Abstract
Centrifugation is a powerful method for solid-liquid separation. It can be applied in numerous ways to simplify solid phase synthetic procedures. At the same time, centrifugation is the only totally parallel technique which can be scaled up for processing volume or number of simultaneously run reactions, without the limitation of overpressure or vacuum-driven filtration-based systems. We have developed synthesizers based on the power of centrifugation — peptide and small organic molecule synthesizers utilizing cotton as the synthetic substrate and inclusion volume chemistry, synthesizers for automation of “tea bag” synthesis, and synthesizers based on “tilted plate centrifugation”. The last technique was employed in an oligonucleotide production facility with the capacity of more than 10 million compounds per year.
Keywords
Introduction
In order to accelerate the synthesis of large single compound arrays, much effort is currently devoted to the implementation of high-throughput synthesis automation (see e.g. 1 ). Numerous companies have started to develop and market instruments for the automated parallel solid-phase synthesis of compound arrays. Most of these instruments are based on solid-phase synthesis technology 2 and use commercially available pipetting robots for the delivery of reagents and wash solutions to the synthetic compartments. The capacity of these synthesizers ranges from 12 to 384 compounds in one run.
A key process in multiple solid-phase synthesis is the parallel removal of excess reagent and wash solutions from the solid support in all synthetic compartments. In most currently available synthesizers, this is achieved through the porous bottoms of the synthetic compartments, either by vacuum filtration (e.g. Argonaut Technologies and Bohdan Automation instruments), or application of pressure from the top of the compartments (e.g. Advanced ChemTech instruments). These methods bear the inherent risk of clogging one or more compartments, resulting in insufficient liquid removal, overflow, and, consequently, contamination of neighboring compartments. An alternative method, which employs aspiration of the liquid from the surface, 3–4 requires dedicated equipment and is not commercially available.
COTTON AS A SUBSTRATE FOR CENTRIFUGATION
Our quest for a simple, parallel, and inexpensive method for synthesis of a multitude of compounds started with an investigation of the properties of cotton. (The full story of cotton as a synthetic substrate is described elsewhere 5 .) Cotton is the purest form of cellulose. Cellulose was one of the first carriers tested (and found unsatisfactory) by Bruce Merrifield in his search for optimal solid support. Cellulose paper was used by Ronald Frank for parallel synthesis of oligonucleotides 6 and later peptides. 7 Cotton is a polysaccharide with a high content of free hydroxyl groups, which, potentially, could be used for the attachment of the first building block or a linker. Moreover, cotton is readily available from multiple sources in various shapes and forms. In the form of textile sheets, it can easily be handled in bulk, used as a continuous strip, 8 or divided into appropriate pieces during the synthesis. 9 With cotton, there is really no need for any reaction vessel. We have performed many syntheses just by placing a cotton strip onto a flat glass plate and soaking the reagents into the cotton fabric. Since there is no excess liquid around, it is not necessary to shake the solid support - distribution of the active reagent by diffusion is adequate. Obviously, if the concentration of the reagent did not exceed the “concentration” of the free amino groups (which were introduced to the carrier by attachment of amino acid glycine to cotton hydroxyl groups) inside the cotton carrier, it would be impossible to achieve a complete reaction. We call this principle “inclusion volume coupling”, and we have tested it with polystyrene type carriers as well. 10
The next step was obvious: We designed a multiple synthesizer using cotton as the carrier, and built the first prototype. Centrifugation is the first choice for the daily life chore of washing clothes — spinning efficiently removes liquid from textile fabrics! Figures 1 shows our first centrifugal synthesizer - a flat grooved rotor mounted on a laboratory centrifuge. Building blocks (amino acids) were pipetted onto the fabric attached in the grooves together with the activator. Capillary forces distributed the solution evenly throughout the carrier and coupling proceeded without any liquid in excess of the amount that was soaked into the carrier. A convenient way of visualizing the process of the coupling was bromophenol blue monitoring. 11 The cotton was first colorized with a dilute solution of bromophenol blue. The solution was centrifuged off, but the cotton remained blue if there were free amino groups available. At the very end of the coupling, the blue color disappeared and the next step of the synthesis could be performed. This type of monitoring is non-invasive, since no sample of the carrier has to be removed and destroyed, and it is also a “real-time” process. We immediately speculated about a camera-based real-time feedback-based synthesizer. 12 The idea was implemented several years later on a more sophisticated machine. After completion of coupling, the reaction solution was centrifuged out and the solvent used for washing was introduced. Washing performed in this way was very efficient, since almost all liquid can be removed from the cotton fabric (only 6% of liquid loaded remained in the fabric after centrifugation). We have been able to perform the whole synthesis using just one wash between coupling and deprotection or between deprotection and coupling.

