Abstract
Background
Purpura fulminans is a rare and extremely severe infection, mostly due to Neisseria meningitidis frequently causing early orthopedic lesions. Few studies have reported on the initial surgical management of acute purpura fulminans. The aim of this study is to look at the predictive factors in orthopedic outcome in light of the initial surgical management in children surviving initial resuscitation.
Methods
Nineteen patients referred to our institution between 1987 and 2005 were taken care of at the very beginning of the purpura fulminans. All cases were retrospectively reviewed so as to collect information on the total skin necrosis, vascular insufficiency, gangrene, and total duration of vasopressive treatment.
Results
All patients had multiorgan failure; only one never developed any skin necrosis or ischemia. Eighteen patients lost tissue, leading to 22 skin grafts, including two total skin grafts. There was only one graft failure. Thirteen patients were concerned by an amputation, representing, in total, 54 fingers, 36 toes, two transmetatarsal, and ten transtibial below-knee amputations, with a mean delay of 4 weeks after onset of the disease. Necrosis seems to affect mainly the lower limbs, but there is no predictive factor that impacted on the orthopedic outcome. We did not perform any fasciotomy or compartment pressure measurement to avoid non-perfusion worsening; nonetheless, our outcome in this series is comparable to existing series in the literature. V.A.C.® therapy could be promising regarding the management of skin necrosis in this particular context. While suffering from general multiorgan failure, great care should be observed not to miss any additional osseous or articular infection, as some patients also develop local osteitis and osteomyelitis that are often not diagnosed.
Conclusions
We do not advocate very early surgery during the acute phase of purpura fulminans, as it does not change the orthopedic outcome in these children. By performing amputations and skin coverage some time after the acute phase, we obtained similar results to those found in the literature.
Introduction
Purpura fulminans is an extremely severe infection, mainly due to Neisseria meningitidis or meningococcus. Meningococcus is a Gram-negative diplococcus found only in human respiratory secretions. While transmitted by aerosolization, it is not yet known how this bacteria penetrates the mucosa in order to infiltrate the blood system and initiate a complex cascade of events leading to disseminated intravascular coagulopathy (DIC). The latter creates microthrombotic processes depriving the more distal and less oxygenated tissues from sufficient blood supply, and also deprivation in coagulation factors, leading to shock and multiple-organ damage [1, 2]. This purpura fulminans condition can seldom also be caused by other Gram-negative bacteria and by hemorrhagic fever viruses or varicella zoster virus (VZV). In France, purpura fulminans accounts for one-fifth of all meningococcal infections, which represents about 1.03 cases per million inhabitants [3].
Despite recent advances in medical management, the mortality rate of fulminant meningococcemia ranges from 15 to 30% [4, 5]. Thus, many children survive the initial phase of shock only to develop vascular insufficiency, compartment syndrome, or peripheral gangrene. Among initial survivors, 10–30% present with skin necrosis, limb ischemia needing orthopedic surgical management such as debridement, skin grafting, muscular flap coverage for limb salvage, and, sometimes, even limb amputation. To our knowledge, there are few studies assessing a pediatric population starting from the first hours of medical and surgical management regarding orthopedic involvements [4, 6].
The aim of our study was to look at predictive factors in orthopedic outcome in light of the initial management of acute purpura fulminans in 19 children who have been referred to our unit over an 18-year period.
Materials and methods
Between 1987 and 2005, 19 patients were referred to our institution, presenting with “acute meningococcal septicemia.” All purpura fulminans were clinically considered due to meningococcal infections and treated by adapted antibiotherapy (third-generation cephalosporins, i.e., ceftriaxone or cefotaxime) after initial blood and cerebro-spinal fluid (CSF) tests. All patients presented with acute shock and DIC. Definitive diagnosis was assessed by positive blood or CSF culture or latex agglutination tests detecting capsular antigens in the more recent cases.
There were 19 patients admitted to the intensive care unit (ICU) presenting initially with purpura fulminans who were referred to our unit for orthopedic advice. Advice was taken by the ICU medical staff when the children were thought to be hemodynamically apt to survive a surgical procedure. This is why there are no deaths in our population.
Eighteen out of 19 identified patients suffered from initial orthopedic sequelae. All patients had multiorgan failure, with absent peripheral pulses. Thus, patients were only included if they required surgical intervention as the result of the complications of meningococcal purpura fulminans.
