Abstract
Boron nitride (BN) nanostructures (= nanoBN) are structural analogues of carbon nanostructures but display different materials chemistry and physics, leading to a wide variety of structural, thermal, electronic, and optical applications. Proper synthesis routes and advanced structural design are among the great challenges for preparing nanoBN with such properties. This review provides an insight into the preparation and characterization of zero dimensional (OD) nanoBN including nanoparticles and nanopolyhedrons from borazine, an economically competitive and attractive (from a technical point of view) molecule, beginning with a concise introduction to hexagonal BN, followed by an overview on the past and current state of research on nanoparticles. Thus, a review of the spray-pyrolysis of borazine to form BN nanoparticles is firstly presented. The use of BN nanoparticles as precursors of BN nanopolyhedrons is then detailed. Applications and research perspectives for these OD nanoBN are discussed in the conclusion.
1. Introduction
Technological progress continuously calls for materials with optimized and novel advanced properties in which the functionality is controlled by nanoscale structures leading to improved and/or new applications [1]. In this category of advanced materials, inorganic compounds stimulate current research activities in many emerging topics ranging from fundamental science to applications such as in sustainable technology, energy conversion, and environmental issues. This is particularly the case for oxide ceramics. However, these materials do not always cover the current technological needs in some applications for which a chemical and/or thermal resistance is required [2,3]. Accordingly, there are growing efforts to prepare nonoxide ceramics that could extend the application of inorganic compounds and one of the most promising categories for that is nitride.
Nitride materials are nitrogen-containing inorganic compounds that contain the nitrogen ion N3− (oxidation state of −3). Nitrides exist in two categories: the ionic-covalent family of nitrides and the transition metal series of the periodic table. In general, they are considered as refractory materials due to the fact that the nitrogen ion strongly attracts the relevant electrons in the chemical bond. Besides this, they display particular properties (electric insulation, thermal conductivity, hardness, etc.) according to their composition to provide an extremely large panel of functions. In the category of covalently bonded nitrides, hexagonal-boron nitride (h-BN but expressed here as BN) has recently attracted considerable attention in relation with its unique combination of key properties. It is considered in the present paper.
BN was discovered in the early 19th century and commercially developed in the latter half of the 20th century [4]. It consists of a layered structure with equal numbers of boron (B) and nitrogen (N) atoms linked by sp2 hybridized orbits and held together mainly by Van der Waals force [5]. Because BN is isostructural to carbon (C) forming a honeycomb-like network analogous to graphite, the researches focused on BN are in general linked to the works performed on carbon. In particular, interest at the academic level has arisen in the synthesis of nanostructured BN (= nanoBN). As an illustration, the discovery of carbon nanotubes (CNTs) by Iijima in 1991 [6] encouraged intense experimental and theoretical research investigations on their BN analogues (= BN nanotubes, BNNTs) [7]. Similarly, the formation of BN fullerenes [8] was inspired by the first fullerene molecule, the buckminsterfullerene (C60), reported in 1985 [9]. Afterward, the discovery of graphene in 2004 [10] inspired researches on their 2D BN counterparts [11]. Finally, like carbon, the layered structure of BN provides useful properties and its modification by various post-synthesis treatments has attracted considerable attention recently [12-19].
BN displays good lubricating properties according to its layered structure (weakly held layers can slide over each other). It proposes a very high thermal conductivity (in the direction of hexagons), a high thermal stability, a high resistance to corrosion and oxidation, as well as a strong UV emission [20-23]. It offers high dielectric breakdown strength and high volume resistivity. BN displays a large band gap (∼5.5 eV) and offers the lowest density (d = 2.26 g cm−3) among nonoxide ceramics. In addition, it can provide superhydrophobicity depending on its shape and synthesis procedure [24–25]. Furthermore, the local polar character of the B-N bonds is present in the BN structure. As an illustration, BN has been demonstrated to exhibit enhanced sorption properties of various substances such as organic pollutants [25-31] and hydrogen [31-34]. However, these properties vary drastically with the synthesis route employed to prepare BN.
