Abstract
Nanostructured TiO2 was successfully synthesized via a facile one-pot solvothermal reaction followed by calcination. Hydrolysis and polycondensation of titanium butoxide (Ti(OR)4) were performed in the presence of sodium dodecylbenzenesulfonate (SDBS). The morphologies, crystallinity and compositions of obtained samples were identified by the methods of X-ray diffraction (XRD), Brunauer-Emmett-Teller (BET) and transmission electron microscopy (TEM). It was found that the nanostructured TiO2 with an average diameter of 10±5 nm had the crystal type of anatase. A good specific surface was also obtained by the standard multipoint BET method (119.2 m2/g). As the anode materials for the lithium-ion batteries (LIBs), the anatase phase TiO2 demonstrated a relatively high gravimetric specific capacity of 264.8 mAh
1. Introduction
The demand for rechargeable batteries with high energy density has become more and more indispensable with the gradual deterioration of the ecological environment. LIBs have been considered as one of the most promising, renewable and intermittent energy sources, owing to their advantage in terms of long-term cost savings, gravimetric and volumetric energy density, and environmental friendliness [1-3]. Whilst the achievement of high power density is hindered by kinetic problems with the electrode materials, graphite/carbon is an anode material widely used in commercial LIBs. The graphite/carbon electrode, however, has its limitations and weaknesses, such as safety properties, structural deformation, electrical disconnection and initial loss of capacity [4,5]. The initial loss of capacity arises from the formation of unstable solid electrolyte interphase (SEI) films [6,7]. Therefore, in recent years, anode materials with higher lithium insertion potential have received a lot of attention.
TiO2 arouses wide interest as an anode material for high rate LIBs due to its good reversibility, small volume change during charge-discharge process, low cost and environmental friendliness [8-10]. For TiO2, eight polymorphs are known: rutile (space group P42/mnm), anatase (I41/amd), brookite (Pbca), TiO2(B) (“bronze”, C2/m), TiO2(R) (“ramsdellite”, Pbnm), TiO2(H) (“hollandite”, I4/m), TiO2(II) (“columbite”, (a-PbO2) Pbcn) and TiO2(III) (“baddeleite”, P 21/c) [11,12]. Thus, anatase TiO2 is the most thermodynamically stable structure and is also the most common natural form [13]. The anatase phase of TiO2 itself is an active material towards lithium insertion, with a theoretical capacity of 335 mAh g−1. However, the usually measured capacity of 168 mAh g−1 derived from the rate of 1 C is used in the literature [14,15]. About 0.6 mol Li+ can be inserted per mol TiO2 at 1.78 V (vs. Li/Li+) [16]. Despite these advantages, TiO2 suffers from low electronic conductivity and Li+ diffusivity [17], which will seriously limit the rate capability and its wide practical application. The mesoporous nanoparticles can improve this problem, which have large surface areas, short diffusion lengths and fast diffusion rates along many of their grain boundaries [18,19]. Good enhanced capacities, high rates of performance, powerful retention abilities and many novel Li storage systems have been found to benefit from nanometre-sized effects [20-23]. Nanostructure materials generally refer to nanometre-sized particles at a range of 1–100 nm. When the size of materials reduces to the wavelength of electrons and optics, these materials perform more efficaciously in catalysis, optics and electrics because of their large specific surface areas and surface atomic numbers. The nanoparticles, whose surface atoms have a higher unsaturation due to the lack of adjacent atoms, possess higher activity. The large surface area of the structure, if present, allows more interfacial contact between the active material and the electrolyte [24]. These nanoscale crystallites, which are building blocks of the hierarchical structure, support fast diffusion of Li+ and electrons [25]. It has shown that organizing nanoparticles is beneficial to the reversible storage of Li+.
Satisfactory results have been obtained by using nanostructured TiO2, which can be beneficial to both power rate and cycle life. For nanostructured rutile TiO2, Pfanzelt et al. [26] recently reported a reversible capacity of about 315 mAh g−1 (0.94 Li/TiO2) within the potential window extending from 0.1 V to 3.0 V. TiO2-B nanowires, as studied by Armstrong et al. [27], which presented a rather interesting reversible capacity of around 200 mAh g−1 (0.60Li/TiO2) for an applied current density of 200 mA g−1.
