Abstract
In chemical development, broad knowledge of solubilities of all reactants, reagents, and products is important because these data are needed for cleaning operations of multipurpose equipment in pilot and production plants. A fully automated workflow on a high-throughput robotic system for determination of solid material solubility in different aqueous and organic solvents is described. Automated solid dispensing, weighing, and solvent addition are performed followed by direct measurement of turbidity with an integrated three-wavelength turbidity probe. Finally, a report is generated containing all solubility information of the solid in a clearly arranged manner. (JALA 2005;10:408–11)
Aqueous solubility of drug candidates plays a major role in bioavailability of drug substances in humans. 1 There are numerous known methodologies for solubility determination such as turbidimetry, 2 nephelometry, 3 ultrafiltration, 4 direct UV, 5 or modified equilibrium method. 6 Methods like nephelometry 7 and direct UV 8 are suitable for high-throughput solubility determination on commercial 96-well plates. Equipments used to determine aqueous solubilities are not necessarily appropriate to measure solubilities in organic solvents due to incompatibility of the hardware. In (early) chemical development, solubility of solids especially in organic solvents is of high interest. Therefore, a different hardware setup is necessary.
Pilot and production plants generally are equipped with multipurpose devices. Cleaning of pilot and production plant equipment has to be in accordance with regulatory requirements in the pharmaceutical industry. 9 For cleaning purposes, it is necessary to determine the solubility of all reagents, reactants, and products employed in the chemical transformation to guarantee an optimal washing of the contaminated equipment. The cleaning of the equipment before and after the reaction is of main importance to guarantee the quality, purity, and yield of the process – thus avoiding “out of spec” results and cross-contamination. Preferentially neutral, acidic, or basic pH waters are the solvents of choice for environmentally friendly equipment cleaning but in special instances also more hazardous organic solvents such as acetone, acetonitrile, tetrahydrofurane as well as dichloromethane and dimethylformamide are used. Normally, solubilities > 100 mg/mL are selected for optimal equipment cleaning. Other selection criteria are recyclability, price, and environmental friendliness.
In 2002, a new high-throughput robotic system (from Zinsser Analytic 10 ) for fast reaction screening and optimization was set up in our laboratories consisting of three connected automated platforms. Up to seven reaction blocks (8–96 reaction vessels per block) can be handled simultaneously giving a large number of possible experiments for process development and optimization. 11 A part of the fully automated system is the solid handling and reaction preparation machine called CALLI (Fig. 1). This platform includes two solid dispensing tools (VARIX), a balance, liquid handling syringes, and needles as well as diverse gripping tools for transportation of different sized vials. We wondered whether it could be possible to use this part of the HTS system for an automated determination of solubility of all intermediates, reagents, and products within process research and development.
CALLI: solid dispensing with VARIX tool on the balance.
For weighing solid materials, a vial is transferred by the appropriate gripping tool onto the balance and tared. Next, the solid is picked up by vacuum from a storage container by the solid dispensing pipette and delivered into the vial on the precision balance. A special feature of this VARIX pipette is the varying volume size, which is controlled by a step motor. After determination of the weight of dispensed materials, the software starts a correlation between pipette volume and dispensed quantity of solid. Finally, the pipette volume is changed to reach the desired amount of solid as fast as possible. Good solids with proper attributes (density, dryness, etc.) can be distributed with a tolerance of ±2 mg.
For solubility measurement, an Accelab turbidity probe was integrated. 12 This probe uses glass fiber optics to measure reflection of light for three different wavelengths (red, yellow, and blue, Fig. 2) and moves through the sample solution. That means, solubility is measured indirectly: more turbidity results in more reflection and greater signal intensity. By comparison of the pure solvent absorbance spectra with a sample mixture, the final turbidity is determined. The Accelab Sensor checker software records all data sent by the turbidity probe and generates three possible results: clear, turbid, or uncertain mixture of the solid in the solvent.
