Abstract
We describe a reagent system and robotic protocol for the isolation of highly purified plasmid DNA from cultured cells. The method is based on the Wizard® MagneSil™ Plasmid Purification System, which purifies sequencing-grade plasmid DNA. Two modifications to the sequencing-grade system were made to create the Wizard MagneSil Tfx™ System. This system provides lower endotoxin and other contaminant levels, giving higher quality plasmid for transfection applications. The Wizard MagneSil Tfx™ System uses MagneSil™ Paramagnetic Particles (PMPs) to clear lysate and bind plasmid, eliminating the need for filtration devices. The endotoxin removal step uses MagneSil™ (PMPs) and a guanidine/isopropanol wash to remove RNA and protein. One 96-well plate may be processed in 45 minutes on the Beckman Biomek® FX robotic workstation. We provide data showing DNA yield, contaminant levels, and transfection efficiency for 5 commonly used cell lines. Comparisons with other systems are also shown.
Introduction
High quality nucleic acid preparation is critical for optimal gene transfer and expression studies. The plasmid DNA should be supercoiled and contain minimal contaminants. Contaminants such as endotoxin carried-over from bacterial lysates, can adversely affect transfection of many cell lines. In addition to the necessity for high quality plasmid DNA, the trend towards miniaturization of transfection protocols is coupled with the need for purification of plasmid DNA in a high-throughput format. Efficient processing of large numbers of DNA samples requires reliable reproducible systems that generate high yields, require minimal human intervention, and give fast results at low cost.
To address the dual criteria of high quality plasmid DNA purification and high throughput formats, Promega developed the Wizard MagneSil Tfx™ System for the purification of transfection-grade plasmid DNA. In this system, MagneSil™ (PMPs) technology 1 is central to the purification of plasmid DNA from bacterial cultures. These silica-based particles (PMPs) are provided in different formulations to first clear bacterial lysates (MagneSil™ BLUE) and then to bind nucleic acids (MagneSil™ RED). Because of their magnetic properties, the particles can readily be separated from initial reaction and wash mixes with specialized magnetic devices, such as the MagnaBot® 96 Magnetic Separation Device, allowing walk-away automation of the procedure. The system builds upon our Wizard® MagneSil™ Plasmid Purification System, but differs in two important aspects. It incorporates a novel magnetic Endotoxin Removal Resin, used after the lysate clearing step, and an isopropanol/guanidine wash step that follows capture of plasmid DNA on the MagneSil™ RED particles. The wash reduces other contaminants such as protein and residual non-plasmid nucleic acids. Addition of these components and steps to the protocol significantly increase plasmid DNA purity and transfection performance with no adverse effects on yield.
The Wizard MagneSil Tfx™ System was tested in combination with 96-well formats. Typically, bacterial cultures containing a high copy number plasmid were grown in flasks overnight; a known amount of cell mass was transferred to the wells of a 2ml 96-well plate, and pelleted to provide starting material for the automated protocol. Consistent, high plasmid yields were obtained from run-to-run. Endotoxin, protein, and RNA levels were lowered as a direct result of the Endotoxin Removal Resin and isopropanol/guanidine wash steps. Transfection performance was tested in commonly used mammalian cell lines using plasmid DNA purified by various methods, including other commercial silica membrane-based and ion-exchange systems.
