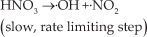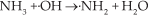Abstract
Raw TiO2 nanoparticles were prepared using the hydrolysis of TiCl4. The nanoparticles were subjected to a surface treatment in diluted sulphuric acid and, subsequently, calcined at different temperatures. Then, a type of super solid acid (SO42−/TiO2) with particle sizes of 20∼30 nm was fabricated. The catalysis of SO42-/TiO2 on the thermolysis of ammonium nitrate (AN) was probed using thermal analysis. For SO42−/TiO2 (AN doped with 3%SO42−/TiO2), the onset temperature decreased by 19°C and the peak temperature decreased by 15.8°C. For TiO2 (AN doped with 3%TiO2), the peak temperature decreased by only 0.5°C. Using the DSC-IR technology, the gas products of the decomposition of 3%SO42-/TiO2-doped AN were detected. We found that the products were mainly N2O (g) and a small amount of H2O (g), and that no NH3 (g) or HNO3 (g) was detected, which ascertained the decomposition reaction of NH4NO3→N2O(g)+H2O(g). In addition, the catalysis mechanism of SO42-/TiO2 on the AN decomposition was discussed in detail.
1. Introduction
Compared with ammonium perchlorate (AP) propellants, ammonium nitrate (AN) propellants have advantages such as nontoxic products, a low signature and insensitivity. These advantages are mainly attributed to the chloride-free composition and low water content in their combustion products. In addition, AN is a type of energetic material with super low insensitivity. For example, pure AN cannot be ignited by flame or heated in the air, and it has a remarkable resistance to stimulation such as friction, impacts, shock waves, etc. Nevertheless, AN has not served as the main oxidizer in propellant formulation and is used only as an ingredient in gas-generating agent [1,2]. This is because AN has three major defects [3]: (1) serious hygroscopicity, (2) phase transition at storage temperature and (3) poor ignition and combustion performance. In particular, the third defect is fatal, i.e., AN-based propellants burn so slowly that their combustion cannot provide sufficient thrust to boost the missile (or rocket) to fly at high speed. Therefore, introducing a catalyst to improve the combustion performance of AN-based propellants is crucial.
At present, with the solving of the first two defects, AN is frequently used in studies of high energy and low vulnerability (HELV) in the field of military science and technology [4-6]. Therefore, over the past five years, there has been an increase in the number of studies about improving the performance of AN-based propellants. In particular, many studies concerning better combustion performance were conducted but the results were not satisfactory. In studies about AP, researchers often find that a good catalyst can always decrease the decomposition temperature by more than 100°C. However,
The number of studies on AN-based propellants has increased since 2011. In particular, researchers from Russia, Japan and India have devoted the most attention to this subject [12-15]. Moreover, it is worth noting that the catalytic effects of nano materials on the thermal decomposition and combustion of AN have not been reported. In this paper, after studying the decomposition mechanism of AN in detail, we propose that solid super acids may exhibit a good catalysis ability because their super high acidity may strongly promote decomposition. Super acids are solid acids whose acidities are much higher than that of 100% H2SO4. Generally, their pKa values are less than −11.93 [16,17]. In industry, many organic synthesis reactions must use acid as a catalyst. Compared with liquid acids, solid acids have advantages such as high catalysis, high selectivity, lack of pollution and ease of separation from the reaction system. Thus, their application would be much suited to the organic industry. The studies of Guo [18] and Wang [19] considered that, due to the strong inductive effects of the S=O group, the adsorbed proton contributes as a Brønsted acid centre. The surface acid sites are associated with metal ions, whose acidic strength can be strongly enhanced by the induction effect of the S=O groups. With these results in mind, we decided to prepare this type of new catalyst and attempt to determine whether it would help to promote the decomposition of ammonium nitrate.
2. Experimental
2.1 Materials and instruments
In this study, analytically pure ammonium nitrate (AN) was produced by Guangzhou Hengyu Chemical Co. Ltd. Analytically pure titanium tetrachloride (TiCl4) was produced by Shanghai Huzhen Industrial Co. Ltd. Strong ammonia (25∼28%) was supplied by Jining Huide Chemical Co. Ltd. Concentrated sulphuric acid (98%) was provided by Chongqing Zhaohui Chemical Factory. Analytically pure ethanol was provided by Concord Tianjin Branch Co., Ltd. Deionized water was provided by Taiyuan Meijiayuan Beverage Co. Ltd. A muffle furnace model LHT04/16 (30°C∼3000°C) was made by Germany RETSCH Company. A vacuum pump model SHZ-CD (≤0.1 MPa) was made by Henan the Great Wall Instrument Factory.
