Abstract
Colloidal semiconductor nanocrystals (quantum dots, QDs), such as CdSe-ZnS core-shell, are highly luminescent and stable inorganic fluorophores that represent a promising alternative to organic dyes for a variety of biotechnological applications. They show size-tunable narrow photoluminescence spectra spanning nearly the full visible region of the optical spectrum for QDs with CdSe cores. We have developed several approaches to conjugate either one type or a combination of biologically distinct proteins to CdSe-ZnS core-shell QDs rendered water-soluble by surface ligation with dihydrolipoic acid (DHLA) groups. QD-protein conjugates prepared using these approaches were found to exhibit high specificity and stability in immunoassays and in Förster resonance energy transfer (FRET) assays as well as in prototype QD bioconjugate sensors. Tunable QD emission over a wide range of wavelengths permitted effective tuning of the degree of energy overlap between the QD donor and an acceptor dye, allowing control over the rate of FRET. Additionally, we have used these QD-bioconjugates in live cell labeling. These hybrid bioinorganic conjugates represent a promising tool for use in many biotechnological applications.
Introduction
Colloidal semiconductor nanocrystals or quantum dots (QDs) have several unique optical and spectroscopic properties, including size-dependent tunable photoexcitation and emission with narrow and symmetric luminescence spectra. 1,2 Advances in achieving surface passivation and additional confinement of excitonic states by overcoating the native core with a thin layer of higher bandgap semiconductor improves the fluorescent quantum efficiency substantially, with resulting core-shell QDs having photoluminescent (PL) quantum yields comparable to organic dyes. 3,4 These materials also possess very strong resistance to chemical and photodegradation, far superior to those exhibited by conventional organic dyes. These properties allow QDs to offer a compelling alternative to traditional organic fluorophores in cellular tagging applications and fluoroimmuno- or Förster resonance energy transfer (FRET)-based assays. QD photoemission spectra can be tuned over a broad region of the optical spectrum from near UV to near IR by simple manipulation of either the nanocrystal dimensions for a given material composition and/or modifying the composition of the inorganic core used. 1,2 These broad tunable absorption properties permit simultaneous excitation of several QD populations below the band edge location of the smallest population while generating distinct emission profiles of each population. Furthermore, the broad absorption spectra allow one the flexibility of choosing the desired excitation wavelength in FRET studies (where QDs are used as donor centers in QD-bioconjugate complexes), far away from the maximum absorption peak of the acceptor dye molecules, such that direct excitation of the acceptor molecule is substantially reduced.
We have previously demonstrated that engineered proteins (such as maltose-binding protein, MBP), terminating in an electrostatic leucine-zipper attachment domain (MBP-zb), tightly bind (via electrostatic noncovalent self-assembly) onto the surfaces of QDs capped with a homogeneous shell of dihydrolipoic acid (DHLA) ligands. 5 We subsequently developed various fluorescence-based immunoassays that employ QDs conjugated to a mixture of MBP-zb and another relevant natural or engineered linker protein, such as avidin or engineered Protein G (PG), to prepare mixed surface QD-protein conjugates. The avidin or PG, in turn, bind and link this QD complex to antibodies. 6,7 The MBP functionality facilitates separation of QD-bioconjugates from unbound proteins over an amylose resin column. Recently, we have extended the above conjugation scheme and have shown that MBP proteins expressing C-terminal oligohistidine sequences can also bind to the surface of DHLA-capped CdSe-ZnS QDs, presumably through histidine-Zn interactions, and provide stable and aggregate-free QD-protein conjugates. 8 In this scientific brief, we present a summary of recent examples of how these QD-bioconjugates have been used in four particular areas: fluoroimmunoassays, QD-FRET assays, prototype QD FRET biosensing, and specific labeling of live cells.