The three-wavelength probe in action.
There are two criteria for assigning a sample as a suspension: all three spectra deviate by greater than 40% from the pure solvent spectrum and at least one spectrum deviates by greater than 80%. In the case that only two light signals have more than 40% deviation, the mixture is identified as “uncertain”, thus a visual control is necessary. All other cases are identified as a clear solution. The overall success rate of the probe with these defined limiting parameter values is greater than 90%. Occasionally problems occur with bigger colorless crystals, which are not always detected by the probe.
Therefore, the described Zinsser High-Throughput System offers an ideal platform for automation of the solubility determination of solids. The Zinsser software is highly flexible, and a fully automated program for solubility determination was developed.
According to our department's internal protocol for solubility determination, discrete amounts of solid (50, 100, 250, 500, and 1000 mg) are dispensed with the VARIX tool into 4 mL glass vials (Fig. 1), and the true weighed amount of material is tracked in the database.
Then, the software does a correction for liquid solvent transfer if the amount of solid deviates from the desired amount. Two available liquid handling needles add the corrected volumes of solvent followed by a 5 min vigorous automated shaking on a vortexer of the vials yielding mainly kinetic solubility information. Routinely the screening includes 8–10 different solvents (see Fig. 4) giving a total number of 40–50 samples (8–10 solvents × 5 weighted samples) in one robotic run. The maximum number of samples is currently limited to 60 due to the rack format. Volatile solvents