2.2 Preparation of Nano SO42−/TiO2
Strong ammonia (25%) was diluted to ammonia solution with a concentration of 6 mol/L, and ethanol was subsequently introduced into the solution as a dispersant. Then, 23.75 g of TiCl4 was slowly added into 84 mL of the ammonia solution and the pH value was adjusted to 9∼10, using ammonia to ensure that Ti4+ completely converted to Ti(OH)4. After 24 h of aging, the precipitation was washed with H2O three times, with ethanol two times and once with acetone. After washing, the precipitate was dried at 50°C. The dried powder was carefully grinded in an agate mortar and tiny particles of amorphous TiO2 were obtained. With ultrasonic stirring, 2 g of amorphous TiO2 was put into the sulphuric acid solution and dipped in 30 min. After dipping, the amorphous TiO2 was dried at 80°C. The dried powders were grinded and calcined at 300°C, 400°C, 500°C or 800°C, and SO42−/TiO2 solid super acids were subsequently obtained.
To investigate the catalysis of SO42−/TiO2, 0.97 g of AN was blended with 0.03 g of SO42−/TiO2 by careful manual grinding and the obtained sample was tagged as [AN +3%SO42−/TiO2]. For comparison, an additional 0.97 g of AN was blended with 0.03 g of nano TiO2 by careful manual grinding and the obtained sample was tagged as [AN +3%TiO2].
2.3 Sample characterization
Morphology was observed with a field-emission scanning electron microscope (SEM, JEOL JSM-7500) and a Philips-Tecnai-12 transmission electron microscope (TEM). The phases of the samples were investigated using an X-ray diffractometer (XRD, Bruker Advance D8) with Cu K_α radiation at 40 kV and 30 mA. XPS analysis was performed using X-ray photoelectron spectroscopy (XPS) and a PHI5000 Versa-Probe (ULVAC-PHI). TG-DSC analysis and DSC-IR analysis were performed using a thermal analyser system (TG/DSC, Mettler Toledo), which was coupled with a Fourier transform infrared spectrometer.
3. Results and Discussion
3.1 Morphology and structure
The calcined powders were analysed using XRD and the results are shown in Fig. 1. The results indicate that the amorphous TiO2 transformed to anatase-phase TiO2 after it was calcined at 300, 400 or 500°C. With the increase in temperature, the intensity of the diffraction peak increased, which indicates that the crystallization degree of the sample gradually increased. In addition, the diffraction peak presented a width phenomenon, which implies that the crystal grain may have had a nano-scale size. According to the Debye-Scherrer equation (D=Kλ/(βcosθ)), the average grain size of SO42−/TiO2 (baked at 500°C) was calculated to be 25.2 nm. Via calcination at 800°C, the anatase-phase TiO2 transformed into rutile-phase TiO2, and its average grain size was more than 100 nm. In Fig. 1b, the XRD patterns of TiO2 and SO42−/TiO2 that were calcined at 500°C were compared. The result shows that TiO2 had a stronger peak intensity than SO42−/TiO2, which implies that surface acidification can delay the crystallization of TiO2. The average grain size of TiO2 was 29.7 nm, which is larger than that of SO42−/TiO2.