Fluoro-immunoassays
Antibody bioconjugates of CdSe-ZnS QDs were prepared using mixed composition protein surfaces containing an MBP-zb for use as a purification tool and a molecular adaptor protein such as avidin or an engineered protein G (PG) derivative (Fig. 1A). The molecular adaptors, PG and avidin, provide specific recognition to either the Fc domain of antibodies or to biotinylated antibodies via avidin–biotin interactions, respectively, while MBP-zb permits removal of any excess unbound antibody through affinity chromatography. 6,7 The engineered PG adaptor protein employs the immunoglobulin G (IgG)-binding domain of streptococcal protein G modified by genetic fusion with the same positively charged leucine zipper interaction domain previously developed and characterized using Escherichia coli MBP. 5 Use of avidin, as an adaptor protein, permits the additional flexibility for coupling QDs to any biotinylated protein. 7,9 These adaptor proteins provide a molecular bridge to successfully attach antibodies to the QD fluorophores and form antibody conjugates that can be used for fluoro-immunoassay development. 6,7 We have employed QDs conjugated to both molecular adaptors, PG-zb or avidin, in direct, sandwich, or competition assay studies to detect toxins, such as staphylococcal enterotoxin B (SEB), cholera toxin, and small molecule explosives. 6,7 The example depicted in Figure 1B describes a fluoroimmunoassay in a competition format for the detection of the explosive RDX [hexahydro-1,3,5-trinitro-1,3,5-triazine], where free RDX molecules added simultaneously with QD-anti-RDX conjugates to the solution compete with BSA–RDX complexes previously immobilized on the substrate. 6,7 Limits of detection for either small molecule explosives or toxins reached using the various assay format were at least as low as what was obtained in similar assay formats employing organic dyes. These data indicate that antigen detection with QDs conjugated to antibodies can be at least as sensitive as detection using antibodies labeled with an organic dye and further confirm that QD-IgG conjugates using the molecular bridging scheme we have developed and refined provide QD-based immuno-reagents that are stable and biologically active. 7,8 Furthermore, QD-avidin complexes prepared as described can be conjugated to nearly all biotinylated antibodies for facile preparation of this fluoroimmuno reagent.
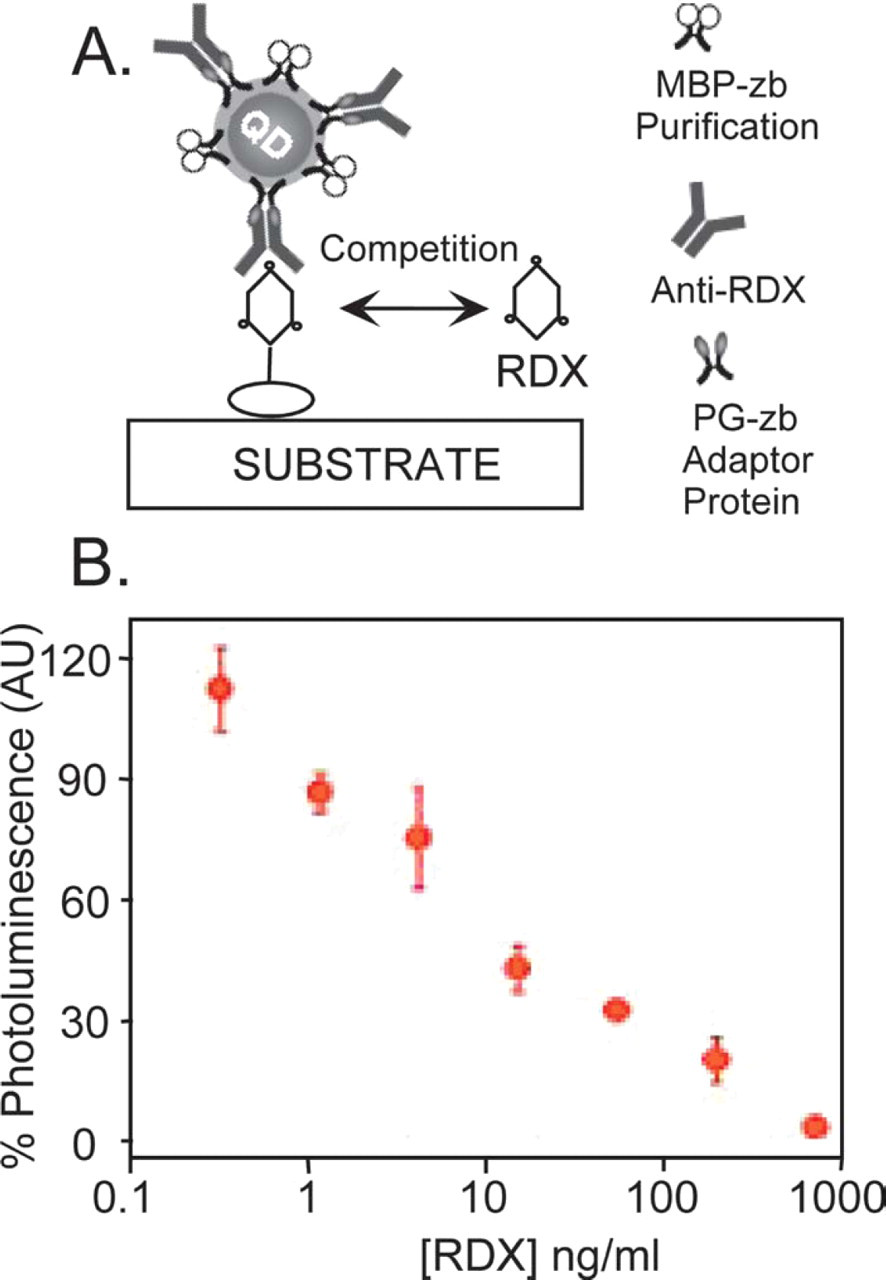
(A) Schematic representation of a competition/displacement assay using mixed surface QD-PG-anti-RDX conjugates in RDX detection. Added free RDX competes with the immobilized RDX on the substrate for binding to QD-anti-RDX. (B) Fluorescence signal (%) measured for increasing concentrations of free RDX added in the assay.
Fluorescence resonance energy transfer in qd-protein conjugates