First centrifugal synthesizer.
Since the first experiments were successful, we built a completely automatic synthesizer capable of synthesizing 24 peptides in parallel. 12 14 This synthesizer evolved through several versions (see Figures 2 and Figures 3) and was the reason behind forming Spyder Instruments to commercialize this machine. The success of Spyder was rather limited; it built five machines and sold four of them. Two of these machines were used rather heavily for several years and produced hundreds of peptides. However, 24 peptide batches simultaneously was not a competitive capacity; there were several competitive instruments capable of the same volume (some of them, however, up to eight times more expensive).


Compas 242 - the first commercially available centrifugal synthesizer for both cotton and “tea bags”.
There is another property of cotton fabric as important as the synthetic support – continuity. It has nothing to do with centrifugation, but I mention it to stress the endless opportunities offered by cotton. We have utilized this feature for continuous solid phase synthesis - synthesis in which the individual steps of synthesis are not separated in time but in space. All steps are performed simultaneously on the different zones of the continuous carrier moving slowly from one zone to another. The principle of this idea is illustrated in Figures 4. This concept is very powerful and a relatively small strip of cotton can produce an impressive amount of peptides, due to the fact that all steps of the synthesis can be performed at once, and continue for days and weeks. 8 Obviously, cotton has its limitation due to the chemical nature of its polysaccharide chains. However, other solid carriers used for solid phase synthesis today can mimic the filamentous structure of cotton, providing this carrier with many unique properties. Inclusion volume coupling in “tea-bags” (polypropylene mesh bags containing polymeric solid support) allowed the use of centrifugation for automation of this powerful technology. 14
Concept of continuous solid phase synthesis. Solid support is continually fed through synthetic compartments – all operations of the whole synthesis are performed at the same time; however, in different parts of the carrier.
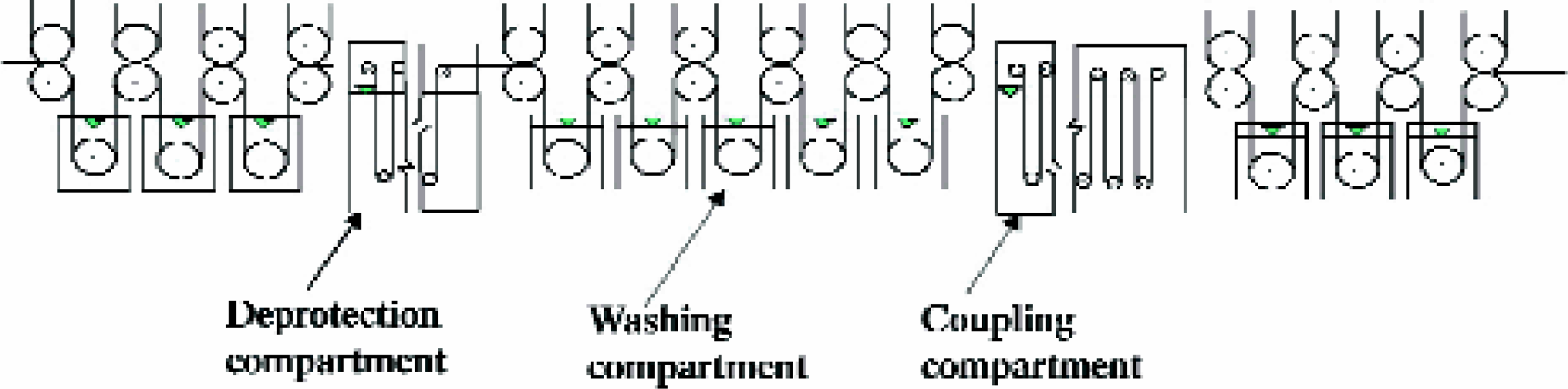
SYNTHESIS AUTOMATION BASED ON TILTED PLATE CENTRIFUGATION
We have devised a new technological concept for the automation of the solid-phase synthesis of large compound arrays. 15 The key feature of this technology is a new method for separation of the solid support from reagent solutions, termed “tilted plate centrifugation”, which uses centrifugation as a means of liquid removal in conjunction with the use of tilted microtiter plates as reaction vessels. The tilted plate centrifugation technology greatly improves the centrifugation method described above by using the wells of microtiter plates as synthetic compartments, thus enabling the parallel synthesis of much larger compound arrays (i.e., 3,072 compounds when eight 384-well plates are used). The plates are mounted on a centrifugal plate and slightly tilted down towards the center of centrifugation, thus generating a pocket in each well, in which the solid support is collected during centrifugation while the supernatant solutions are expelled from the wells (Figures 5).
