Abstract
A new method was used to prepare nanocomposites (copper oxide, copper iron oxide). The copper oxide and copper iron oxide were characterized by powder X-ray diffraction, transmission electron microscope, and X-ray photoelectron spectra. Copper oxide/graphene oxide nanocatalyst was prepared during which copper oxide nanoparticle was simultaneously anchored on graphene oxide sheets. Copper iron oxide/graphene oxide was also prepared. In this work, the catalytic performance of the synthesized materials on the thermal decomposition of ammonium nitrate was investigated by thermogravimetric differential scanning calorimetry and thermogravimetric mass spectrometry. The results of thermogravimetric differential scanning calorimetry experiments indicated that nanocomposites catalyzed thermal decomposition of ammonium nitrate significantly, especially copper oxide/graphene oxide. The synergistic effect of copper iron oxide was not found. The activation energy of ammonium nitrate mixture was calculated respectively by Kissinger method. The initial temperature, peak temperature, and activation energy were significantly decreased. A new phenomenon, nitrous oxide formed at a very low temperature, was observed by mass spectra. Two stages of thermal decomposition phenomenon of ammonium nitrate catalyzed by copper oxide/graphene oxide were first observed according to thermogravimetric mass spectrometry results. The thermal decomposition mechanism of ammonium nitrate with copper oxide/graphene oxide was proposed according to thermogravimetric differential scanning calorimetry and thermogravimetric mass spectrometry results.
Introduction
Ammonium perchlorate (AP) as the main oxidizer is using in most of the solid rocket propellants currently, but its products of combustion are toxic and not environmentally friendly. To overcome these environmental problems, it’s necessary to investigate the use of the so-called clean burning propellants. Thus, many researchers are going to develop environmentally friendly oxidizers. Ammonium nitrate (AN) has attracted attention many years ago as a potential propellant oxidizer for no HCl presented in production of AN decomposition. However, poor ignitability, hygroscopicity, low energy, low burning rate, and polymorphic transitions around room temperature limited the application of AN in solid rocket propellants. 1 –4 Many researchers have attempted to overcome these problems to develop a wide range of practical applications for AN-based propellants. It’s known that transition metal oxides (TMO) were effective additives to increase the burning rate and ignitability of AN propellants. Many researchers have reported that TMO was successfully used in AN-based propellant thermal decomposition. 5 –13
Graphene has attracted a great interest in both fundamental and applied areas. 14,15 It has excellent properties, especially huge surface area. 16 –18 In recent years, many papers have reported graphene or graphene oxide (GO) to catalyze AP, 19 –24 especially GO, which can easily absorb polar molecules to form different nanocomposites on carbon nanosheets. 25,26 However, a few literature has reported graphene- or GO-supported nanocomposites to catalyze AN. Therefore, it attracted great interests in developing GO-supported nanocomposites to catalyze AN thermal decomposition.
Ferric oxide (Fe2O3) and copper oxide (CuO) have been studied for excellent catalytic activity in solid propellants. 27 –31 Copper iron oxide (CuFe2O4) has also been reported in AP catalytic decomposition for its synergistic effect. 32 –34 It has been reported that nano-CuO and Fe2O3 can catalyze decomposition in AN-based propellants. 3,7,8,11–13 However, no literature reported CuFe2O4 composite oxides catalyzed AN thermal decomposition. For synergistic effect of composite oxides, thermal decomposition of AN can also be catalyzed by CuFe2O4 composite oxides.
Since pure nanoparticles were likely to aggregate, the catalytic activity would be decreased notably. Therefore, a facile and reliable method was to anchor nanocomposites onto a supporter to prevent aggregation. Graphene/GO may be an ideal catalyst supporter. Based on this viewpoint, we expected that graphene/GO can dramatically improve the catalytic activity of nanoparticles as a catalyst supporter. Herein, we report a facile synthesis of CuO and CuFe2O4 nanocomposites by an emulsion combustion method. Firstly, nanoparticles were characterized by powder X-ray diffraction (XRD), transmission electron microscope (TEM), and X-ray photoelectron spectra (XPS). The catalytic effect of CuO, CuO/GO, CuFe2O4, and CuFe2O4/GO nanocomposites on the thermal decomposition of AN was investigated by thermogravimetric differential scanning calorimetry (TG-DSC). Thermal decomposition kinetics of AN mixture were analyzed. The thermal decomposition mechanism was also analyzed by thermogravimetric mass spectrometry (TG-MS). The probable mechanism was also proposed.
