Abstract
IQ® Technology, a homogeneous, universal-detection platform, originally designed for high-throughput screening (HTS) of kinases and phosphatases, has now been applied to protease screening. Representative enzymes from the major classes of proteases have been assayed in the IQ® format. Enzyme activity and compound inhibition data are presented for such enzymes as Trypsin, Matrix Metalloproteinase 3 (MMP-3) and Calpain 1. The technology has been tested in 96- to 384- to 1536-well microplate formats and is universally suited for automated screening. IQ® Technology is a direct, noncompetitive assay that does not require antibodies or radioisotopes. Fluorophore-labeled peptides are used as enzyme substrates. Kinase or phosphatase activity is quantified by direct measurement of the phosphorylation state of the substrate. For protease activity, cleavage is quantified with a peptide substrate containing a phospho-residue distal to the fluorphore. Cleavage of the substrate liberates the fluorphore-labeled terminus from the terminus containing the phospho-residue. Protease activity is measured by the change in fluorescence intensity that occurs when a proprietary compound binds specifically to phosphoryl groups on peptides and quenches the fluorescence. Iq® Technology can be used with any peptide sequence and is insensitive to high concentrations of ATP and substrate. The IQ® Technology has been validated against a large number of detergents, organics, and other reagents found in reaction mixtures and has been optimized for HTS applications exhibiting representative Z' values of 0.7. (JALA 2004;9:171-6)
Keywords
Introduction
Pierce has developed the IQ® Assay Platform, a novel, patent pending, homogeneous assay format for determination of protein kinase, phosphatase or protease activity. IQ® Technology is based on the ability of an iron-containing compound to quench the fluorescent emission of fluorophore-labeled phosphorylated peptides. In a protein kinase assay the IQ® Reagent Working Solution (WS) binds specifically to phosphoryl groups on the peptide end-product. Binding of the iron-containing compound to the phosphoryl group on the peptide, results in a quantitative decrease in the observed fluorescent emission of the phosphorylated peptide relative to a nonphosphorylated peptide. The iron-containing compound thereby serves as a specific dark quencher for phosphorylated, fluorophore-labeled peptides. In a protein phosphatase assay, dephosphorylation of the peptide results in an increase in the observed fluorescent emission intensity, compared to a phosphorylated peptide, after addition of the IQ® Reagent WS. In a protease assay, peptide substrates are C-terminally modified with a phosphorylated amino acid. The peptide sequence includes a specific cleavage site unique for the protease being studied. The IQ® Reagent WS binds specifically to the phosphoryl group on the peptide substrate. The binding of the iron-containing compound to the phosphoryl group on the undigested peptide results in a quantitative quenching of the observed fluorescence of the peptide. When the protease cleaves the peptide, the dye is no longer in proximity to the bound IQ® Reagent and the observed fluorescence increases in direct proportion to the molecules of cleaved product. A pictorial of an IQ® Kinase, Phosphatase, and Protease Assay is shown in Figure 1.
