Abstract
Automated techniques and philosophies, which have become increasingly important in modern laboratories, are not traditionally covered in undergraduate chemistry curricula. To this end, we have been incorporating automated sample preparation methods using standard robotic workstations in our undergraduate analytical chemistry laboratory. Using a Benchmate™ II Workstation, an automated method has been developed and implemented for the solid phase extraction of capsaicins from commercial hot pepper sauces prior to liquid chromatographic analysis. This paper reports on pedagogical aspects of incorporating automation into the undergraduate curriculum as well as results obtained for manual and automated extractions conducted by students.
Keywords
Introduction
Today, virtually every modern laboratory utilizes some level of automation to perform routine tasks and procedures. These tasks cross many fields of biology and chemistry and can include sample analysis, sample preparation, electronic capture of data, and tracking of samples, just to name a few. Many future scientists (i.e., students) are not exposed to automation in their undergraduate education due to various circumstances. Establishing collaborations with industrial colleagues to incorporate automation in undergraduate laboratories, however, can help offset both the high cost of implementation and time constraints in the curriculum. The objective of this paper is to document one such experience in incorporating automation in our undergraduate analytical chemistry laboratory using a model experiment, and to discuss the advantages as well as challenges encountered during the process.
The process of identifying the appropriate experiment for this collaboration led us to consider some of the same questions that one asks for any automation project. First, what processes or procedures do the students/customers find most mundane and therefore least desirable to perform? Second, do any of these processes match with current automated solutions that are commercially available? And third, how do we implement such an experience without requiring additional time in our existing laboratory curriculum?
To address the first question, we considered the sample preparation process required for an existing experiment, based on the extraction of capsaicins from various commercial sources followed by quantitation by high performance liquid chromatography (HPLC). 1 – 3 A variation of this laboratory, specifically designed as an undergraduate analytical chemistry experience, is used in our curriculum to teach students about solid phase extraction (SPE) and liquid chromatography. Two weeks of the semester (one three-hour period each week) are allocated for this experiment. The sample preparation portion of the laboratory, traditionally requiring most of the first laboratory period to complete, involves the extraction of capsaicins from commercial hot pepper sauces followed by SPE cleanup, where the extracts are manually driven through SPE cartridges with syringe plungers. The second laboratory period is devoted to calibration and analysis of extracts by HPLC and data analysis. The down time associated with the manual SPE cleanup for young scientists can quickly diminish their interest in the exercise if they feel as though they are completing tasks requiring little thought. Because this is a favorite laboratory experiment in our analytical course, it is important for us to continue the general theme of the experiment without sacrificing the excitement of the students.
Our response to the second question was yes; there is commercially available equipment capable of completing all the operations required for the sample preparation portion of the experiment. The Zymark Benchmate™ II workstation is equipped with a solid phase extraction station, a balance, and basic liquid handling functions such as dilution, liquid transfers, vortexing, and filtration. These capabilities, in conjunction with the fact that many of these units are currently being replaced by the next generation model (and subsequently available for donation by industrial users), rendered this particular robotic workstation an excellent fit for the project described here.
The final, more complex question of practical development and implementation, and how to expose undergraduates to automated techniques without requiring a substantial amount of time in the existing lab course, was addressed as follows. First, upperclassmen interested in learning about automation helped to develop the sample preparation method as an independent study/research project separate from the intended analytical course. These students were directly involved in the collaboration, troubleshooting, design, and optimization of the automated sample preparation method. Once developed, the new procedure was integrated into the existing undergraduate laboratory course. We were able to successfully accomplish all of these steps and implement the experiment with advantages and disadvantages as we discuss here.
In addition to the obvious advantages of exposing students to new technologies, we realized other benefits as we proceeded with the project. One important aspect that was considered in this experience was safety issues associated with students working with hazardous substances. In the case of toxic capsaicins, automation minimizes handling of such materials. Another practical advantage is that although we were concerned with using valuable time in the laboratory curriculum, in fact, we were able to accomplish more in terms of content in the same (or even less) in-lab time. With the new automated method students now have the opportunity to explore more variables for optimization, because rather than spending time preparing samples, instead, they are designing experiments and writing associated method programs for the workstation to be executed while, and well after, they leave the laboratory. It should be noted, however, that it is easier for students to lose the fundamental understanding of the experiment if the instrument is treated as a black box where a button is pressed, and a sample is generated. Careful designing of experiments, as well as having the students involved in writing method programs, helps alleviate this drawback. In addition, students in this experiment watch one entire cycle of the robot to ensure consistency with their written program.