Materials and Methods
E. coli JM109 cells were transformed with the pGL3 Control Vector (Promega Cat.# E1741). Five milliliters of a seed culture was transferred to 2 liter flasks containing 500ml Terrific Broth plus 100μg/ml ampicillin. Cultures were grown at 37°C with shaking at 275rpm overnight. After determining the O.D.600 of the culture, equivalent amounts of cells (usually 1ml of 4 O.D./ml cells) were aliquoted into each well of a 2ml, 96-well plate. Plates were centrifuged in a tabletop centrifuge at 3000rpm (1500 × g) for ten minutes to pellet cells, and media was decanted. Reagents used in the purification were from the Wizard Magnesil Tfx™ System (Promega Cat.# A2380 & A2381). Purifications were done on a Biomek™ FX (Beckman Coulter, Inc., Fullerton, CA) equipped with a single multichannel pod and 96-well pipetting head. After purification, wells were analyzed for plasmid yield using gel electrophoresis measured against pGL3 Control plasmid standards using densitometry analysis on an Alpha Innotech Digital Imaging System (San Leadro, CA). Protein levels were determined using Coomassie Plus Protein Assay Reagent (Pierce Cat.# 23236 & 23238). All RNA levels were determined by alkaline hydrolysis, followed by HPLC separation from other materials that absorb at 260nm. Quantitation of RNA was based on peak area, minus a blank that accounted for any material other than RNA that co-eluted with RNA and absorbed at 260nm. All endotoxin levels were determined using Pyrochrome® Limulus Amebocyte Lysate kits (Associates of Cape Cod Cat.# C0180). Plasmids preparations purified using Qiagen Turbo (Qiagen Cat.# 27191) and QIAwell 96 Ultra (Qiagen Cat.# 16191) Plasmid Kits were performed manually, as directed.
For transfection, purified plasmid was transfected into five cultured mammalian cell lines (NIH 3T3, CV-1, HeLa, 293, and Chinese Hamster Ovary) using TransFast™ Transfection Reagent (Promega Cat.# E2431) at a reagent to DNA charge ratio of 1:1. The cells were lysed and assayed for presence of Renilla Luciferase activity after 2 days growth using Renilla Luciferase Assay System (Promega Cat.# E2810).
ROBOTIC WORKSTATION SETUP
This method was developed on a Biomek® FX workstation fitted with a single multichannel pod and 96-channel disposable tip pipetting head. The deck setup for single 96-well plasmid purification used the following:
One Tip Loader ALP (Beckman Coulter Cat.# 719856) A 96-well Tipwash ALP (Beckman Coulter Cat.# 719804) Three single position labware ALP (Beckman Coulter Cat.# 719360) One 16 position labware ALP (Beckman Coulter Cat.# 719360) One heating/cooling ALP (Beckman Coulter Cat.# 719361) A Linear Shaker ALP (Beckman Coulter Cat.# 379410) A Device Controller Unit (Beckman Coulter Cat.# 719366) Three P250 Tip Rack Assembly (Beckman Coulter Cat.# 717251)
The method is available for download at: www.promega.com/automethods
Additional required equipment included the following:
A circulatory water bath capable of 65°C heating (for heating/cooling ALP) One Promega MagnaBot® 96 Magnetic Separation Device (Promega Cat.# V8151)
One Promega Heat Transfer Block (Cat.# Z3271) Labware used for this method development is listed below*:
Greiner 96-well U-bottom microplates (Cat.# 650101) Innovative Microplate reagent reservoirs (Cat.# S30014) Beckman Coulter square well titer plate (Cat.# 140504)