BN was synthesized by Balmain [35] in 1842, applying the reaction between molten boric acid (H3BO3) and potassium cyanide (KCN). It is nowadays produced by conventional powder technology, requiring nitridation or carbothermal reaction of boric acid (H3BO3)/boric oxide (B2O3) with melamine (C3H6N6) or urea (CO(NH2)2) and the use of additives during the further sintering process [36]. BN is used in various fields of chemistry, metallurgy, high-temperature technology, electronic and in thermal management applications. However, the design of BN in which the structure is designed at nanoscale to generate nanoBN such as nanoparticles (NPs) is complex to investigate for future industrial challenges. Proper synthesis routes and advanced structural design are among the great challenges for preparing nanoBN.
Among the category of nanoBN, NPs have received less attention than BNNTs despite their wide-spectrum uses (as functional materials for high thermal conductivity and electrically insulating composites or as precursors for others materials). In the present paper, we have restricted ourselves to the synthesis of BN NPs starting with a state-of-the-art focused on BN particles and NPs. It is followed by the preparation and characterization of borazine-derived BN NPs in particular using spray-pyrolysis, and the thermal behavior of the BN NPs at high temperature leading to generate BN nanopolyhedrons (NPHs).
2. General Routes for the Preparation of BN Particles and NPs
The first intensive efforts on the preparation of BN particles and submicronic particles were made by Paine et al. [37-41]. Firstly, they used an aerosol-assisted vapor synthesis (Figure 1) from liquid ammonia solutions of poly(borazinylamine) to provide BN particles. However, this route was considered to be not convenient and relatively expensive [37].

Schematic diagram of the aerosol process [39]
Later, they investigated the same process using aerosol droplets containing water-soluble H3BO3 at 1200°C in an ammonia/nitrogen atmosphere [38]. The particles were spherical and displayed a diameter of 1–2 μm. In 2005, the same authors were able to reduce the size of BN particles through the pyrolysis of aqueous aerosols containing a guanidinium borate ([C(NH2)3]2[B4O5(OH)4]•2H2O) under ammonia/nitrogen flow up to 1600°C [39]. One year later, Paine et al. used trimethylborate ((MeO)3B) in neat form in methanol as an aerosol precursor to produce, by reaction with ammonia, spherical BN particles with a high production rate, low oxygen and carbon contents (as a consequence of a post-pyrolysis annealing at 1600°C), and with a diameter 0.2–2 μm (Figure 2) [40]. These BN powders could be prepared as mesoporous materials [41].

SEM image of BN particles produced from (MeO)3B aerosol and post-pyrolyzed at 1600°C [40]
Bando et al. prepared submicronic spherical BN particles with diameters ranging from 50 to 400 nm (Figure 3) by using a two-step synthetic process [42–43].

SEM image of BN particles synthesized by CVD of (MeO)3B (a) then annealed at 1100°C (b) [42]
They combined chemical vapor deposition of solutions of (MeO)3B under ammonia using two types of systems (Figure 4) and annealing treatment. Authors showed that they could generate two types of BN particles according to the used CVD systems: using ammonia at 1400°C during annealing, oxygen-containing BN with a diameter around 90 nm and BET Specific Surface Area (SSA) of 26.8 m2/g could be obtained. Oxygen-free BN particles with a diameter of 30 nm and at BET SSA of 52.7 m2/g could be generated using argon instead of ammonia during the annealing process [43]. Following a two-step process, Shi et al. synthesized BN particles in the diameter range 0.5–1.5 μm by spray-drying a mixture of hydrogen borate (HBO3), Sodium tetraborate decahydrate (Na2B4O7) at 250°C which was subsequently mixed with (NH2)2CO to be heated in air at 900°C. As-made particles were nitrided at different temperatures from 1200 to 1550 °C [44]. In 2010, monodisperse BN NPs were prepared by a modified solid-state reaction route, using barberiite (NH4BF4) and sodium azide (NaN3) as the reactants. As-prepared BN NPs displayed good thermal stability and high specific surface area [45]. More recently, BN NPs with diameters from 20 to 100 nm have been prepared from B2O3 by high-energy ball-milling followed by annealing at 1200°C [46]. These NPs were mainly elliptical plate-like with diameters ranging from 20 to 100 nm. Impurities were detected. In a last report, Xiong et al. produced water-dispersible BN NPs with diameter of around 30 nm by direct reaction of H3BO3 and ammonium chloride (NH4Cl) [47]. Copper oxide (CuO) has been identified in the final product.