are placed in the first line to avoid evaporation of solvent out of the open vials. This instance limits the application of this methodology to ambient temperature. But in general, solubility of solids increases at elevated temperature, and pilot plants are normally “boiled out” with hot solvent to maximize effectiveness. Next, the handler picks up the turbidity probe and starts measuring all mixtures and saves the turbidity results in the raw database (Figs. 2 and 3). The turbidity measurement takes place without further shaking of the samples. The whole procedure including weighing, solvent distribution, and turbidity measurement, takes about 3 h and is performed overnight.
Raw data acquisition of the Accelab turbidity probe during measurement. Report generation: final document for the batch record preparation.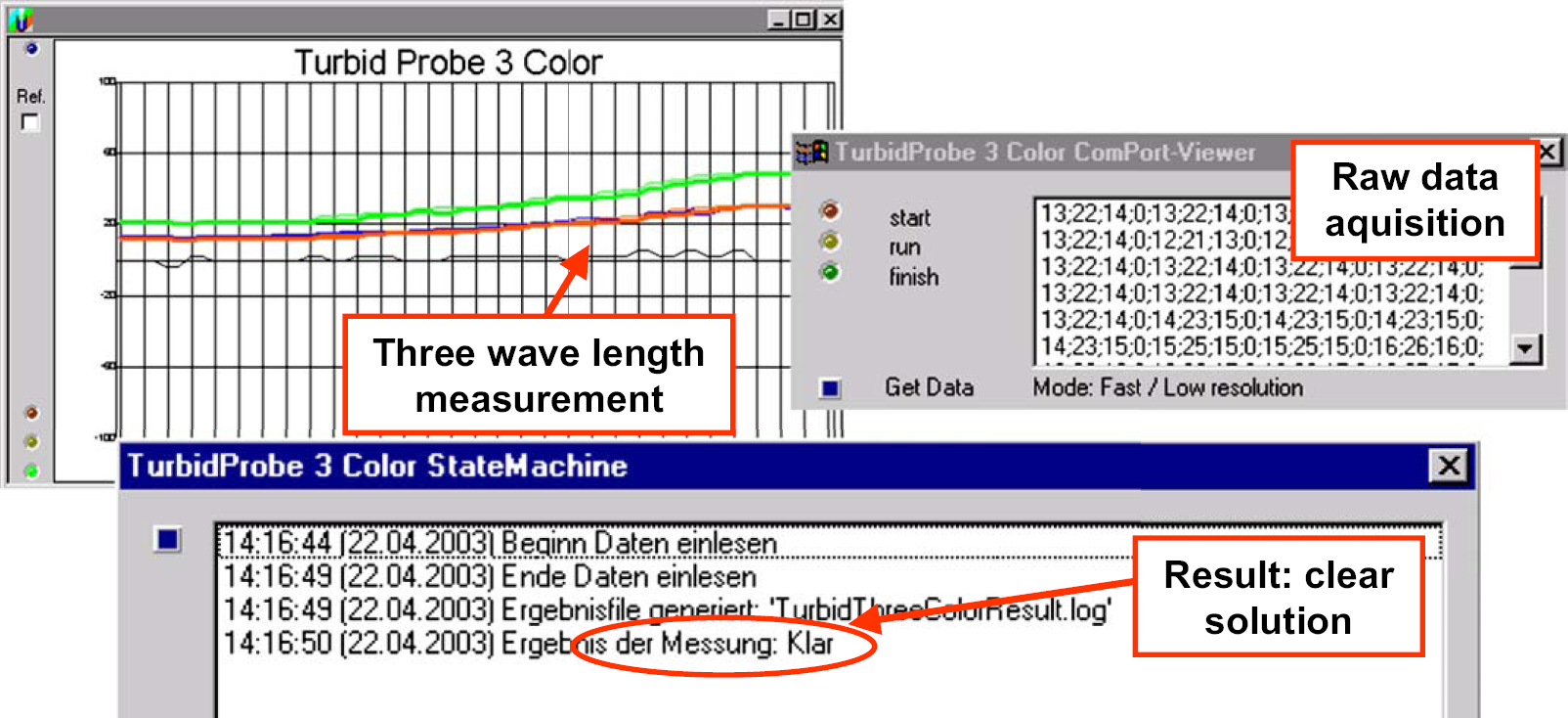
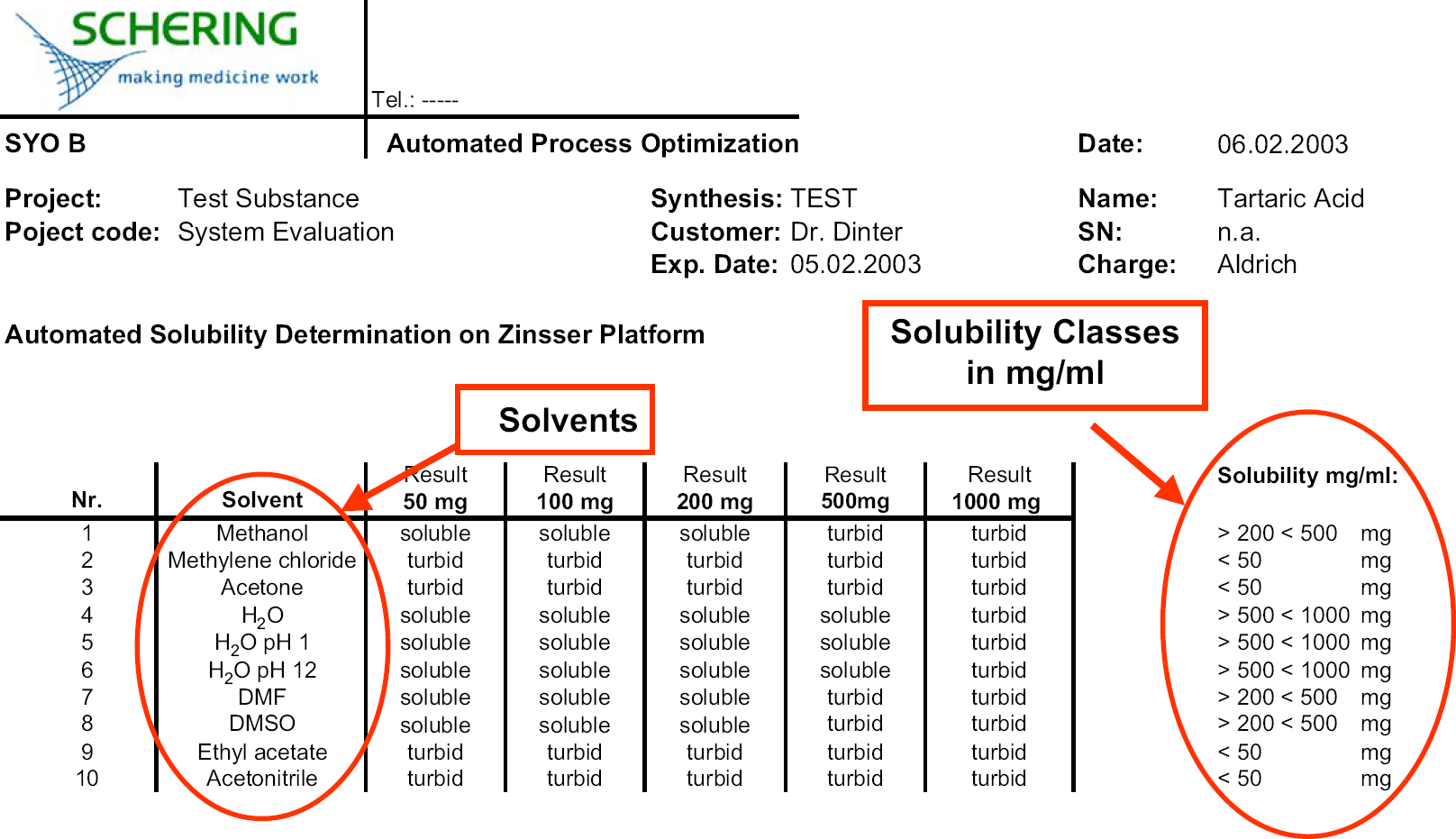
The raw data from the CALLI database (including sample number, concentration, solvent, recording date, and time, as well as the absorbance values for all three wavelengths) are exported into an Excel worksheet. Using a customized Excel macro, the raw data set is extracted and imported into the final report file. The Excel sheet is the final report with detailed solubility information on each of the 10 solvents and directly created after raw data import. Finally, the name of the material, internal registration number, and batch number are added into the document, and the report is sent to the pilot plant chemist for batch record preparation (Fig. 4).