QD-protein bioconjugates formed using a noncovalent self-assembly scheme were further employed in FRET assays, where the QD served as energy donor to an acceptor dye attached to the protein at a fixed residue along its amino acid sequence. 9,10 This allowed us to explore criteria such as size tuning of the spectral overlap integral and control of the FRET cross section via titration of the number of acceptor dyes interacting with a single QD donor. The latter is achieved by varying the number of dye-labeled proteins attached to the QD surface for a given total number of proteins in each QD-protein conjugate. 10 Samples consisted of Cy3-labeled MBP proteins that had been immobilized onto the QDs via the C-terminal-polyhis, and control of the number of dyes interacting with a single donor was realized by changing the dye-labeled protein to unlabeled protein ratio while keeping the total number of proteins attached to each QD constant during the initial conjugate prepation. 8,10 Using this conjugate configuration, where a single donor is made to interact with several acceptors placed at equal separation distance from the conjugate center (Fig. 2), we observed a progressive and substantial enhancement in the energy transfer efficiency with increasing number of MBP-Cy3 attached to a single nanocrystal for a given conjugate and a given population of QDs (thus a given emission spectrum). 10 Figure 2 shows a steady and progressive quenching of the QD emission coupled with a steady increase in the Cy3 PL with increasing number of dyes per conjugate for 530 nm QDs conjugated to a mixture of MBP and Cy3-labeled MBP (contribution from direct excitation to the dye emission is minimal compared to the overall signal resulting from FRET). Furthermore, we found that FRET efficiency varied with the degree of spectral overlap. Conjugates made with QDs having a larger spectral overlap integral with the acceptor provided proportionately more efficient energy transfer or QD quenching. Using Förster theory, we extracted measurements of the average donor-acceptor separation distance for each QD-dye pair, and found that, in general, distance measurements varied proportionally with average QD core size from one set of conjugates to another. The total number of unlabeled and dye-labeled proteins around the QD donor in each titration is unchanged. 10 These results provided an in-depth analysis of a QD-protein bioconjugate FRET system and demonstrated the utility of QDs as efficient FRET donors, where the ability of tuning (via size) the nanocrystal photoemission properties allowed for efficient energy transfer with a conventional organic dye. Furthermore, the large size of QD fluorophores compared to organic dyes allowed design of QD bioconjugates where multiple acceptor dyes could coordinate around and interact with a single QD donor; the resulting enhancement of FRET suggests that this strategy can improve assay sensitivity. 10 This also suggests the possibility of realizing multicolor multiple analyte detection using a single QD donor and multiple acceptors in a FRET assay format.