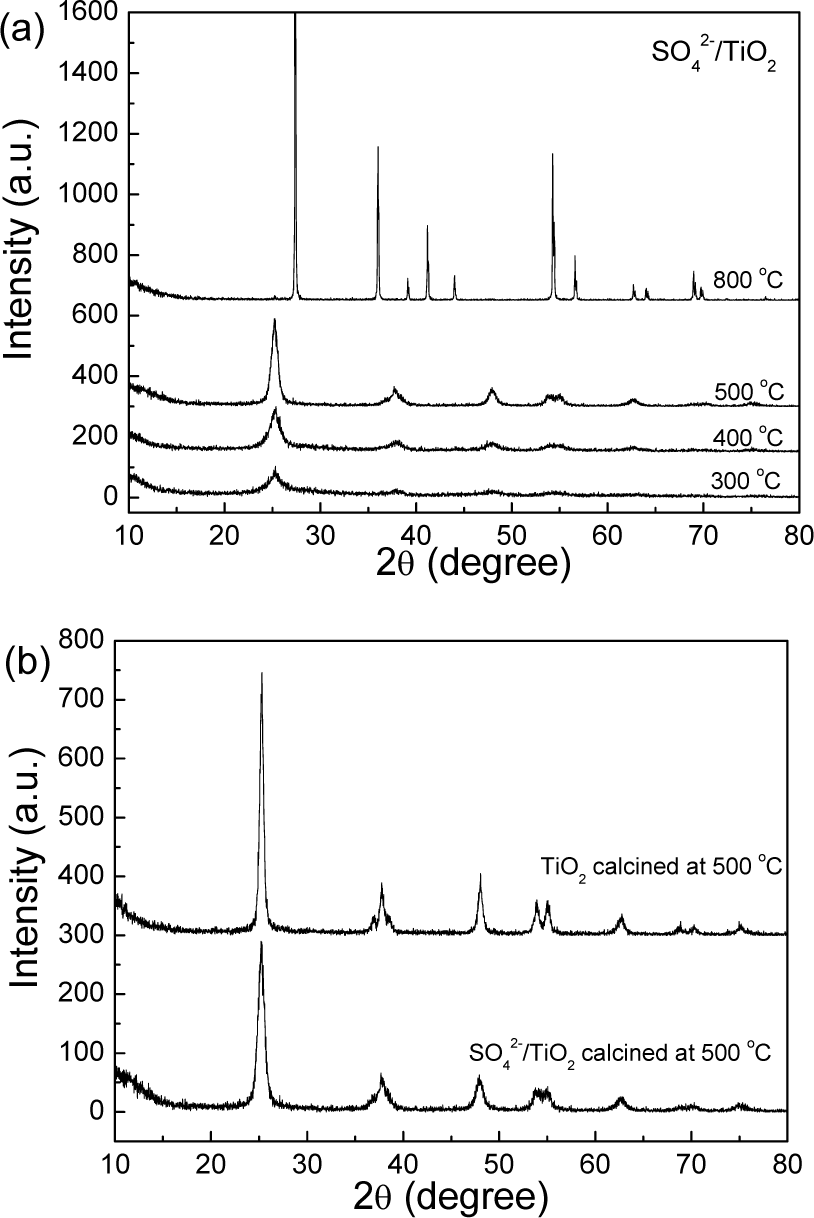
XRD patterns of samples calcined at different temperatures
Fig. 2 shows the XPS spectrum of SO42−/TiO2 calcined at 500°C. Three elements (i.e., S, Ti and O) were detected in the spectrum. The O and Ti peaks are located at the binding energies of 529.9 eV and 458.7 eV (463.8 eV), respectively. These peaks should reflect the elements in TiO2 because their intensity was notably strong. The peak at the binding energy of 168.5 eV is related to the 2p electron transition of the S element. The S element should originate from the SO42− anion on the surface of SO42−/TiO2, which implies that SO42− was fixed on the surface of TiO2 after acidification.

XPS spectrum of nano SO42−/TiO2 super acid
Fig. 3 shows the morphology and particle size of the non-acidification samples and acidification samples that were calcined at 500°C. Fig. 3a indicates that pure TiO2 (with a particle size of 25∼40 nm) showed a certain agglomeration. Fig. 3b shows that SO42−/TiO2 (with a particle size of 20∼30 nm) also massively agglomerated. Therefore, we had sufficient reason to reconsider the drying process in the fabrication of SO42−/TiO2 nanoparticles. Freeze-drying or supercritical drying may be more feasible. The powder was added into ethanol, and strong ultrasonic was used to disperse the agglomerate particles. Then, the obtained suspension was subjected to a TEM analysis. The TEM images are shown in Figs. 3c and d, and the particle sizes were measured and recorded. The specific size of each particle in Figs. 3c and d was carefully measured, and the statistical data are displayed in Figs. 3e and f. Fig. 3 shows that the average particle sizes (X̄) of TiO2 and SO42−/TiO2 were 33 nm and 29 nm, respectively, i.e., SO42−/TiO2 were somewhat smaller than TiO2 in terms of particle size, which is consistent with the XRD analysis results, i.e., surface acidification slightly delays the growth of particles (or grains).