Schematic diagram of the synthetic systems used in the traditional (leading to samples labeled NT204) (a) and modified CVD processes (leading to samples labeled NT202) (b), and the appearance of the samples NT202 and NT204 [43] (c). Arrow points to the area of the sample
Based on the previously described studies, it appears that the selection of the BN precursor is important to avoid impurities in the final materials. It is clear that precursors with the stoichiometric B:N ratio and the preformed B3N3 ring, while hydrogen (H) is the only element added to B and N, are required to prepare BN. Borazine (BZ, H3B3N3H3) appears to be an appropriate precursor of BN NPs. Borazine-derived BN NPs have been reported by Hidalgo et al. [48]. In particular, they described the formation of BN NPs (mixed with cubic BN NPs) using laser CVD. However, few data have been provided concerning the nanostructure of these materials. Besides this report, our group demonstrated in 2009 the possibility to produce pure BN NPs from the spray-pyrolysis of BZ [49], which is the subject of the next section.
3. Spray-pyrolysis Process of Borazine – Preparation of BN NPs
3.1 Synthesis and Characterization of Borazine
BZ represents a highly pure synthetic low-molecular weight precursor with the planar six-numbered hexagonal ring in which uniform chemical composition is established at the molecular scale [50]. BZ is isolectronic and isostructural of benzene and has the chemical formula H3B3N3H3. BZ was first isolated by Alfred Stock in 1926 [51]. It displays a melting point of −58 °C and a boiling temperature of +55 °C.
BZ can be used as a BN precursor following two strategies. In a first strategy, it can be polymerized into a macromolecule (= inorganic or preceramic polymer called polyborazylene) by self-condensation; then this polymer offers many opportunities to produce BN (with a control of the composition at the molecular scale) in various morphologies and porosities following the Polymer-Derived Ceramics (PDCs) route [52-58]. BZ offers the advantage of being a colorless liquid with an adequate vapor pressure. As a consequence, it can be used in a second strategy in various vapor-phase processes [48,59-65]. In the present section, the preparation of BZ-derived BN NPs by spray-pyrolysis is reported from the synthesis and characterization of BZ to the characterization and high-temperature thermal behavior of BN NPs. In the following section, their use as precursors for preparing nanopolyhedrons (NPHs) [66–67] has been selected for discussion.
First of all, BZ can be prepared through different pathways with probably the most economical one being from sodium borohydride (NaBH4) and ammonium sulfate ((NH4)2SO4) [68]. The 1H NMR of BZ consists of a BH quadruplet between 3.40 and 5.20 ppm and an NH triplet between 5.20 and 5.95 ppm (Fig. 5a).

(a) 1H NMR spectrum recorded in CDCl3 and (b) FTIR spectrum of BZ
The IR spectrum in Fig. 5b is consistent with the formation of BZ: it is dominated by the vB-H (2515 cm−1), the characteristically sharp vN-H (3452 cm−1), the B-N stretch near 1445 cm−1, and the B-N-B bending mode at ∼900 cm−1; the two last bands are characteristic of the borazine ring [69–70]. The deformation of N-H bending is characterized by the band at ∼715 cm−1, whereas the smallband emerging around 1170 cm−1 could be attributed to B-H bending [71]. As previously mentioned, BZ is volatile and can be used in a spraypyrolysis (SP) apparatus to produce NPs.