There were 11 males and eight females, with a mean age of 3.97 years (range 0.1–16 years, standard deviation 3.3 years). Germs identified by the blood tests were collected, along with all initial surgical procedures, initial estimation of skin necrosis extension using the same topographic criteria as in the Lund and Browder method [7] to assess the surface area involved in pediatric burns, and, more generally, all other orthopedic complications, such as vascular insufficiency, compartment syndrome, or peripheral gangrene. Thus, skin necrosis was evaluated to be minor when under 5% of the overall body surface was involved, moderate between 5 and 15% of the overall body surface, and major over 15%.
Vasopressive treatment duration and all other medical treatments that were initiated at the same time were noted. The total duration of treatment was noted.
All of these case reports are summarized in Table 1.
Clinical features of the 19 patients included in the series
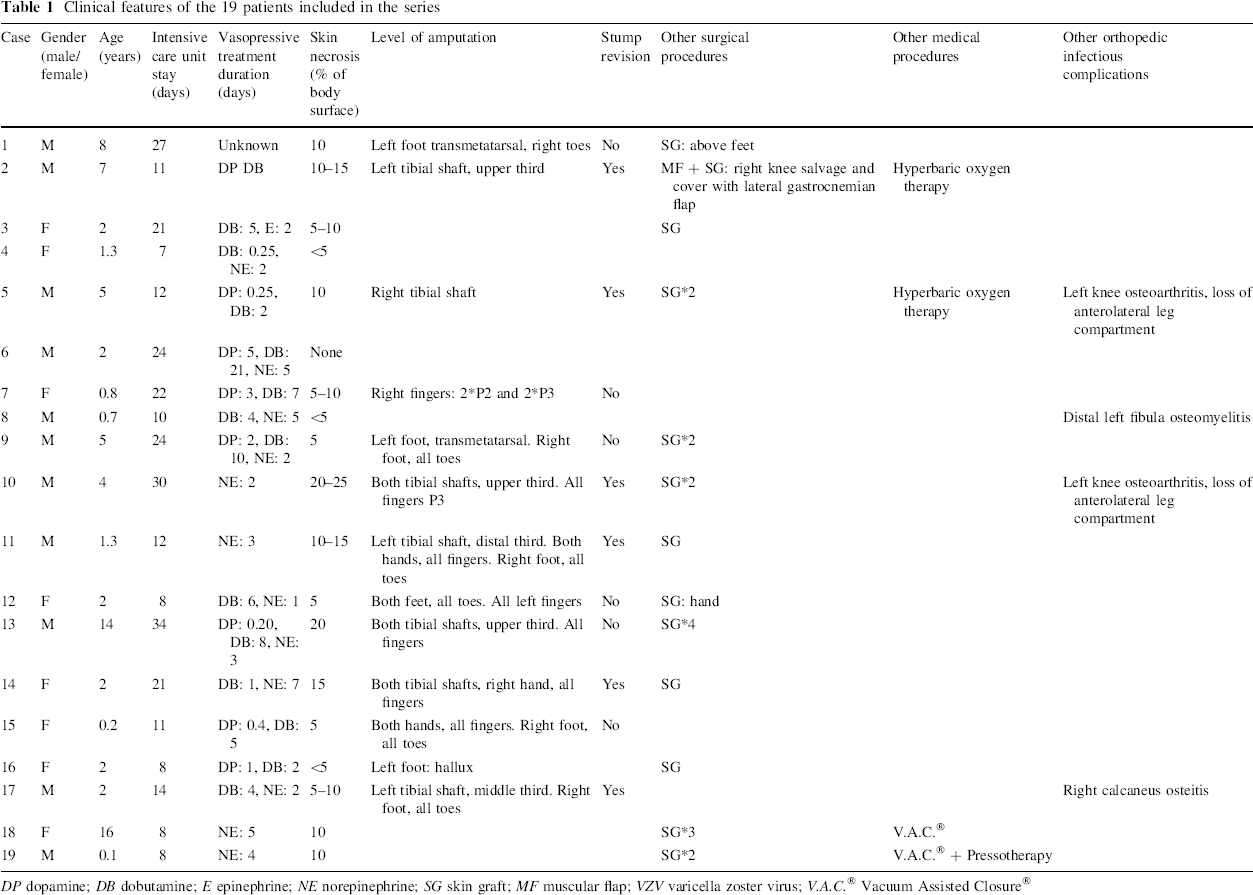
DP dopamine; DB dobutamine; E epinephrine; NE norepinephrine; SG skin graft; MF muscular flap; VZV varicella zoster virus; V.A.C. ® Vacuum Assisted Closure®
Results
All patients had multiorgan failure, with absent peripheral pulses; however, patient #6 never developed any skin necrosis or peripheral ischemia, unlike all of the other patients. Compartment pressures were not systematically measured, even when skin necrosis was found to be extensive. It was done when absent peripheral pulses coincided with another clinical sign of ischemia.
