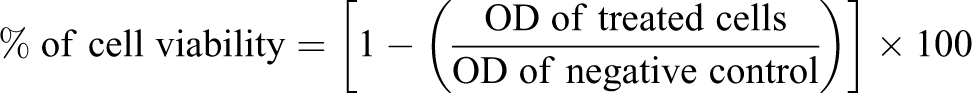Abstract
Rutin has been enzymatically oligomerized by laccase from Trametes versicolor. Five fractions of oligomers were obtained from the monomers having high solubility in water, which can reach 351-times that of rutin. Cytotoxicity of rutin and oligorutin fractions was evaluated towards K562 cells. Oligorutin fractions showed a lower antiproliferative effect compared with its monomer. The genotoxic potential of rutin and oligorutin fractions was assessed, at the limit of the solubility of each molecule, using the comet test. None of the tested concentrations of either rutin or oligorutin fractions has showed a genotoxic effect. Similarly, the antigenotoxic effect of these flavonoids was tested using the same assay. The obtained results showed a higher ability of oligorutin fractions to reduce the genotoxicity induced by hydrogen peroxide compared with monomeric rutin.
Introduction
Flavonoids attracted researchers, food manufacturers and consumers due to their antioxidant properties as well as their role in the prevention of various diseases such as cancer and cardiovascular diseases. 1,2 Recently, attention has been focused on the identification of natural products exhibiting antigenotoxic and/or anticarcinogenic effects. 3 –7 The genotoxic and antigenotoxic activities of monomeric flavonoids were reported by some authors. 7 –10 However, few studies have focused on flavonoid oligomer antigenotoxicity. Dhawan et al. 11 studied the antigenotoxic effects of monomeric tea polyflavonols (catechin, epicatechin, epicatechin gallate, epigallocatechin and epigallocatechin gallate) and their dimers using the comet assay. These authors have reported that neither the monomeric nor the dimeric flavonols can reduce the lymphocyte DNA damage induced by the 3-amino-1-methyl-5H-pyrido[4,3-b]indole. In the case of rutin, one of the major representatives of flavonoids, it has been demonstrated that it caused cell cycle arrest and induced apoptosis in many types of human cancer cell lines. 5 –12 The oligorutins, synthesized enzymatically via oligomerization reactions of rutin, have high biological activities than rutins. 13 –15 In fact, rutin oligomerization by laccases from Pycnoporus coccineus and Pycnoporus sanguineus led to oligomers with improved superoxide radical activity. 15 Moreover, oligorutins obtained by laccase from Myceliophthora, compared with its monomer, present a better inhibition of 2,2′-azobis (2-amidinopropane) dihydrochloride radical that is responsible for cell damage. 14 In a recent work, we have shown a high inhibitory effect of oligorutin fractions against the mutagenicity induced by methylmethanesulfonate and 2-aminoanthracene, using the ‘Ames’ assay’ with two Salmonella typhimurium strains TA 104 and TA102. 16
Previous data have shown that rutin and its oligomers have an interesting activity like antioxidant and anticarcinogenic effects. To complete these data, we have investigated, in this article, the cytotoxicity of oligorutins against K562 cells on the one hand and their genotoxic and the antigenotoxic activities in a eukaryotic system using the comet test on the other hand. The obtained results were also compared with those obtained with rutin.
Methods
Obtention of oligorutin fractions
Oligorutin fractions were obtained and characterized as described by Anthoni et al. 13,17 and by Ben Rhouma et al. 16
Solubility study
The maximum solubility of rutin and oligorutin fractions was evaluated at 20°C in water. Monomer and oligomers (in excess) were added to 50 ml of water, and the reaction media were stirred in agitated reactor at 600 r/min at a constant temperature.
Samples of the solution were withdrawn at several intervals and placed in vials at the same temperature as the solution temperature in order to avoid any precipitation. The samples were then filtered (0.22 μm) and analyzed by SEC-UV (HPLC LaChrom, UV 280 nm LaChrom L-7400, Tosoh TSKgel α 3000 column, 60°C). In all cases, it was found that after 9 h of stirring, the equilibrium of dissolution was reached.