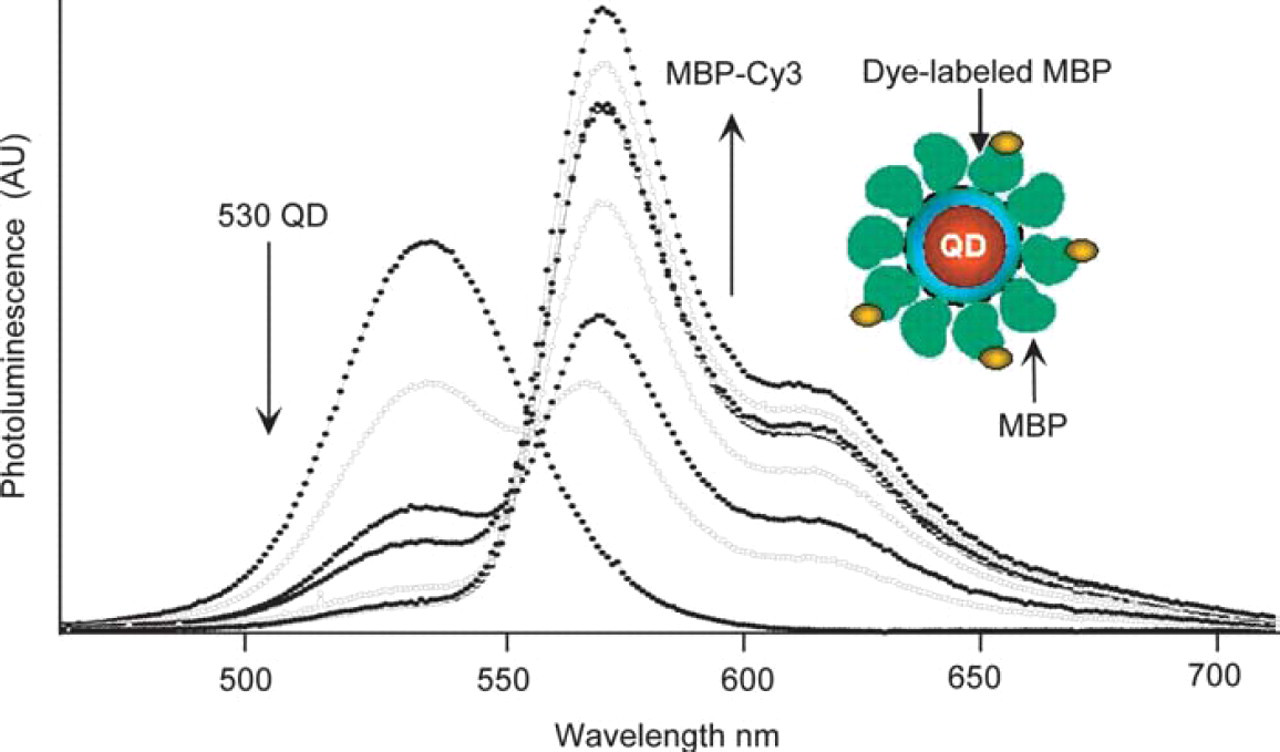
530 QD-MBP-Cy3 FRET. Photoluminescence (PL) spectra of a QD donor—multiple acceptor titration. 530 QDs were conjugated with an increasing ratio of MBP-Cy3 (Absorption max ∼550 nm, emission max ∼570 nm), and PL was measured at each MBP:MBP-Cy3 ratio. Total number of MBPs is kept constant. Ratios of MBP:MBP-Cy3 displayed: 10:0, 9:1, 8:2, 7:3, 6:4, 3:7, and 1:9 (Direct Cy3 dye excitation not subtracted). Inset shows a schematic of a single QD surrounded by 10 MBPs, three of which are dye-labeled. This represents a QD surrounded by both dye-labeled and unlabeled proteins.