The overall goal of this work is not to have students determine capsaicin concentrations in hot pepper sauce, nor is it to train undergraduates on the specific robotic workstation employed in the project. Instead, the motivation is to use these tools to expose students to realistic automated techniques that may be encountered in their future scientific careers. This goal is achieved in two phases. First, junior and senior level independent study/research students are directly involved in developing and writing the automated method. Then, the new automated method is implemented in the laboratory portion of a curriculum course (in this case, instrumental analysis).
Materials and Methods
Reagents
Distilled, deionized water and HPLC grade methanol and acetonitrile were used without further purification. A mixture of natural capsaicins (65 % capsaicin, 35 % dihydrocapsaicin) was purchased from Aldrich and used to make standards ranging from 1–20 ppm total capsaicins in HPLC mobile phase (75 % methanol, 25 % water). 3-mL, 500-mg BAKERBOND spe™ Octadecyl disposable extraction columns were purchased from VWR (Cat. No. JT7020–3) and Tabasco® brand pepper sauce (McIlhenny Co., Avery Island, LA) was used as purchased from a local grocer.
MANUAL SAMPLE PREPARATION
Students performed manual methods for the solid phase extraction of capsaicin and dihydrocapsaicin from pepper sauces similar to those described previously. 1,4 First, 10 mL of acetonitrile was added to a 2–3g sauce sample and mixed well. Then, 1 mL of this acetonitrile extract was diluted with 9 mL water. After conditioning an SPE cartridge with 5 mL acetonitrile followed by 5 mL water, the above solution was passed through the cartridge, disposing of the aqueous phase. The capsaicins were eluted with 4 mL acetonitrile, and then with 1 mL of 1 % acetic acid in acetonitrile. High performance liquid chromatography was used for analysis of this combined extract.
AUTOMATED EXTRACTIONS
A Zymark (Hopkinton, MA) Benchmate™ II Workstation (shown in Figure 1) with DOS-based software version 3.0 was employed for the automated sample preparation methods. The manual SPE procedure was modified in order to conduct the extraction using the robotic workstation. Undergraduate students, as independent study projects, developed the robotic method used for this experiment, realizing that rather than simply writing the program just as the manual method was carried out, several aspects had to be addressed based on the limitations of the workstation. For example, volume limitations of the test tubes required that the method be scaled down, which although trivial to experienced scientists, was an excellent exercise for beginning researchers. Another change in the method was mandated when backpressure limitations in the system caused a leak during the SPE step. Therefore, a filtering step was added prior to SPE to minimize particulate matter that would otherwise be passed through the cartridge. In addition, the order of individual steps, vortex time and speed, and solvent flow rates, as well as other set up parameters, were all realized as important optimization variables in the sample preparation phase of the experiment. The method program, which was performed on ∼1 gram of sauce (weighed accurately and loaded onto the sample tray), for the laboratory experiment reported here is shown below:

The total time for completion of this method program is 37.4 minutes.

Zymark Benchmate™ II Workstation used for acetonitrile extraction, filtration and SPE cleanup of samples prior to liquid chromatographic analysis.
CHROMATOGRAPHIC ANALYSIS
All standards and capsaicin extracts were analyzed by high performance liquid chromatography during the second three-hour laboratory period. Capsaicin and dihydrocapsaicin were quantified by reversed-phase HPLC using a Kratos Spectroflow 400 dual piston solvent delivery system equipped with a 20-μL injection loop and a Kratos SF 769Z variable wavelength UV/Vis absorption detector set at 205 nm. A ZORBAX 4.6 × 150 mm column with 5 mm C-18 particles was used (Agilent Technologies, Palo Alto, CA). The mobile phase was 75:25 methanol:water at a flow rate of 1.0 mL/min, and data was collected using a custom program written in LabVIEW® software version 4.0 (National Instruments, Austin, TX). Extracts (as prepared above) were injected and chromatographic peak heights were compared to calibration curves generated from a series of capsaicin standards.