3.2 Spray-pyrolysis of Borazine and Preparation of BN NPs
SP is a method in which a precursor is delivered as an aerosol, i.e., tiny droplets carried by nitrogen. Then, the precursor droplets are carried by a gas and the aerosol is carried out in a pyrolysis reactor to be converted into solid nanoparticles. Here, BZ droplets are carried out by nitrogen into the pyrolysis reactor fixed at 1400 °C to be fully evaporated (according to their relatively high vapor tension) and decomposed into BN powders that are collected in two traps. Nucleation from the vapor phase and growth of BZ are involved in the process of nanoparticle formation. Figure 6 presents the SP process using BZ to produce BN NPs.

Schematic diagram of the spray-pyrolysis of BZ leading to BN NPs
3.3 (Nano)structural Characterization of BN NPs
The XRD pattern and the solid-state 11B NMR spectrum of as-obtained powders are presented in Fig. 7. The XRD pattern of commercially available BN is in general composed of peaks at 26.76° (002), 41.60° (100), 43.87° (101), 50.15° (102), 55.16° (004), 75.93° (110), 82.18° (112), and 85.52° (105) [72]. However, it is extremely difficult to form the hexagonal phase of BN and to identify all the peaks characteristic of this phase in the XRD pattern of precursor-derived BN. Two disordered BN phases, turbostratic-BN (t-BN), and amorphous-BN (a-BN), are in general identified [73-75]. t-BN shows a random stacking sequence of the (002) layers and a disorientation of these layers around the c-axis. a-BN represents a structure disordered at atomic level. XRD patterns of such phases are significantly distinct from that of BN. This is the case of BZ-derived powders. As an illustration, the XRD pattern of powders showed broadened (002) peaks and diffuse (100 and 110) peaks which are significantly shifted to the Bragg angles of BN [49]. The pattern of BZ-derived powders is representative of a t-BN structure. The formation of t-BN most probably results from the SP process. The extremely fast heating rate involved by this SP process (∼102 °C/s) associated with the short residence time of BZ at 1400°C (3 s) inherently leads to the low crystallization of BN powders. We investigated solid-state 11B NMR of powders. 11B solid-state NMR spectrum of the sample (Fig. 7 inset) shows a large signal in the region of tricoordinated boron atoms that could be assigned to B–N bonds in planar BN3 groups [54,56] within BN graphitic layers. The second small signal at δ = 0.6 ppm (no quadrupolar shape) is indicative of the presence of tetragonal BO4 groups [76–77] in a low portion.

XRD pattern and 11B NMR spectrum of BN NPs prepared by spray-pyrolysis of borazine at 1400°C
Transmission Electron Microscopy (TEM) was investigated to estimate locally the size of powders as well as their agglomeration level and to confirm their low degree of crystallization. The low-magnification TEM bright field image of BN powders (Fig. 8) showed that samples are composed of agglomerated NPs, but their easy dispersion in solvent proved that agglomeration level is low. The particle population was relatively homogeneous and the NP size ranged from 55 to 120 nm [49]. Data provided by the corresponding selected area electron diffraction (SAED) pattern, inset in Figure 8 highly reflected XRD results. The amorphous halo imposed on the diffuse and continuous (002) arcs and the poorly resolved (004) ring confirm the low level of crystallinity of BN NPs [79].

TEM micrographs and SAED pattern of BN NPs prepared by spray-pyrolysis of BZ at 1400°C
The high-resolution TEM (HRTEM) image (Fig. 9) showed that the long-range ordering is minimal and the crystallinity is inhomogeneous. The lattice image in Fig. 9 highlighted the presence of grains with a low thickness and layers with high tortuosity in their stacking sequence as commonly observed in precursor-derived BN [80–81]. It was interesting to observe that a certain portion of the smallest particles (≤10 nm) seems to be hollow.