Cell culture
The human chronic myelogenous leukaemia (CML) cell line (K562) was maintained in RPMI 1640 (Invitrogen Life Technology, Cergy Pontoise, France) medium supplemented with 10% foetal bovine serum,
Assay for cytotoxic activity
To control whether the tested concentrations of rutin and its oligomer fractions impacted cell viability, we estimated the viability of K562 cells by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT; Sigma Aldrich, Courtaboeuf, France) assay, which is based on the cleavage of a tetrazolium salt by mitochondrial dehydrogenase in viable cells. The in vitro test against the human CML cell line K562 was performed essentially according to the method described previously. 18 Cells were seeded in 96-well microtitration plates and incubated overnight. The test samples were dissolved and diluted in RMPI. Twenty four hours after seeding, 100 µl of 1% dimethyl sulfoxide (DMSO; Sigma Aldrich, Courtaboeuf, France) in new media or test compounds were added before incubating the plates for a supplementary 48 h. Cells were washed once before adding 50 µl foetal bovine serum-free medium containing 5 mg/ml MTT. After 4 h of incubation at 37°C, the medium was discarded and the formazan blue formed in the cells was solubilized in 50 μl DMSO. Negative control without the tested compound but with DMSO was treated in the same manner. Optical density was measured at 540 nm. The cytotoxicity index (%) was calculated according to the following equation 19
Comet assay
The comet assay with human lymphocytes was used to detect DNA damage. Before each experiment, frosted microscope slides were precoated with two layers (100 μl) of normal agarose (1% in milli-Q water) and left at room temperature to allow agarose to dry. In order to evaluate whether rutin and its oligomer fractions exhibited or not a genotoxic effect on human lymphocytes, cells were treated during 24 h with different concentrations of the tested substances (100, 200 and 400 µg/ml). They were harvested in phosphate-buffered saline (PBS). The cell dilution (5 × 105 cells in 60 μl) was mixed with an equal volume of low-melting-point agarose (1.2% in PBS). This agarose cell suspension (120 μl) was spread onto each precoated slide and covered with a cover slip. After 10 min on ice, the cover slip was gently removed and the slides were placed in a tank filled with the lysate buffer (2.5 M NaCl, 100 mM EDTA, 10 mM Tris-HCl, 1% sodium sarcosinate pH 10, 1% of Triton X-100 and 10% DMSO). They were immersed in this buffer for 1 h (at 4°C, in the dark). The slides were then transferred into the electrophoresis buffer (NaOH 10 N, EDTA 200 mmol/l, pH 13 in deionized water) during 20 min at room temperature in the dark. Electrophoresis was carried out for 15 min at 25 V and 300 mA. Finally, the slides were gently rinsed with neutralization solution (0.4 M Tris-HCl, pH 7.5) for three times of 5 min each. Staining of DNA was accomplished using 50 μl of ethidium bromide (Sigma Aldrich, Courtaboeuf, France) solution at 20 μg/ml in PBS per slide. 20 The slides were examined using an epifluorescence microscope (Zeiss Axioskop 20; Carl Zeiss, Microscope Division, Oberkochen, Germany).
In order to study the antigenotoxic effect of rutin and its oligomer fractions, cells were treated in the same manner as for genotoxic study and with the same concentrations, except an additional step with the tested samples after 24 h of incubation, which consists of stressing the treated cells with 75 µM hydrogen peroxide (H2O2) during 5 min in ice.
Quantification of the comet assay
A total of 100 comets on each scored slide for each concentration of each molecule were visually scored according to the relative intensity of fluorescence in the tail and classified as belonging to one of the five classes. We utilize three slides for each concentration of each sample, and the experiments were repeated three times. Each comet class was given a value of 0, 1, 2, 3 or 4 (from undamaged (0) to maximally damaged (4)) as described previously by Collins et al. 21 The total score of DNA damage was calculated by the following equation
Total DNA damage (TDD) = (percentage of cells in class 0 × 0) + (percentage of cells in class 1 × 1) + (percentage of cells in class 2 × 2) + (percentage of cells in class 3 × 3) + (percentage of cells in class 4 × 4).
Consequently, the total score was ranging from 0 to 400.
The inhibition percentage of tail DNA (%) was calculated relative to DNA damage in the control group cells treated with H2O2 only, using the following formula
The inhibition percentage of tail DNA (%) = [1 − (TDD of cells treated with the sample and H2O2 − TDD of untreated cells)/(TDD of H2O2 treated cells − TDD of untreated cells)] × 100
Statistical analysis
Data were collected and expressed as a mean ± SD of three independent experiments. Statistical analyses were performed by means of analysis of variance test followed by Duncan’s multiple comparison test using Statistica (Version 6.0, Statsoft, Inc., Maisons-Alfort, France) to compare data on DNA damage in cells treated with different concentrations of the tested samples and the genotoxic agent H2O2 in the antigenotoxic assay. In the genotoxic assay, damage data of cells treated with different concentrations of rutin and its oligomer fractions were compared with those of the negative control (untreated cells). Statistical differences were determined at p < 0.05.