Prototype QD fret biosensors
We utilized the insights gained from the studies of FRET in QD-protein-dye bioconjugates described above, namely, the constraints imposed by distance and overlap requirements and the advantage of using conjugates, where several acceptors interact cooperatively with a single QD donor to achieve efficient FRET signal, to build a prototype QD nanoscale sensing assembly. This sensing assembly employed QDs as energy donor and functioned in sugar detection. 6,10 Figure 3A schematically depicts how this prototype FRET sensor functions. Approximately 15 to 20 MBPs are first immobilized on the surface of each QD via His-Zn coordination. The QD-MBP-His conjugates are then incubated with a solution of β-cyclodextrin (β-CD) adduct covalently labeled with a QSY-9 quencher, which results in the capture of QSY9-labeled β-CD within the centrally located sugar binding pocket of these MBP moieties because of the favorable interactions between MBP and β-CD. 8,11 The absorption of this dark quencher has an optimal overlap with the QD emission at 555 nm. Since the β-CD-QSY9 is located in the MBP binding pocket, its separation distance from the QD center is well within the range of effective FRET and is thus able to induce a substantial concentration-dependent quenching of the QD emission. Figure 3B shows homogenous sensing of this solution phase QD biosensor, where added maltose displaces the β-CD-QSY9 complex, disrupting FRET quenching in a concentration-dependent manner. An apparent maltose binding constant (Kapp) of ∼7.0 μM can be derived from the above titration curve with maltose. When this sensing assembly was tested against several different sugars, only oligosaccharides containing α-1,4-linked glucosyl units elicited a response, indicating that QD-MBP conjugate assembly retained its functional specificity. 6 These results showed that QDs can be exploited as FRET donors for sensitive and specific biosensing. 6
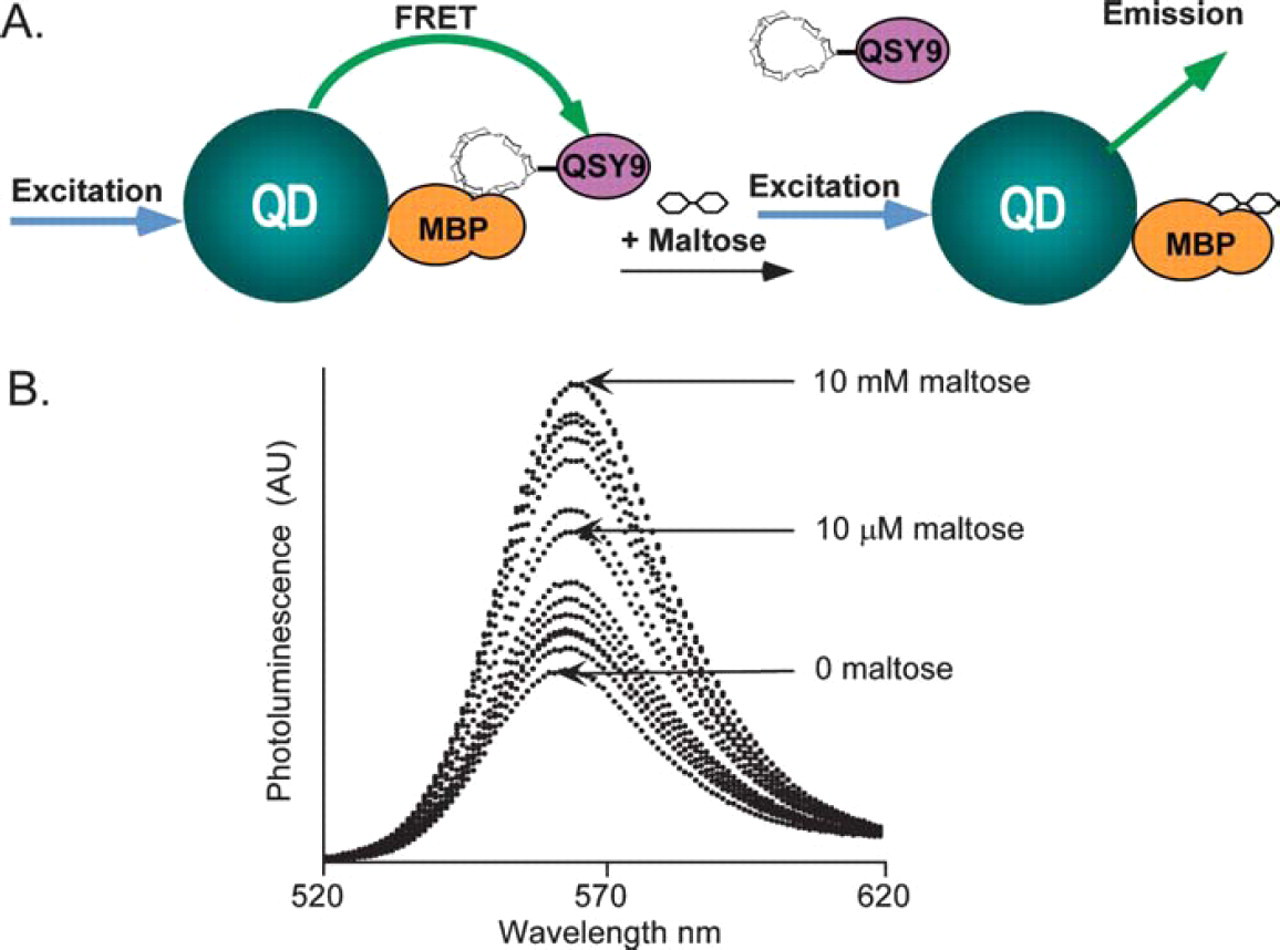