HRTEM micrographs of BN NPs prepared by spray-pyrolysis of BZ at 1400°C
3.4 Characterization at the Mesoscopic Scale of BN NPs
The NPs have been characterized at the mesoscopic length scale by nitrogen gas adsorption–desorption measurement at 77 K [49]. Based on IUPAC classification [82–83], the adsorption–desorption isotherms of the BN NPs (Fig. 10) depict a type-I/II shape, which strongly suggests the formation of nonporous NPs, which was confirmed by the low BET SSA of samples (<35 m2/g). However, the adsorption hysteresis of type H4 could indicate the presence of micropores in the NPs.
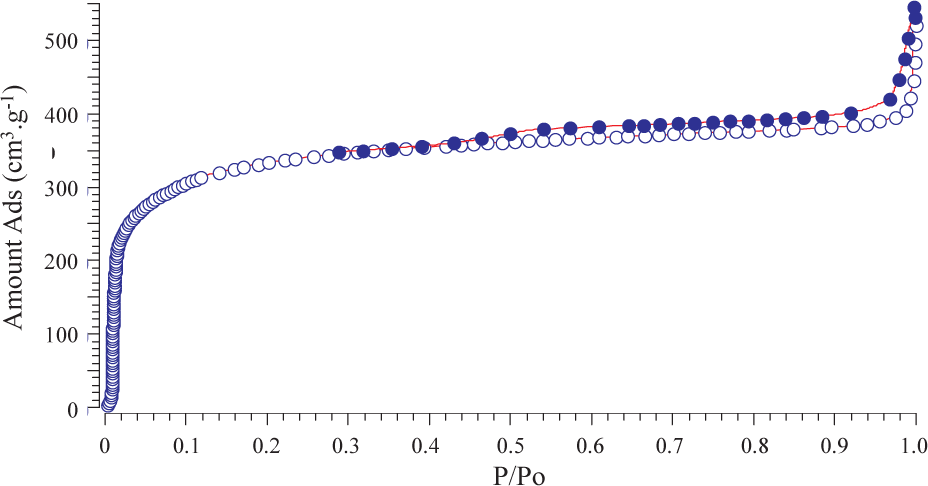
N2 adsorption–desorption isotherms recorded at 77 K of BN NPs prepared by spray-pyrolysis of BZ at 1400°C
3.5 Characterization of BN NPs by XPS
In the present paper, the chemical environment around the B, N, C, and O atoms in powders has been investigated by X-ray Photoelectron Spectroscopy (XPS). Figure 11 displays the general XPS graphs recorded on the surface and after argon sputtering and Fig. 12 reports the characteristic individual peaks attributed to B 1s and N 1s environments. Figure 11 shows that XPS spectroscopy is oversensitive to surface groups: BN powders are very prone to surface passivation with atmospheric oxygen. The detection of carbon and oxygen, which are considerably reduced after argon sputtering, clearly results from contamination. XPS results (B, 41.6 at%; N, 41.2 at%; C, 9.0 at%; O, 8.2 at%) for the surface of the sample give evidence that the bulk composition of the material is close to stoichiometric BN. The B 1s peak at 190.8 eV corresponds to the B-N bonding, whereas the one at 191.9 eV (Fig. 12a, only at the surface of the bulk sample) corresponds to B2O3 [78]. The N 1s peak (Fig. 12b) at 398 eV is typical for nitrogen shifts in BN [55]. However, we can suggest that contamination is extremely low and the SP process of BZ leads to pure BN NPs.