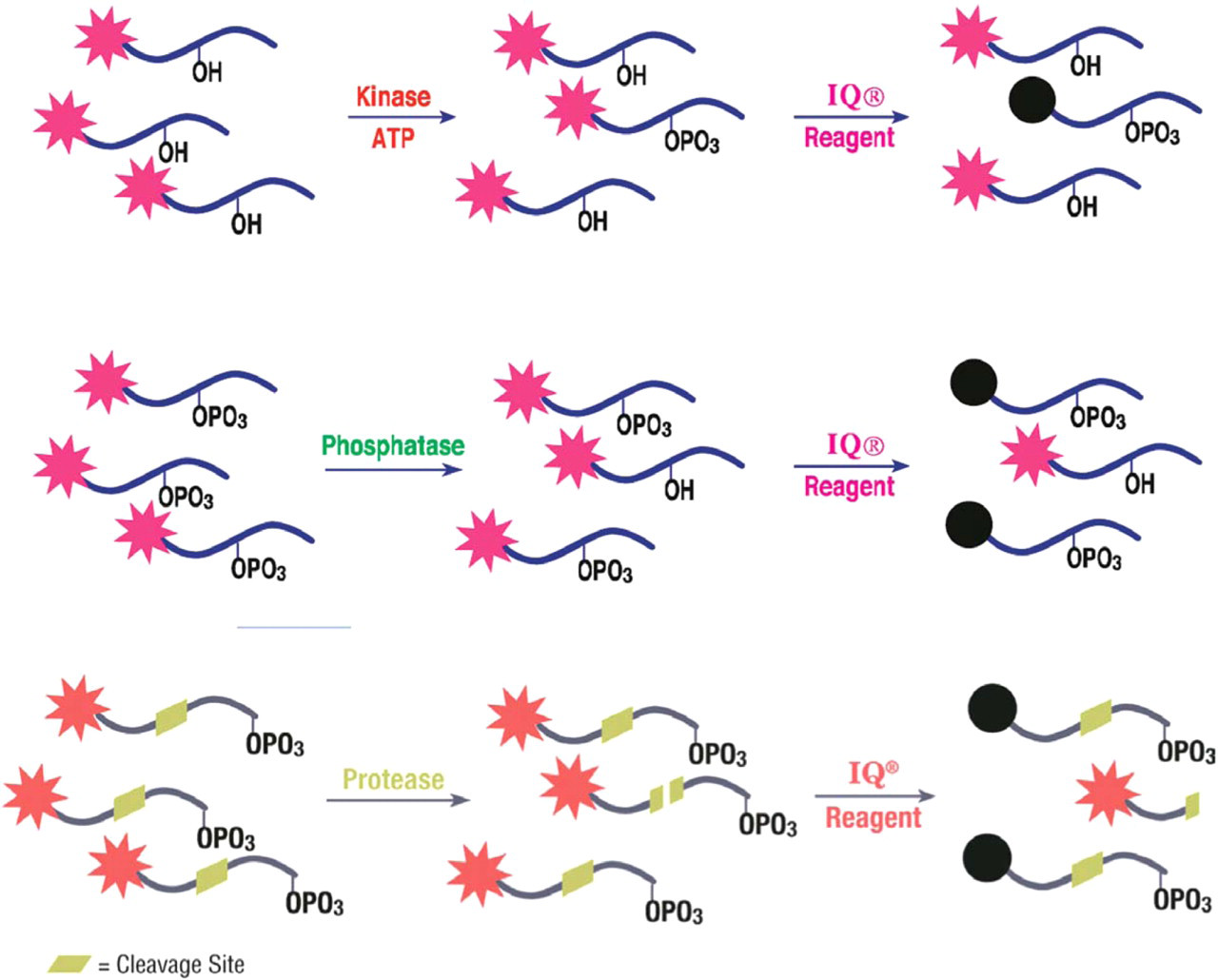
Schematic representation of IQ® Kinase, Phosphatase, and Protease Assays. The IQ® Reagent binds specifically to the phosphoryl group on the peptide. The result is a reduction of the observed fluorescence proportional to the extent of phosphorylation in a kinase reaction mixture, an increase of the observed fluorescence proportional to the extent of dephosphorylation in a phosphatase reaction mixture and an increase of the observed fluorescence in direct proportion to the cleaved product in a protease reaction mixture.
Materials and methods
Enzymatic assays were conducted at room temperature in black Greiner 96 microwell plates. Protein kinase C (PKC) purified from rat brain, purified recombinant cyclic AMP-dependent protein kinase (PKA) catalytic subunit, and TPCK Trypsin are Pierce enzymes; other enzymes were purchased from Upstate Biotechnology (Charlottesville, VA) or EMD Biosciences (La Jolla, CA). PKC assays (Figure 2) were conducted in a final enzyme reaction mixture consisting of 20 mM HEPES, 1 mM CaCl2, 5 mM MgCl2, 1 mM disodium ATP, 1 mM dithiothreitol (DTT), 0.2 mg/mL phosphatidyl-L-serine, pH 7.4. PKA assays (Figure 2) were conducted in a final enzyme reaction mixture of 20 mM HEPES, 5 mM MgCl2, 1 mM disodium ATP, 1 mM DTT, 0.1 mM cAMP, pH 7.4. Src, PKB, and Src kinase assays (Figure 2) were conducted in a final enzyme reaction mixture of 20 mM HEPES, 5 mM MgCl2, 2 mM MnCl2, 1 mM disodium ATP, 1 mM DTT, pH 7.4. Phosphatase assays (Figure 3) were conducted in a final enzyme reaction mixture of 20 mM HEPES, 5 mM MgCl2, 1 mM DTT, pH 7.4. The amount of Recombinant MMP-3 catalytic subunit (Figures 4A, 5B, and 6) used reflects a common unit definition where 1 U hydrolyzes 1.0 mmol of MCA-RPKPVE-Norvaline-WRK-(DNP)-NH2 per minute at 37 °C, pH 7.0. The amount of Calpain 1 (Figures 4B, 5A and 6) from human erythrocytes used reflects a common unit definition where 1 U will increase the absorbance at 750 nm by 1.0 in 30 minutes at 30 °C using casein as a substrate. The amount of TPCK Trypsin (Figure 7) used reflects a common unit definition where 1 U hydrolyzes one mmole of p-toluene-sulfonyl-L-arginine methyl ester (TAME) per minute at 25 °C, pH 8.2, in the presence of 0.01 M calcium. Reaction conditions, reaction incubation times, substrate identity, and substrate concentration for each enzyme reaction are described in Figures 2–8, respectively. Reactions were stopped by addition of a 0.25x IQ® WS. Fluorescence was measured using a Tecan Safire with excitation/emission of 560/590 and bandwidth of 2.5 or an ULTRA® microplate fluorometer with excitation/emission of 560/590 with bandwidths of 10 and 7, respectively. Data were plotted using GraphPad Prism® either as observed relative fluorescence units (RFU) or after normalization of the RFU (net RFU) to control wells.