GENERAL PROCEDURAL STEPS OF THE PURIFICATION METHOD
The following steps detail the addition of reagents and sample handling:
90μl of Cell Resuspension Buffer is added to a 2ml, 96-well plate containing pelleted E. coli with plasmid. Pellets are suspended with a mixture of pipetting and shaking. 120μl of Cell Lysis Buffer is added to each suspension. Cells are lysed by shaking. 120μl of Cell Neutralization Solution is added to the lysate. The lysate is mixed by shaking. MagneSil™ BLUE resin is resuspended by pipetting, and 25μl is added to the lysate. The resin is mixed with the lysate by shaking, followed by pipetting. The resin/lysate mixture is transferred to a 96-well plate on a MagnaBot® 96 Magnetic Separation Device, and the resin is allowed to migrate to the magnet. The resulting cleared lysate is transferred to a 96-well plate containing Endotoxin Removal Resin. The Endotoxin Removal Resin and cleared lysate are mixed by pipetting. The plate is then transferred to the MagnaBot® device, and the resin is allowed to migrate to the magnet. The cleared lysate is transferred to a 96-well plate containing MagneSil™ RED resin. The Lysate and MagneSil™ RED are mixed by pipetting, allowing the plasmid to bind to the resin. The mixture is then transferred to the MagnaBot® 96, and the resin is allowed to migrate to the magnet. The lysate is discarded, and the plate containing the MagneSil™ RED pellet is removed from the MagnaBot® 96. 100μl of 4M guanidine hydrochloride/40%isopropanol (4/40 wash solution) is added to the MagneSil™ RED pellet, which is resuspended by shaking. The mixture is then transferred to the MagnaBot® 96, and the resin is allowed to migrate to the magnet. The 4/40 wash solution is discarded, and the plate is removed from the MagnaBot® 96. 100ul of 80% ethanol is added to the MagneSil™ RED pellet, which is resuspended by shaking. The mixture is then transferred to the MagnaBot® 96, and the resin is allowed to migrate to the magnet. The ethanol solution is discarded, and the plate is removed from the MagnaBot® magnetic separation device. Steps 11 and 12 are repeated twice, for a total of 3 ethanol washes. The MagneSil™ RED pellet is dried at 55–60°C for 7.5 minutes. 100μl of nuclease-free water is added to the MagneSil™ RED pellet, which is resuspended by pipetting. The mixture is then transferred to the MagnaBot® 96, and the resin is allowed to migrate to the magnet. The solution containing the plasmid is removed from the pellet and transferred to a clean 96-well plate.
Results
PLASMID YIELD
The ability to reproducibly recover plasmid over the entire plate during the purification is critical. The 96-well head of the Beckman FX minimizes interplate differences by adding solutions to the entire plate simultaneously. As shown in Figure 1, examination of yields by gel electrophoresis and densitometry give well-to-well variations of less than 20%. Run to run variations in yield, using identical amounts of cells, are also minor (Figure 2).
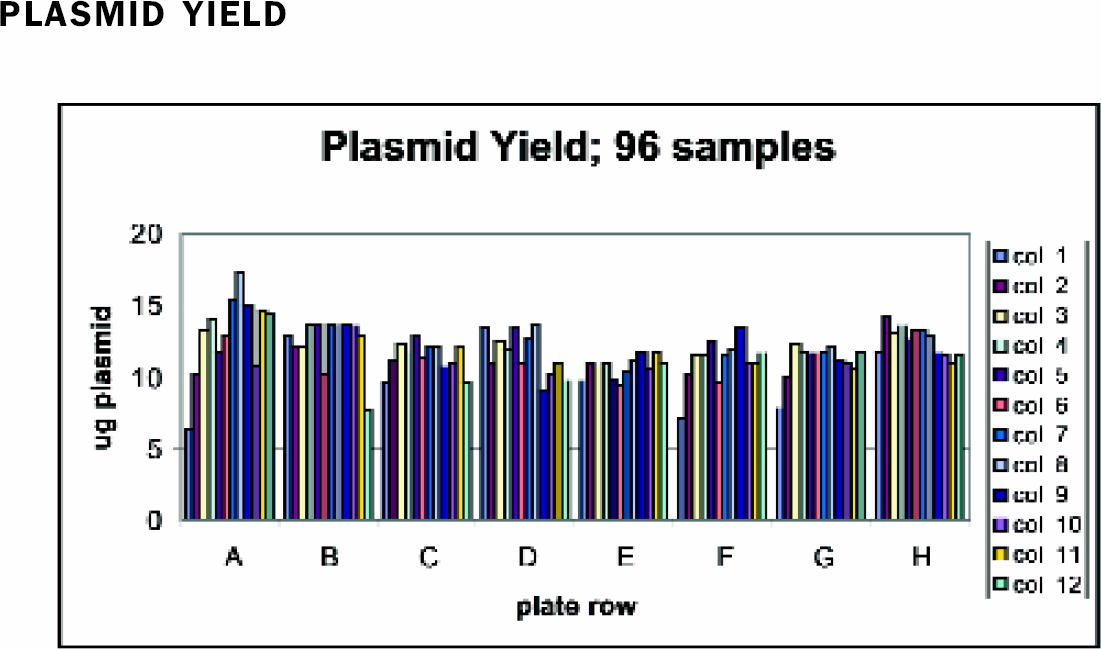
Yield: 4 O.D. of JM109 cells containing pGL3 Control Vector were purified in a 96-well plate using Wizard® MagneSil Tfx™ as described. Samples were analyzed by gel electrophoresis and densitometry for plasmid yield using pGL3 Control Vector standards. The average value of all 96 samples is 11.8 +/-1.7 ug.