Results
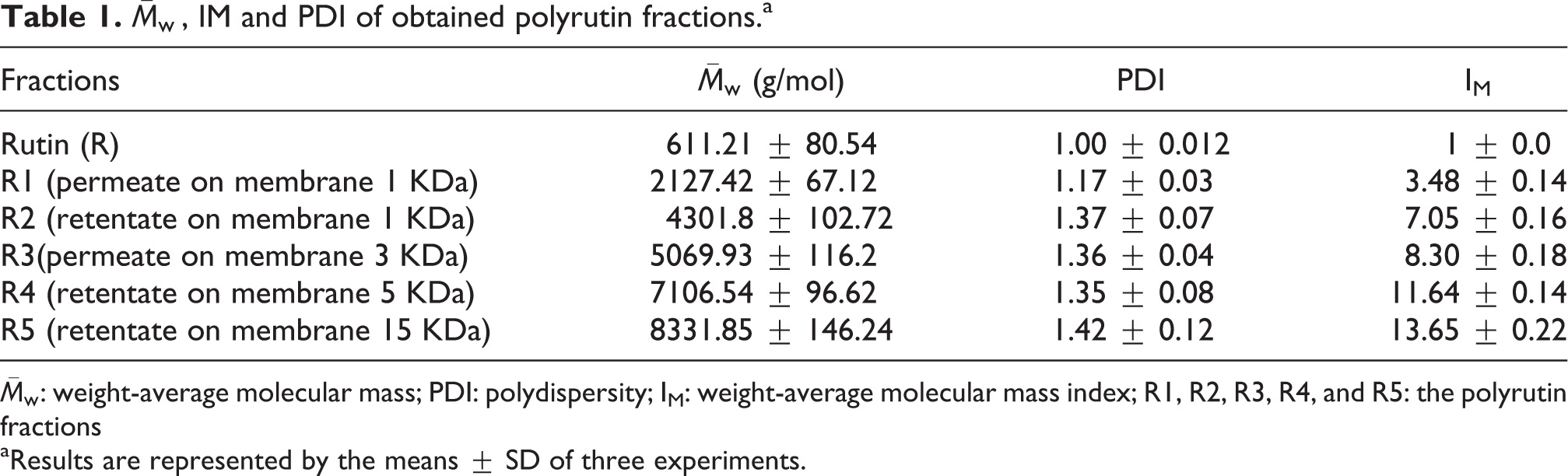
Oligorutin fractions obtaining
Solubility study
The dissolution kinetics of rutin and its oligomer fractions presenting different weight-average molecular mass (

Pictures of different classes of comet in human chronic myelogenous leukaemia K562 cells after treatment with different tested extracts. These comets illustrate examples of the visual scoring classification.
aResults are represented by the means ± SD of three experiments.
Cell viability assay
The relationship between the concentrations of tested flavonoids and their effects on K562 cell viability were investigated by MTT assay. MTT is a yellow water-soluble tetrazolium salt. Metabolically active cells are able to convert the dye to water-insoluble dark blue formazan by reductive cleavage of the tetrazolium ring. Cells were treated with rutin and oligorutin fractions (R2, R3 and R5) at the concentrations ranging from 50 to 800 µg/ml for 48 h. The results are summarized in Figure 2. Oligorutin fractions exhibited low inhibition of K562 cells viability, which did not exceed 32% at the highest tested dose (800 µg/ml) compared with rutin, which showed a considerable cytotoxicity towards K562 cells at the highest tested dose (800 µg/ml) with a half maximal inhibitory concentration (IC50) of 400 µg/ml. We observed a weak relationship between the inhibition of K562 cell viability and the molecular weight of oligorutin fractions (Figure 2). However, rutin and its oligomer fractions inhibit K562 cells viability in a dose-dependent manner.

Solubility of rutin and oligorutin fractions in water. R2: permeate obtained after diafiltration of oligorutin on a 3-kDa membrane; R3 fraction: permeate obtained after diafiltration of oligorutin on a 5-KDa membrane; R5: retentate obtained after diafiltration of oligorutin on a 15-KDa membrane. Values were expressed as mean ± SD of three experiments. *p < 0.05 as compared to rutin using ANOVA followed by Duncan’s multiple comparison test. ANOVA: analysis of variance.