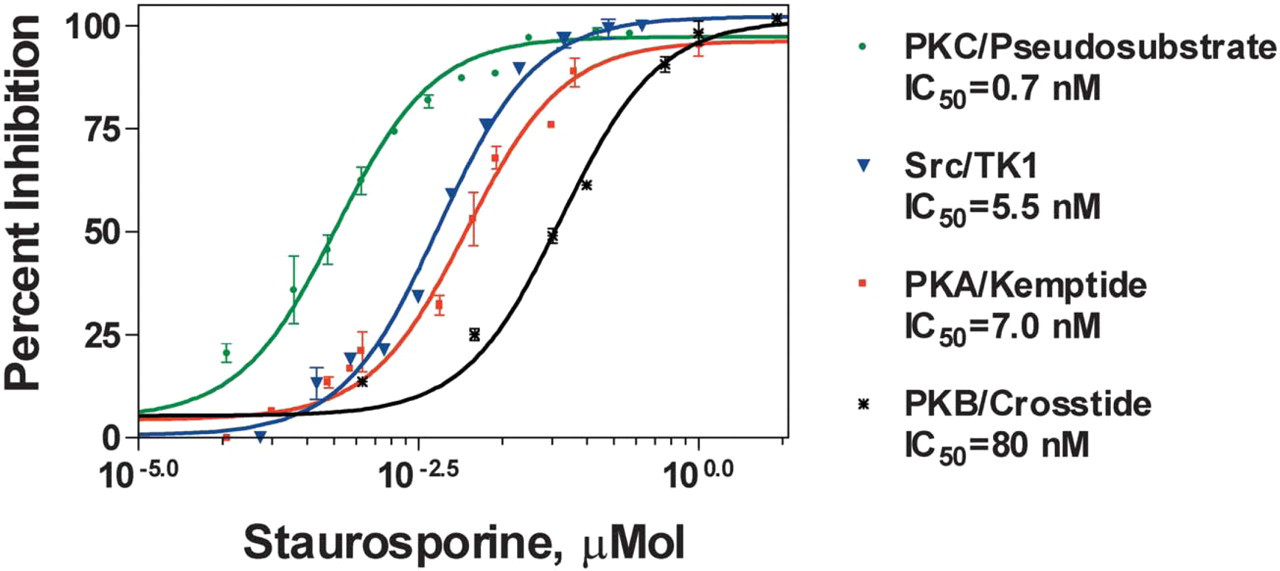
Dose-dependent inhibition of kinases using the IQ® Platform. IC50 values for the kinase inhibitor staurosporine were determined using the specific fluorophore-labeled peptide substrates and kinases listed in the graph. Inhibitor studies were carried out in triplicate using pre-determined substrate and enzyme concentrations. Staurosporine was diluted in 10% DMSO and incubated with each enzyme for 20 minutes at room temperature, after which the enzymatic reaction was initiated by addition of ATP at a final concentration of 20 μM. The enzymatic reaction was carried out for 1 hour at room temperature followed by addition of IQ® WS. Fluorescence was then measured and data were analyzed using nonlinear regression. Measured values correlate with published IC50 values.