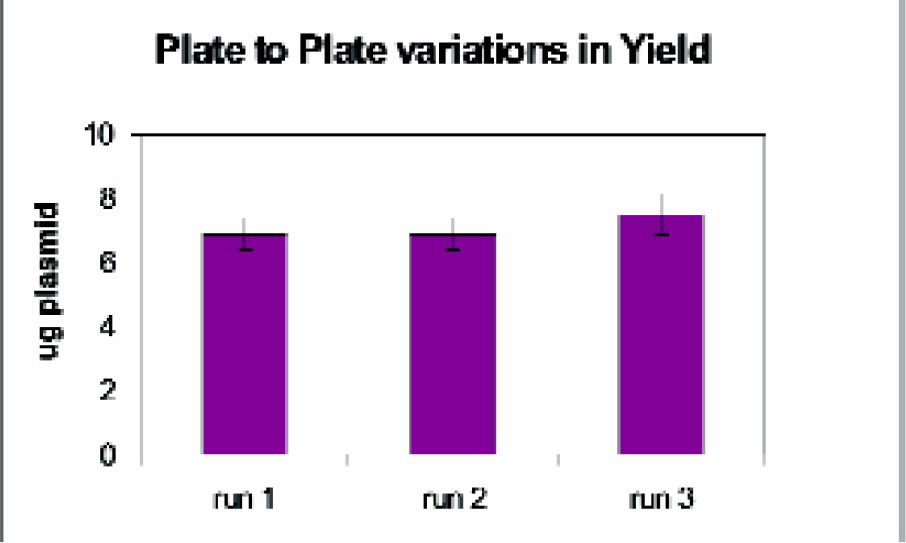
Three plates containing identical amounts of JM109 cells with pGL3 plasmid were processed sequentially using Wizard® MagneSil Tfx™ as described. Eight samples from across each plate were analyzed by gel electrophoresis and densitometry for plasmid yield using pGL3 Control Vector standards.
Use of a high copy plasmid such as pGL3 usually yielded more than 10ug of plasmid in a preparation. Since transfections usually require less than 1 ug of plasmid, yields are sufficient to do several transfections, even with well-to-well variations in yield. The amount of MagneSil used in the protocol (5mg) is sufficient to bind over 30ug of plasmid. 2
PLASMID YIELD
REMOVAL OF CONTAMINANTS
Contaminants present in plasmid preparations can have a negative impact on transfection. Endotoxin especially has been implicated in low transfection efficiency, 3 and while it has been reported that many cell lines can transfect in the presence of 2000 EU/ug, 4 in our experience, lower levels of endotoxin can have a negative effect. Other cell lines, such as Huh7, show greater sensitivity. Sequencing-grade plasmid preparations can tolerate much higher levels of endotoxin and remain viable. As shown in Figure 3, the addition of an endotoxin removal step using a paramagnetic particle lowered endotoxin levels by up to 90%. An additional wash step using guanidine HCl and isopropanol lowered RNA, protein, and endotoxin levels.
