Abstract
Anticoagulant rodenticides are frequently a cause of poisoning of domestic animals, wildlife, and human beings. A toxicosis in 6,000 laying hens caused by the malicious addition of unknown amounts of coumatetralyl bait as well as the insecticides aldicarb, methomyl, and imidacloprid in the drinking water, was investigated in the current study. In order to determine a possible carryover of coumatetralyl into eggs, a rapid and reliable analytical method was developed and fully validated for the simultaneous detection of 6 anticoagulant rodenticides (warfarin, coumatetralyl, coumachlor, bromadiolone, difenacoum, and brodifacoum) in yolk and albumen using high-performance liquid chromatography (HPLC) with fluorescence detection. The method developed was reproducible, sensitive, accurate, and linear within the range of 0.01–1 mg/kg, which is the concentration range of bromadiolone and warfarin found in yolk in previously reported studies. The coefficient of variations of within and between days was 1.0–8.5% for yolk and 0.6–3.8% for albumen, while recoveries from spiked albumen and yolk samples were all in the range of 79–99% and 51–95%, respectively. Limits of detection in yolk were 0.01 mg/kg for warfarin and 0.003 mg/kg for the remaining compounds; in albumen, the limit of detection was 0.003 mg/kg for warfarin, coumatetralyl, and coumachlor, and 0.0015 mg/kg for difenacoum and brodifacoum. The application of the validated method revealed the presence of coumatetralyl in the yolk only at levels of 0.0057 mg/kg and 0.0052 mg/kg on the second and fourth day of the poisoning. In conclusion, the HPLC method demonstrated suitability for application in official analysis of anticoagulants in hen eggs.
Introduction
Anticoagulant rodenticides are widely used and are highly effective in controlling rodent populations (Jackson WB, Ashton AD: 1992, A review of available anticoagulants and their use in the United States. In: Proceedings of the 15th Vertebrate Pest Conference, pp. 156–167. University of California, Davis, CA). 26 Their fundamental mechanism of action is the inhibition of vitamin K epoxide reductase, thereby causing deterioration of blood clotting eventually leading to widespread hemorrhages and death. 21 Anticoagulant rodenticides are generally classified as first and second generation. The first-generation compounds (warfarin, coumatetralyl, chlorophacinone) display a moderate toxicity to rodents with a 50% lethal dose (LD50) of 10–50 mg/kg of body weight (BW) and need a repeated bait exposure in order to exert a lethal effect. 21 Moreover, due to their relatively short half-life in blood and liver, and a lower bioaccumulation potential, a widespread resistance has been developed to the first-generation anticoagulants. 21 Consequently, second-generation anticoagulant rodenticides (bromadiolone, difenacoum, brodifacoum, difethialone, flocoumafen) with increased toxicity (LD50 0.2–4 mg/kg BW) and higher bioaccumulation were developed.21,26 However, their long half-life and high persistence in animal tissues constitutes a major risk of secondary chronic intoxication of wildlife, as was demonstrated by numerous studies (Erickson W, Urban D: 2004, Potential risks of nine rodenticides to birds and nontarget mammals: a comparative approach. Washington DC, U.S. Environmental Protection Agency. Available at: http://pi.ace.orst.edu/search/getDocketDocument.s?document=EPA-HQ-OPP-2006-0955-0005).5,9,13,23
Ready-to-use cereal-based baits containing anticoagulant rodenticides are readily available over the counter and are often colored with blue, green, or red dyes. The use of bait may present a practical risk for public health, domestic animals, and the environment, due to accidental exposure of nontarget animals, passage into the food chain resulting in secondary toxicosis, and potential contamination of water sources.11,12,15 Poisoning by anticoagulant rodenticides in human beings, domestic and livestock animals, and wildlife has been extensively reported worldwide.2–4 Although fatal intoxications in poultry have been previously described, ingestion of anticoagulant baits rarely leads to clinical signs of intoxication because poultry are less susceptible to these compounds than mammals (Erickson W, Urban D: 2004, Potential risks of nine rodenticides).17,20 The available acute oral and dietary toxicity data for Leghorn chickens (layers of edible eggs) are very limited, and a precise acute LD50 value (942 mg/kg) was determined empirically only for warfarin, which is a rodenticide that is rarely used at present. 