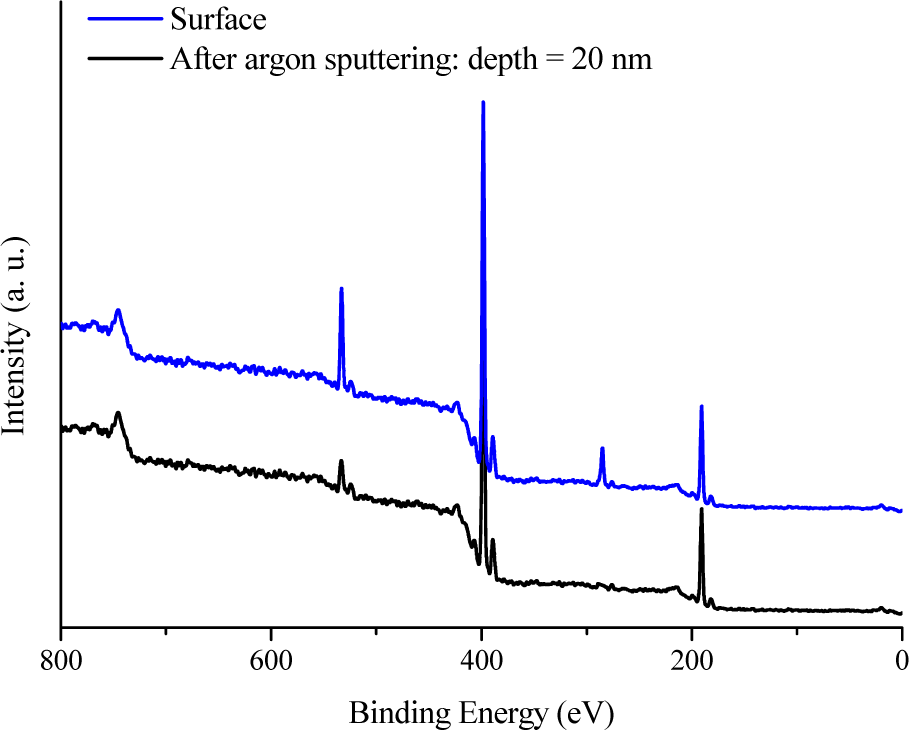
XPS spectra from BN NPs prepared by spray-pyrolysis of BZ at 1400°C after argon sputtering corresponding to a depth profile of 20 nm

High-resolution B 1s (a), (b) N 1s XPS spectra of BN NPs prepared by spray-pyrolysis of BZ at 1400°C
3.6 High-temperature Behavior of BN NPs by XPS
Because the BN NPs displayed a low degree of crystallinity, we investigated their behavior at high temperature in the temperature range 1000–1800°C under nitrogen atmosphere (Figure 13) [66–67], especially their weight change by High-Temperature ThermoGravemetric Analysis (HT-TGA). In addition, we investigated the same high-temperature range under argon. The profile of both curves is similar: BN NPs underwent a first weight loss from RT to 800°C, followed by a plateau from 800 to 1100°C, and then a second weight loss was identified.

HT-TGA of of BN NPs prepared by spray-pyrolysis of BZ at 1400°C (RT-1800°C, argon and nitrogen atmospheres)
As previously suggested, the conversion of BZ into BN is not fully achieved by SP and some hydrogen atoms remain at the periphery of the BN network. Therefore, we suggested that the first weight loss identified in Fig. 13 results from evolution of residual hydrogen. It represents 1.2% of weight loss. The second weight loss is more complex to interpret, whereas BN is known to be stable up to 2000°C as shown in our previous paper [67]. We concluded that it probably results from the poor crystallinity of BN NPs. Within this context, we have investigated the structural evolution on BN NPs by heat-treatment under nitrogen in the temperature range 1400–1800°C, leading to the formation of the materials we labeled
4. Annealing of BZ-derived BN NPs – Preparation of BN NPHs
Interestingly, the heat-treatment of BN NPs in the temperature range 1400–2000°C modified their nano-/microstructure as shown on the TEM images of samples
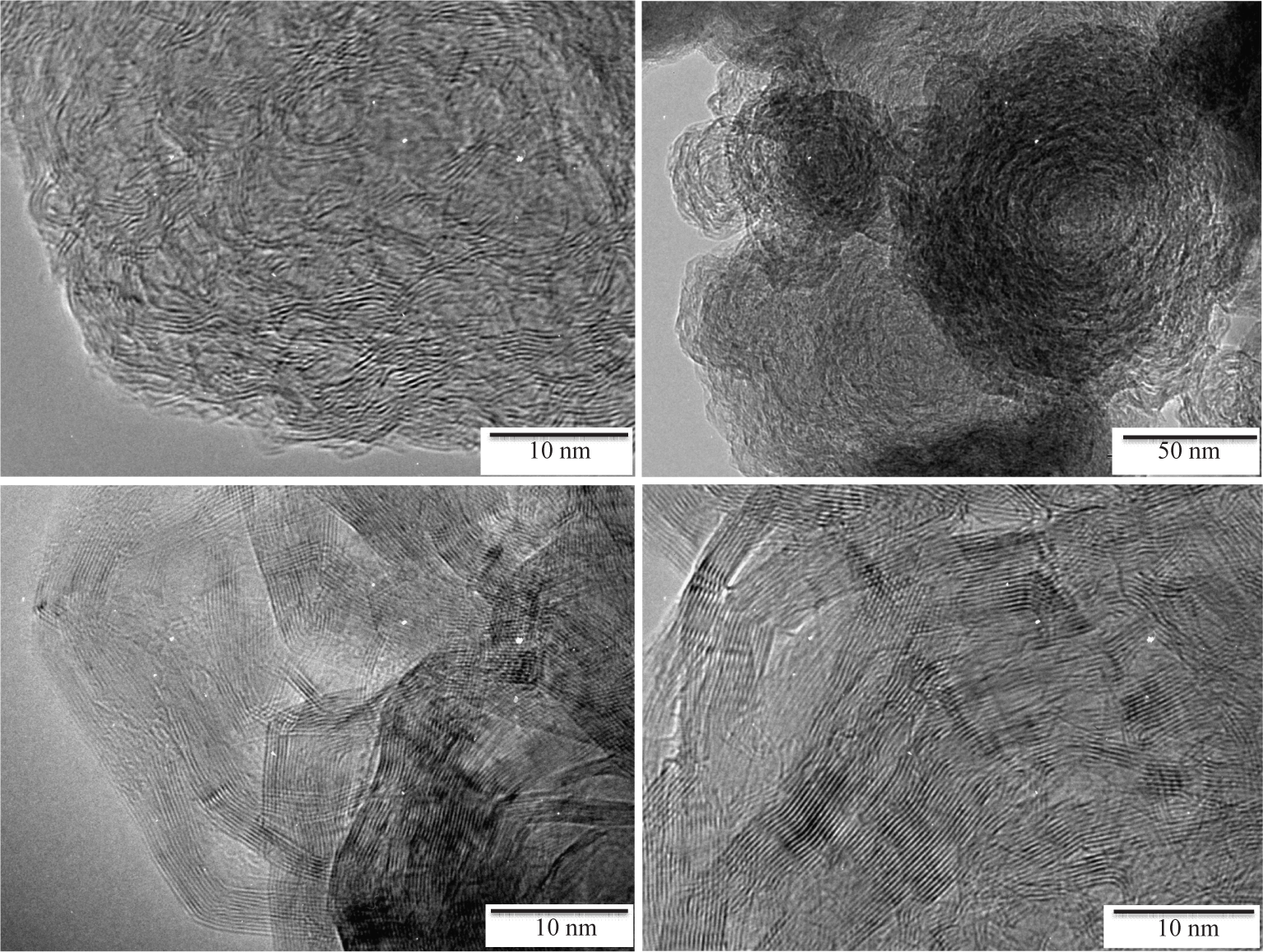
High-magnification TEM bright field images of the samples
It should be mentioned that the NPs annealed at 1450°C did not show strong differences in comparison to as-obtained BN NPs. In contrast, in the sample