SEM and TEM images of the samples: (a, c) nano TiO2; (b, d) nano SO42−/TiO2; (e, f) size distribution of nano TiO2 and nano SO42−/TiO2/ which was calculated from Fig. 3 (c and d)
3.2 Thermal analysis
Thermal analyses were performed using the TG-DSC technique and the results are shown in Fig. 4. Pure AN has no exothermic peak and four endothermic peaks. The first phase transition occurs at 52.9°C, which corresponds to the transformation from phase III to phase II (ΔH=13.9J g−1). The second phase transition (at 126.1°C) is the transformation from phase II to phase I (ΔH=40.1J g−1). The third endothermic peak (at 167.6°C, ΔH=55.4J g−1) reflects the melting of AN. The strongest endothermic peak depicts the decomposition process of AN. The decomposition begins at 239.2°C and has its peak point at 277.4°C (ΔH=888.9J g−1). In theory, liquid NH4+NO3− dissociates via a proton transformation (from NH4+ to NO3−) to generate NH3(g) and HNO3(g) (Eq. 1), which is the first decomposition step of almost all onium salts. This dissociation course does not spontaneously proceed (ΔG=+90.6 kJ mol−1) but it can occur after 2.18 kJ g−1 of heat is absorbed [20]. After the dissociation, the redox reactions (exothermic) between the oxidizer and the reducer begin. However, the amount and rate of heat release from the redox reactions are notably low. Thus, the presented decomposition course in the DSC traces is endothermic.

DSC, TG and DTG traces of the samples
The results in Fig. 4a also show that the main decomposition process of AN did not change with the doping of either SO42−/TiO2 or TiO2. The presented processes in the DSC traces remained endothermic, which implies that the addition of a nano catalyst cannot increase the reaction heat in thermodynamics. However, the onset and peak temperatures (To and Tp) decreased with the addition of nano catalysts. The catalytic effect of TiO2 is notably limited because, compared with pure AN, To decreased by only 7.4°C, and Tp seldom changed. In comparison, SO42−/TiO2 had a much stronger catalytic effect than TiO2, where To and Tp decreased by 19°C and 15.8°C, respectively. This result may imply that the SO42− ions on the surface of the catalyst particles are favourable for the thermal decomposition of AN. Fig. 4b shows the TG-DTG curves of pure AN, [AN +3%TiO2] and [AN+3%SO42−/TiO2]. In addition to the onset temperature, there was no obvious distinction among these TG curves. After the derivation calculus, the DTG curves were obtained and show the decomposition rates of the samples. These DTG peaks are similar to the DSC peaks.
3.3 Catalytic mechanism
The decomposition products of [AN+3%SO42−/TiO2] were detected using the DSC-IR technique and the results are shown in Fig. 5. Fig. 5a shows the stack plots of the gas products and Fig. 5b shows the infrared spectra of the gas products at the times of 528.9 s, 706.6 s, 770.5 s, 856.9 s and 920.8 s. It is clear that the decomposition products are mostly N2O and a small amount of H2O. This result indicates that the redox reactions between the dissociation products of 3% SO42−/TiO2-doped ammonium nitrate completely proceeded because no NH3(g) or HNO3(g) was detected. In theory, the thermal decomposition of AN has two mechanisms: ionization reaction (Eqs. 1–4) and radical reaction (Eqs. 1 and 5–7). Note that the final product of both channels is N2O, which is consistent with our experimental results. At a low temperature, the decomposition reaction should proceed according to the ionization mechanism because the rupture of the HO-NO2 bond (i.e., Eq. 5) only occurs at elevated temperatures (approximately 1300°C).