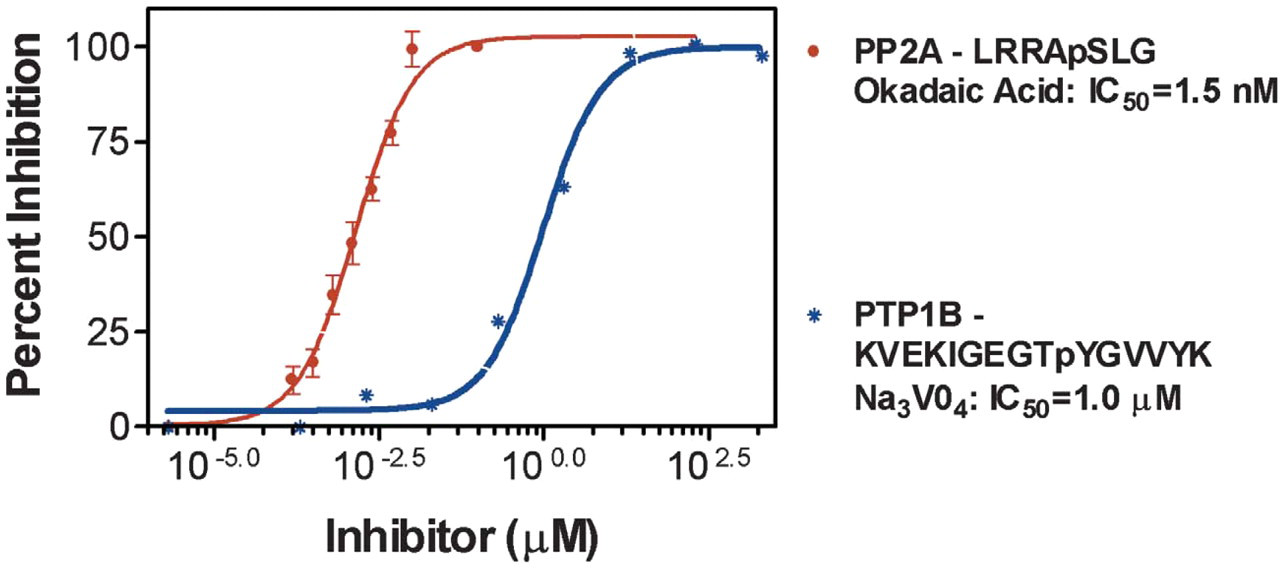
Dose-dependent inhibition of phosphatases using the IQ® Platform. IC50 values of two known inhibitors (shown in the graph) were determined using the specific fluorophore-labeled phosphopeptide substrates and serine/threonine or tyrosine phosphatases listed in the graph. Inhibitors were diluted and incubated with each enzyme in the presence of the listed phosphopeptide with reaction buffer for 1 hour at room temperature. IQ® WS was added and fluorescence was measured. Data were analyzed using nonlinear regression. N = 3. Measured values correlate with published IC50 values.
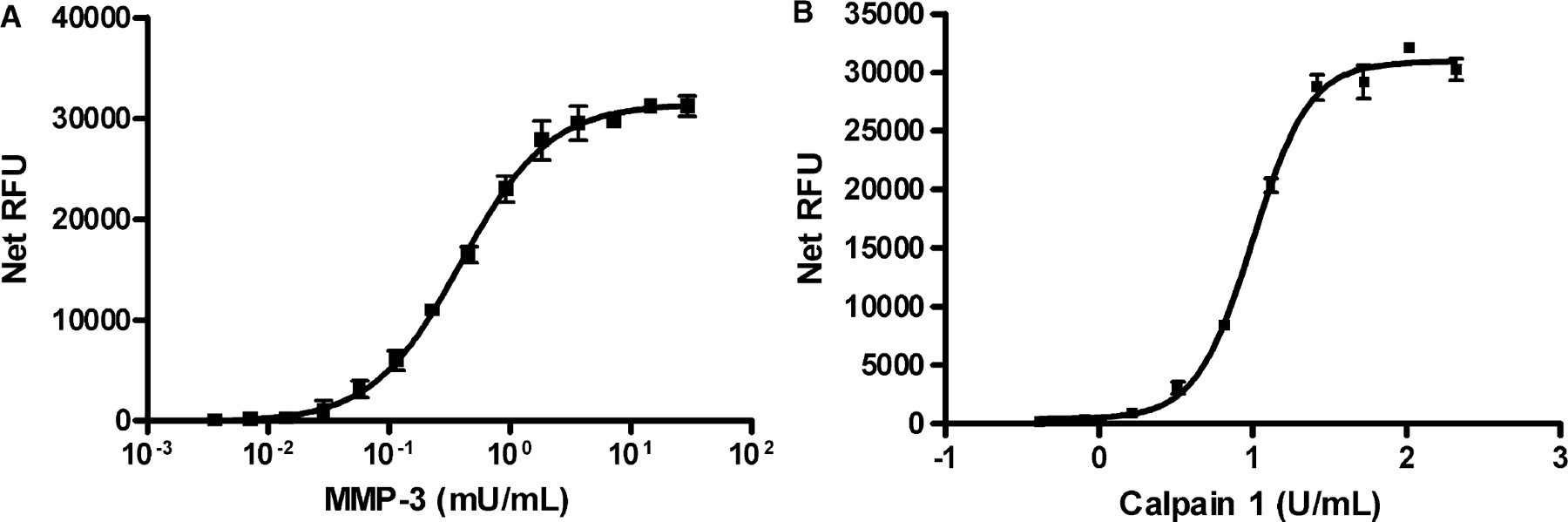
(A) MMP-3 titration using the IQ® Protease Assay. MMP-3 was serially diluted two-fold over 14 wells in triplicate. Dye-labeled peptide substrate (Dye-RPKPVE-Norvaline-WRKpSA) was added at a concentration of 10 μM final in a reaction buffer of 100 mM MES, pH 6.0, 10 mM CaCl2, 150 mM NaCl, 10 μM ZnCl2, 0.05% Brij-35. The mixture was incubated for 1 hour at room temperature and stopped by the addition of 0.25 × IQ® WS. The linear range of MMP-3 activity was 0.014–1.84 mU/mL. (B) Calpain I titration using the IQ® Protease Assay. Calpain I was serially diluted two-fold over 10 wells in triplicate. Dye-labeled peptide substrate (Dye-QQQEVYGMMPRDpSA) was added at a concentration of 10 μM final in a reaction buffer of 25 mM Tris, pH 