In this paper, we report a simple synthesis of nanostructured TiO2 with a diameter of 10±5 nm, which has been successfully synthesized via a facile one-pot solvothermal reaction followed by calcination. It only uses Ti(OR)4, nitric acid, absolute ethanol and SDBS as raw materials. What is novel about our work is that we improve the preparation procedures of TiO2 nanoparticles. In general, direct synthesis of TiO2 nanostructures under mild reaction conditions, without using any templates or additives, is still difficult. An additional problem is the fact that titanium alkoxide precursors tend to hydrolyse in the presence of moisture [28]. To prevent such hydrolysation of titanium alkoxides, acids are usually used. In this article, low water component, excessive alkoxide and slow hydrolysis rate contribute to the formation of a Ti-O-Ti chain [29]. We need partial hydrolysis of titanium butoxide before the solvothermal process, rather than complete hydrolysis. The proportion of deionized water and nitric acid is significant for the preparation of nanostructured TiO2. During the solvothermal process, the coulombic force and van der Wals force will lead to the reunion among nanoscale particles. SDBS, which is a kind of surfactant, can improve the system interfacial state and reduce the solid-liquid interfacial energy. By reducing reunion effectively, SDBS contributes to the preparation of nanostructured TiO2 with a narrow particle-sized distribution and good dispersibility. By taking advantage of nanostructured TiO2 (anatase) as anode materials for LIBs, we have demonstrated that the nanostructured TiO2 (anatase) possesses high electrochemical activity.
2. Experiment
2.1 Synthesis of Nanostructured TiO2 (Anatase)
The preparation of nanostructured TiO2 was based on the combination of the hydrolysis and crystallization of Ti(OR)4 (Beijing Reagent Factory, China) in the presence of SDBS, followed by the solvothermal crystallization at 150 °C. In a typical procedure, 0.2849 g SDBS, 0.6 mol nitric acid and 1.8 mol distilled water were dissolved in 7 ml absolute ethanol to form a solution. Then this solution was added slowly to the other solution, which was composed of 9 ml titanium n-butoxide and 15 ml absolute ethanol at room temperature with vigorous stirring. The sol was then stirred for at least 30 minutes. After that, the mother liquor was transferred to a Teflon -lined autoclave, which was filled to 80% and tightly closed for solvothermal reaction. The solvothermal treatment was carried out at 150 °C for one day, three days or six days. In this way, the white powder product was obtained. The prepared precipitate was washed with distilled water and then dried at 60 °C for 24 hours. Finally, the obtained sample was thoroughly grinded by an agate mortar and annealed at 450 °C for four hours in order to increase the crystallinity of the sample.
2.2 Characterization and Electrochemical Property
The crystal structure of samples was determined by XRD (D/Max2500PC X-ray diffractometer with Cu radiation λ=0.15406 nm). The microstructures and morphologies of samples were investigated with transmission electron microscopy (TEM, JEOL JEM-2100F, JEOL JEM-2100HR). The standard multipoint BET method, which presented the nitrogen adsorption–desorption isotherms, was used to calculate the specific surface areas of samples.
The electrochemical performances were tested by CR2025 coin cells. A mixed slurry of the as-prepared TiO2 materials (80 wt%), carbon black (10 wt%), polyvinylidene fluoride (PVDF) binder (10 wt%), and N-methyl pyrrolidone (NMP) solvent was coated on a sheet of Cu foil using a doctor blade and then dried at 120 °C under vacuum conditions for 12 hours.
Coin cell assembly was carried out in a glovebox filled with ultra-pure argon. The button cell comprised the TiO2 sample, the separator, the lithium foil as the counter electrode and LB315 as the electrolyte. Galvanostatic charge and discharge tests were performed at room temperature on a LAND CT-2001 battery tester, while extraction tests were carried out between 1.0 V and 2.5V. Cyclic voltammetry (CV) and electrochemical impedance spectroscopy (EIS) tests were performed on a CH650D electrochemical workstation. CV tests were carried out at a scan rate of 0.1 mV s−1 within a test voltage range of 1–2.5 V (vs. Li/Li+) and the frequency range of 30 mHz-100 KHz. Electrochemical impedance spectroscopy (EIS) studies for the TiO2 batteries were carried out in the frequency range 0.01Hz to 100 KHz, with an amplitude of 5 mV.