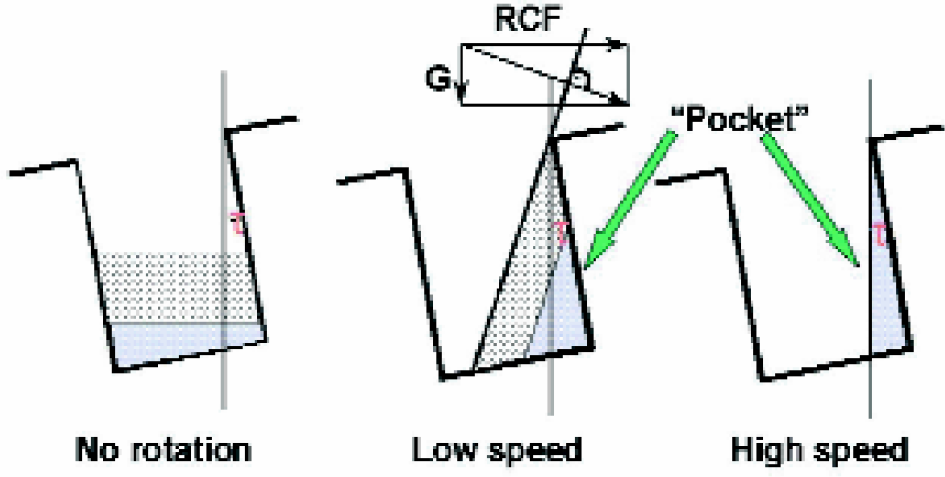
Concept of tilted plate centrifugation: Formation of the pocket in the well of a tilted plate during centrifugation (direction: left to right). The solid support (lower layer) is collected in the pocket, while the liquid (upper layer) is expelled from the well. The liquid surface angle is perpendicular to the resulting force vector of the relative centrifugal force (RCF) and gravity (G).
In order to ensure efficient liquid removal (i.e., no solution remaining in the wells after centrifugation) and at the same time to avoid any loss of solid support during centrifugation, the volume of the well-pockets should be equal to the volume of swollen resin in each well. This can be achieved by adjusting the pocket size by using plates with varying well volumes and/or modifying the tilt angle, as well as the speed of rotation.
An essential feature of this approach is that well to well cross-contamination with reagent solution or resin is avoided by the fact that the plates are tilted, while the direction of centrifugation is horizontal. Consequently, any liquid or resin expelled from the wells is either captured in the inter-well space of the plate, or collected on the wall of the centrifugal drum. The fact that the cross-contamination was not an issue has been proven by analyzing all products prepared on the microtiter plate by HPLC/MS. The originally speculated trajectory of the expelled liquid from individual wells — that the liquid detaches from the upper edge of the well, flies across the inter-well space, and lands on the outer wall of the neighboring well — was proven erroneous based on the modeling of the dynamics of the fluid leaving the well. Per the model, the liquid clearly adheres to the wall and follows the surface of the plate until it reaches the edge of the plate (see Figures 6).