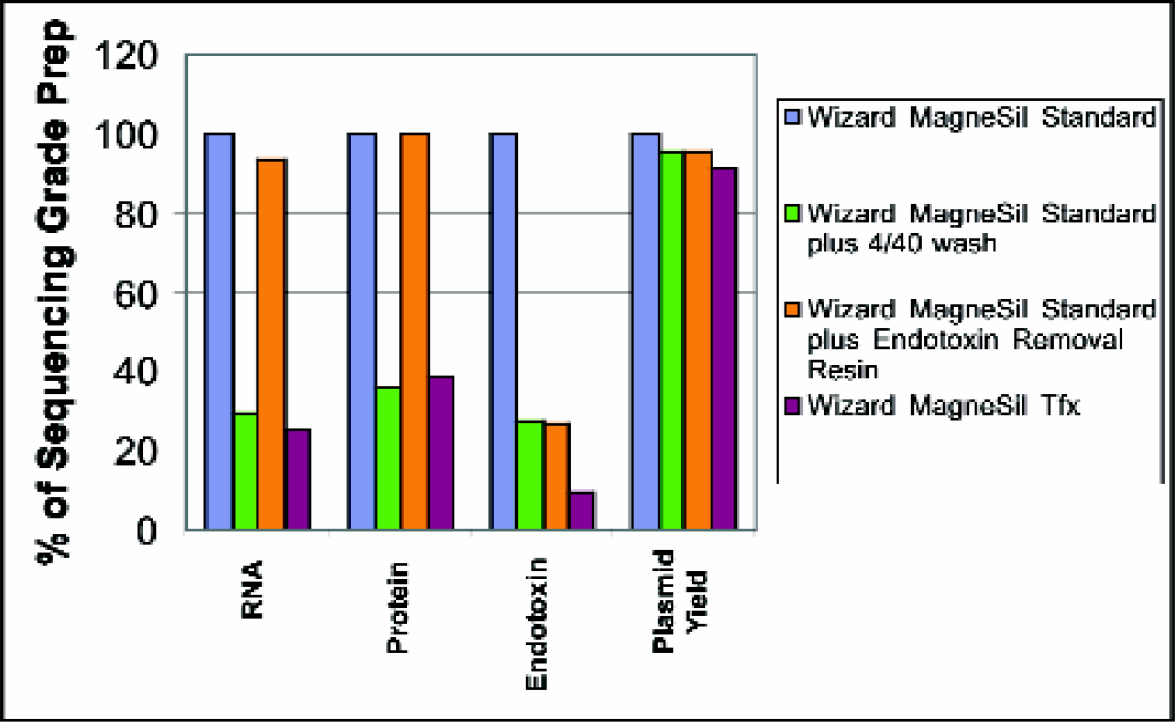
Effectiveness of Modifications of Wizard® MagneSil™ Plasmid Purification Protocol on Removal of Contaminants: Four preparations of plasmid were isolated from the same batch of bacterial cells. Wizard® MagneSil Standard (sequencing grade), Wizard® MagneSil™ Standard with a 4/40 wash; Wizard® MagneSil™ Standard with an Endotoxin Removal Resin step; and Wizard® MagneSil Tfx™ System were purified using a Beckman FX. Samples from each preparation were tested for contaminants and plasmid yield as described.
Combining the two modifications results in far lower concentrations of endotoxin, RNA, and protein, as illustrated in Figures 3 and 4. Levels of contaminants in the modified MagneSil™ Tfx method were greatly reduced without a loss of yield. The level of impurities seen in the Tfx™ preparations is closer to the levels seen in anion exchange methods such as Qiagen Ultra.