Experimental sections
Materials
Copper nitrate (Cu(NO3)2·3H2O; AR), ferric nitrate (Fe(NO3)3·9H2O; AR), and ammonium nitrate (AN; AR) were used without further purification. Sorbitan monooleate (SMO) and diesel were industrial materials. GO was self-made in laboratory from purified natural graphite (230 mesh, Qingdao Zhongtian Company, China) according to the method reported by Hummers and Offeman. 35
Synthesis and characterization of nanocatalyst
Synthesis of CuFe2O4 nanocatalyst by new method
Nano-CuO and CuFe2O4 were synthesized by a new method. This process of synthesis of CuO and CuFe2O4 nanoparticles was not reported in the previous study. It has two steps: (1) preparation of emulsion and (2) calcination of emulsion in furnace. The formation of emulsion was as follows: Water (H2O):AN:Cu(NO3)2·3H2O:Fe(NO3)3·9H2O:SMO:diesel = 16:38:9.19:30.81:1.5:4.5 H2O:AN:Cu(NO3)2·3H2O:SMO:diesel = 16:38:40:1.5:4.5 Preparation of emulsion: The procedure of emulsion prepared involved two steps. First, the oxidizer solution (H2O, AN, Cu(NO3)2·3H2O, and Fe(NO3)3·9H2O) was prepared by dissolving the ingredients in a large stainless steel beaker heated to a temperature of approximately 80°C. The oil phase (SMO and diesel) was then poured into the mixer bowl, when it was heated to 60°C, and maintained. Then, it was mixed with a speed of approximately 200 r/min, and the hot oxidizer solution was slowly poured into the bowl. After pouring, the speed of mixing was accelerated (approximately to 1200 r/min) and continued for a few seconds to achieve the final refinement. Calcination of emulsion in furnace: The emulsion sample was then calcined in furnace at a temperature above 600°C. After heating for 2 h, CuFe2O4 nanoparticles were collected. The solid was washed with ethanol and then used for further experiments. The nanoparticles were characterized by XRD and TEM. The preparation method of nano-CuO was similar to that of CuFe2O4.
Synthesis of CuFe2O4/GO nanocatalyst and sample preparation
The CuFe2O4/GO nanocomposite was synthesized in the following procedure. Hundred milligram of GO powder and 5 mL of n-butylamine were dispersed in 100 mL H2O by sonication, forming stable GO colloid. After 1 h, a dispersion of 200 mg CuFe2O4 was sonicated in 100 mL H2O, and 5 mL ethylenediamine was added to the GO colloid solution. Subsequently, the mixture was heated to 60°C with magnetic stirring for 6 h. The bulk samples can be obtained by centrifugation of the mixture, and then washed with deionized H2O and ethanol for several times. The CuFe2O4/GO nanocomposite was obtained after the precipitate was dried at 60°C under vacuum for 12 h. The preparation method of nano-CuO/GO was similar to that of CuFe2O4/GO.
For the catalytic activity studies on CuO, CuFe2O4, CuO/GO, and CuFe2O4/GO, nanocatalyst was mixed with AN separately in a weight ratio 98:2 (wt%) by blending on a mechanical stirrer. The samples were named as follows: 1#: 2% CuO + 98% AN; 2#: 2% CuFe2O4 + 98% AN; 3#: 2% CuO/GO + 98% AN; and 4#: 2% CuFe2O4/GO + 98% AN.