20 However, eggs laid by poultry unintentionally exposed to anticoagulant rodenticides potentially pose a consumer health risk due to carryover of the rodenticides into the eggs as was demonstrated for warfarin and bromadiolone in earlier studies.10,14,22 Such studies indicated that both anticoagulant rodenticides were partially excreted into the eggs and were still detectable in the yolk after 9–14 days.10,14,22 A consistent kinetic excretion pattern of warfarin and bromadiolone into eggs was observed, in which warfarin reached maximum concentration in the albumen 24 hr post–oral exposure, while in the yolk, warfarin and bromadiolone reached peak concentrations 5 days postexposure.10,14,22 Due to the high bioaccumulation properties of the more persistent second-generation anticoagulant rodenticides such as bromadiolone, difenacoum, and brodifacoum, chronic exposure to low levels of anticoagulants may lead to significant bioaccumulation in the liver over a longer period of time and eventual intoxication cannot be ruled out. 21 Such toxicoses have been frequently reported in wildlife (Fisher P, O’Connor C, Wright G, Eason CT: 2003, Persistence of four anticoagulant rodenticides in the livers of laboratory rats. Department of Conservation, DOC Science Internal Series 139, Wellington, New Zealand. Available at: http://www.connovation.co.nz/~connovat/images/stories/pdfs/researchpapers/anticoagsgeneral/persistanceoffouranticoags.pdf).6,24
In February 2013, an attempted poisoning of 6,000 laying Leghorn hens by malicious contamination of the drinking water was reported to the Israeli Veterinary Services and Animal Health. An investigation of the poisoning by the Laboratory of Toxicology of the Kimron Veterinary Institute (Bet Dagan, Israel) revealed the presence of aldicarb, methomyl, imidacloprid, and the anticoagulant rodenticide coumatetralyl in the drinking water of the hens. On the first 2 days following exposure, 120 hens died, while on the following days the rate of hen mortality returned to normal. The pathological examination of the hens revealed no pathological manifestations of coagulopathy due to coumatetralyl exposure. Moreover, no residues of aldicarb, methomyl, or imidacloprid were found in the livers of the dead hens, whereas coumatetralyl was found in the liver of 5 hens at a level of 0.004–0.005 mg/kg. Consequently, a question was raised by the Veterinary Services whether the eggs laid by the hens exposed to the contaminating drinking water contained coumatetralyl and at what level.
Numerous high-performance liquid chromatography (HPLC) methods for multiple anticoagulant rodenticides have been described in the literature for various biological matrices.1,8,18,25 However, only a few analytical methods for anticoagulant determinations in eggs have been reported in the literature and all of them have been developed and validated for the determination of a single anticoagulant rodenticide, namely warfarin or bromadiolone.10,14,22 At present, the use of 3 anticoagulant rodenticides (bromadiolone, brodifacoum, and coumatetralyl) are regulated and approved by the Israeli Ministry of Agriculture. However, no officially validated analytical method existed for the detection and quantification of the aforementioned anticoagulants in eggs. The aim of the present study was to develop and validate a multiresidue analysis of 6 commonly used first- and second-generation anticoagulants (warfarin, coumatetralyl, coumachlor, bromadiolone, difenacoum, and brodifacoum) in egg yolk and albumen and to quantitate coumatetralyl levels in eggs laid by the hens exposed to the contaminating drinking water.
Materials and methods
Apparatus and chromatographic conditions
A commercial HPLC system a was used for the analysis of the anticoagulant rodenticides, utilizing a C18 analytical column, b 300 mm × 4.5 mm with 5 µm particle size together with a 5 µm guard column. c The HPLC analysis of the anticoagulant rodenticides was performed using 50 mM of sodium acetate d (pH 6.5) as solvent A and methanol e as solvent B at a constant flow rate of 1 ml/min in a gradient run. The elution was as follows: a linear gradient of solvent B from 30% to 65% during 0–17 min followed by a linear gradient of solvent B from 65% to 90% during 17–24 min was applied. The column was washed with 90% solvent B for 5 min followed by 4 min equilibration to the initial conditions. The fluorescence detector was set at 318 nm excitation and 400 nm emission, and the excitation spectrum was recorded at 240–360 nm. The injection volume was 30 µl, and the column temperature was adjusted to 25°C.
Standard solutions
Stock solutions of the 6 anticoagulant rodenticides (warfarin, f coumatetralyl, f coumachlor, f bromadiolone, f difenacoum, f and brodifacoum f ) as well as of the internal standard (4-methyl-umbelliferone f ) were prepared in a mixture of acetone e and methanol e (1:1 v/v) at a concentration of 1.0 mg/ml and were found to be stable for 3 months when stored at −20°C. Working standard solutions were prepared by dilution in methanol e to give the following concentrations: 20, 10, 2, 1, 0.4, 0.2 mg/l. The internal standard solution was diluted to give a 1 mg/l working solution.