3. Results and Discussion
The crystal structure of the samples, which were obtained via a simple solvothermal process and subsequently via a thermal treatment, had been determined by XRD. Fig.1 shows the XRD patterns of the nanosized TiO2 with a different reaction time during the solvothermal process. All samples, for which the sharp and clear diffraction peaks in the XRD patterns could be well indexed based on the anatase phase of TiO2 (JCPDS No. 21-1272), presented good crystallization. Compared with one day and three days, the sample for six days of solvothermal reaction had the smaller grain size. The average grain size of the nanocrystals was calculated based on the widths of the major diffraction peak (2θ= 25.3°) (along the (101) plane) using the Scherrer formula. The result showed that TiO2 (six days) was smaller than TiO2 (three days), which agreed well with the display of the crystallite sizes measured with the TEM images.
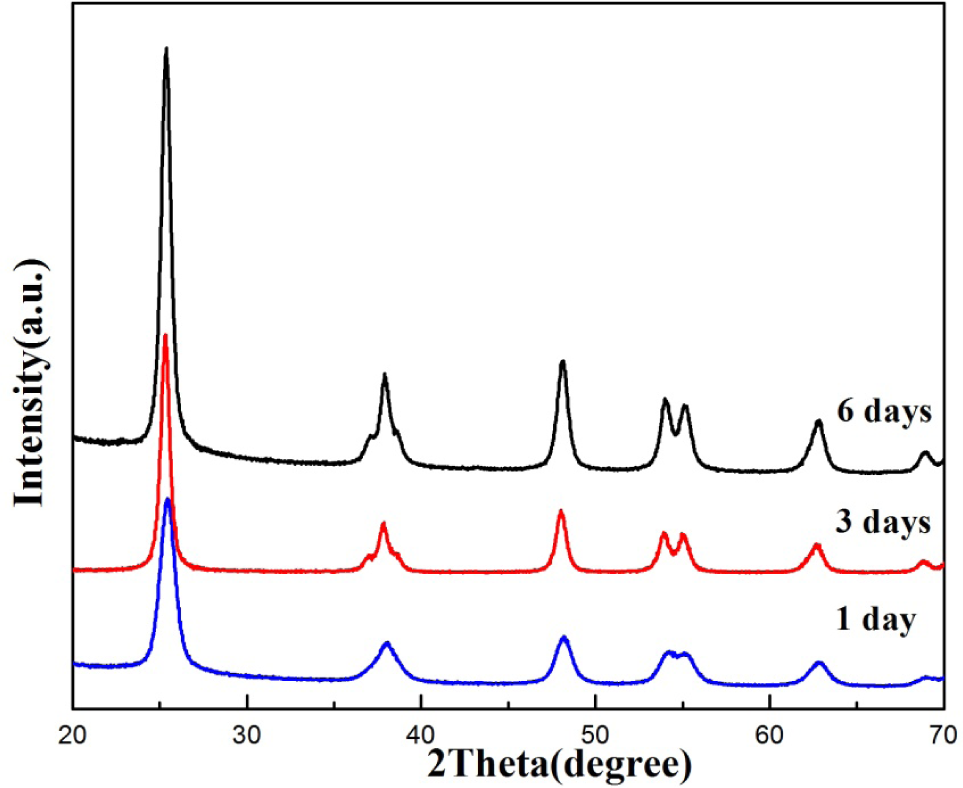
XRD patterns of nanostructured TiO2 were based on the combination of the hydrolysis for different times
As an anode material, samples with a high BET surface-area-to-density ratio was a perfect choice. The specific surface area and pore volume are summarized in Table 1. From the table, the TiO2 sample prepared by hydrolysis for six days had a higher BET surface area (119.2 m2/g) than the others. In sharp contrast, Hong-En Wang et al. [22] reported that TiO2 nanospheres for LIB applications received a BET surface area of 64 m2/g. Yassine Bessekhouad et al. [29] obtained a specific surface area of 44.5 m2/g for photocatalytic TiO2 nanoparticles. The nitrogen adsorption/desorption isotherms of the TiO2 nanoparticles for six days, as shown in Fig.2, can be identified as a type-IV isotherm with H2-type hysteresis loop, caused by filled pores between uniform nanoparticles. Pore-size distribution of the filled pores in the sample for six days, which was obtained from braches of the nitrogen adsorption/desorption isotherm according to the Barrett–Joyner–Halenda (BJH) model, can be seen in Fig.2 according to the inset image. We note that the BET surface area depends on materials and preparation conditions, such as preparation method and temperature. Among the samples with the same morphologies, TiO2, with a smaller particle size, has a higher BET surface area.