XRD, TEM, and XPS analyses
The powder XRD analysis of the samples was carried out with a Bruker D8 (Bruker, Germany) SuperSpeed apparatus operating with a copper (Cu) Kα radiation. The morphologies of the samples were investigated by JEM-2100 TEM (JEOL, Japan). XPS recorded on a Thermo ESCALAB250 (Thermo Fisher Scientific, USA) X-ray photoelectron spectrometer, using aluminum K α (hυ = 1486.6 eV). The binding energies were calibrated using C1 s peak of contaminant carbon (BE5284.6 eV) as an internal standard.
Thermal decomposition of AN samples
Thermal decomposition experiments of AN samples were carried out under a nitrogen atmosphere with a NETZSCH STA449C (NETZSCH, Germany) instrument. In all experiments, 10 mg sample was loaded in a closed ceramic crucible with a pinhole in the cap and heated up from room temperature to 400°C. Nitrogen was used as a carrier gas with a flow rate of 20 mL/min. The heating rate was 2.5 K/min, 5 K/min, 10 K/min, and 20 K/min.
TG-MS experiments
The TG-MS test was performed with NETZSCH STA449C system and MS (NETZSCHQMS403C, NETZSCH, Germany) system. NETZSCHQMS403C has the following conditions: ionizing electron energy of 70 eV, quartz capillary gas connector, and pressure injection 1000 mbar. Approximately 2 mg of sample was heated from 35°C to 400°C. The heating rate was 10 K/min, and the sample was carried in aluminum oxide crucibles. High-purity argon was used as purge gas with a gas flow rate of 20 mL/min.
Decomposition kinetic analysis
The kinetics of the AN thermal decomposition with and without additive can be determined using the Kissinger method, 36 which uses the peak temperature (T m) at which the reaction rate is at a maximum as obtained from the DSC curves of samples decomposition in experiments with different heating rates. The equation was as follows
where β is the heating rate (K/min), Tm is the temperature, A is the pre-exponential factor, R is the gas constant, and Ea the is activation energy. The slope and intercept from the line plot of against 1/T m were used to obtain the activation energy and pre-exponential factor, respectively.
Results and discussion
Characteristic of nanocatalysts
The nanocatalysts were characterized by XRD, TEM, and XPS. The XRD results were listed in Figure 1. The diffraction peaks located in the range from 20° to 80° were all ascribed to the characteristic Bragg reflections of CuO (JCPDS 34-0394), α-Fe2O3 (JCPDS 33-0664), and CuFe2O4 (JCPDS 30-1346), respectively. No obvious diffraction peaks of GO or graphene were observed. It indicated that CuO, α-Fe2O3, and CuFe2O4 particles were all attached to GO surface. Figure 2 showed the TEM images of as-synthesized CuO, CuFe2O4, CuO/GO, and CuFe2O4/GO. The results found a slight aggregation of metal oxide particles (CuO and CuFe2O4). The average sizes of CuO and CuFe2O4 were 83 and 13 nm, respectively. The GO prevented the aggregation of metal oxide particles , and most CuO and CuFe2O4 nanoparticles distributed randomly on GO sheets.

XRD of as-synthesized (a) Fe2O3, (b) Fe2O3/GO, (c) CuO, (d) CuO/GO, (e) CuFe2O4, and (f) CuFe2O4/GO composites. XRD: X-ray diffraction; Fe2O3: ferric oxide; GO: graphene oxide; CuO: copper oxide; CuFe2O4: copper iron oxide.