Extraction and cleanup
Blank yolk and albumen (4.0 ± 0.1 g) from anticoagulant-free eggs were weighed into a disposable 50-ml polypropylene centrifuge tube. For the validation assay, the blank samples were spiked with the working standard and internal standard solutions. One gram of anhydrous magnesium sulfate d and 0.5 g of sodium chloride d were added to the tube followed by extraction with 10 ml of acetonitrile e by vortex-mixing for 2 min at high speed. The extraction was continued on a shaker g for 10 min at a speed of 6 cycles/min. The tube was then centrifuged at 4,000 × g for 10 min and the supernatant transferred to a 15-ml tube h and vortexed for 10 sec at high speed. The tube was then centrifuged i at 4,000 × g for 5 min and the supernatant transferred to a 15-ml glass test tube and evaporated to dryness at 60°C under a N2 stream. The residue was dissolved in 0.6-ml solution composed of double distilled water j and methanol e (1:1 v/v) and vortex-mixed for 10 sec. Lipids were removed by adding 0.1 ml of ice-cold hexane e to the reconstituted samples, vortexed for 5 sec, and finally centrifuged k at 14,000 × g for 10 min. The clear methanol–aqueous phase was transferred into an autosampler vial for analysis.
Validation assay methodology
An overall validation of the method was performed using blank egg yolk and albumen spiked with working standard and internal standard solutions. The validation parameters were determined in accordance with the recommendations defined by the European Union (European Commission: 2005, Volume 8: notice to applicants and guideline, veterinary medicinal products: establishment of maximum residue limits (MRLs) for residues of veterinary medicinal products in foodstuffs of animal origin: development and validation of a proposed regulatory method. Available at: http://ec.europa.eu/health/files/eudralex/vol-8/pdf/vol8_10-2005_en.pdf). The following set of parameters was included: recovery, linearity, accuracy, precision, limit of detection (LOD), and limit of quantification (LOQ). 7 A daily calibration curve was obtained by spiking 4-g samples at 6 concentrations of the anticoagulant rodenticides mixture (4, 2, 0.4, 0.2, 0.08, 0.04 µg) and analyzing 5 replicates for 3 consecutive days. All quality control samples as well as the unknown samples were fortified with the internal standard (0.2 µg). The extraction recoveries from yolk and albumen of all 6 anticoagulant rodenticides were calculated for low (0.02 mg/kg), middle (0.1 mg/kg), and high (0.5 mg/kg) concentration levels in accordance with the guidelines defined by the European Union (European Commission: 2005). Concentration of each analyte was calculated using the internal standard method, according to the equation: normalized analyte area = (plotted analyte area/plotted internal standard area) × 100.
Analyte concentrations were eventually determined by linear regression using the formula Y = mX + b, where Y indicates the normalized analyte area and X indicates the analyte concentration in µg/kg. The data was used to determine the validation parameters. The extraction recoveries of the analytes were determined by comparing the results of the analysis of the spiked samples with those of the working solutions. The LOD and LOQ were calculated utilizing a specific calibration curve covering samples in the concentration range of 0.005–0.05 mg/kg according to the equation: LOD = 3 × (SD/m); where SD indicates the residual standard deviation of the regression line and m indicates the slope of the calibration curve.
Collection and analysis of samples retrieved from the affected chicken coop
Eggs (n = 50) were collected from the most severely affected chicken coop that contained 1,600 layers, in which 36 hens had been found dead the morning after the toxicants were thought to be added into the drinking water. The eggs were collected by inspectors of the Israeli Veterinary Services only on the second and fourth day of the poisoning event, while the eggs collected on the remaining days were disposed of by Veterinary Services and therefore were not available for residual analysis. The eggs were analyzed for pesticides and anticoagulants by the Laboratory of Toxicology and the National Residue Control Laboratory. For the analysis of coumatetralyl in the eggs, the yolk and albumen of 5 randomly chosen eggs from different hens were individually homogenized, and a 4.0-g aliquot of each sample was fortified with 0.2 µg of internal standard. All samples were analyzed on the same day. The analysis of methomyl, aldicarb, and methiocarb (LOQ 0.003 and 0.004, respectively) in the eggs was performed by an in-house multiresidue method utilizing liquid chromatography tandem mass spectrometry (MS/MS).
Results
Chromatographic conditions
A gradient solvent delivery system was used for the separation of the anticoagulant rodenticides, and different proportions of methanol and sodium acetate buffer (pH 6.5) were tested. A linear gradient of 30% methanol to 65% methanol during 17 min, followed by a linear increase to 90% methanol during 7 min and fluorescence detection at 318 nm excitation and 400 nm emission, yielded the optimal resolution and sensitivity of the 6 anticoagulant rodenticides (Fig. 1). An excitation spectrum ranging from 240 to 360 nm was recorded for the identification of all 6 analytes, and the emission wavelength of 400 nm was used for analyte quantification (Supplementary figure). Warfarin, coumachlor, and coumatetralyl displayed excitation maximum at 315 nm, whereas the internal standard, bromadiolone, difenacoum, and brodifacoum showed excitation maximum at 318 nm (Supplementary figure).