Comet assay
The induction of DNA damage in human leukaemia K562 cells after exposition to different concentrations of rutin and oligorutin fractions (R2, R3 and R5) for 24 h was studied using the comet assay. This method was reported to be sensitive for detecting DNA strand breaks in individual eukaryotic cells. 4 Quantification of the comet data is reported as TDD in Table 2. No significant difference was detected between the TDD obtained by incubation with the monomer or its oligomer fractions (TDD ranges from 106 to 110.5) at any tested concentrations (from 50 to 400 µg/ml) on one hand, and the negative control (vehicule-treated cells; TDD = 108) on the other hand. In contrast, a significant increase in the TDD (TDD = 223.5) was observed with the positive control corresponding to K562 cells exposed to 75 µM of H2O2 compared with the vehicle-treated cells. This result indicated that tested molecules induced no genotoxicity in K562 cells. The alkaline single-cell gel electrophoresis (comet) assay was also performed in order to elucidate the antigenotoxicity potential of the rutin and its oligomers fractions, against H2O2-induced DNA damage in K562 cells. It appears that oligorutin fractions were more effective in reducing the genotoxicity induced by 75 µM of H2O2 than the corresponding monomer. Rutin decreased the TDD of H2O2-stressed cells in a dose-dependent manner, with a maximum inhibitory percentage of 26% at the highest tested concentration (400 µg/ml). Likewise, R2, R3 and R5 fractions decrease the TDD of H2O2-stressed cells to an extent of 39, 45 and 60%. It appears that R5 fraction is the most efficient in reducing H2O2-induced DNA damages on human lymphocytes (Figure 3).

Effect of different concentrations of rutin and oligorutin fractions (R2, R3 and R5) on K562 cell viability. R2: permeate obtained after diafiltration of oligorutin on a 3-kDa membrane; R3 fraction: permeate obtained after diafiltration of oligorutin on a 5-KDa membrane; R5: retentate obtained after diafiltration of oligorutin on a 15-KDa membrane. The percentage of cell viablility was measured by MTT assay. Results are represented by the means ± SD of three independent experiments. *p < 0.05 as compared to the control group using ANOVA followed by Duncan’s multiple comparison test. ANOVA: analysis of variance; MTT: 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide.
Genotoxic study of rutin and polyrutin fractions (R2, R3, and R5) against K562 cells.a

R2: permeate obtained after diafiltration of oligorutin on a 3-kDa membrane; R3 fraction: permeate obtained after diafiltration of oligorutin on a 5-KDa membrane; R5: retentate obtained after diafiltration of oligorutin on a 15-KDa membrane; T: untreated control group; ANOVA: analysis of variance; H2O2: hydrogen peroxide.
aComet assay of genomic DNA of K562 cells treated with different concentration of rutin and polyrutin fractions and H2O2. Values were expressed as means ± SD of three experiments.
b p < 0.05 as compared to the control group using ANOVA followed by Duncan’s multiple comparison test.
Discussion
Larger molecules such as the flavonol oligomers, which are also present in significant amounts in food and beverages, have only recently attracted attention. 22,23 In a recent study, grape procyanidins such as B5-3′-gallate were shown to possess strong antioxidant activity. 24
Many of these compounds are already used in pharmaceutical, cosmetic and food preparations. Unfortunately, the use of some of them is limited by their low solubility and stability in both lipophilic and aqueous media. Therefore, to improve these properties, different techniques of derivatization are suggested, among them is the enzymatic polymerization.
Our study showed that oligomerization of rutin increases its solubility in water. Anthoni et al.
13
reported that this solubility reached 4200-folds that of rutin in water. Unlike Anthoni et al.,
13
who studied the solubility of total obtained oligorutin without separation, we tried to elaborate the study of oligorutin solubility by focusing on the effect of oligorutin fractions
The high water solubility of oligorutin may be due to the structure of oligorutin; in fact, it has been shown through a molecular modelling study that, in water, rutin showed a folded structure where the rhamnose and the ring A became closer together, while oligorutin (hexamer) revealed an unfolded structure where sugars offer a large contact with the surrounding solvent. 13
The biological activities of monomeric rutin and in particular its cytotoxicity were well established by many authors.
9,25,26
The objective of the present study was to evaluate the effect of polymerization of flavonoids on their solubility in water in a first step, then on their capacity to protect biological systems against oxidative DNA damages. In fact, free radicals are frequently associated with DNA damage induction in cells, and thus with carcinogenesis processing. We chose to evaluate whether oligomerization of flavonoids should imply better antigenotoxicity than monomer and thus better roles in cancer chemopreventive. In fact, different flavonoids were reported to modulate DNA repair in cells treated with a genotoxicant
6,27
–29
at non-toxic concentrations. The mutagenic and antimutagenic activities of rutin and oligorutin fractions (R2, R3 and R5) were studied in our laboratory using the Ames’ test on a prokaryotic system.