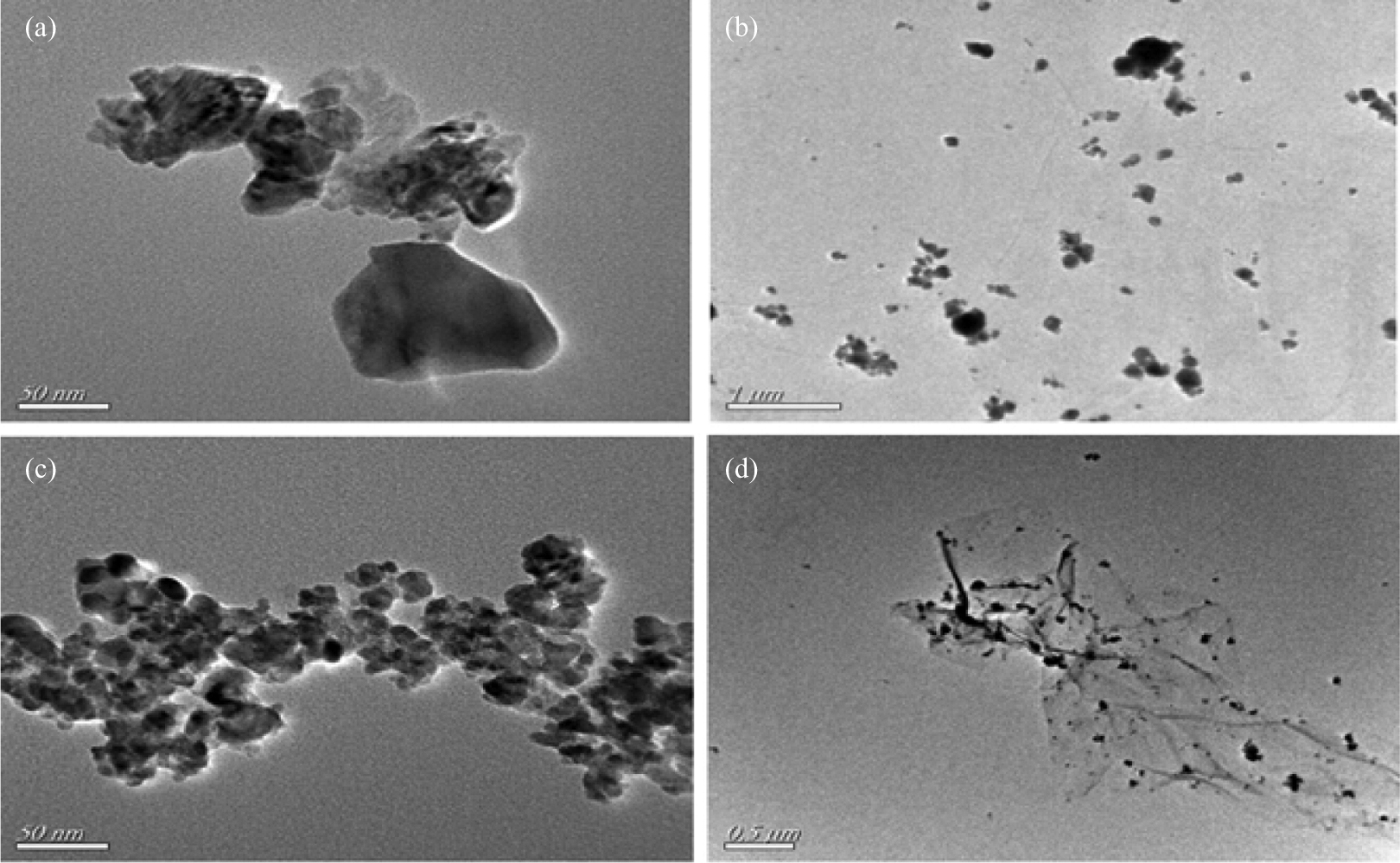
TEM images of (a) CuO, (b) CuO/GO, (c) CuFe2O4 and (d) CuFe2O4/GO composites. TEM: transmission electron microscope; CuO: copper oxide; GO: graphene oxide; CuFe2O4: copper iron oxide.