Physical properties from N2 sorption on the synthesized TiO2 for different times


Nitrogen adsorption–desorption isotherms and pore-size distribution (in inset) of nanostructured TiO2 synthesized by sol-solvothermal for six days
The microstructure and morphology of TiO2 were characterized using a TEM operated at 200 kV and equipped with a scanning unit. The results show that the structures of both the precursor and the product were nanosheets. Fig.3 shows the typical TEM images of the as-prepared TiO2 samples for different times. Fig.3(a,b) shows that the sizes of TiO2 nanoparticles for one day or three days of the solvothermal treatment were not uniform. Besides, TiO2 nanoparticles for three days at 150 °C had a smaller size to that of TiO2 for one day. Fig.3(c) indicated that nanostructured TiO2 samples with a diameter of 10±5 nm had been successfully synthesized during the six days of the solvothermal treatment. The HRTEM image (Fig.3d) displayed clear crystal lattices with spacings of 0.351 nm, 0.247 nm and 0.193 nm, corresponding to the (101), (004) and (200) planes of anatase TiO2. The selected area electron diffraction (SAED) (inset, Fig.3d) pattern also showed that the diffraction rings 1–3 (from the centre) had a lattice-spacing related to the (101), (004) and (200) planes. It also complemented the XRD results in the presence of the anatase phase. The chemistry mechanism of the sol–gel progress is under discussion with regard to providing directions in order to specifically prepare the nanostructured TiO2, which is equipped with a high degree of regularity and a large surface area [30]. TiO2 nanocrystals own the high rate capability and reversible electrochemical capacity due to their large specific surface area, which can offer more contact positions for electrochemical reactions. We propose that the SDBS as the dispersing agent is used to enhance the homogeneity of the metal cations [31,32]. Control experiments have shown that the nanostructured TiO2 can only be prepared with the right combination of reagents and reaction conditions. First, the presence of SDBS is necessary for the reaction. Only the nanostructured TiO2 with the homogeneous size is obtained through the existence of SDBS. Second, the time of solvothermal treatment significantly influences the morphologies of the final TiO2 products.

TEM of TiO2 for different reaction times: (a)(b) three days and HRTEM; and (c)(d) six days and HRTEM
Coin-type LIBs (2025-type) were fabricated to evaluate the electrochemical properties of the TiO2 samples as working electrodes. Fig.4 shows the cycle performances and charge/discharge abilities (insets) of TiO2 (one day), TiO2 (three days) and TiO2 (six days). Fig.4a of TiO2 (one day) displays an initial discharge capacity of 237.5 mAh g−1 at 0.2 C, with capacity decreasing to 147.7 mAhg−1 after 100 cycles. Fig. 4b shows that TiO2 (three days) exhibits good cyclic performance, while capacity decreases to 185.7 mAhg−1 after 100 cycles. Moreover, the coulombic efficiency of TiO2 (three days) during the first cycle was 62.5% (inset, Fig.4b). Increasing the time of solvothermal reaction improved the electrochemical performance of TiO2 remarkably. Fig.4c shows that TiO2 (six days) exhibited good cyclic performance. A reversible capacity could still be retained as high as 196.4 mAh g−1 after 100 cycles at a rate of 0.2 C. The coulombic efficiency of TiO2 (six days) during the first cycle was 74.2% (inset, Fig.4c). It is apparent that TiO2 hardly shows a significantly irreversible capacity loss during the first cycle and low capacity retention after 100 cycles. Such irreversible capacity loss is usually attributed to a reduction in the electrolyte on the electrode surface. In the process of battery charging and discharging for the first cycle, electrochemical reduction reaction will occur in electrolyte solvent, while gases are produced under a certain potential along with an increase in charging voltage and negative shift of negative potential.
