Abstract
Haemophilia A patients who develop factor VIII inhibitors pose a challenge with respect to bleeding and orthopaedic management. This is particularly relevant in cases requiring amputation. We present here a case of a patient with severe haemophilia A and inhibitors who had a history of multiple surgeries due to periprosthetic joint infection and a non-healing wound which led to above-knee amputation. Following the implementation of appropriate and suitable transfemoral prosthesis and emicizumab therapy, the patient experienced a significant improvement in mobility and quality of life without any adverse events or bleeding episodes. Additional studies are required to more fully understand treatment options for lower limb amputations in the haemophilia population.
Background
Haemophilia A is a bleeding disorder that results from a coagulation factor VIII deficiency, and places patients at risk of spontaneous bleeding into joints, muscles, and other organs, which can lead to chronic pain, joint deformity, and disability. 1 In addition, patients may develop autoantibodies (inhibitors) to factor VIII, which poses a high risk of bleeding and requires unique treatment strategies. 2 However, emicizumab has emerged as a prophylactic treatment option for patients with haemophilia A with inhibitors.3,4 Studies have shown that major surgery in patients receiving emicizumab can be safely performed using appropriate replacement therapy protocols. 5 To-date, limited information exists on amputation in haemophiliacs and the use of transfemoral prostheses in patients with haemophilia A and factor VIII inhibitors. This present case report describes a patient with a history of haemophilia A and inhibitors who successfully underwent above-knee amputation and received a transfemoral prosthesis following emicizumab therapy.
Case report
A 60-year-old man had visited our outpatient department for regular follow-ups over the past 18 years. He had been diagnosed with congenital haemophilia A at the age of 12. He had severe haemophilia caused by an intron 22 inversion mutation and over the years had experienced bleeding events like swelling and ecchymosis over his arms. His factor VIII inhibitor level fluctuated around 5 to 7 Bethesda units (BU) per ml. Due to haemophilic arthropathy, the patient had undergone several surgeries, including right knee replacement surgery (1999), revision arthroplasty because of prosthesis loosening (2006), and left knee replacement surgery (2015). Bypass treatment with activated prothrombin complex concentrate (aPCC; FEIBA) was applied whenever he experienced bleeding.
In February 2020, the patient developed periprosthetic joint infection of his right knee (Figure 1–a), leading to prosthesis removal and multiple debridement procedures. Due to soft tissue damage and ongoing bleeding (Figure 1–b), the plastic surgeon also performed a gastrocnemius-propeller extended myocutaneous flap procedure. During a follow-up appointment in January 2021, the patient still had an unhealed wound with discharge, indicating osteomyelitis (Figure 2). Microbiological culture identified methicillin-resistant Staphylococcus aureus (MRSA) and antimicrobial therapy (i.e., ciprofloxacin, trimethoprim/sulfamethoxazole, and fusidic acid) was applied continuously from that date. Furthermore, considering the patient's osteomyelitis, failure of previous interventions and multiple debridement procedures, and the potential benefits of amputation in resolving the infection and improving the patient's quality of life, after a discussion with the patient and his family, the decision was made for amputation.

Imaging findings of the right knee joint before amputation. (a) A radiograph of the right knee shows gas shadows in the suprapatellar recess, periprosthetic bone resorption, and implant loosening and (b) Computed tomographic (CT) angiography of lower limbs shows hypervascularity, bone sequestrum, and soft tissue defect at the right knee joint. This was probably caused by the post-removal of the fixator with an infectious process.

Bone scintigraphy using technetium 99m-methyl diphosphonate showing the infectious and inflammatory process involving the right knee, right distal femur, and proximal tibia.
From February 2021, the patient received emicizumab (i.e., loading dose 3 mg/kg subcutaneously once weekly for 4 weeks, followed by maintenance dose of 3 mg/kg every 2 weeks). Factor VIII inhibitors were monitored regularly using the chromogenic Bethesda assay which accounts for potential effects of emicizumab on conventional assays. 6 In November 2021, after receiving emicizumab for eight months, the patient's factor VIII inhibitor titer had decreased from 7 to 2 BU/ml. In December 2021, following further discussions with the patient and his family, an above-knee amputation was performed without complications (Figures 3–a). During the perioperative period, administration of recombinant factor (r) VIIa (NovoSeven) was carefully guided by haematologists experienced in the management of haemophilia and was tailored to the patient's needs. During surgery, the patient received rFVIIa 90 mcg/kg every 2 hours and then every 4 hours on the first day after surgery. From Days 2–3, he received rFVIIa 50 mcg/kg every 4 hours, from Days 4–6, 50 mcg/kg every 6 hours and from Days 7–8, 50 mcg/kg every 8 hours.