Table 1 summarizes the results in our series of 19 patients. Tissue loss secondary to ischemia concerned all but one patient.
Twenty-two skin grafts were performed in 13 patients (68.4% of our patients), including 20 thin skin grafts and two total skin grafts. Skin was harvested in unharmed locations, such as the inner aspect of the thigh, the back, and, sometimes, the scalp in young infants. The two total skin grafts were performed because of a thin skin graft failure in patient #9 and in order to give greater elasticity and better appearance to patient #12's hand. The mean delay between the first signs of purpura fulminans and the carrying out of skin grafts is 10 weeks (ranging from 5 to 14 weeks).
Other soft tissue necrosis was noted predominantly at the lower limbs, with 13 patients concerned by at least one amputation procedure (68.4% of our patients). The level of amputation and the involved limb are summarized in Table 1. Amputations were done after necrosis had demarcated itself; thus, only surgical amputations were performed, even concerning the fingers, and there were no auto-amputations. All amputations were performed after the first signs of purpura fulminans with a mean delay of 4 weeks (ranging from 4 days to 8 weeks).
Because of extensive skin and soft tissue necrosis of the left knee in patient #2, a muscular flap using the lateral gastrocnemian muscle was performed in order to protect the knee joint 7 weeks after the first signs of purpura fulminans.
Four patients developed an associated peripheral joint infection. All of these infections were treated with adequate antibiotherapy.
Patients stayed in the ICU for a mean of 16.4 days (range 7–34 days). Dopamine, epinephrine, and norepinephrine were used as vasopressive drugs during the resuscitation phase, in addition to dobutamine. All 18 patients in whom data could be accurately collected were found to have had vasopressive drugs over a period of a few days most of the time, seldom during a longer period, and most patients received in addition dobutamine as a cardiac stimulant. After this initial management in ICU, patients were transferred to our Pediatric Orthopedics Unit for a mean of 45 days (range 5–106 days).
Discussion
This oversight of all of our patients’ outcome after the first signs of purpura fulminans illustrates that there are no predictive factors that can help give an orthopedic prognosis.
Some children never or hardly ever developed any soft tissue necrosis, despite the initial severity of their hemodynamic condition (patients #4, #6, and #16), whereas all of the other patients presented ultimately with a great variety of lesions. Like all other studies, the number of children is too small and the outcomes too varied to allow proper statistical analysis. All children underwent an adapted antibiotic treatment based on third-generation cephalosporins, a vasopressive therapy to sustain proper blood pressure, and oxygenotherapy to obtain normal oxygen blood saturation, but, nonetheless, they developed necrosis in a totally non-predictable way. Antibiotics consisted of ceftriaxone or cefotaxime and were adapted to N. meningitidis antibiograms for all patients.
Vasopressive therapy differed between the different patients with regards to both inotropic agents prescribed and the duration of treatment. The treatment chosen depended on the cardiovascular status of each patient according to the ICU medical staff protocols. All patients were treated with inotropic agents for between 1 and 21 days. These treatments all have peripheral vasoconstrictive effects, but it seems that the duration of these vasoconstrictive effects is not linked to the severity of the soft tissue necrosis. Cremer et al. [8], in a similar study focusing on medical treatments including 44 patients with purpura fulminans, could not find any particular factors influencing positively or negatively the short-term outcome of purpura fulminans, apart from factor VII levels on admission. Antihemostatic treatments were not evaluated in this study, as they were never used early in the medical treatment protocol in this series of patients. It is still being discussed whether it is beneficial for all meningococcal septic shocks [9], even though heparin may limit digit and extremity necrosis [10].