(A) Prototype QD-based sensor function. A 555-nm emitting QD is surrounded by ∼15–20 MBP molecules. MBP coordinates to the QD via His-Zn. Combining β-CD-QSY9 (absorption max. ∼565 nm) with MBP forms the QD-MBP-β-CD-QSY9 sensor complex, which quenches the QD-MBP emission (indicated in green). Added maltose displaces the β-CD-QSY9 away from the QD-MBP sensor. This displacement causes an increase in QD emission due to recovery of the QD PL (blue arrow indicates excitation). (B) Titration of a 555-nm emitting QD-MBP conjugate preassembled with β-CD-QSY9 (∼10-fold molar excess) with increasing concentrations of maltose. An apparent Kd of ∼7 μM for maltose can be estimated from this set of data.
Cellular Labeling
QDs capped with DHLA ligands were used in live cell-imaging studies conjugated to antibodies via molecular bridging proteins. We have also developed a generalized labeling scheme that allows one to tag any cell capable of endocytosis. 12 Cells can be loaded with QDs using two different strategies. Incubation with DHLA-capped QDs allowed cells (mammalian HeLa and Dictyostelium discoideum AX2) to endocytose QDs present in the growth media. QD-avidin or QD-PG conjugates were also used to specifically label cells and regions of cells. For example, we have been able to demonstrate labeling of specific proteins on the surface of HeLa cells by exposing the cell surfaces to a biotinylatied antibody (anti Pgp) followed by incubation with preassembled QD-avidin conjugates (Fig. 4). In particular, in a mixed population of cells expressing or not expressing the green fluorescent protein-tagged P-glycoprotein (Pgp-GFP) on their surface, only the cells that express Pgp-GFP were labeled with the avidin conjugated QDs. No labeling was observed for the cells that did not express Pgp-GFP. 12
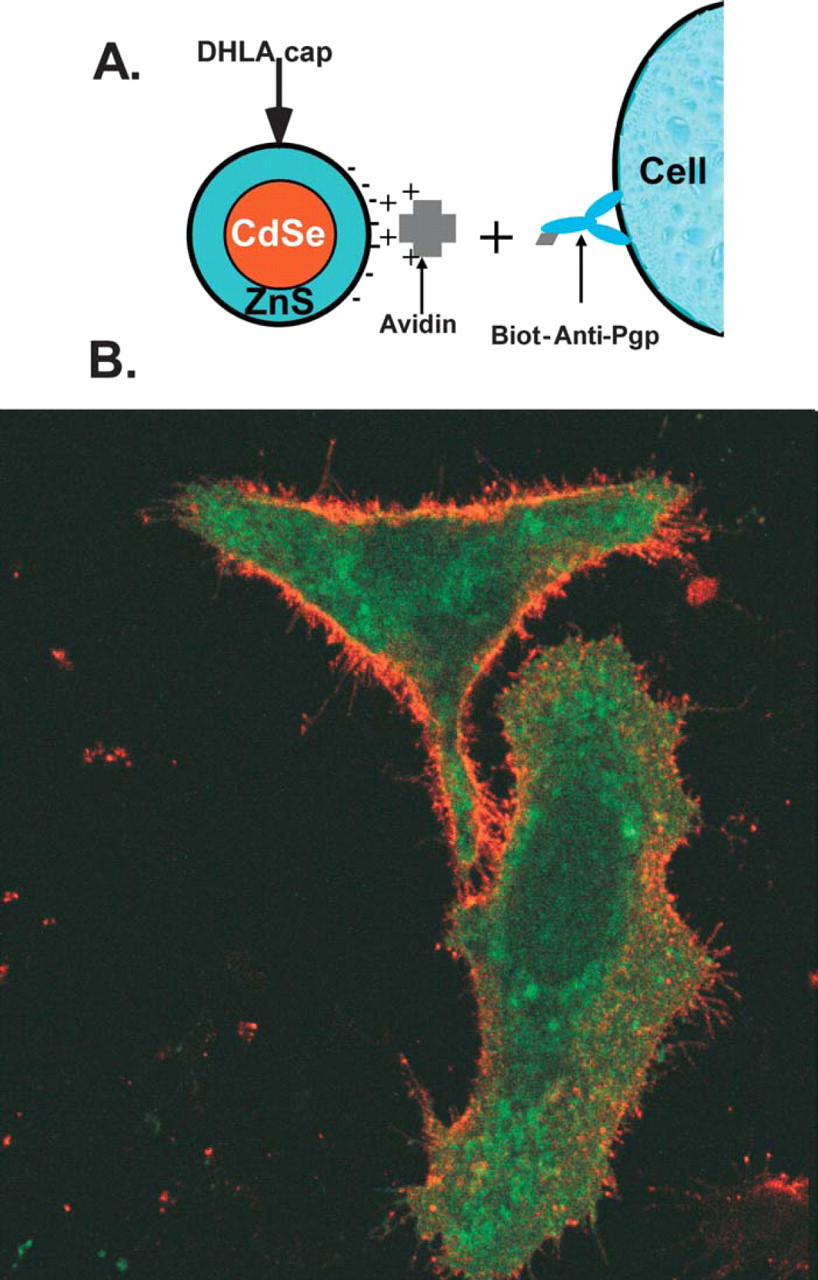
Overlay of Pgp-GFP and QD fluorescence images displaying HeLa cells expressing P-glycoproteins fused to green fluorescent proteins (Pgp-GFP) incubated for a short period with biotinylated anti-Pgp antibody and QD-avidin conjugates, respectively. Rinsing steps of excess unbound antibody and QD-avidin conjugates were carried out between incubations. Images were taken from fixed cells. Images clearly indicate that while green emission characteristic of Pgp-GFP is homogeneously distributed inside the cells; orange QD emission is limited to the cell membranes.