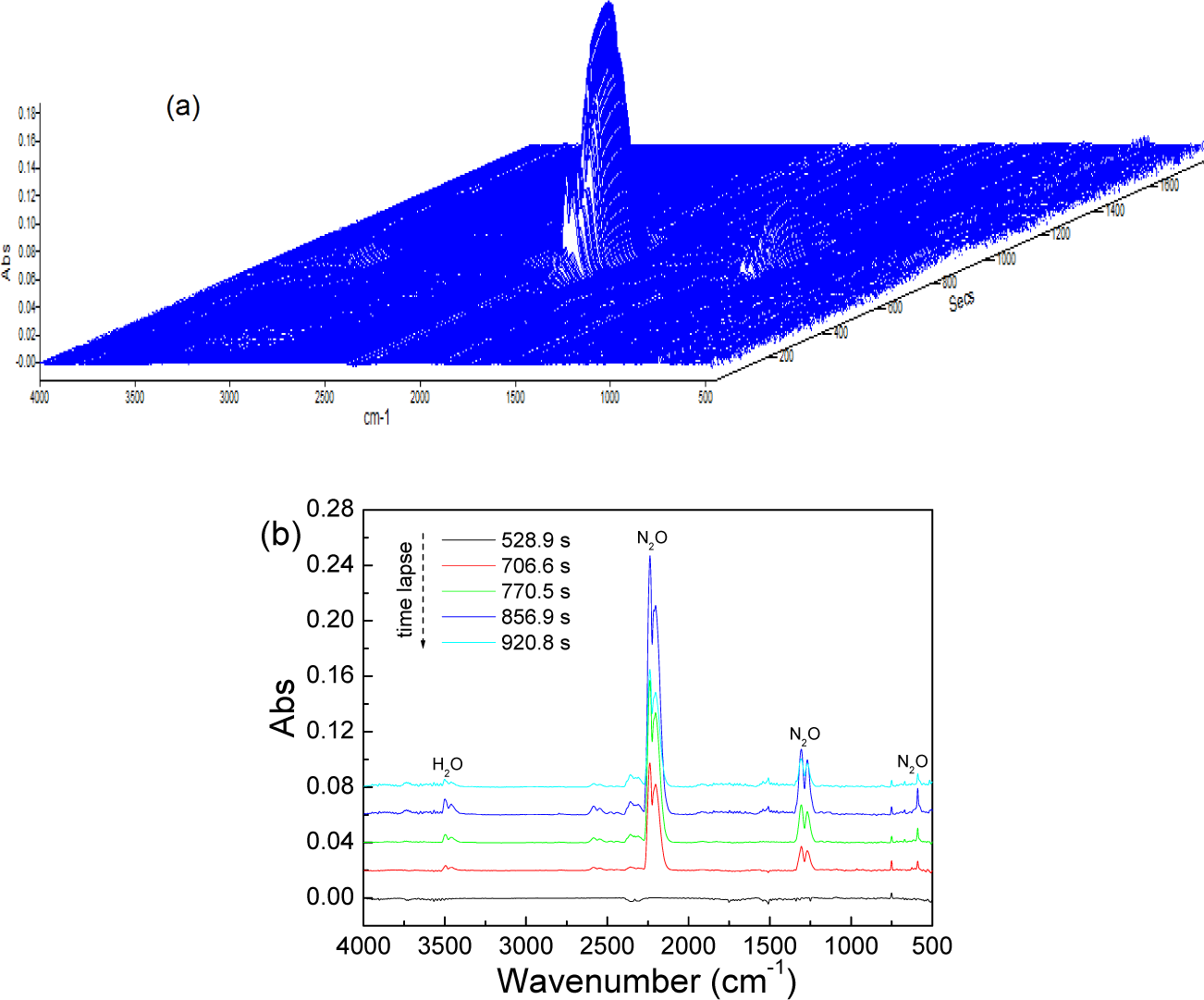
Decomposition products of the AN+3%SO42−/TiO2 mixture: (a) stack plots of gas products; (b) IR spectra of the gas products intercepted at different times
Generally, the reactions (Eqs. 2 and 5) are controlled steps for ionization reaction and radical reaction, respectively, because their rate constant is notably small. Hence, the issue of promoting AN decomposition may be addressed by finding a method to accelerate the limiting steps. Herein, we attempted to promote decomposition using simple chemical processes, based on the principle that a stronger acid can replace a weak acid. First, a type of acid, “HA”, was introduced (“H” means proton; “A” means anion), whose acidity was quantified by pKα. When we added HA into the decomposition system of AN, the reaction NH3+H +⇌NH4+ would occur if pKα(HA) was less than pKα(NH3). This process helped to fix HN3(g) as NH4+ to avoid the cessation of the decomposition by NH3 poisoning. Likewise, if pKα(HA)<pKα(HNO3), the reaction NO3−+H3O +⇌HNO3+H2O would begin. This process considerably increases the concentration of HNO3 in the decomposition system and accelerates the controlled steps (Eqs. 2 and 5). In particular, when pKα(HA)«pKα(HNO3), the decomposition of HNO3 is changed (see Eq. 8). Note that the decomposition of HNO3 is important for the thermolysis of ammonium nitrate [12].