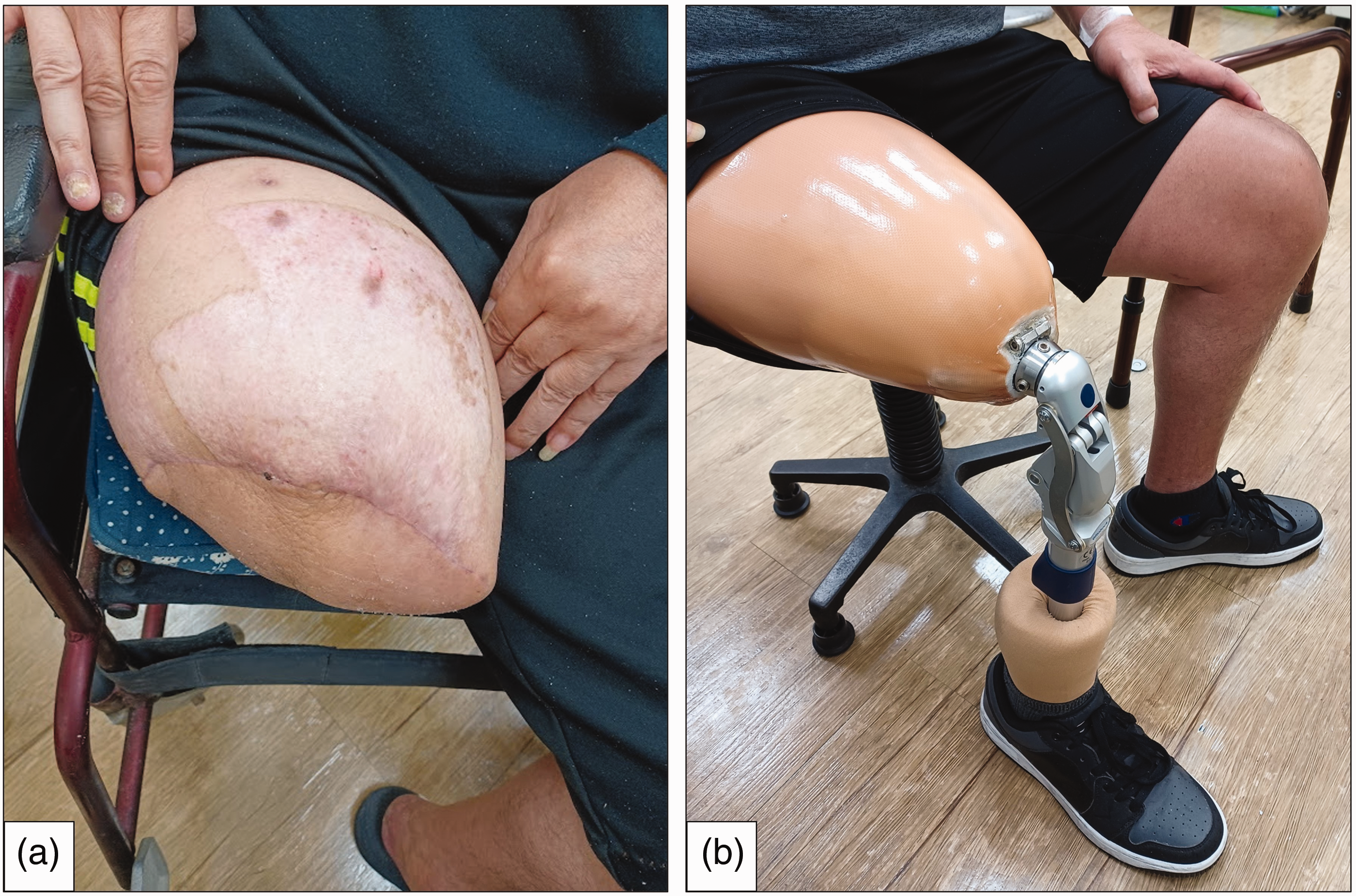
Photographs following the above-knee amputation. (a) Appearance of the residual limb and (b) The transfemoral prosthesis.
Following amputation, the patient received a transfemoral prosthesis using Ottobock Skeo Skinguard liner, ischial ramus containment, a modular polycentric knee joint with pneumatic swing phase control, and a modular prosthetic single-axis foot (Figure 3–b). The prosthesis was customized to ensure proper alignment and weight-bearing support, and the fitting process was carefully monitored to prevent any bleeding complications. The patient was directed to our rehabilitation centre for various follow-up physical therapies (e.g., infrared therapy; interferential current therapy; prosthesis training; therapeutic exercise; stretching exercise; core muscle strengthening training).
The patient received continuous prophylaxis treatment with emicizumab 6 mg/kg every 4 weeks and underwent rehabilitation with the support of a team of professionals, including a haematologist, physiatrist, and physiotherapist. To-date, the patient has not experienced any bleeding or adverse reactions to the prosthesis. Additionally, he reported a substantial enhancement in his mobility and overall well-being. He now can perform daily tasks independently, such as walking and climbing stairs. During the follow-up period from 2022 to 2023, his factor VIII inhibitor titer remained consistently low at around 1 BU/ml and with no signs of relapse.
The reporting of this study conforms to CARE guidelines. 7 Written informed consent was obtained from the patient to publish his anonymised data. The study was approved by the Institutional Review Board of Tri-Service General Hospital, School of Medicine, National Defense Medical Center, Taipei, Taiwan (TSGHIRB No. B202315074, approved on 17 May 2023).