To explore Cu and iron (Fe) elements on the surface of CuFe2O4 composites, XPS spectra were measured. Figure 3 depicted the Cu 2p and Fe 2p XPS spectra of different samples. The Cu 2p spectrum of CuO and CuFe2O4 samples showed the typical structure. The Cu 2p signal was composed of peak at 933 eV. The result was assigned to the presence of CuO species. The Cu 2p3/2 and Cu 2p1/2 peaks were accompanied by distinct shake-up at about binding energies of 944 eV and 963 eV, which were the characteristic of Cu2 +. The results testified that a part of Cu species were incorporated into the framework in the form of CuFe2O4. Comparing CuO with CuFe2O4 XPS spectra, we can confirm the Cu species was on the external surface of CuFe2O4 catalysts. The XPS spectra of Fe 2p of CuFe2O4 catalysts were showed in Figure 3(b). For Fe 2p XPS spectra, each peak can be split. One of the Fe 2p3/2 doublet peaks was at 710.8 eV and the second one was at 713.8 eV. Hence, from XPS results, the peak of Cu2p and Fe2p almost has no change. From XPS results, we can also confirm the Fe species was on the external surface of CuFe2O4 catalysts. The species ratio of Cu:Fe = 16.62:33.1. The result was the same with mole ratio of CuFe2O4. The XPS results showed that ferric and Cu species were distributed well proportionly on the surface of CuFe2O4 sample.

(a) Cu 2p XPS spectra of samples; (b) Fe 2p XPS spectra of samples. Cu: copper; XPS: X-ray photoelectron spectra; Fe: iron.
The influence on the thermal decomposition of AN with catalysts
The CuO, CuFe2O4, CuFe2O4/GO, or CuO/GO composite was explored as a catalyst to the thermal decomposition of AN, respectively. The reference has found that GO has little influence on thermal decomposition of AP even at high temperature (>350°C). 20,21 Hence, GO was not investigated to catalyze thermal decomposition of AN again for AN decomposition temperature lower than 350°C. The performances of CuO, CuFe2O4, CuFe2O4/GO, and CuO/GO composite on the thermal decomposition of AN were investigated by TG-DSC. The results were showed in Figure 4. The DSC curve of Figure 4(a) revealed that prior to 170°C, appeared endothermic peak was phase transition of AN; after 170°C, thermal decomposition of AN was carried out. The other samples also have the same tendency. As can be seen from Figure 4, it’s shown that, compared with pure AN, decomposition temperature of AN mixture with 2% CuO and CuO/GO shifted to a low temperature notably. Aforementioned papers found that synergistic effect of CuFe2O4 presented in catalyzed AP. 32 –34 However, the present article found no synergistic effect of CuFe2O4. The reference found that Cu-based catalysts have been found to be highly active at relatively low temperatures (<350°C). But Fe-based catalysts were at high temperatures (>300°C). 37 For thermal decomposition temperature of AN about 300°C at the present experimental condition, the Cu activity was stronger than Fe. The XPS results showed that Fe2O3 distributed on the surface of CuFe2O4 sample was well proportioned. Therefore, we can explain the synergistic effect of CuFe2O4 was not found in AN mixture thermal decomposition process.

DSC curves for: (a) pure AN, (b) AN + CuFe2O4/GO, (c) AN + CuO/GO, (d) AN + CuFe2O4, and (e) AN + CuO at a heating rate of 10 K/min. DSC: differential scanning calorimetry; AN: ammonium nitrate; CuFe2O4: copper iron oxide; GO: graphene oxide; CuO: copper oxide.
When GO was added as a catalyst supporter, decomposition temperature of AN with CuO/GO shifted to a low temperature about 5°C. It means the catalytic activity of catalytic agent increased. However, to our surprise, the peak temperature of AN with CuFe2O4/GO shifted to a low temperature about 7°C. It must combine with activation energy to confirm catalysis results.
