Abstract
Peripheral neuropathy is a common complication in patients with diabetes. However, the appropriateness of administering nerve block anesthesia to these patients remains uncertain. Key concerns include the potential for prolonged block duration, an increased risk of local anesthetic toxicity, and the possibility of further damaging already compromised peripheral nerves. We herein report a case involving a patient with diabetic peripheral neuropathy who underwent finger amputation under ultrasound-guided nerve block anesthesia and subsequently lost pain and temperature sensation in both hands 1 month later. For critically ill patients undergoing surgery, regional anesthesia, such as a nerve block, may be a more suitable option than general anesthesia. When performing nerve block procedures in patients with diabetes, using ultrasound guidance is recommended to ensure precise targeting and reduce the risk of complications. However, it remains unclear whether nerve block anesthesia exacerbates peripheral neuropathy.
Keywords
Introduction
Peripheral neuropathy is a common complication in patients with diabetes. 1 However, whether it is appropriate to administer nerve block anesthesia to these patients remains uncertain. Key concerns include the potential for prolonged block duration,2,3 an increased risk of local anesthetic toxicity, 4 and further damage to already compromised peripheral nerves. 5 With the patient’s written informed consent, we herein report a case of diabetic peripheral neuropathy in a patient who underwent a finger amputation under ultrasound-guided nerve block anesthesia. This report adheres to the CARE guidelines. 6
Case report
A woman in her early 40s was admitted to the intensive care unit with a >1-day history of left lower abdominal pain and an 8-hour history of dizziness. She was diagnosed with septic shock, multiple organ failure, cardiogenic shock, severe pneumonia, type I respiratory failure, acute respiratory distress syndrome, hydronephrosis with kidney stones, and type 2 diabetes. Upon admission, she received aggressive treatment, including anti-infection therapy, anti-shock measures, organ protection, improvement of circulation disorders, and bedside continuous renal replacement therapy to remove inflammatory mediators. After stabilizing her internal environment and other treatments, she suddenly lost consciousness with a corresponding drop in her heart rate and blood pressure. Following emergency resuscitation, her heart rate and blood pressure gradually stabilized. However, she subsequently developed gangrene at the ends of both hands, characterized by increased skin temperature, swelling, and blister formation. After active disinfection, dressing changes, and hyperbaric oxygen therapy, her skin temperature decreased and the swelling and blister formation improved. Physical examination revealed dry necrosis in the middle and distal segments of all five fingers of the right hand; in the middle and distal segments of fingers 1, 2, 3, and 5; and at the distal end of finger 4 on the left hand (Figure 1).
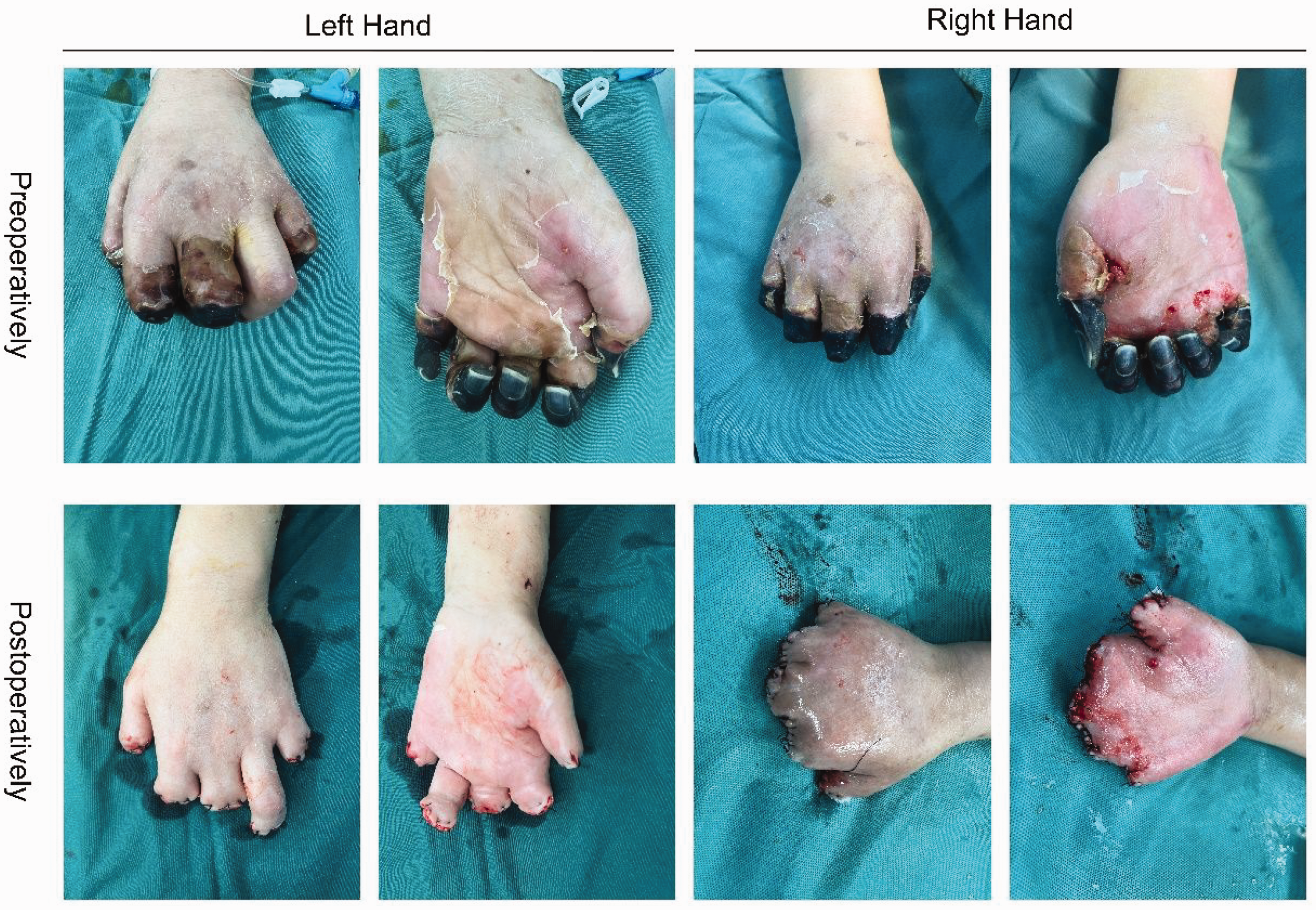
Preoperative dry necrosis of multiple fingers of the patient’s hands and postoperative changes.