7.2, 150 mM NaCl. The dye-labeled peptide was prepared in 25 mM Tris, pH 7.2, 150 mM NaCl, 10 mM CaCl2 and the Calpain I was prepared in 25 mM Tris, pH 7.2, 150 mM NaCl. Reactions were initiated by the addition of Calpain I. The reaction was incubated for 1 hour at room temperature in black Greiner 96 microwell plates, then stopped by the addition of 0.25 × IQ® WS. The linear range of Calpain I activity was 0.8–13.2 U/mL.
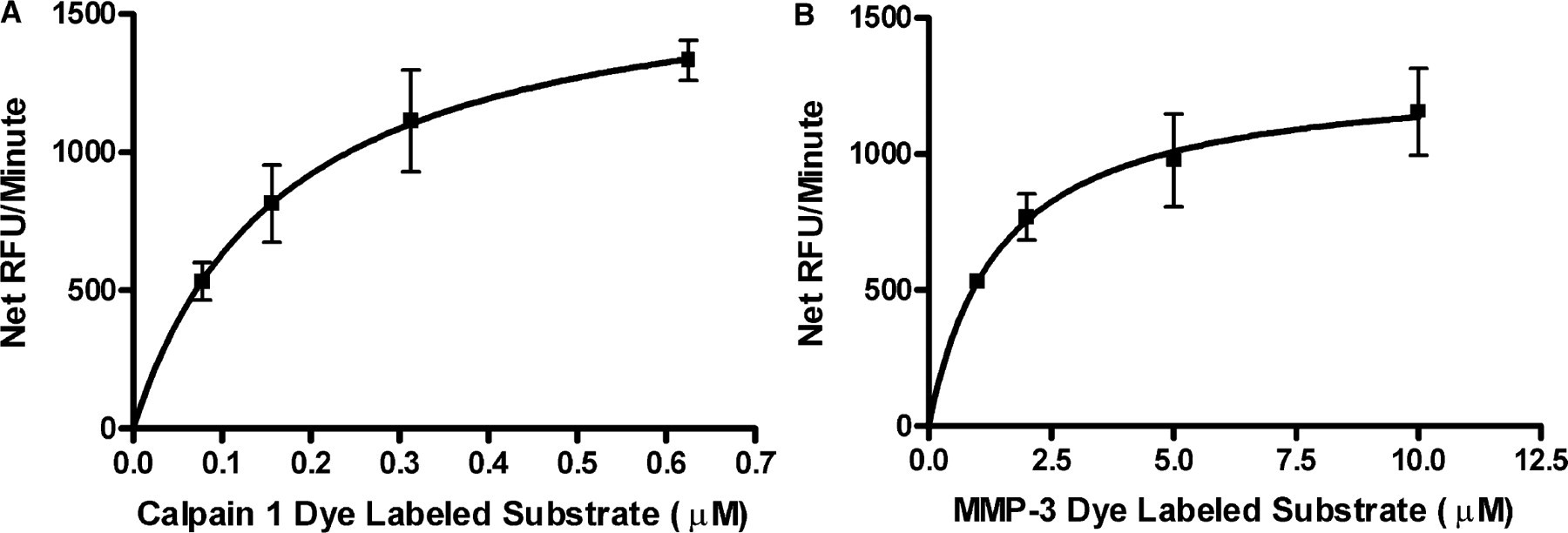
(A) Calpain 1 substrate Km determination using the IQ® Protease Assay. Calpain 1 dye-labeled peptide (Dye-QQQEVYGMMPRDpSA) at 0.078, 0.15, 0.31, and 0.62 μM were digested with 13.15 U/mL Calpain 1. The dye-labeled peptide was prepared in 25 mM Tris, 150 mM NaCl pH 7.5, and 10 mM CaCl2, and the Calpain 1 was prepared in 25 mM Tris, 150 mM pH 7.5. Reactions were initiated by the addition of Calpain 1 and stopped after 5 minutes at room temperature with 0.25 × IQ® WS. Km was 0.17 ± 0.01 μM and N = 3. (B) MMP-3 substrate Km determination using the IQ® Protease Assay. MMP-3 dye-labeled peptide substrate (Dye-RPKPVE-Norvaline-WRKpSA) at 1, 2, 5, and 10 μM were digested with 0.88 mU/mL of MMP 3 in a reaction buffer of 100 mM MES, pH 6.0, 10 mM CaCl2, 150 mM NaCl, 10 μM ZnCl2, 0.05% Brij-35. The reactions were stopped after 5 minutes at room temperature with 0.25x IQ® Working Solution. Km was 1.44 ± 0.15 μM and N = 3.