In order to analyze the catalysis of CuO, CuFe2O4, CuO/GO, or CuFe2O4/GO to AN thermal decomposition, the activation energy and initial decomposition temperature were listed in Table 1. From Table 1, the initial temperature and peak temperature significantly decreased when CuO/GO was added to AN, and also CuFe2O4/GO has excellent catalysis but catalytic activity was lower than CuO/GO. We also have found the activation energy required for the thermal decomposition of pure AN to be 162.7335 kJ/mol. Although the reported activation energy for pure AN decomposition varies from 86.2 kJ/mol to 206.9 kJ/mol, previous researchers in this area agreed that the overall decomposition reaction of AN was described by the first-order reaction kinetics. 39 The activation energy of AN mixture decreased notably, especially in 3#. In other words, the reaction kinetics process of AN with additives perhaps was changed. The reaction kinetics process was not first-order reaction kinetics. Hence, according to initial temperature, peak temperature, and activation energy, we can obtain that additives catalyzed AN thermal decomposition significantly, especially CuO/GO. The difference of CuO/GO and CuFe2O4/GO was also observed. The activation energy of CuFe2O4 with and without GO almost has no discrepancy. But CuO was very notable. The reason perhaps was that Fe2O3 has little effect on AN thermal decomposition. 8 Hence, the effective content of catalytic activity materials of CuFe2O4/GO was fewer than CuO/GO. In other words, CuO/GO has more remarkable catalytic activity than CuFe2O4/GO to AN thermal decomposition.
Thermal properties of AN with additives.a

AN: ammonium nitrate; CuO: copper oxide; CuFe2O4: copper iron oxide; GO: graphene oxide; T i: initial decomposition temperature; T p: peak temperature; E: activation energy; lnA: pre-exponential factor; R 2: correlation coefficient.
a T i, and T p were valued at a heating rate of 10 K/min. T i was temperature at the 5% mass loss according to Li et al. 38
In the experiment process, we also found the sample mass notably effected the AN thermal decomposition. The results were listed in Online Supplementary Figure S1. Li et al. 38 also found the same results. Koga found that the activation energy of AN decomposition decreased slightly with increasing sample mass in the range of 5–15 mg using TG. 40,41 Hence, we selected less mass sample to analyze TG-MS in the next TG-MS experiments.
Evolved gas analysis
In order to analyze the decomposition mechanism of AN with additives, samples were chosen to perform the evolved gas analysis. Real-time mass spectral analysis of the evolved gases was also performed. The DSC curves of samples were listed in Online Supplementary Figure S2. The MS results of the evolved gases were listed in Online Supplementary Figures S3 to S6. It showed the ion current curves for mass-to-charge (m/z) ratios = 16, 17, 18, 30, 44, 46, 63. The peaks were assigned to ammonia (NH3; m/z = 16, 17), H2O (m/z = 17, 18), nitic oxide (NO; m/z = 30), nitrous oxide (N2O; m/z = 44), nitric acid (HNO3; m/z = 30, 46, 63), and nitrogen dioxide (NO2; m/z = 46).
For dissociation reaction of AN presented even at low temperature, 42 the species (m/z = 17) at low temperature appeared, and it was ascribed to NH3. From Online Supplementary Figures S3 to S6, the curve change showed the presence of NH3. Then, NH3 can further undergo pyrolysis but needs high temperature. The HNO3 (g) (m/z = 63) was also detected, and it can demonstrate dissociation reaction of AN presented at very low temperature. The NH3 was found at 130°C in isothermal experiments. 43 Liu 44 also detected species (m/z = 15, 16, ·NH, ·NH2) at low temperature (100°C). However, Liu thought it presented at very high temperature. 45,46 During the reaction process, NO2 (m/z = 46) was also detected. The species (m/z = 44) was N2O not CO2 at present experimental condition.
Based on above analysis of the results from the present study, the thermal decomposition of AN with additives has been changed. And it needed to further analyze in the next paragraph.
Catalytic mechanism of the thermal decomposition of AN catalyzed by nanocomposites
Thermal decomposition of AN has been researched widely. 1 –4 Many researchers have been proposed thermal decomposition mechanism. Oxley proposed a widely accepted mechanism. Oxley 47,48 showed that the decomposition mechanism of AN followed two pathways: an ionic reaction and a radical reaction. The ionic reaction occurred at low temperature and has relatively low speed. The radical reaction occurred at high temperature.