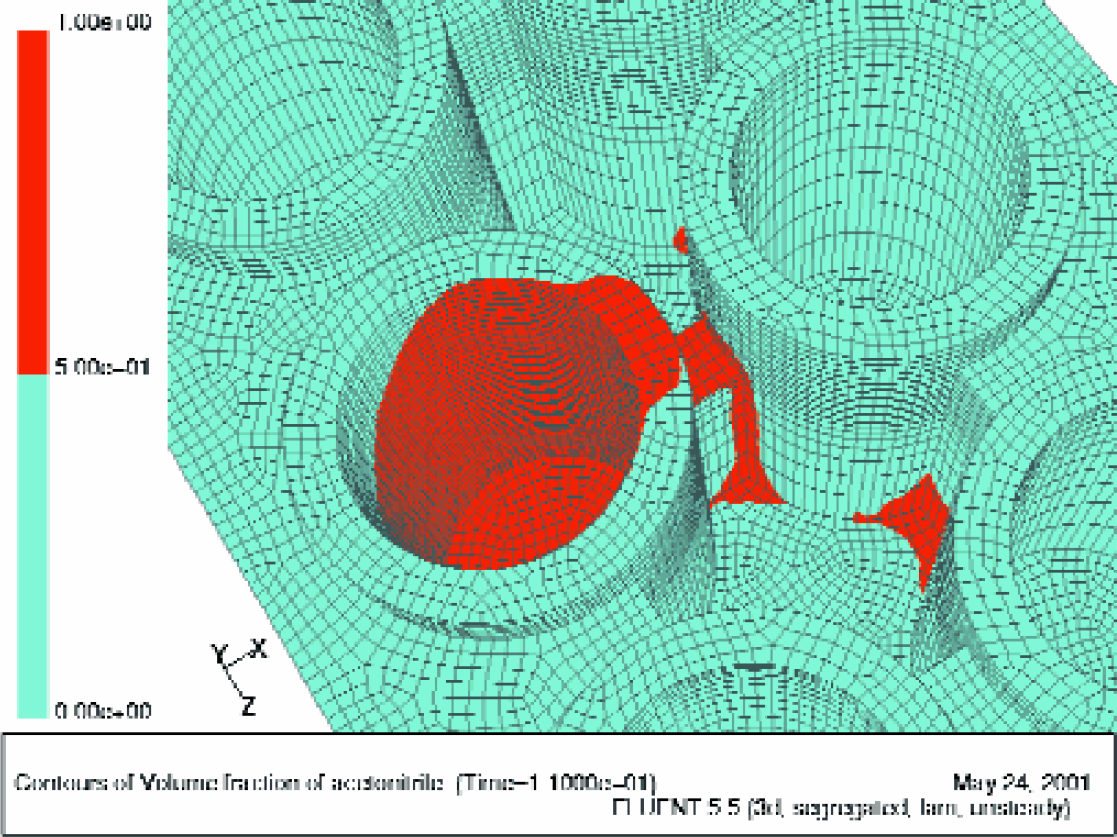
Fluid dynamic simulation of the liquid (acetonitrile) being expelled from the individual polypropylene well by centrifugal force.
Centrifugation-based synthesis technology was proven on the syntheses of arrays of peptides and small organic molecules. 3 The apparent danger of cross-contamination of individual reaction compartments due to the fact that reactions are performed in the open environment was suspected. However, no contamination was observed. The inner volume of the reaction chamber is continuously flushed with inert gas (nitrogen) and only components of high volatility would have a chance of building concentration, which may potentially contaminate other reaction compartments. (Nitrogen flushing is needed not only as the protection of reaction from moisture and oxygen, but also as the safety blanket preventing the creation of flammable gas mixture.)
Tilted plate centrifugation as a method for high-throughput synthesis was first presented at LabAutomation 98 in San Diego. 16 It was developed as part of the high-throughput synthesis program at Trega Biosciences and was aimed at the synthesis of libraries of small organic molecules, presented as individual compounds in the wells of microtiter plates. Since this methodology was understood by Trega as secondary to the methodology of surface suction 3 4 developed at the same time, centrifugal synthesizer development was pursued by Spyder Instruments, Inc. with the funding provided by the National Institutes of Health. A patent for tilted plate synthesis technology was issued in 2000. 17 This technology is applicable not only to solid phase synthesis but also to fluorous synthesis, solid and liquid-liquid extraction, and in-plate washing.
OLIGONUCLEOTIDE SYNTHESIZER BASED ON TILTED PLATE CENTRIFUGATION
In 2000, Illumina, Inc. became interested in Spyder's technology for high-throughput DNA synthesis, and later, after successful verification of its feasibility, Illumina, Inc. acquired Spyder Instruments. The challenge of high-throughput DNA synthesis using this technology was not in the chemistry, which is one of the most optimized chemistries available today, but in the design of the instrument which would be capable of parallel processing of hundreds of reaction vessels simultaneously. Instruments capable of parallel synthesis of oligonucleotides were developed by ProtoGene Laboratories, 18 Stanford University, 19 and recently by Institute of Biomedical Sciences at Taiwan. 20 These synthesizers use 96- or 384-well microtiter plates equipped with filters at the bottom of each well. Unfortunately, their capacity was deemed to be too low to efficiently meet the demands of the Illumina technology.