More specifically, it seems that immediate outcome is non-related to the age of the patient in this study. Regarding skin necrosis and leg or foot amputation, it seems that younger children under the age of 2 years could be less at risk. It should be stressed that, because it is as yet impossible to determine what short-term sequelae each particular child will develop in the following days or hours, a pediatric orthopedic surgeon should always be involved very early.
However, all of the lesions encountered are the same as that found in the literature [4, 11]; that is, mostly damage to the lower limbs and only very distal necrosis to the upper limb. Purpuric lesions, regardless of the etiology of the purpura, tend to develop at the lower part of the body because of orthostatism. Moreover, in purpura fulminans, lesions tend to be more extensive on the body surfaces with lower blood flow [12], explaining the predominance of lower limb damage and the coexistence of necrosis adjacent to viable tissues. It seems that, in children who survive the initial resuscitation, there is seldom the need to perform upper limb or above-knee amputations. While this study supports this hypothesis, some authors have described forearm amputation [4, 13] or above-knee amputation [14].
In such cases, our main concern was to spare these children the consequences of unnecessary or extensive amputations. Hamdy et al. [13] advocated bone scintigraphy as a reliable means to assess non-perfusion in ischemic limbs, but they could not draw any conclusion and no further studies were carried out. Other authors [4, 12] measured intracompartment muscular pressures in order to avoid amputations by performing early fasciotomies, but it was thought that performing fasciotomies under clinical grounds rather than solely on measurements might be less detrimental to survival [4]. It is true that clinical grounds are not always available easily to such patients in the ICU. Careful palpation of the muscles, Doppler evaluation of underlying pulses, along with pain monitoring (which can be evaluated by morphine requirement) can help evaluate the qualitative increase in intracompartment muscular pressure, even in sedated children. In our series, no compartment pressure measurements and no fasciotomies were performed. Unlike post-traumatic limb swelling, the increase in muscular intracompartment pressure is histologically proven by small-vessel thrombi, inflammation due to acute infection, and ischemia [15]. Bache and Torode [16] underlines, and we greatly support this advice, that fasciotomies performed in children with compartment syndrome without ischemia have the same role as in traumatology, whereas in children with compartment syndromes associated with ischemic thrombotic condition, the benefits of fasciotomy are dubious, as it does not allow better tissular perfusion. Boeckx et al. [17] noted in a series of 14 patients that fasciotomy, while decompressing the subcutaneous edema, never restored blood supply to the muscles. They highly recommend that arteriolysis be additionally performed at the bedside in the ICU in order to obtain proper adventitial sympathectomy of the artery leading to vasodilation. Arterial dissection also allowed the removal of constricting bands, leading to refilling of the distal arterial bed and reperfusion of the distal tissues. Interesting results were obtain by Boeckx et al. in seven patients, especially regarding the most distal structures. Between 76 and 82% of ischemic toes and fingers survived in the seven patients, whereas less than 2% survived in a comparable group without arteriolysis.
Two patients underwent hyperbaric oxygen (HBO) therapy. It was not felt that the HBO therapy modified the outcome in these patients, probably because a hyperoxygenated patient is not the solution to a condition mainly due to distal thrombosis. Waisman et al. report a favorable experience in children treated with HBO [18]. Recently, HBO has been described as a conservative surgical management of soft tissue necrosis in a baby [19]. There has, to date, only been a few cases reporting the use of HBO therapy in children surviving meningococcal sepsis.
Amputation procedures were performed with a mean of 4 weeks delay, after having always waited for the necrosis to demarcate in order to determine more accurately the level of amputation (Fig. 1). Some authors advocate such management [8], since it has been proved that early amputation leads to greater rates of stump revision. Despite our waiting for tissues to demarcate properly, five patients needed stump revision for ischemic changes.
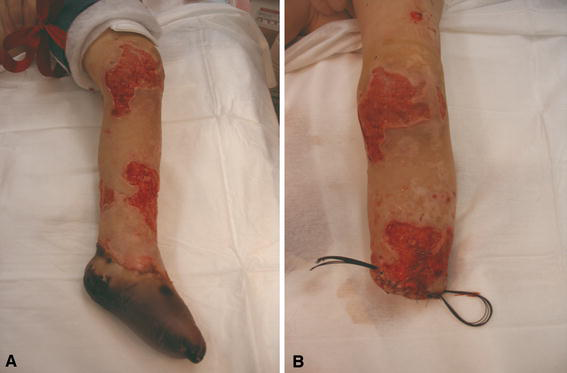
Patient # 11.