A similar arrangement of the BN layers is identified, i.e., formation of hollow core-shell structures, in the sample
The flower-like architecture was kept after annealing the BN NPs at 1800°C (
There are some reports focused on the polyhedral morphology. Polyhedral BN-nanocapsules (BN-NCs) less than 80 nm in size have been synthesized by nitridation of Fe–B NPs obtained by reaction between NaBH4 and Iron trichloride (FeCl3) and treatment at 900°C under ammonia/nitrogen [84]. Treatment of the as-prepared product with dilute nitric acid (HN03) was required to dissolve the Fe–B NPs and generate the polyhedral BN-NCs. Finally, most of the BN-NCs encapsulate nonmetallic, oxide or nitride NPs to form hybrid materials [85-89]. Therefore, the encapsulated particles need to be removed to obtain NPHs, which usually requires a complex purification process. Facile formation of hollow BN powders with spherical primary particle morphology has been achieved through an aerosol process [41,90]. However, such powders were produced at the microscale and the heat-treatment did not allow the formation of NPH structures. It is clear that the above conventional synthetic methods are not fitted to the synthesis of NPHs, especially with hollow cores. Here, the structural organization of the BN NPs produced by SP of BZ allowed the developing of a convenient route for the synthesis of gramscale BN NPHs at high temperature.
5. Experimental Part
5.1 Materials
The synthesis of BZ was carried out in an argon atmosphere, using argon/vacuum lines, and Schlenk-type flasks. Argon (>99.995%) was purified by passing through successive columns of phosphorus pentoxide (Sigma-Aldrich, Saint Quentin, France), sicapent (Millipore S.A.S, Molsheim, France), and copper-oxide-based catalysts (Sigma-Aldrich, Saint Quentin, France). Schlenks were dried at 120°C overnight before pumping under vacuum and filling them with argon for the synthesis. Sodium borohydride (NaBH4, ≥ 98.5%, powder from Sigma-Aldrich, Saint Quentin, France), ammonium sulfate ((NH4)2SO4, ≥ 99.0% from Sigma-Aldrich, Saint