Results and Discussion
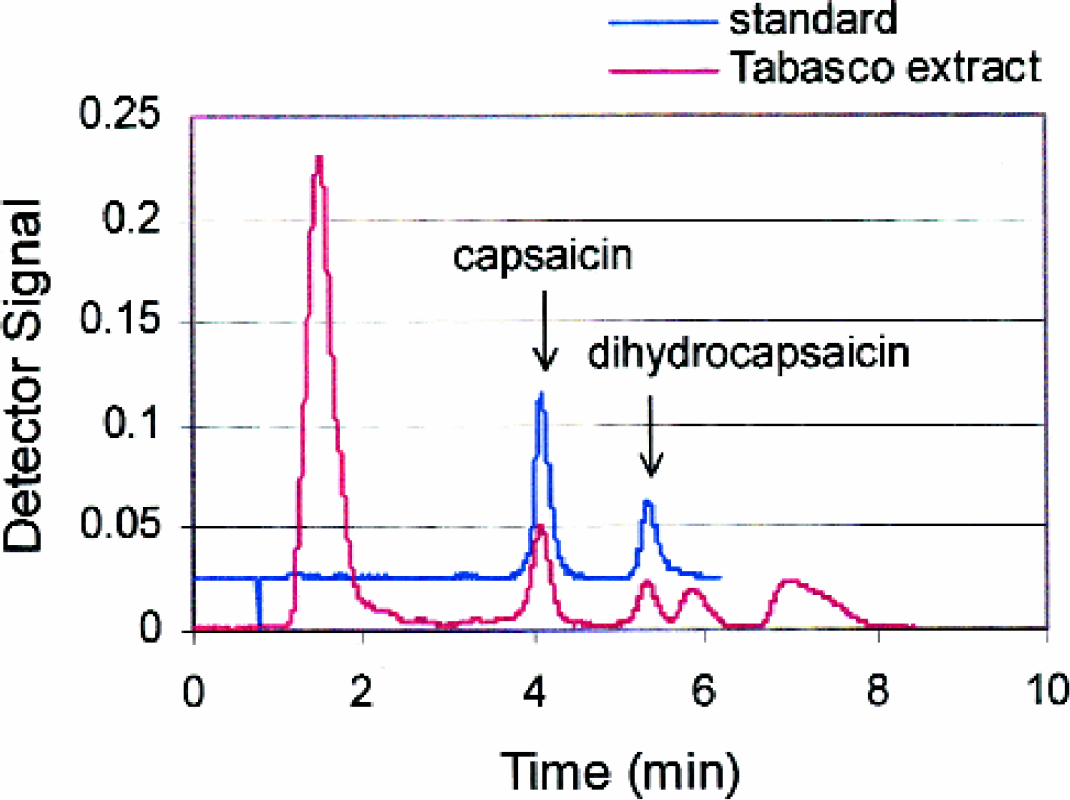
Although many hot sauces have been used in this work, results reported here are for Tabasco® brand pepper sauce due to its availability and ability to achieve a homogeneous solution compared to other oil-based sauces that have a tendency to separate into distinct phases. Figure 2 represents example liquid chromatograms for a capsaicin standard and for a Tabasco extract prepared by the automated method. The baseline of the standard is offset for clarity. Capsaicin and dihydrocapsaicin were identified in the sauce extract by their chromatographic retention times of 4.1 and 5.3 min, respectively. Other peaks in the extract chromatogram are consistent with previous reports, such as the peak at 1.5 minutes being a polar constituent not removed by the SPE cleanup, 1 and the peaks at 5.8 and 7 min being other capsaicin compounds 4 that were not a focus in this work. Figure 3 presents student calibration curves generated from standards for capsaicin and dihydrocapsaicin used to quantitate the two compounds in the sauces. Plotting peak height as a function of capsaicin concentration yielded straight lines with acceptable correlation coefficients (r 2 = 0.9995 and 0.9989 for capsaicin and dihydrocapsaicin, respectively) for quantitation.