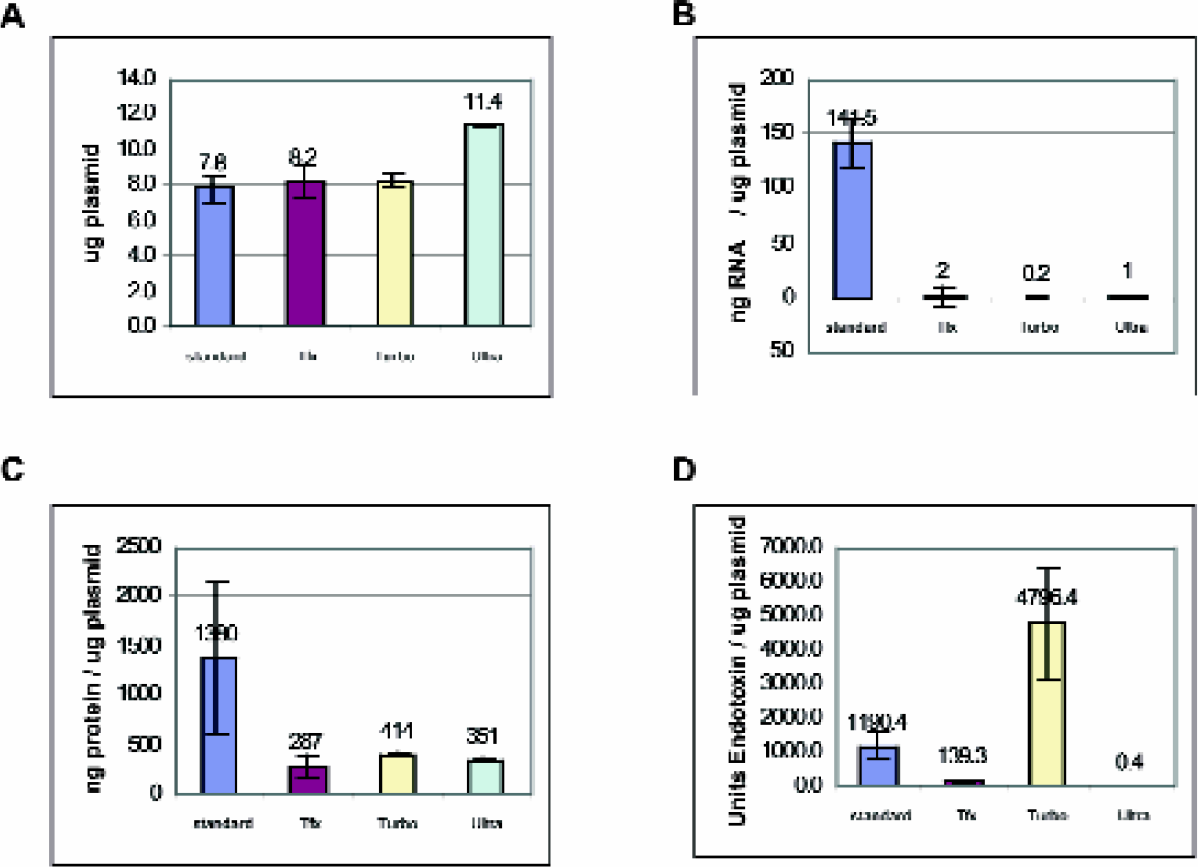
Four 96-well plates, each well containing identical cultures of JM109/pGL3 Control Vector, were processed in one of four ways: Wizard® MagneSil™ Plasmid Purification System (standard), Wizard® MagneSil Tfx™ System (Tfx™), QIAGEN Turbo, or QIAGEN Ultra Plasmid Kits. Eight Samples from each 96-well prep were analyzed for yield and contaminants as described; the numbers represent average values +/- standard deviation.
TRANSFECTION
While there are literally hundreds of mammalian cell lines into which DNA can be transfected, most researchers confine themselves to a select few. We studied the transfection efficiency of DNA purified by various methods into five common cell lines.
The results shown in Figure 5 indicate that the changes made to the Wizard® MagneSil™ Plasmid Purification protocol improves the transfections of these cell lines. The large error bars for the standard protocol are indicative of the unreliability of transfecting samples containing large amounts of endotoxin. By removing the contaminants with the Wizard® MagneSil Tfx™ System, the reliability of assay-to-assay transfection is improved.