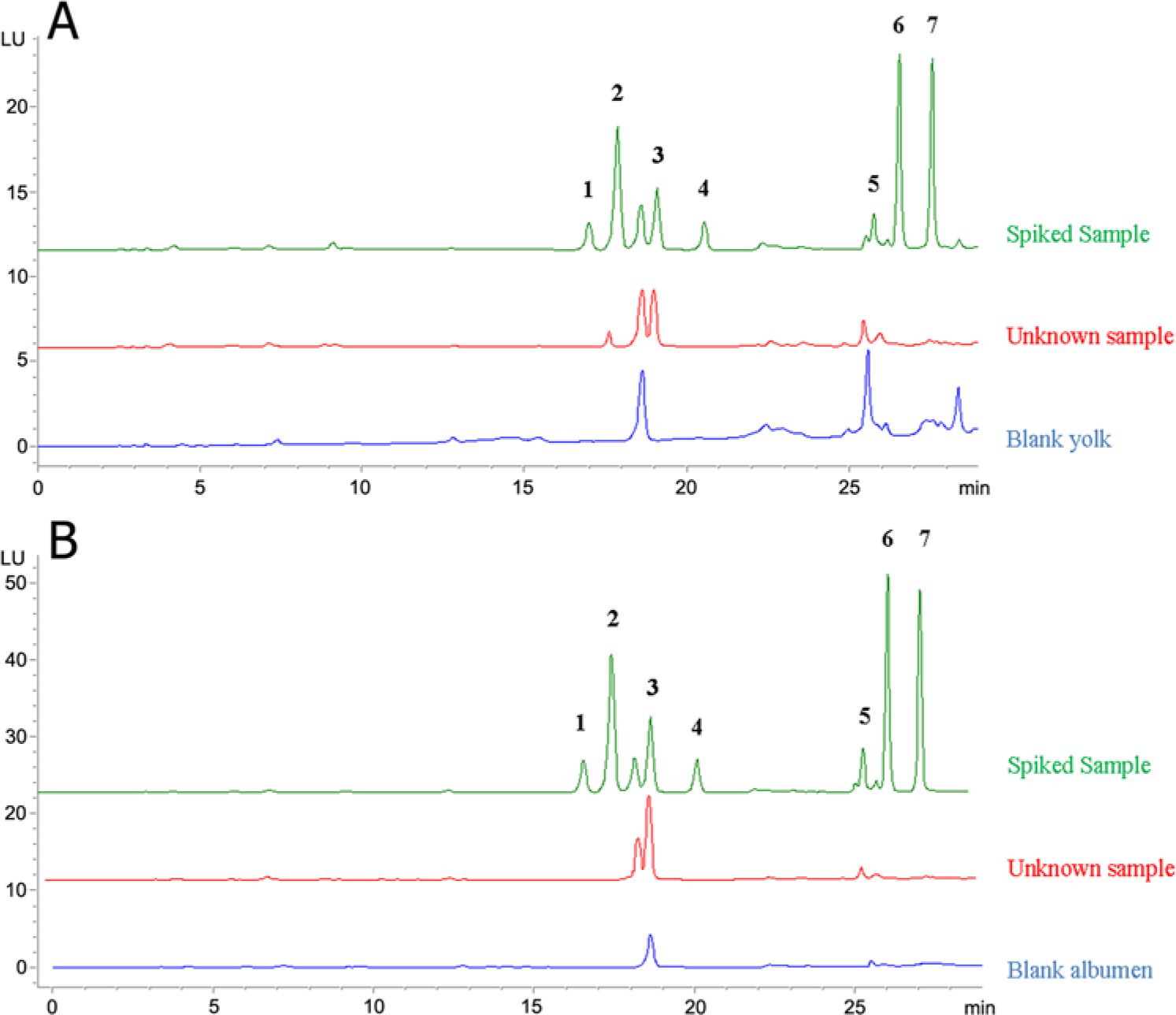
Chromatograms of a spiked standard mixture of anticoagulant rodenticides (0.1 mg/kg), an unknown sample collected from poisoned hen eggs, and a blank sample in yolk (
Sample preparation
The method specificity was determined by analyzing 10 spiked yolk and albumen samples and verifying the lack of interfering matrix components. An unidentified matrix component was eluted (retention time [RT] = 18.6 min; Fig. 1) in both yolk and albumen matrices in proximity to the internal standard (RT = 19.06 min; Fig. 1). However, the peak separation was sufficient to obtain excellent intra- and interday repeatability (% coefficient of variation [%CV]) of 2.5 and 3.1 in yolk and 3.2 and 3.9 in albumen, respectively. In addition, the RTs of the 6 anticoagulant rodenticides as well as the RT of the internal standard remained constant when the working standard solutions or the spiked samples were chromatographed. Therefore, the analytical method could be considered to be specific.