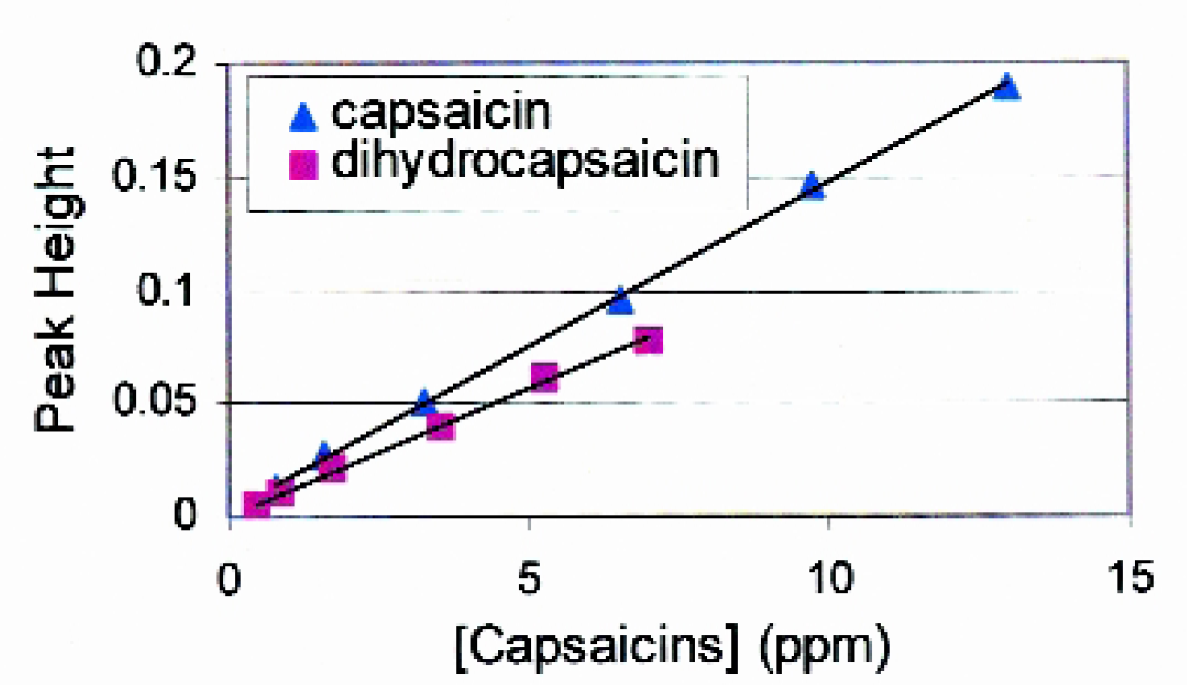
HPLC calibration plots generated from capsaicin-dihydrocapsaicin standards. The equations of the lines were y = 0.0145x + 0.0025 (r2 = 0.9995) and y = 0.0111x + 0.0010 (r2 = 0.9989) for capsaicin and dihydrocapsaicin, respectively.
Figure 4 presents typical data obtained by students for the concentrations of capsaicin and dihydrocapsaicin in Tabasco® sauce, comparing manual and automated sample preparation methods. The error bars on the plot represent ± the standard deviation of several independent extractions (n = 7 for the manual extractions and n = 6 for the automated extractions). The two methods yielded statistically similar results in terms of concentrations (107 ± 12 ppm capsaicin and 66 ± 12 ppm dihydrocapsaicin and 117 ± 8 ppm capsaicin and 73 ± 7 ppm dihydrocapsaicin for preparations done manually and by the Benchmate, respectively), but better reproducibility was achieved with the automated preparation method. The results obtained here are consistent with capsaicin concentrations reported (in terms of Scoville Heat Units) for Tabasco®. 5 It should be noted, however, that capsaicin concentrations in this, and other, pepper sauces can vary from bottle to bottle, so these reported results are from the same bottle.