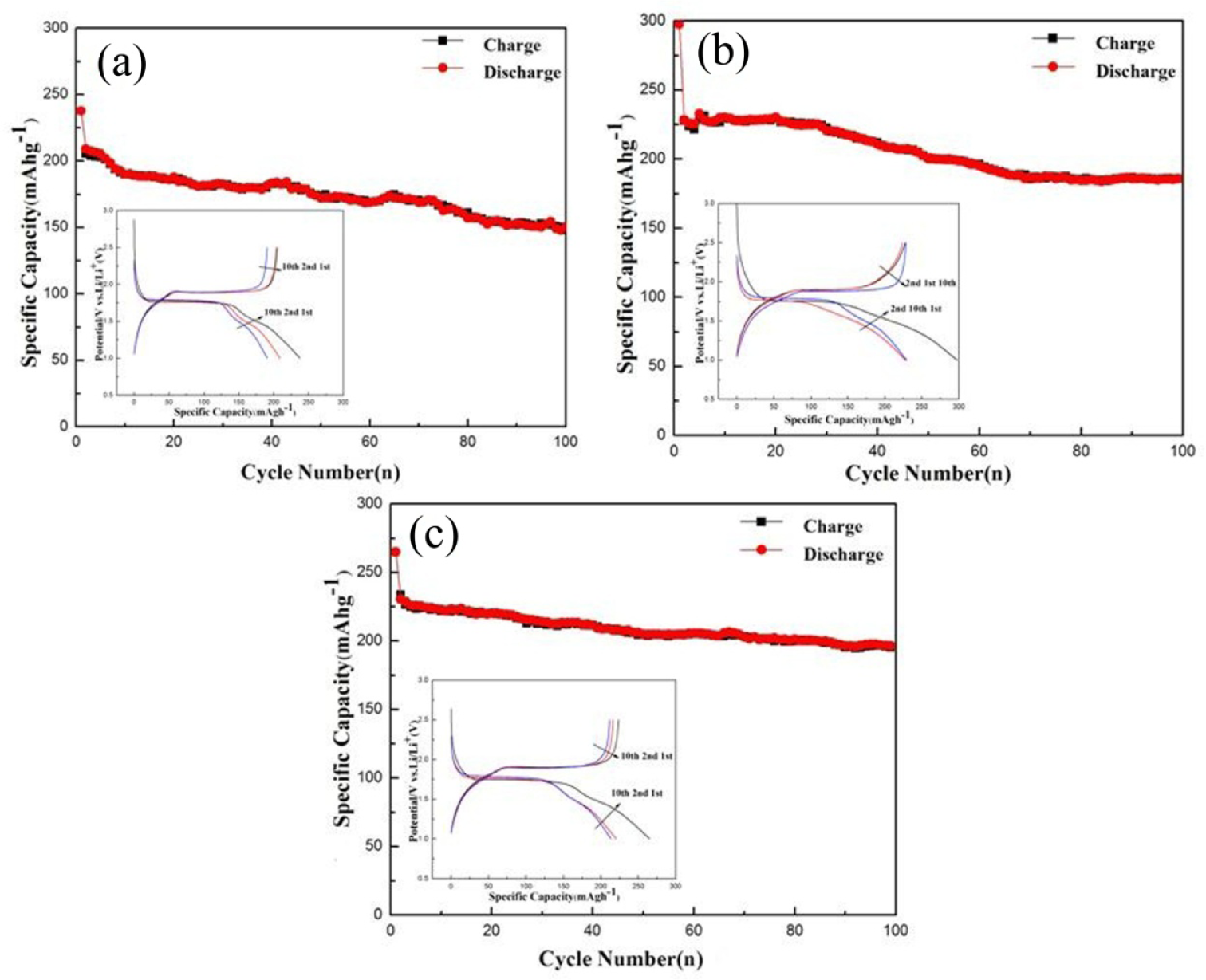
Cycling performance of TiO2 for different times: (a) sol-solvothermal for one day; (b) sol-solvothermal for three days; and (c) sol-solvothermal for six days
As shown in Fig.5, the electrochemical properties of the TiO2 samples were evaluated. The initial charge/discharge abilities of the TiO2 anode material between 1–2.5 V at an increasing rate from 0.2 to 10 C are shown in Fig.5. Fig. 5(a,b) are the cycle performances of TiO2 samples (one day and three days) from 0.2 C to 10 C in several steps after increasing every 10 cycles. Increasing the rate to 0.2, 0.5, 1, 2, 5, and 10 C, capacities of 176.1, 155.2, 125.5, 110.2, 87.4, 67.7, 206.5, 173.2, 152.9, 125.3, 101.4, and 82.5 mAh g−1 were obtained after 10 cycles, respectively. After the current density was increased gradually to 10 C, it was then decreased abruptly to 0.2 C of current. The reversible capacity of 180.5 mAh g−1 and 201.8 mAh g−1 could be delivered at the rate of 0.2 C finally. In comparison with the reported values of TiO2-based electrodes (one day and three days), the discharge capacities of the TiO2 samples (six days) were higher at a low rate (0.2 C) and comparable at higher rates. Fig.5c shows the influence of the discharge rate on capacity retention, while the inset shows the cycle performance of TiO2. The specific discharge capacity of about 221.7, 204.6, 190.4, 174.5, 149.3 and 124.7 mAh g−1 was obtained at a rate of 0.2, 0.5, 1, 2, 5 and 10 C. The final discharge capacity was about 210.3 mAh g−1 when the current density abruptly decreased to 0.2 C. These results respectively suggest that the TiO2 samples (six days) are promising anode materials for the application of LIBs. There are two reasons for