After multidisciplinary consultations, it was determined that the patient had clear necrosis of multiple fingers on both hands. The necrotic areas exhibited well-defined boundaries, making the timing suitable for surgery. Conservative treatment for dry necrosis in multiple fingers was unlikely to succeed, so finger amputation was deemed necessary to remove the necrotic tissue, promote healing, and reduce the risk of infection. The plan was to perform finger amputation surgery for multiple finger necrosis in both hands under nerve block anesthesia. In the operating room, the patient underwent ultrasound-guided bilateral axillary brachial plexus block (0.25% ropivacaine + 0.5% lidocaine, 20 mL per side) with a satisfactory effect. She then underwent bilateral debridement of necrotic tissue and repair of the residual ends of multiple fingers (Figure 1). Her vital signs remained stable throughout the procedure, and she was returned to the intensive care unit postoperatively before being transferred to the rehabilitation department for recovery. One month later, her hand motor function was normal, but she had lost sensation of pain and temperature. We obtained the patient’s written consent for treatment and written consent for publication.
Discussion
The patient developed dry gangrene at the fingertips of both hands during diagnosis and treatment, primarily due to poor glucose control in diabetes and microcirculatory disorders caused by septic shock. Choosing the most appropriate anesthesia method for this patient is critical. General anesthesia may lead to intraoperative hemodynamic instability, especially after septic shock, necessitating increased fluid administration to maintain stability and correct hypotension, 7 This increased fluid load can burden the heart and contribute to edema. However, the potential for nerve block anesthesia to cause additional damage to already compromised peripheral nerves remains a concern.
The differences in regional anesthesia for patients with diabetic neuropathy are noteworthy. First, the threshold for nerve electrical stimulation is significantly higher, 5 increasing the risk of needle injury during nerve block procedures guided by a nerve stimulator. Additionally, the nerves affected by diabetic neuropathy exhibit heightened sensitivity to local anesthetics, leading to prolonged retention of these anesthetics,2,3 which can extend the duration of the nerve block. This prolonged effect may result from delayed clearance of local anesthetics in neurons, increased sensitivity of diabetic neurons to the blocking effects, or both. Although diabetic neuropathy patients are more susceptible to local anesthetic toxicity during nerve block, the evidence is inconclusive and should not preclude the use of local anesthesia when clinically indicated. 4
Given the patient’s poor overall condition and the necessity of surgery, ultrasound-guided nerve block anesthesia was considered the best option. This approach allows for precise targeting of the nerve plexus, effectively reducing intraoperative stress and enabling careful adjustment of local anesthetic dosage. Ultrasound guidance improves clinical efficacy and safety by minimizing stress responses and lowering the risk of postoperative complications. 8
Considering the patient’s dry gangrene at the fingertips due to poorly controlled diabetes and septic shock-induced microcirculatory disorders, the local anesthetic should be administered at the lowest effective concentration during nerve block. Ultrasound guidance is crucial for accurate nerve block placement, reducing the risk of nerve injury. 9 Furthermore, ultrasound-guided nerve blocks significantly reduce the total fluid requirement during surgery, promoting hemodynamic stability. This method also decreases postoperative pain and reliance on analgesics, which is critical for patient recovery. While complications such as nerve and vascular injury or intravascular injection of local anesthetic, as well as toxicity due to large doses, are possible with bilateral axillary nerve blocks, these risks are outweighed by the benefits. However, 1 month later, the patient experienced a loss of pain and temperature sensation in both hands, likely due to the progression of diabetic neuropathy. Whether the nerve block contributed to worsening peripheral neuropathy remains uncertain.
In summary, for critically ill patients undergoing surgery, regional anesthesia such as nerve block is generally more suitable than general anesthesia. When performing nerve block in patients with diabetes, it should be guided by ultrasound to ensure precise targeting, thereby reducing complications and injuries. However, it remains uncertain whether nerve block exacerbates peripheral neuropathy.
Footnotes
Acknowledgements
We gratefully acknowledge the assistance of Dr Li Yang in preparing the photographs. We also gratefully acknowledge the funding provided by the Shaanxi Association for Science and Technology Foundation and Shaanxi Provincial People’s Hospital Foundation.
Author contributions
Yan Cheng and Minmin Yi proposed the ideas for the report, collected and organized the data, and wrote the manuscript. Jianwei Guo developed the assessment indicators, provided overall supervision, and reviewed the manuscript. All authors have approved the final version of the article for publication.
Data availability statement
Not applicable (this manuscript does not involve data generation or analysis).
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Ethics statement
This study was approved by the Ethics Committee of Shaanxi Provincial People’s Hospital. (No. 2024K-186).
Funding
This work was supported by the Natural Science Basic Research Program of Shaanxi Province (2023-JC-QN-0961) and the Science and Technology Development Incubation Fund of Shaanxi Provincial People’s Hospital (2023YJY-46).