Method validation
The lowest recoveries obtained from yolk were 51–65% for brodifacoum, and the highest recoveries obtained were 86–95% for coumatetralyl (Table 1). Moreover, the recoveries from albumen were generally higher for all tested analytes compared with recoveries from yolk. The calibration curves presented excellent correlation between analyte concentration and peak area (R2 for all curves were >0.995; Table 2). The LOD and LOQ values for the tested anticoagulant rodenticides in yolk were 0.003 and 0.01 mg/kg, respectively, except for warfarin, exhibiting 3 times higher LOD and LOQ values (Table 2). In albumen, the LOD and LOQ values for warfarin, coumatetralyl, coumachlor, and bromadiolone were the same, namely 0.003 mg/kg and 0.01 mg/kg, respectively, while difenacoum and brodifacoum displayed LOD and LOQ values of 0.0015 and 0.005 mg/kg, respectively. The %CV of within and between days was 1.0–8.5% for yolk and 0.6–3.8% for albumen (Table 3). The accuracy values for the analytes tested were in the range of 90–110%. Hence, in the examined range of concentrations analyzed, the optimized analytical method was considered accurate and precise.
Recovery data of fortified yolk and albumen samples at low, middle, and high analyte concentrations within the tested calibration curve range of 0.01–1 mg/kg.

Linearity parameters, limit of detection (LOD), and limit of quantification (LOQ) obtained from blank yolk and albumen spiked with anticoagulant rodenticides.*
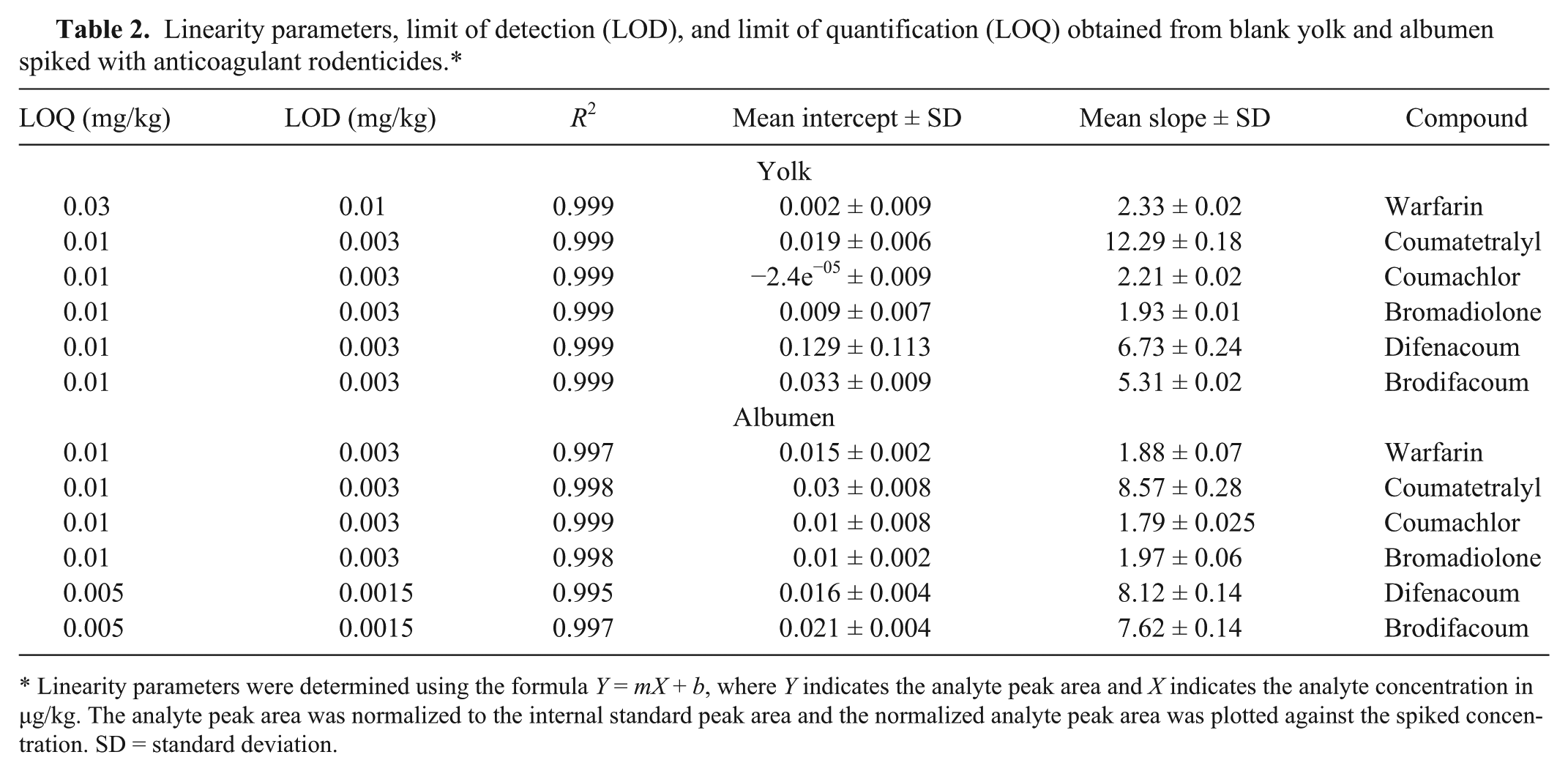
Linearity parameters were determined using the formula Y = mX + b, where Y indicates the analyte peak area and X indicates the analyte concentration in µg/kg. The analyte peak area was normalized to the internal standard peak area and the normalized analyte peak area was plotted against the spiked concentration. SD = standard deviation.
Accuracy and precision data obtained from yolk and albumen spiked with anticoagulant rodenticides.*
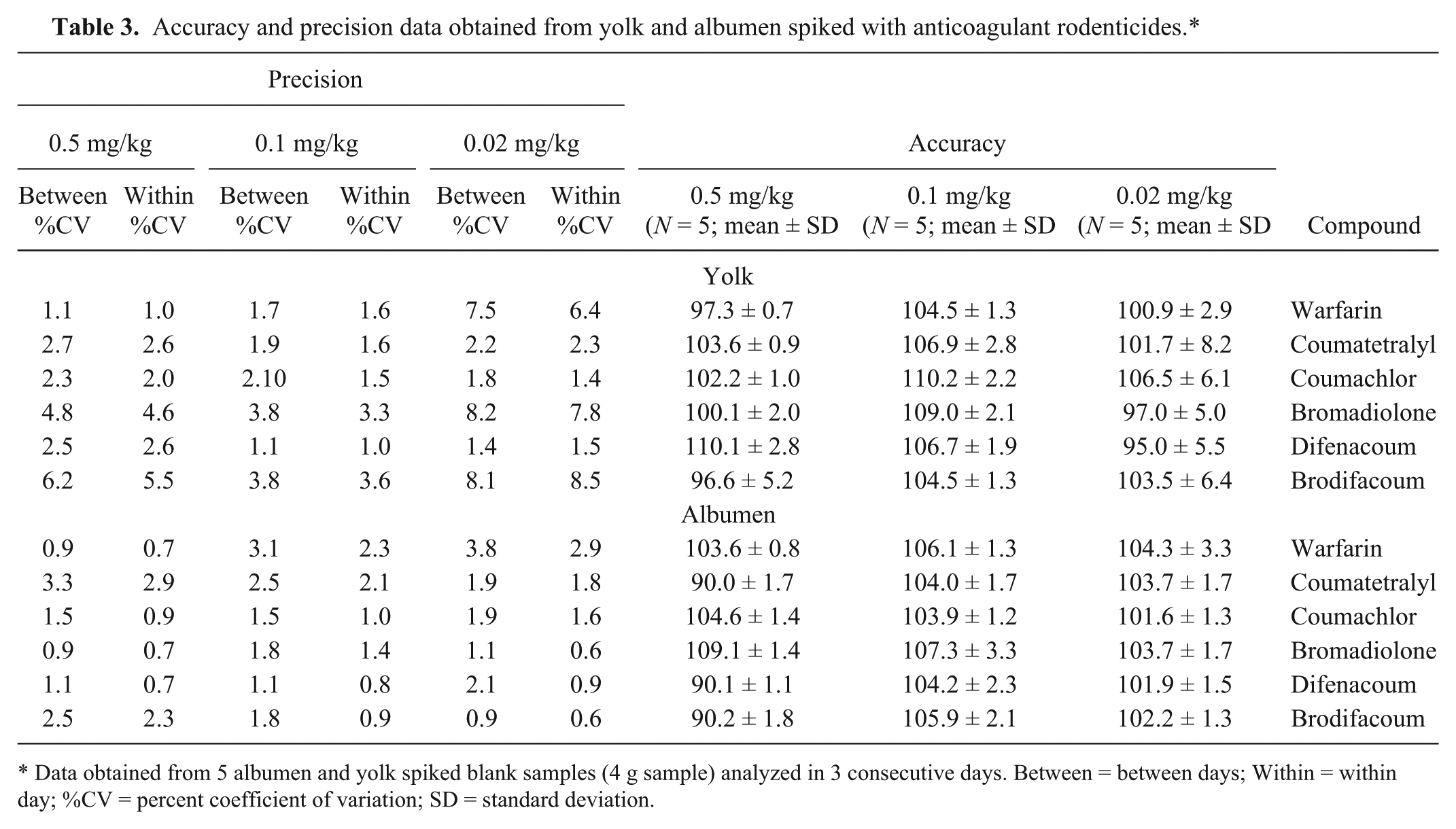
Data obtained from 5 albumen and yolk spiked blank samples (4 g sample) analyzed in 3 consecutive days. Between = between days; Within = within day; %CV = percent coefficient of variation; SD = standard deviation.