Either approach of labeling cells (via antibody or endocytosis) allowed effective labeling of cells with QDs. 11 We further investigated effects of loading cells with QD fluorophores on the growth and development of cell. We could image HeLa cells labeled with QDs as they grew for up to 2 weeks. Additional investigations of QD-labeled live cells indicated that development, differentiation, signaling, and chemotaxis of QD-labeled cells was indistinguishable from that of unlabeled cells. 12 We were also able to use multiple color QDs to fluorescently tag and simultaneously image individual cells over long periods. These methods of labeling cells with QDs thus open up the possibility of using the nanocrystals for a variety of biological applications requiring specific and generalized labeling of cells and proteins.
Conclusions
In this scientific brief, we have shown examples of how QD-protein bioconjugates can be applied to a variety of biolabeling techniques, both in active or passive roles. Examples employing QD-antibody conjugates in fluoroimmunoassays to detect target analytes and for specific labeling of live cells were presented. In addition, we showed the design and implementation of a simple solution phase FRET-based nanoscale sensing assembly using QDs as energy donors. These initial results indicate that incorporation of QDs into biological labeling techniques can overcome many problems inherent in using organic dyes. If fully exploited, the unique spectroscopic and physio-chemical properties of QDs will facilitate numerous multiple analyte format studies not only in in-vitro assays, but also in biosensing and cellular labeling.
Acknowledgments
We thank H. Hellinga (Duke University) for providing the MBP plasmid. ARC and ILM acknowledge National Research Council Fellowships. JKJ and SMS acknowledge grants NSF BES 0110070 and NSF BES 0119468 for financial support. JMM and HM thank K. Ward at the Office of Naval Research (ONR) for research support, grant N001400WX20094. The views, opinions, and/or findings described in this report are those of the authors and should not be construed as official Department of the Navy positions, policies, or decisions.
