility of medicinal substances in aqueous solutions, hence potentially augmenting their absorption and bioavailability. This enables researchers and pharmaceutical enterprises to develop diverse dosage forms, including oral solutions, suspensions, and injectable formulations, so augmenting the adaptability of drugs. The inherent flexibility of this system enables the customization of drug administration to cater to the specific requirements of each patient.The utilization of hydrotropic agents in the process of solubilization presents advantages in terms of cost-effectiveness and time efficiency, as it allows for the avoidance of the resource-intensive task of developing new pharmaceutical compounds. This methodology yields cost reductions and expedites the timelines for drug development. Hydrotropic agents possess notable attributes such as biodegradability and biocompatibility, rendering them a viable and ecologically sound substitute for solubilization methods reliant on harmful solvents. Combination therapies encompass the integration of hydrotropic solubilization with other drug delivery methods, such as nanoparticles or liposomes, in order to establish sophisticated and accurate drug delivery systems. This has the potential to result in a significant shift in the field of pharmacological therapy and the development of innovative therapeutic approaches. Nevertheless, it is crucial to acknowledge that the utilization of hydrotropic solubilization remains a rather nascent domain, necessitating additional investigation to comprehensively comprehend the fundamental mechanisms and relationships implicated. Furthermore, the broad implementation of hydro-tropic solubilization in the pharmaceutical sector would heavily depend on regulatory factors and the approval of health authorities.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.