16
This study demonstrated an interesting antimutagenic activity in the presence of oligorutin fractions on two Salmonella strains TA104 and TA102.
16
However, it seems essential to confirm these results on a prokaryotic system; therefore, we used the comet test to evaluate the genotoxic and antigenotoxic activities in a eukaryotic system. Our purpose of this study is to ameliorate protective capacities of flavonoids by increasing their solubility using polymerization process. As continuous cell lines represent an attractive easy-to-use model, we adopted this eukaryotic system to evaluated non-toxic range concentrations of both the tested monomer and its corresponding oligomer fractions. It appears from the results that oligomer fractions with high
In accordance to previously reported studies, 9,25,26,30 we showed that the monomer rutin exhibited a cytotoxic effect especially at the highest tested dose (800 µg/ml).
The cytotoxicity of flavonoids was studied by Plochmann et al. 31 These authors had established the structure–activity relationship of flavonoids cytotoxicity. They reported that the presence of a C2=C3 double bond on C-ring of flavonoids structure could increase 3- to 10-fold the cytotoxicity compared with flavonoids without a C2=C3 double bond. The important role of the C2=C3 double bond in enhancing the cytotoxicity was also supported by other authors. 32 –35 Moreover, flavonoids with a C4 carbonyl group were found to be 30-fold more cytotoxic than flavonoids without this functional group. 33,34 Decrease in cytotoxicity in the presence of oligorutin fractions compared to the monomeric rutin could be attributed to the fact that the C2=C3 double bond and/or the C4 carbonyl group were affected during the rutin oligomerization.
Flavonoids are known to protect cellular compounds due to their antioxidant and free radical scavenging effects. 1,12,13 However, we can not exclude the existence of flavonoids with pro-oxidant properties. 2
Rutin and its oligomer fractions (R2, R3 and R5) did not damage DNA at different tested concentrations. Indeed, there is no difference between the tail DNA of the untreated cells and those treated with the monomer or its oligomer fractions; this indicates that DNA does not seem to be relevant target for these flavonoids. The absence of genotoxicity of rutin using the comet assay is in accordance with the results of many studies. 3,7,31,36
The protective effect of rutin and oligorutin fractions against the DNA damage induced by H2O2 in K562 cells was studied using the comet assay (Figure 4). Oligorutin fractions revealed significant antigenotoxic activity compared with rutin. This activity was dose dependant and proportional to the

Inhibitory effects of rutin and oligorutin fractions R2, R3 and R5 on the genotoxicity induced by H2O2 in K562 cells. R2: permeate obtained after diafiltration of oligorutin on a 3-kDa membrane; R3 fraction: permeate obtained after diafiltration of oligorutin on a 5-KDa membrane; R5: retentate obtained after diafiltration of oligorutin on a 15-KDa membrane. Values were expressed as mean ± SD of three experiments. *p < 0.05 as compared to the control group using ANOVA followed by Duncan’s multiple comparison test. ANOVA: analysis of variance; H2O2: hydrogen peroxide.
Several works reported the structure–activity relationship of different flavonoids. Hatch et al. 36 indicated that the 4-keto substituent at C4 of the C ring, the presence of a C2=C3 double bond and the planarity of the C ring were required for antimutagenic activity of flavonoids. The structure–activity relationship advanced by Hatch et al. 36 was confirmed by Edenharder and Tang, 10 who investigated the antimutagenic effect of 56 flavonoids against the mutagenicity induced by 2-nitrofluorene, 2-nitrofluoranthene and 1-nitropyrene in S. typhimurium TA98. Thus, the high antimutagenic activity of oligorutin fractions against toxicity induced by H2O2 reinforces the assumption that the 4-keto substituent at C4 and the C2=C3 double bond were not affected during the rutin oligomerization.
Conclusion
Oligorutin fractions (R2, R3 and R5) were characterized by high solubility in water especially in the presence of oligorutin fractions having the highest
Footnotes
Acknowledgements
The authors are grateful to the Lorraine region in France.
Funding
This work was supported by Ministère Français des Affaires Etrangères (Institut Français de Coopération (IFC) and the Ministry of Higher Education and Scientific Research in Tunisia.
Conflict of interest
The authors declared no conflicts of interest.