Analysis of egg samples collected from the field toxicosis
No lesions of intoxication due to anticoagulant exposure were visible upon pathological examination but typical clinical manifestations of carbamate poisoning were clearly visible in the surviving hens. Yolk and albumen of 5 randomly selected eggs were separately processed according to the final validated method described herein. A typical chromatogram of incurred yolk and albumen samples is shown in Figure 1. Coumatetralyl was found in all yolk samples on the second and fourth day of hen exposure, displaying an average concentration of 0.0057 ± 0.0011 and 0.0052 ± 0.0007 mg/kg, respectively. No detectable coumatetralyl levels were found in the albumen. Carbamates were not detected in any of the egg samples.
Discussion
The most common analytical methods for anticoagulant rodenticide determination in various biological matrices are based on reversed-phase HPLC utilizing ultraviolet, fluorescence, or electrospray-ionization MS/MS detection techniques.8,18,25 At the present, only a limited number of methods have been published in the literature regarding anticoagulant rodenticide determination in eggs, and all of them were developed and validated for a single anticoagulant determination, namely warfarin or bromadiolone.10,14,22 Therefore, the aim of the present study was to develop and validate a timely and cost-effective multiresidue analysis of 6 different anticoagulant rodenticides and to determine residue levels in eggs collected from hens exposed to unknown dosage of coumatetralyl in their drinking water.
In the present study, a fully validated multiresidue analytical method of 6 anticoagulant rodenticides (warfarin, coumatetralyl, coumachlor, bromadiolone, difenacoum, and brodifacoum) was established in the range of 0.01–1 mg/kg in albumen and yolk. The concentration range chosen covered residual levels found in egg yolk and albumen analyzed from hens fed with 10 mg/kg BW of warfarin and bromadiolone, a dosage corresponding to the maximum risk and to 2-fold the maximum risk for common baits containing 0.025% warfarin and 0.005% bromadiolone.10,14,22 The newly developed method met the requirements for specificity, linearity, accuracy, precision, and recovery in accordance with the method performance recommendations defined by the European Union. The calculated LOD and LOQ values in the current study were lower than the values reported for liver and blood matrices utilizing a HPLC method with fluorescence detection and are suitable for the diagnosis of anticoagulant rodenticides residues in eggs.1,8
Most published HPLC assays use a mobile phase consisting of methanol as the organic solvent and a moderately acidic to neutral buffer (pH 3.5–7) as the aqueous phase. Preliminary studies performed by the authors to evaluate the optimal chromatographic conditions by modifying the pH value of sodium acetate buffer (3.5, 5, or 6.5), solvent strength (0.5, 5.0, or 50 mM) and organic eluent (methanol or acetonitrile), clearly indicated that a mobile phase consisting of methanol and 50 mM of sodium acetate buffer at pH 6.5 led to the highest sensitivity, resolution, and symmetry factors.