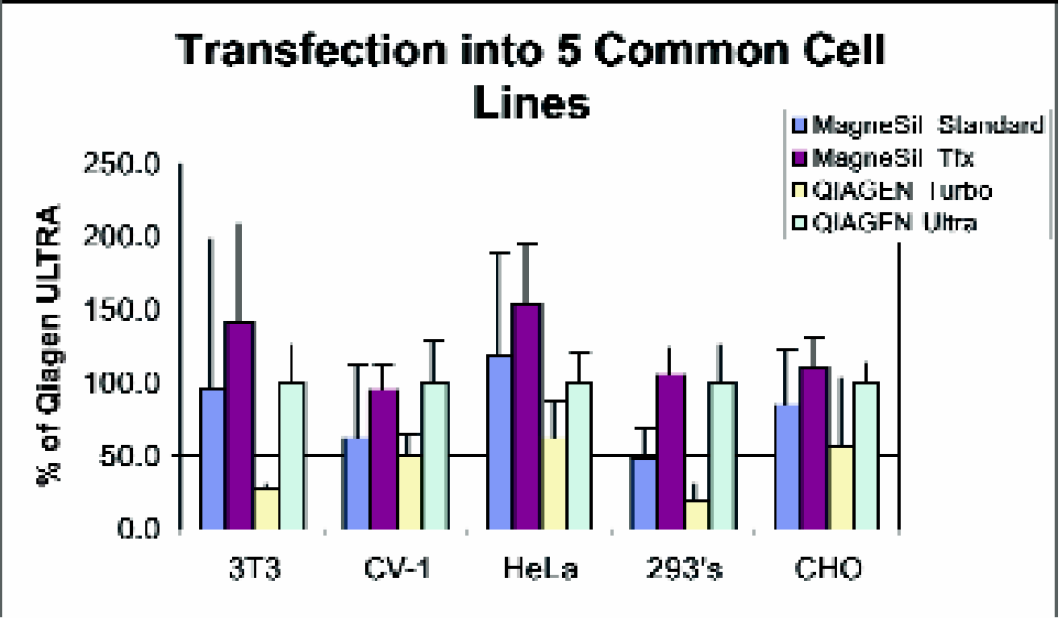
Five common cultured mammalian cell lines were transfected with pGL3 Control Vector purified using Wizard® MagneSil™ (Standard) Plasmid Purification System (silica) Wizard® MagneSil Tfx™ System (silica), QIAGEN Turbo (silica), and QIAGEN Ultra (anion exchange). MagneSil™ samples were taken from three separate preparations; for Qiagen samples, one preparation was done. Plasmid DNAs were transferred into cells using the Transfast Transfection Reagent under standard conditions. Cell lysates were harvested 2 days post-transfection and assayed for luciferase activity. Relative transfection efficiency was assessed by light units per standard lysate volume measured and normalized for QIAGEN Ultra.
The data also shows that plasmid purified using the Wizard® MagneSil Tfx™ System transfects the five cell lines shown as well or better than the anion-exchange method used by QIAGEN Ultra. This is a dramatic improvement over standard silica based systems, which typically transfect poorly.
Conclusions
We have enhanced the Wizard® MagneSil™ Plasmid Purification System to enable production of transfection grade plasmid in high throughput formats. The Wizard MagneSil Tfx™ System protocol uses MagneSil™ Paramagnetic Resins to clear bacterial lysates and bind plasmid DNA. Endotoxin Removal Resin acts to remove endotoxin lipopolysaccharide, which has been shown to have a detrimental effect on transfection. A 4M Guanidine HCl/40% isopropanol wash reduces levels of endotoxin lipopolysaccharide, RNA, and protein. The resulting plasmid DNA transfects mammalian cell lines more efficiently than traditional silica-based systems and efficiencies equal to or better than plasmid purified by an anion exchange method such as provided in the QIAGEN Ultra Plasmid Kit.
The method developed for the Beckman® FX robotic workstation requires 45 minutes to process one 96-well plate. In addition, we have adapted the protocol to work on the Beckman Biomek® 2000. The Biomek® 2000 requires 150 minutes to process one plate; however, the isolated DNA transfects at the same high efficiency as plasmid purified on the Biomek® FX. The protocol is adaptable to other robotic workstations, both in single and multiplate formats.
All trademarks are property of their respective owners.