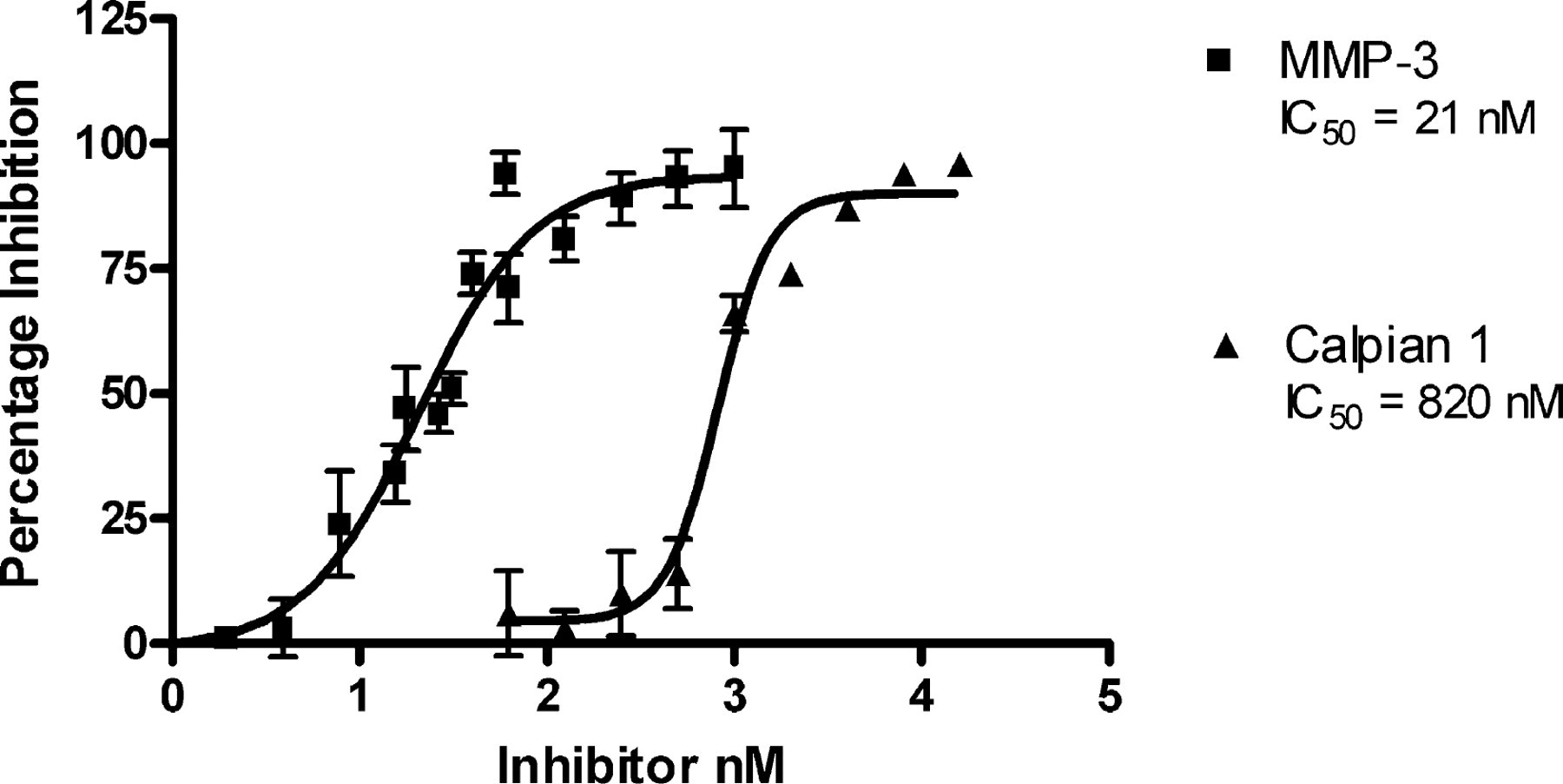
Dose-dependent inhibition of proteases using the IQ® Platform. The IC50 value of MMP-3 Inhibitor II (N-isobutyl-N-(4-methoxyphenylsulfonyl)-glycylhydroxamic acid) or Calpain 1 Inhibitor 3 (Carbobenzoxy-valyl-phenylalanial) were determined using the substrates described in Figure 4. Reactions were incubated in the presence and absence of Inhibitor for 1 hour followed by addition of 0.25 × IQ® Working Solution. Fluorescence was measured with a Tecan Safire, and data were analyzed by nonlinear regression. The resulting IC50 values correlate well with published values.