AN melted at 169°C and began to decompose as soon as it melted. Reaction (2) was generally accepted as the thermal decomposition initiating reaction, which was an endothermic proton transfer. However, Izato and Miyake found that the molten AN was first dissociated to









However, since the activation energy of the homolysis of HNO3 was very high at about 190 kJ/mol, it was the rate-controlling step. Hence, at high temperature, the ionic mechanism of AN decomposition was overtaken by radical reactions,
51
with homolysis of HNO3 being the rate-controlling step
3
as shown in reaction (4). The MS also detected NO2. The reference also found the energy barrier was lowest in different reaction pathways.
52
Then amidogen radical (
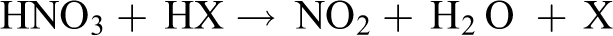


Fe2O3 has showed excellent catalysis in NH3-Selective Catalytic Reduction (SCR). It was widely accepted that it can interact with NH3. 57 The reaction was as follows (reaction (12))

CuO has also been found to have same reaction. When NH3 was absorbed on the nanoparticle surface, the reaction carried out by the formation of CuO and NH3 and amine (NH2).
58
However, Vargeese and Muralidharan proposed new viewpoint.
59
There was no NH2 formed during the reaction process. If NH2 formed, the curve of ion current would change. The results of ion current curve of NH2 were listed in Figure 5. The discrepancy of different samples was little except CuO/GO. At about 190°C, the ion current notably changed, which means NH2 formed. It can be oxidized easily by oxidizing gas. To our surprise, ion current curve of N2O was very strange. From Figure 6, we can find that N2O formed at very low temperature of 3# at about 130°C. Chen et al.
60
thought N2O was formed when NH3 reacted with NOx at less than 200°C. According to Andersen et al.’s
50
results, the NO2 must be presented to form N2O. However, at present condition, NO2 was very difficult to form below 200°C or little amount. Oxidizing gas was just only HNO3, it can also be found at very low temperature.
42
Another oxidizing species was
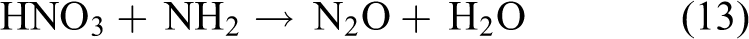

Ion current curve of NH2 (m/z = 16). NH2: amine; m/z: mass-to-charge ratio.

Ion current curve of N2O (m/z = 44). N2O: nitrous oxide; m/z: mass-to-charge ratio.
However, from Figure 6, we also found that N2O was formed in two stages. When temperature was heated to more than 200°C, the NOx was detected. It means NH2 can be oxidized by NOx. Hence, the two stages of N2O mean the mechanism of AN thermal decomposition was changed. The mechanism of AN thermal decomposition was proposed. At low temperature (<200°C), most likely mechanism was that NH2 was oxidized by HNO3 or
Zhang et al. thought NO played an important role in the NOx and NH3 catalytic reaction by CuO. 37 However, Figure 7 showed no change in the ion current of NO. Hence, in the present research, the mechanism of AN thermal decomposition might be different from the results of Zhang et al. 37 and Janssens et al. 59

Ion current curve of NO (m/z = 30). NO: nitric oxide; m/z: mass-to-charge ratio.
Conclusion
In summary, CuO/GO and CuFe2O4/GO nanocomposites were successfully obtained by a new emulsion combustion method and they showed an intensive catalytic effect on the thermal decomposition of AN. The catalytic agent was characterized by XRD, TEM, and XPS. The initial temperature, peak temperature, and activation energy were notably decreased when CuO/GO was added to AN. Fe2O3 distributed on the surface of CuFe2O4, and Fe2O3 has little effect on AN thermal decomposition. Therefore, the synergetic effect was not found when CuFe2O4/GO was added to AN. An important phenomenon was that N2O was formed at very low temperature around 140°C during the AN thermal decomposition process. We considered that it was formed by HNO3 and NH2 reaction at low temperature. The mechanism of thermal decomposition of AN with CuO/GO was proposed. The formed intermediate of NH2 was found to be a very important factor to progress the catalytic reaction of AN decomposition.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The article was supported by Inner Mogolia Sheng An Chemical Ltd Co. and the Foundation of Jiangsu University of Advanced scholars (15JDG159), Natural Science Foundation of Jiangsu Colleges and Universities (16KJB150010).