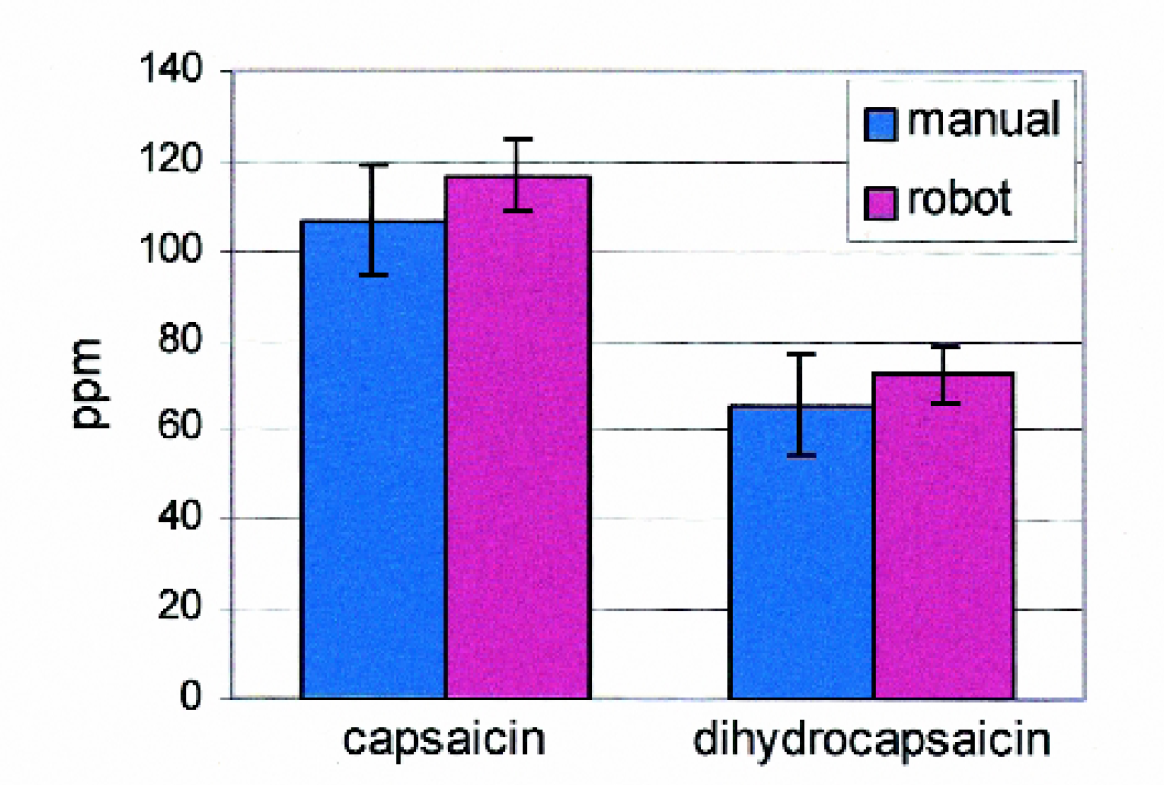
Comparison of capsaicin and dihydrocapsaicin concentrations and reproducibility for Tabasco® samples prepared by students manually (n = 7) and by the automated method (n = 6).
Overall, students enjoyed working with the robotic workstation rather than preparing the samples by hand and some students even proposed new ideas, not only on improving this particular method, but also on applications of the workstation in other areas of chemistry and biology.
Conclusion
An automated sample preparation method was developed by students for the extraction and SPE cleanup of capsaicins from commercial pepper sauce using a robotic workstation. In addition, automation was integrated into the undergraduate instrumental analysis laboratory by modifying an existing experiment without utilizing additional lab time, and students were eager to work with the workstation rather than preparing the samples by hand. The authors hope to increase awareness of the readers of this Journal about the important role of teaching institutions in implementing automation, and will also realize the pedagogical benefits of
industrial-academic collaborations and donations of equipment that might otherwise be discarded.
Acknowledgements
The authors thank Johnson & Johnson Pharmaceutical Research and Development for the donation of the robotic workstation, Zymark Corporation, and students in the Analytical Chemistry II (CHM 312) class of spring 2003.