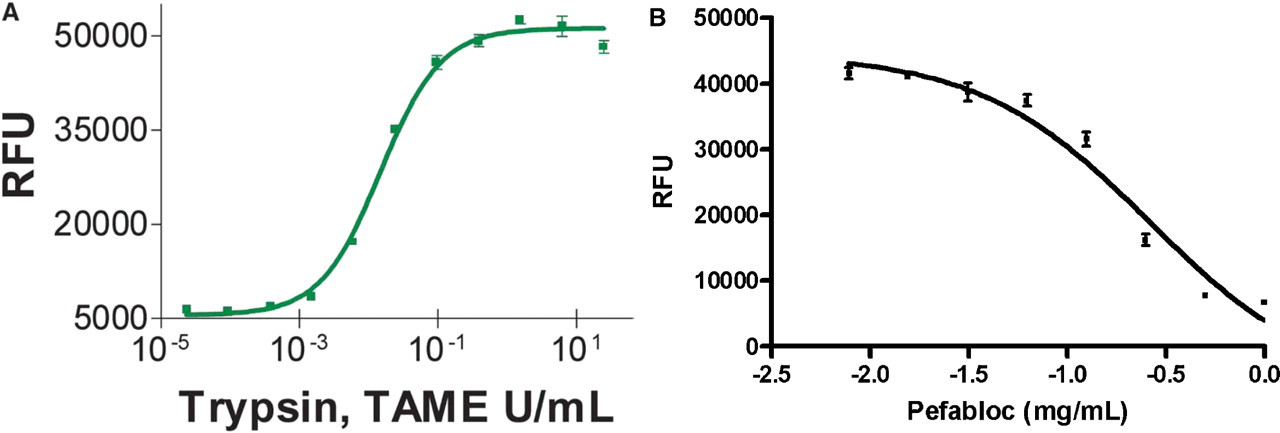
(A) Trypsin titration using the IQ® Protease Assay. Trypsin was serially diluted four-fold over 11 wells in triplicate. Dye-labeled peptide substrate (Dye-KRWVEVPKPRpSA-OH) was added at a concentration of 20 μM final in a reaction buffer of 25 mM Tris, pH 7.2, and 150 mM NaCl and a final volume of 30 μL. The mixture was incubated for an hour at room temperature in black Dynex 96 microwell plates then stopped by the addition of the 120 μL of a 0.25 × IQ® WS. (B) Dose-dependent inhibition of trypsin using the IQ® Assay. The IC50 value of Pefabloc (Roche) was determined using the substrate described in (A). TPCK Trypsin (33 U/mL) reactions were incubated in the presence and absence of Pefabloc (1.0–1000 μg/mL) for 1 hour followed by addition of 0.25 × IQ® WS. Fluorescence was measured with a Tecan Safire and data were analyzed by nonlinear regression. The resulting IC50 value of 260 μg/mL correlates well with published values.
CONCLUSIONS
The IQ® Homogeneous Assay Platform is a novel method for HTS of kinase, phosphatase, and protease activity. Key benefits of this technology include the ability to utilize a common reagent for the direct detection of enzymatic end-product without the need for an antibody or radioisotope, wide flexibility in substrate (and ATP) concentration and low enzyme requirements. For IQ® Protease Assays, a primary benefit (Figure 8) compared to typical FRET-based protease assays is the additional distance that can be utilized between the fluorophore and the quencher in the IQ® Assay. The IQ® Platform is versatile, flexible, and robust. Assays are readily scaled from 96- to 384- to 1536-well plate formats. A large number of organics, detergents, and other reagents that might be used in an enzyme reaction have been tested and shown not to interfere in the detection of activity. The technology provides Z' factors greater than 0.7 at low levels of substrate conversion. Custom peptide synthesis is available to enable analysis of any kinase, phosphatase or protease target.