To prove the basic concept of tilted plate centrifugation works for DNA synthesis, we first modified semi-automated peptide synthesizers and carried out a series of syntheses (Figures 7). We realized that the throughput of the synthesizers in cases utilizing fast chemistries critically depended on the speed of the reagent delivery, and we centered the design of the new synthesizer on liquid handling. The central part of the new instrument remains a centrifuge with a rotor diameter of 50 cm. Eight polypropylene microtiter plates were placed on the perimeter of the rotor. However we made significant modifications to: (i) the original rotor design, (ii) the building block delivery system, (iii) bulk reagent delivery system, and (iv) the computer control system.

Commercially available (Spyder Instruments, Inc.) centrifugal synthesizer Compas 768 (on the left), and the same instrument modified for proof of concept oligonucleotide synthesis (on the right).
Based on the results obtained with the prototype synthesizers, we built 12 fully automatic machines and enabled Illumina's Oligator Farm™ facility (see Figures 8). The technical details were described earlier, 21 so we will recapitulate here only the major features of the design.

ROTOR DESIGN:
The delivery into smaller openings of high-density plates requires bringing the nozzles of the delivery banks very close to the plate. However, achieving the tilt needed for the centrifugation step and placement of the nozzles close to the plates are two contradictory requirements. We decided to solve this problem by the application of “dynamic tilt”. Plates are placed in the holder supported by a spring, which retains them in the horizontal position when centrifugal force is not applied (Figures 9). However, as soon as the centrifugation starts, the weight located under the axes of plate holder hinge rotation pulls the plate into tilted position, the limit of which is defined by locating nuts placed on the screw supporting the springs. Plates can thus be centrifuged in the tilted position and serviced in horizontal position.
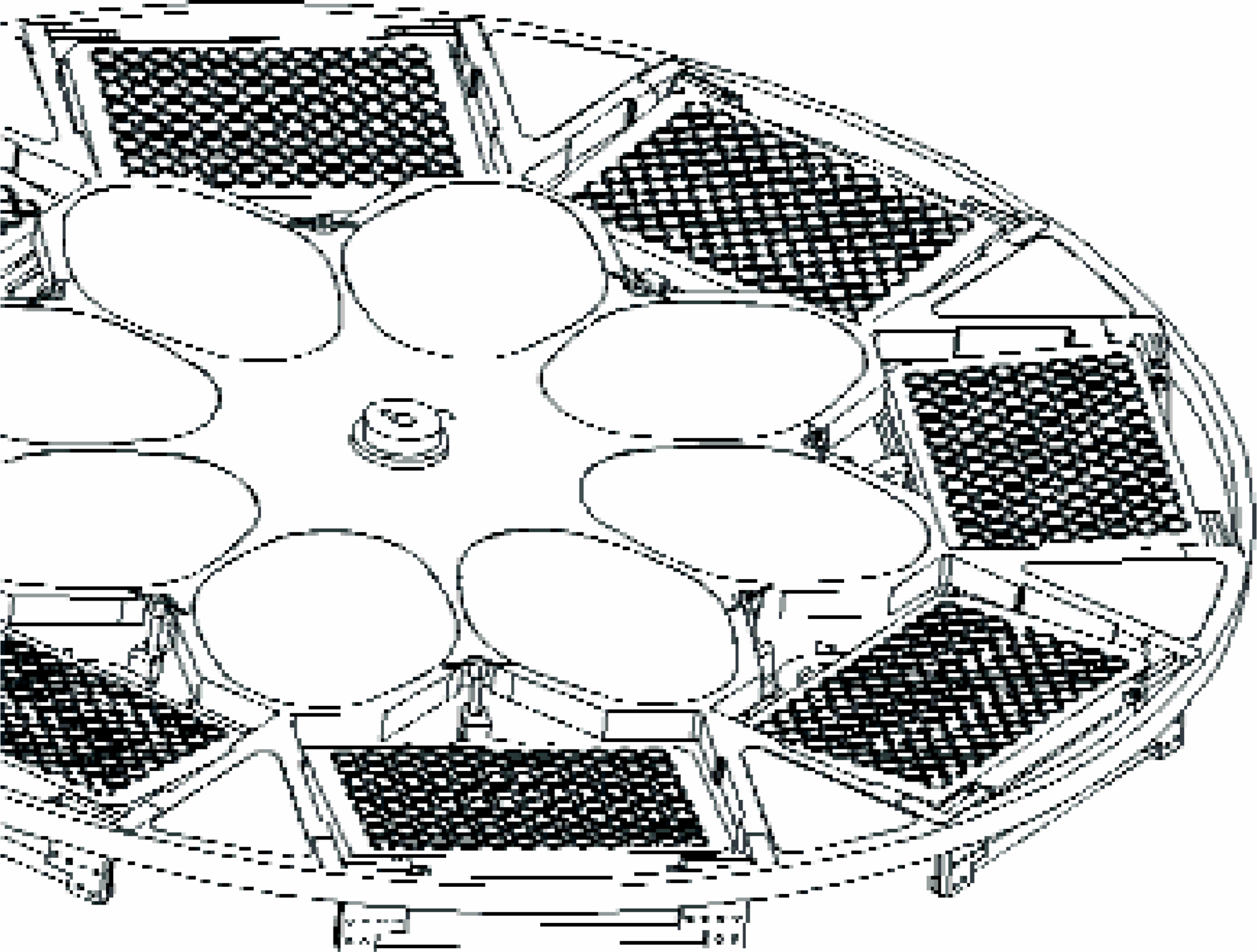
Drawing of the rotor equipped with plate holders capable of “dynamic tilt”.