In fact, many Lewis acid sites and Brønsted acid sites coexist on the surface of SO42−/TiO2 (Fig. 6). The studies of Guo [18] and Wang [19] considered that, due to the strong inductive effects of the S=O group, the proton contributes as a Brønsted acid centre. The surface acid sites are associated with metal ions, whose acidic strength can be strongly enhanced by the induction effect of S=O groups. Hence, we believe that, instead of Lewis acid sites, Brønsted acid sites are important in this process. The experimental result confirms that TiO2 did not present catalysis in the AN decomposition. Please note that there are many Lewis acid sites on the surface of TiO2 (Fig. 6). Some papers have reported that Lewis acid sites (on the surface of anatase titanium dioxide) could catalyse the decomposition of ammonia (2NH3→N2+3H2) [21-23]. According to the results, although the decomposition of NH3 (catalysed by Lewis acid sites) is a feasible process in thermodynamics, the reaction(s) processed notably slowly in kinetics. Meanwhile, please note that this dissociation of NH3 is endothermic (exactly like the pyrolysis of ammonia in industry). Moreover, for ammonia pyrolysis in industry, a high reaction temperature and a noble metal catalyst are indispensable.

Lewis acid sites and Bronsted acid sites on the surface of nano SO42−/TiO2
Molecular H2O, which is generated from AN decomposition and absorbed on the surface of SO42−/TiO2, can enable the transformation (Lewis acid transforms to Brønsted acid). However, liquid H2O can also cause a loss of SO42− from the surface because of the lixiviation of sulphate groups. Thus, in the condensed phase, H2O can weaken the catalysis of SO42−/TiO2. However, this factor is not important because the temperature of the AN decomposition is more than 210°C. In fact, Sun and MacNeil studied the catalysis of some inorganic acids in the AN decomposition [24, 25]. They found that hydrochloric acid had a distinctly higher catalysis ability than sulphuric acid [24]. Their reported catalysis mechanism of HCl and H2SO4 is not repeated here. Briefly, they used the function of the anion of the acid, whereas we used the function of the proton of the acid. In addition, a rocket designer would not introduce liquid acids into the formulation of any type of propellant because they would seriously hinder the preparation process. Hence, using solid acid may be a better choice. Moreover, in terms of acidity, solid super acids have a much lower pKα (<-12) than sulphuric acid (pKα=-3.0) and hydrochloric acid (pKα=-8.0).
4. Conclusion
To promote the thermal decomposition of ammonium nitrate, we prepared a nanometre solid super acid (SO42−/TiO2) as the catalyst. The best baking temperature of 500°C was determined using XRD analysis. SEM and TEM analyses show that the average size of SO42−/TiO2 particles is 29 nm. There is an S element on the catalyst surface, as confirmed via XPS analysis.
The thermolysis properties of pure AN and AN doped with 3%SO42−/TiO2 were probed using TG/DSC technology. Meanwhile, for comparison, the thermolysis of AN doped with 3%TiO2 was also analysed. The results indicate that SO42−/TiO2 helps to decrease the decomposition temperature by 15.8°C, but TiO2 did not show catalysis activity in the decomposition. Using DSC-IR technology, we conclude that the main decomposition products of [AN+3%SO42−/TiO2] are massive amounts of N2O and few H2O. Please note that there was no NH3 in the products. In the mechanism analysis, the function of SO42−/TiO2 was proposed based on the principle that a stronger acid can replace a weak acid. Due to the significantly low pKa value, the protons on the surface of SO42−/TiO2 cause the reactions NH3+H+⇌NH4+ and NO3−+H3O+⇌HNO3+H2O to occur. As a result, the concentrations of NH4+ ions and HNO3 increase on the surface of the catalyst, which promotes AN decomposition.
Footnotes
5. Acknowledgements
This research was supported by the National Natural Science Foundation of China (Grant No.: 51206081).