Various solvents and solvent mixtures (acetone, chloroform, acetone-to-chloroform [30:70], dichloromethane, methanol, and acetonitrile) were tested to find the most suitable solvent to extract anticoagulant rodenticides from egg yolk and albumen. Acetonitrile was found to be the most suitable extraction solvent, yielding cleaner extracts and better recoveries. No significant differences regarding recovery were found between 1 or 2 extraction steps consisting each of 10 ml of acetonitrile and between extraction times ranging from 10 to 30 min. Therefore, 1 extraction step of 10 ml of acetonitrile and 10-min extraction time on a rotary shaker was chosen for the method validation. Many biological matrices contain lipids that are co-extracted together with the analytes of interest. Yolk contains high concentration of various lipids that need to be removed from the extract prior to analysis.8,22 Several different sorbents and sorbent combinations were investigated for sample clean-up, namely primary and secondary amines (PSAs; 50 mg), Florisil (100 mg), basic alumina (100 mg), and MgSO4 (175 mg).16,25 The sorbent combination of 50 mg of PSA, 100 mg of Florisil, and 175 mg of MgSO4 yielded the most satisfying specificity and recovery. The addition of 0.1 ml of ice-cold hexane to the reconstituted sample (water-to-methanol mixture [1:1]) followed by centrifugation at 14,000 × g yielded 2 phases consisting of a lower aqueous–methanolic phase in which the anticoagulant rodenticides were dissolved and an upper lipid phase. The lower aqueous–methanolic phase was eventually used for injection. The lower recoveries obtained from yolk in comparison with albumen are most probably due to the high lipophilic nature of the rodenticides conferring them higher affinity to the lipophilic components in yolk (Pesticide Properties Database, available at: http://sitem.herts.ac.uk/aeru/footprint/index2.htm).
The predominant partitioning profile of coumatetralyl in yolk was similar to the partitioning profile of bromadiolone following oral administration (10 mg/kg) to laying hens. 10 As both anticoagulant rodenticides share similar physicochemical properties, such as the partition constant (log P), water solubility, and logarithmic measure of the acid dissociation constant (pKa) value, it is reasonable to assume that other 4-hydroxy-coumarin–related rodenticides possessing similar physicochemical properties, such as coumachlor, difenacoum, and brodifacoum, will exhibit similar partitioning behavior (http://sitem.herts.ac.uk/aeru/footprint/index2.htm).
Previous studies clearly demonstrated the passage of warfarin and bromadiolone into eggs at various dosages.10,14,22 The maximal carryover, calculated as the maximal amount of anticoagulant rodenticide found per egg divided by the total single oral dosage, was found to be within the range of 0.06–0.04%. The highest level of bromadiolone and warfarin found in a single egg collected from adult hens (average BW of 1.7–2.2 kg) dosed with a lethal amount of 120 mg of bromadiolone or a sublethal amount of 180 mg of warfarin, were 64 and 65 µg, respectively, assuming an average yolk and albumen weight of 16 and 36 g, respectively.10,19,22 Ingestion of 4 eggs containing the maximum amount of bromadiolone or warfarin would still be significantly below the toxic dosage for mammals the size of cats, dogs, and sheep and, therefore, an acute intoxication due to anticoagulant effect in human beings seems highly improbable (Erickson W, Urban D: 2004, Potential risks of nine rodenticides). Moreover, the total amount of coumatetralyl found in the eggs in the current study was 0.091 µg, an amount well below any dosage being capable of inducing an acute toxic response (Erickson W, Urban D: 2004). On the other hand, the adverse effects associated with chronic consumption of low levels of anticoagulant rodenticides over a longer period of time are unknown. Due to the long half-lives and persistence of the second generation rodenticides in animal tissues and the lack of knowledge associated with long-term exposure to residue levels of anticoagulant rodenticides, long-term chronic toxicity studies are needed to determine toxicity threshold for residual levels for each rodenticide. In conclusion, the proposed HPLC method was found to be suitable for determining anticoagulant rodenticide residues in the albumen and yolk of eggs from hens exposed to anticoagulant baits as was demonstrated in the above-described poisoning case.
Footnotes
Acknowledgements
The authors gratefully thank Mr. Yossi Hofi and Dr. Oran Erster for their excellent technical assistance.
a.
Agilent HPLC 1100 series system equipped with degasser, auto sampler, diode-array detector and fluorescence detector, Agilent Technologies Deutschland GmbH, Waldbronn, Germany.
b.
Allure organic acids column, Restek Corp., Bellefonte, PA.
c.
Trident Direct in-line guard column, Restek Corp., Bellefonte, PA.
d.
Sigma-Aldrich Handels GmbH, Vienna, Austria.
e.
LC-grade, Merck KGaA, Darmstadt, Germany.
f.
Sigma-Aldrich, St. Louis, MO.
g.
Promax 2020, Heidolph, Kelheim, Germany.
h.
QuEChERS tubes containing 50 mg PSA, 100 mg Florisil, and 175 mg MgSO4, Agilent Technologies Inc., Santa Clara, CA.
i.
Eppendorf Centrifuge 5810 R, Eppendorf AG, Hamburg, Germany.
j.
Elga Purelab ultra analytic system, Veolia Water, Buckinghamshire, UK.
k.
Biofuge 13 with Hereaus Sepatech rotor (24 x 1.5 ml), Heraus, Hanau, Germany.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