REAGENT DELIVERY SYSTEM:
We designed and built the liquid delivery system based on moving banks of nozzles. An array of nozzles was placed on the actuator (single or double axis robotic arm). Each nozzle in the array was connected through the solenoid valve to the motorized syringe pump or pressurized solution container and can be positioned (as a part of the array) above the particular well of a given microtiter plate. We have used up to seven arrays of eight nozzles for the delivery of up to seven building blocks simultaneously. Each array serves in parallel the wells of one column of the microtiter plate. The arrays are placed in the grid, which covers seven columns of the microtiter plate.
We have designed the nozzle array holder to allow quick assembly of the multiarray and make it easy to maintain and troubleshoot. This array is capable of reproducible delivery of the amounts of liquid at the range of 1 to 500 ul. We have tested this assembly in both syringe pump operated systems and with timed delivery through solenoid valves. In the latter arrangement, the solutions are stored in pressurized (argon, 3 psi) glass vessels and solenoid valves in the bank are actuated according to the computer algorithm depending on the position of the nozzle above the microtiter plate.
Due to the fact that all seven arrays of nozzles can be operated at the same time, the delivery of the reagents is very fast - one pass over the standard microtiter plate delivering 3 ul of reagent can be achieved in less than 2 seconds.
BULK DELIVERY SYSTEM:
Since the syntheses can use several different solvents and reagents, their separate delivery must be arranged appropriately. One option was to use a separate delivery head for each solvent/reagent. This would require dedicated pumps for delivery of each reagent. Another option was to have one delivery head connected to the alternate reagent syringes via a motorized selector valve. The simplest solution, although requiring the most thorough washing of lines between operations, uses only one motorized syringe and selector valve. This last option was implemented in the first model of the synthesizer. The reagents were delivered by two coupled pairs of motorized syringe pumps simultaneously addressing two 96-channel delivery heads to speed up the delivery cycle. Later we replaced syringe pumps by a single gear pump. This modification resulted in significant time reduction due to the gear pump delivering the liquid directly without the need for charging before each delivery, which is necessary in the syringe pump.
THE CONTROL SYSTEM:
The synthesizer control system utilizes a standard PC architecture. The top-level software is composed of two main components — the run-time component and the direct control component. The run-time component allows the creation and editing of synthesis protocols, that is the step-by-step instructions for the machine's hardware components. This is accomplished by a user interface that incorporates buttons and dialog boxes that allows facile generation of English text statements representing the instruction and parameters associated with that instruction. The interface also allows the loading, editing, saving, and running of protocols once they have been created.
The PC communicates with two sub-controller systems to control the entire machine. The first system used a Compumotor 6K2 (Parker Hannifin, Compumotor Division, U.S.A.) real-time motion controller. Instructions are sent via Ethernet from the PC to the 6K2. The 6K2 contains software that operates as subroutines, and can execute simple or complex operations without continuous supervision from the top-level PC. The 6K2 performs operations, i.e., moving the rotor, operating the dispensing head, etc., and then simply reports back the results, success or error, to the PC. This removes the load from the PC to the 6K2 which is much more capable of real-time, multitasking operations. The 6K2 also provides for interfacing discrete components, such as solenoid valves and sensors, for controlling the various mechanical systems within the machine.