this phenomenon. At first, the diffusion of Lithium-ions is facilitated because the nanocrystal subunits are small enough to shorten the Li+ diffusion path. Secondly, the unique structure, which has a relatively large surface area, endows them with a higher capacity and provides the sufficient contact interface between electrode and electrolyte [33].

Rate performance of TiO2 for different times: sol-solvothermal for one day; (b) sol-solvothermal for three days; and (c) sol-solvothermal for six days
To further gain the effect of nanostructured TiO2 (anatase) on the electrochemical property of LIBs, Fig.6 shows the cyclic voltammetry (CV) curves of the TiO2 sample within the scanning potential range of 1–2.5 V and the scanning rate of 0.1 mVs−1. Fig.6(a,b) shows a pair of oxidation/reduction peaks at 2.07 and 1.69 V on the CV curves of the TiO2 samples (one day and three days), which was characteristic of the Li+ intercalation/deintercalation reaction in anatase TiO2. As shown in Fig.6c, for TiO2 (six days), oxidation/reduction peaks existed at 2.06/1.68 V, which was corresponding to the extraction and insertion of lithiumions in anatase TiO2. For TiO2 (six days), the CV curves showed better reversibility than TiO2 (one day and three days).
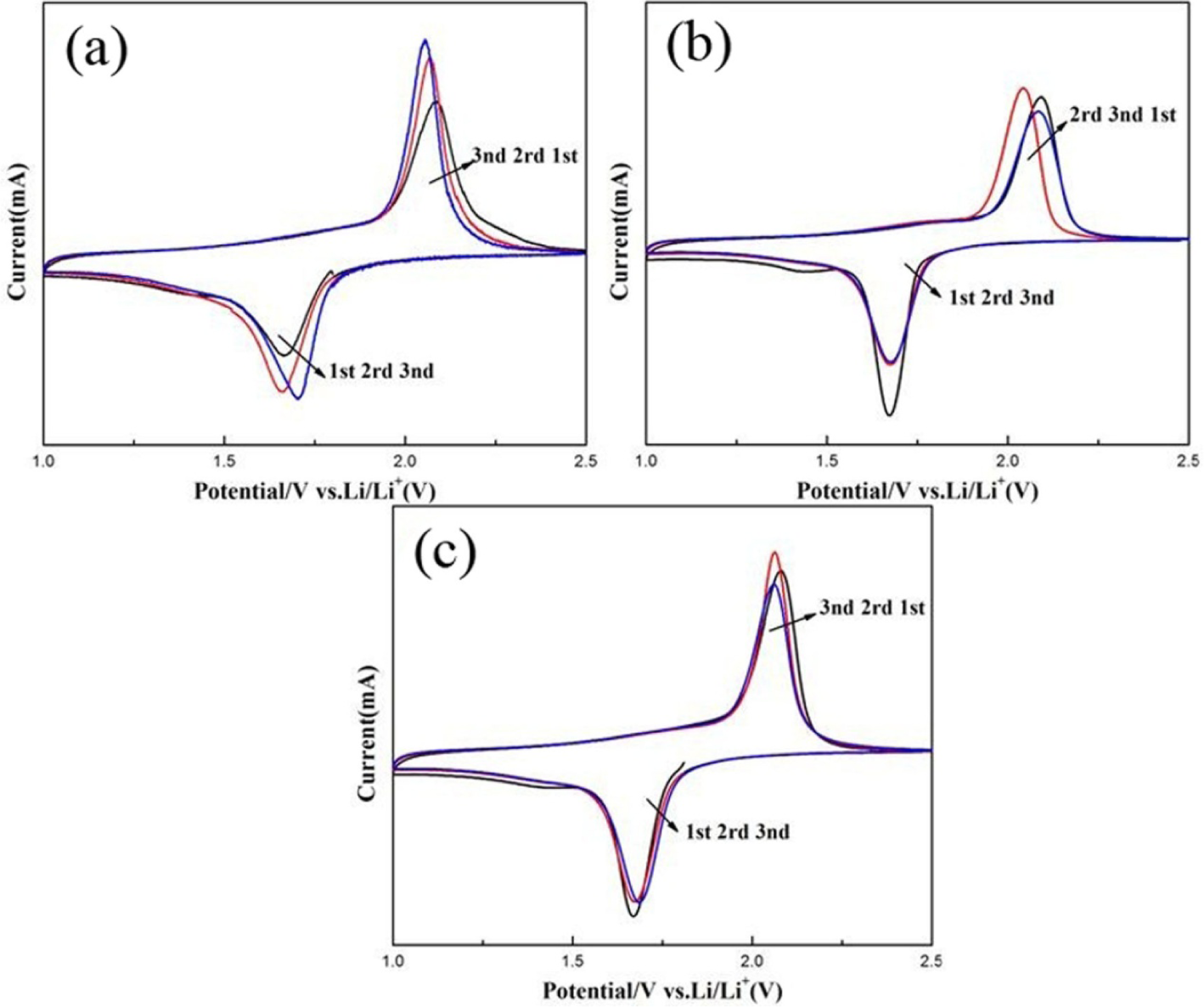
CV curves of TiO2 for different times: (a) sol-solvothermal for one day; (b) sol-solvothermal for three days; and (c) sol-solvothermal for six days
EIS measurements were also conducted to gain additional insights into the good rate performances of nanostructured TiO2 (anatase) electrodes. The Nyquist plots of the electrodes in Fig.7 show similar trends, composed of the semicircles in relation to higher frequency range and oblique lines