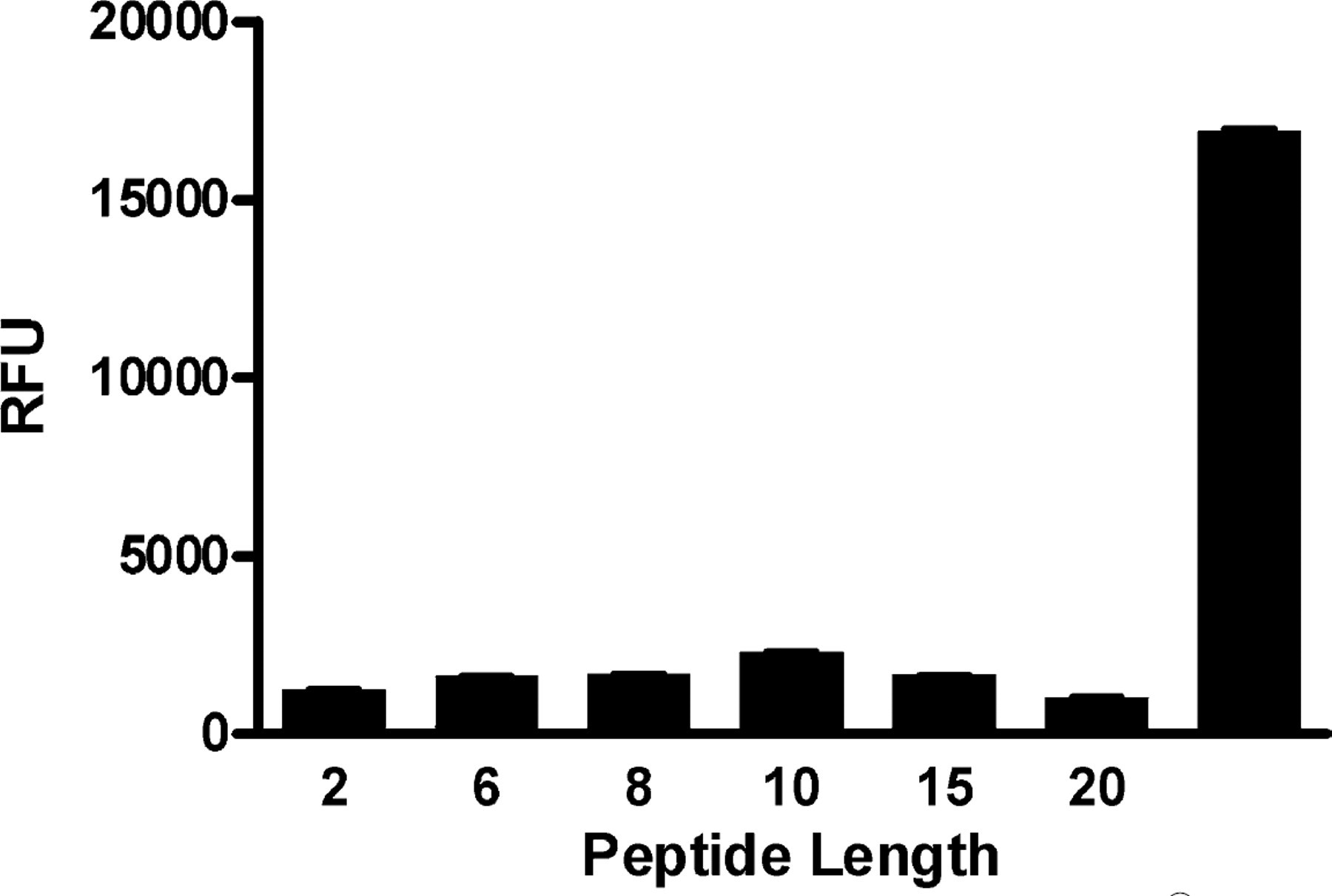
Distance requirement for quenching in the IQ® Assay. Six derivatives of a peptide were synthesized and dye-labeled at various amino acid distances between the dye and the phosphorylated residue. Maximum quench distance was determined by incubating 30 μM of each derivative with 0.25 × IQ® WS. Signal was generated for each phosphopeptide derivative post-IQ® Reagent addition. The maximum distance between the phosphoryl group and the dye tested was 20 amino acids with no difference in signal. Longer peptides may also work. The distances that work with the IQ® Assay are longer than typical FRET-based quench molecules, making this ideal to utilize more physiologically relevant substrates.