The second control subsystem uses a valve controller board (VCB) to control the fast solenoid valves during the base dispensing process. This dedicated 8-bit controller downloads the sequence information and dispenses volumes from the PC via an RS-232 link. The controller calculates the actual dispense time based upon an empirically determined calibration curve that correlates pressure and delivery volume to time (the amount of time the valve must be kept open to deliver the desired volume). The dispense cycle is composed then of moving the dispensing head across the microtiter plate while the 6K2 signals the VCB when the head is correctly positioned over sets of wells. At each trigger interval the VCB opens the valves that correlate the desired base in the desired well, for the pre calculated time interval, all without stopping the dispensing head motion over the plate.
POST-SYNTHETIC PROCESSING:
After the conclusion of repetitive operations (coupling, capping, oxidation, deblocking, repeated up to hundred times) of the synthesis, the plates are removed from the centrifuge and cleavage reagent is added (ammonia solution or ammonia - methylamine mixture) and plates are heated for several hours (detailed conditions depend on the linker used in the particular synthesis). Plates are removed from the oven, cooled down, and the product is extracted using an automatic multichannel pipettor. Obtained aqueous solution from deep well microtiter plates is analyzed and prepared for the delivery to the customer (as dried down film or concentration normalized solution). The whole process is controlled by a barcode driven LIMS system. However, due to the fact that the synthesis is performed in batches of given size (up to eight microtiterplates), no scheduling of individual samples is necessary.
A BENCH-TOP SYNTHESIZER FOR RESEARCH LABORATORIES (“PET OLIGATOR”)
During the evaluation of the high-throughput synthesizer, we realized that it would be extremely beneficial to have a smaller automated synthesizer. This synthesizer mimics the performance of the big machine on much lower throughput, allowing us to optimize the protocols and respond very quickly to the requests of researchers requiring only small numbers of oligonucleotides. We found the optimal number of compounds synthesized in one batch to be 24 to 48. This number of synthetic compartments, or wells, can be placed on the perimeter of a small rotor with a diameter of 14 cm. This rotor is then placed in the drum of the centrifuge covered with a lid having an integrated array of nozzles connected through the solenoid valves with vessels containing the particular reagents. During one rotation of the rotor, all synthetic compartments are placed under the appropriate nozzle and the reagent is delivered. Removal of the solvent is achieved again by centrifugation. Since the construction and performance of the prototype “Pet Oligator” system (Figures 10) was very promising, we decided to evaluate this instrument as a possible alternative to commercially available synthesizers. The Pet Oligator system can achieve similar throughput, but due to the simplicity of its concept, it can be built for a fraction of the cost.