Twenty-two skin grafts were performed, with only one thin skin graft failure. This is quite surprising given that the underlying tissues have suffered from prolonged ischemia and necrosis. Again, this high success rate can probably be explained by the long delay that we observe before grafting our patients, thus, allowing acceptable revascularization of all underlying soft tissues (Fig. 2). Before grafting, several dressings were done under general anesthesia every 48 h, consisting of necrosis excision and the application of vaseline mixed with povidone–iodine, as is routinely performed in our Burns Unit.
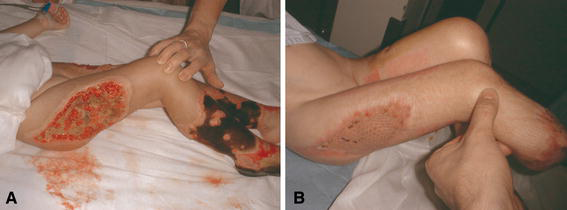
Patient # 14.
In our latest patients #18 and #19, negative pressure wound therapy (NPWT) using V.A.C.® GranuFoam™ therapy (KCI, Kinetic Concepts, Inc.) was used. NPWT allowed dressings to be done only every 5 days without using anesthetics, which was very convenient for our youngest patient (#19), who was aged 1.5 months. Moreover, it allowed skin grafting after only 2 weeks, with highly granulated underlying soft tissue. There is not enough follow-up and experience to tell if aesthetics are improved, but it seems that subjective qualitative appearance is greater than with standard bolster dressings. NPWT was started in our experience immediately after necrosis excision and it seems that the skin surface needing grafting after such treatment was smaller than after standard dressing. To our knowledge, only one case is reported to date besides this one, treated by NPWT after purpura fulminans [20]. Our case suggests that V.A.C.® therapy could reduce the delay between necrosis and the time of grafting. This point needs to be assessed more accurately in further studies, as it could reduce hospital stay, which is usually prolonged in this pathology.
The only muscular flap in this series was performed soon after the acute phase of the purpura fulminans, because of extensive necrosis that left the entire knee joint uncovered and in open air. Because the other leg needed below-knee amputation, it was felt to be very important for this child to recover with at least one functional lower limb. There are few particular recommendations with regard to muscular flaps in such conditions in the literature [6, 12]. While caution is advocated by Potokar et al. because of the underlying microvascular damage, Wheeler et al. report nine local fasciocutaneous and myocutaneous flaps with good results, arguing that distinction between viable and non-viable tissue can be confidently made at the time of surgery. It happened that, again, surprisingly enough, the flap managed to protect the knee joint and this child recovered with good knee function.
Some authors recommend the utilization of skin allograft and keratinocyte culture [6, 21, 22]. These treatments could be useful to maintain a healthy graft-bed until definitive autologous grafting can be performed.
This study highlights the occurrence of osteitis and osteomyelitis in these patients, as it complicates the medical management and they are often not seen in the early stages of the disease. Few studies mention osteo-articular infections during the acute phase of purpura fulminans. These patients need to be examined thoroughly very frequently by an orthopedist, especially when blood tests (white blood cell count, C-reactive protein) indicate that infection is not being properly controlled. In our series, four patients developed additional bone infections. These infections did take place preferentially in regions where there was extensive skin necrosis, thus, allowing direct contamination. Despite early diagnosis and adequate antibiotherapy, three children presented years later with incomplete physeal growth arrest, but it could also be a consequence of ischemic effects.
In conclusion, young patients presenting with purpura fulminans should systematically be referred to an orthopedist trained in pediatrics. It is, to date, impossible to predict what will be the short-term sequelae in such children. Soft tissue necrosis and distal ischemia are the two major surgical challenges during the acute phase of purpura fulminans. There is strong recommendation that surgery be delayed without acting rashly in case of compartment syndrome or distal ischemia. Unlike many studies, our choice was to wait for the medical management (that is, antibiotherapy, ventilation, and inotrope support) to help solve what remains, above all, a major DIC. By performing selective and carefully planned amputations and skin grafting long after the initial acute phase of purpura fulminans, the clinical results were found to be as good as in the literature.