This methodology was applied very successfully on some substances used for system testing, as well as on many reaction intermediates and reagents used for production purposes. For example, the solubilities of tetrazole, anthracene, fluorobenzene derivatives, aminobenzenesulfonamide, diverse pyrimidines, and steroids (a core business of Schering AG) were determined. In special cases, information about solubility in less commonly used solvents (e.g., dichloroethane, dimethylformamide, benzotrifluoride, glyme) is also of interest. Easily, additional solvents can be added in the screening (e.g., heptane, methyl tert-butyl ether, octanol). In addition, this technique is easily extendable to liquid mixtures (e.g., water/methanol) for determination of precipitation properties. Finally, results of some investigations were compared with those obtained from manual operations, and no major deviation was found.
In conclusion, the determination of solubility for cleaning of pilot and production equipment offers an ideal field for automation. Time consuming manual operations by lab technicians are avoided. Overall the results are very reliable, and the gained information about the solid materials is of high value for process and pilot plant chemists. Future steps will be the introduction of bar coded raw material containers to further reduce manual work and mixing up of data on the final document. Furthermore, a decrease in the amount of needed material by miniaturization is planned.
Footnotes
Acknowledgement
We thank Claudia Merten and Thomas Wessa for realization of the automation project at Schering as well as Evelyn Adam for excellent preparative work and technical assistance with the Zinsser HTS robot. Ursula Ganzer is acknowledged for fruitful discussions and information about solubility measurements in general. We also thank all people from Zinsser and Accelab who were involved in the realization of this project.