“Pet Oligator” - economical synthesizer capable of 24 parallel syntheses. On the left - one of the first prototypes. On the right - final version.
The Pet Oligator system was used in hundreds of separate syntheses of up to 24 oligonucleotides in a batch, with lengths spanning from 25-mers up to 125-mers. We have used mass spectroscopy and HPLC for evaluation of quality of produced compounds and we found this small synthesizer invaluable in evaluating the new protocols for high throughput machines. Analysis of one run of 24, 25-mers is given in Figures 11.
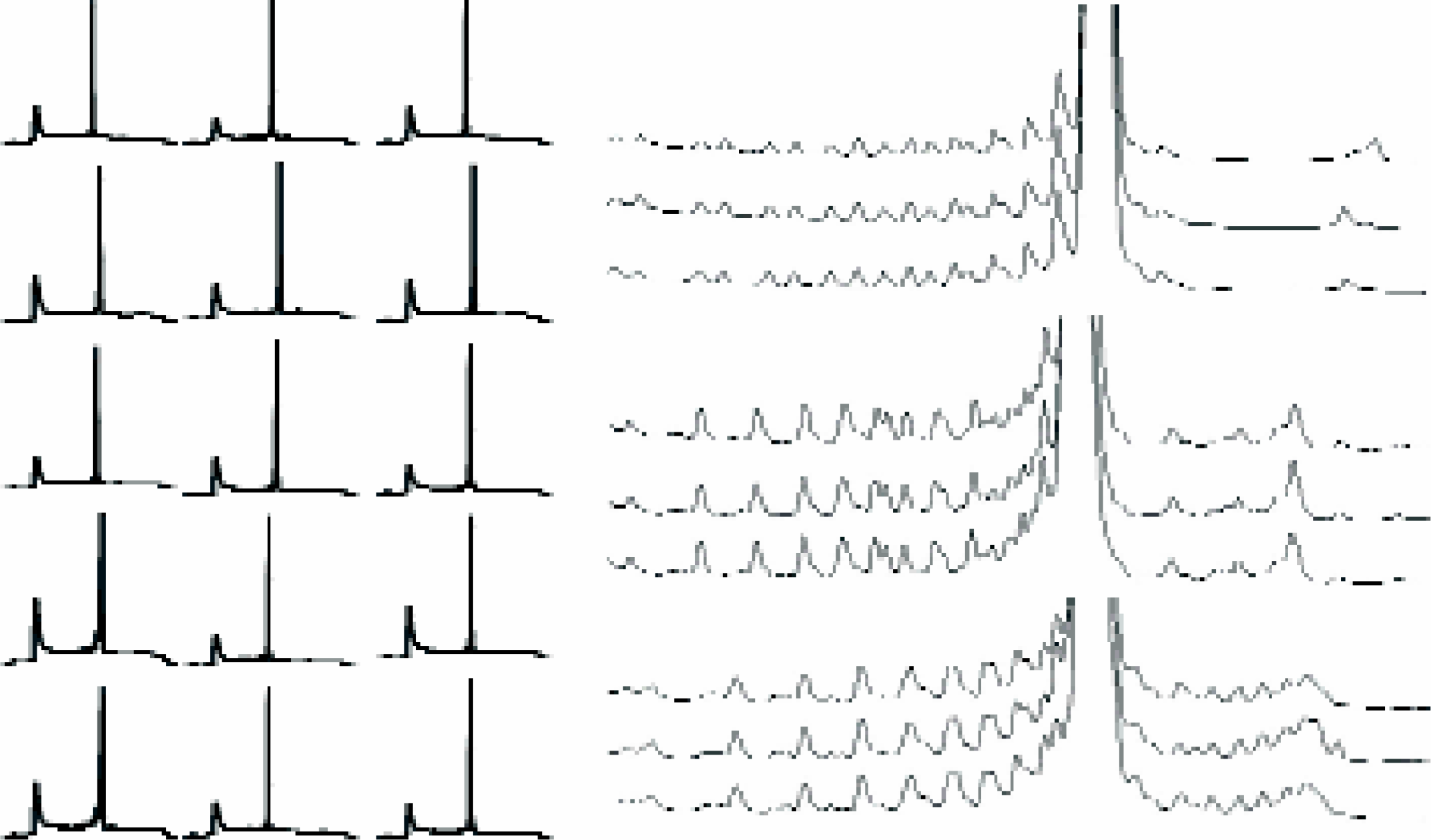
HPLC traces of 25-mers synthesized on Pet Oligator. On the left: the full traces of 15 oligos. On the right: superimposed traces of repeated syntheses of the same oligonucleotides synthesized in the same batch in different positions of the rotor (X scale expanded to focus on the main peak; Y scale extended to visualize the side products.) Note the single base resolution of the HPLC analysis. Stepwise yields: upper traces − 99.3%; middle and lower traces − 99.0%.
Conclusion
Centrifugation can be used as a basis for construction of extremely economical high-throughput solid phase synthesizers. The greatest advantage of centrifugation, in contrast to filtration, is the independence of individual wells in the solvent removal operation, allowing for basically unlimited scaling of the operation. As a result of the efficiencies gained by these synthesizers, Illumina has adopted a price leadership strategy, substantially reducing the cost of oligonucleotides used by researchers.
Acknowledgments
The successful development of these automated synthesizers required a dedicated team of engineers and chemists. I was lucky to work with Christine Burger, Steve Burgett, Brett Ellman, Steve Fambro, Vladimir Gut, John Hachmann, David Heiner, Georges Ibrahim, Aaron Jones, Jan Kalousek, Sam Kim, Viktor Krchnak, Jiri Lebl, Mark Nibbe, Jaylynn Pires, Petr Mudra, Marcel Patek, Vaclav Pesek, David Podue, Vit Pokorny, Pavel Poncar, Marketa Rinnova, Pavel Safar, Chris Santos, Magda Stankova, Alena Stierandova, Sean Touhy, and Karel Zenisek, to name just a few. Without the collaboration of these people on two continents (Prague, Czech Republic, and San Diego, USA), these synthesizers would have remained only a nice idea. However, it was Richard Houghten, with his financial support and Jutta Eichler with her endless enthusiasm, who were courageous enough to jump with me into the adventure called Spyder Instruments, Inc. Projects were financially supported by NIH SBIR Phase I and II Grants #R43 GM61511–01 and #R43 GM58981-01.