Discussion
To the best of our knowledge, this is the first reported case study demonstrating that a transfemoral prosthesis can be successfully used in a patient with severe haemophilia A with inhibitors receiving emicizumab therapy who had undergone an above-knee amputation. Despite the patient's complicated medical history, he experienced no bleeding complications or adverse events during the fitting of the prosthesis. The patient also reported a significant improvement in mobility and quality of life. This case highlights the safe and effective use of prostheses as a treatment option for patients with severe haemophilia who require limb amputation.
The management of patients with haemophilia A and inhibitors who undergo major orthopaedic surgeries or invasive procedures can be difficult. While prophylaxis is becoming more widespread for the treatment of haemophilia, and the number of patients requiring surgery is increasing, there is a lack of information and guidance on choosing the ideal prosthetic component for those patients who have above-knee amputation. 8 Although transfemoral prostheses have been successfully used in many patients, 9 reports of their use in individuals with haemophilia, especially those with inhibitors, are scarce. However, several studies have documented the importance of multidisciplinary care and appropriate prophylaxis in achieving successful surgical outcomes in the management of haemophilic arthropathy in patients with inhibitors.10,11 Our case report is novel, in that it showcases the successful and complication-free implementation of a transfemoral prosthesis in a patient with severe haemophilia A and inhibitors. Moreover, our successful outcome is consistent with findings from another study that reported low rates of complications in patients with bleeding disorders undergoing major surgeries at a haemophilia treatment center. 12 Importantly, our case study emphasises the significance of using a collaborative team approach involving specialists to manage patients with haemophilia.
Emicizumab is a recombinant, humanized, bispecific monoclonal antibody that bridges activated factor IX and factor X to restore the function of missing activated factor VIII, which is needed for adequate haemostasis. 4 Due to its unique structure, emicizumab is not affected by existing factor VIII inhibitors. 4 Studies have shown that minor and major surgeries can be performed safely in patients with haemophilia A receiving emicizumab prophylaxis irrespective of inhibitor status. 3 The drug has been proven to be well tolerated during perioperative care, and there is growing evidence of successful surgical experiences for patients taking emicizumab outside clinical trials, as demonstrated by various case reports and case series. 13 Furthermore, one study reported real-world evidence that in patients treated with emicizumab, surgery, including thigh amputation, can be safely carried out using appropriate replacement therapy protocols. 5 In addition, because emicizumab has a long half-life and is administered subcutaneously, it has advantages for the long-term treatment of outpatients, and so may be more cost-effective than other options. 14 Importantly, there is a high risk of bleeding complications during and after surgical amputation procedures and when using prostheses in a patient with severe haemophilia A and inhibitors. In our case, emicizumab was administered before the amputation and was continued during the prosthetic fitting, successfully preventing bleeding complications and facilitating healing and rehabilitation. Therefore, our study demonstrated that emicizumab is a suitable treatment choice for patients who undergo amputation and have prosthetics fitted.
The fitting and use of a prosthesis can potentially increase the risk of adverse events like stump bleeding. Therefore, it is essential to ensure proper alignment and weight-bearing support of the prosthesis to minimize the risk of injury. With excellent elasticity and good fit on the residual limb, the Ottobock Skeo Skinguard liner reduces friction, enhances the connection between the liner and skin, and spreads suspension forces all over the stump surface. 15 As a consequence, it may decrease the risk of hematoma formation in patients with severe haemophilia. The knee joint, with pneumatic swing phase control, has a polycentric design that allows for a natural walking motion while using less energy and increasing ground clearance during the swing phase. Its wide flexion angle and lightweight structure also provide a comfortable experience. 16 The prosthetic single-axis foot, with a natural shape and smooth surface, quickly provides a confident stance, making it particularly suitable for transfemoral fittings. 17 Combining these prosthetic elements, helped our patient regain mobility and achieve a comfortable and natural gait, crucial for maintaining independence and improving his quality of life.
In summary, using prostheses after transfemoral amputation in a patient with haemophilia A presents unique challenges. These challenges arise from the underlying bleeding disorder, the increased risk of complications, and the need to select and carefully fit prosthetic components. It is important to consider factors such as the liner material, suspension system, knee joint design, and foot component to optimize comfort, stability, and mobility whilst minimizing the risk of bleeding complications. Regular follow-up appointments and ongoing communication among the patient, prosthetist, haematologist, and physiotherapist are necessary to ensure optimal function and safety of the prostheses. Close collaboration among healthcare professionals and tailored rehabilitation programs are essential to address these challenges and optimize the functional outcomes for patients with haemophilia A and transfemoral prostheses. In addition, individuals with severe haemophilia A and inhibitors who undergo above-knee amputation may be able to improve their quality of life through proper medical management and rehabilitation; this should include the use of emicizumab as prophylaxis and a properly selected lower limb prosthetic. This case report highlights the need for collaboration among multiple disciplines in managing complex haemophilia cases. Additional studies are required to more fully understand the treatment options for lower limb amputations in the haemophilia population.