in the low frequency end. The oblique lines can be attributed to the Warburg impedance, caused by Li+ diffusion onto the interface of electrodes in the actual electrode process [34]. The semicircles in relation to higher frequency range arose from the charge transfer reaction in the electrolyte/electrode interface. The radius of the semicircle usually represented the degree of electrode impedance, which was connected to the structure of electrode materials. The diameters of the semicircles in the following order: six days< three days< one day. It indicated that TiO2 nanoparticles for six days possess the lowest overall charge transfer resistance and interparticle contact resistance.

Nyquist plots of nanostructured TiO2 for different times
4. Conclusions
In summary, nanostructured TiO2 (anatase) samples were successfully synthesized via a one-pot solvothermal method with the assistance of SDBS as a surfactant. TEM images confirmed that the diameters of these nanostructured TiO2 samples ranged from 10±5 nm. The as-prepared TiO2 (six days) exhibited a capacity of 181.5 mAh g−1 at a rate of 0.2 C after 100 cycles and a capacity of 119.5 mAh g−1 at a very high rate of 10 C with excellent capacity retention performance. The remarkable electrochemical performance of TiO2 (six days) can be ascribed to the uniform size structure, which can facilitate an excellent electron transport path upon the cycling of anatase TiO2 particles.
Footnotes
5. Acknowledgements
The work is supported by the Key Scientific and Technological Project of Jilin Province, Project Grant No. 20140204052GX; the Open Project of the State Key Laboratory of Inorganic Synthesis and Preparative Chemistry, College of Chemistry, Jilin University (201505).