Quentin, France), and tetraethylene glycol dimethyl ether (CH3O(CH2CH2O)4CH3, 99.0 % from Sigma-Aldrich, Saint Quentin, France) were used as-received. It should be mentioned that ammonium sulfate was dried at 120°C inside an oven for three days, then put under vacuum during cooling for 1 h. Manipulation of the chemical products was made inside an argon-filled glove box (Jacomex BS521; Dagneux, France) dried with phosphorus pentoxide.
5.2 Synthesis of Borazine
The operating procedure, adapted from the literature [68], is reported in our previous paper [52].
5.3 Synthesis of BN NPs
The experimental setup (Fig. 6) is composed of a nebulized spray generator (RBI, Meylan, France), in which the spray is generated by a piezoelectric device (barium titanate). Frequency (800 kHz) and power (100 W) alimentations are adjusted to obtain the aerosol. The aerosol temperature is first held at 15°C by a regulated water circulation to avoid borazine evaporation and/or condensation. The piezoelectric device generates an ultrasound beam, which is directed to the liquid–gas interface; a fountain formed at the surface followed by the generation of the spray resulting from vibrations at the liquid surface and cavitations at the gas–liquid interface. BZ was directly introduced in the aerosol-generating chamber under nitrogen, then aerosolized and carried to the pyrolysis furnace with a 0.5 mL min−1 nitrogen flow rate. The thermal decomposition of borazine was performed in a hot alumina tube containing an isothermal zone of 0.1 m in length. The fast heating rate implies gaseous species generation leading to powder formation by a chemical vapor condensation route. The particles were finally trapped into two collectors placed before the vacuum pump and containing filter-barriers made of microporous alumina (pore size of 1 μm). Yield was estimated to be 0.22 g min−1. After their synthesis, powders were stored inside an argon-filled glove-box. In a typical experiment, 27 mL (21.9 g) of BZ are used to produce 6.5 g of white-color powders. However, the exact yield is difficult to estimate because of the design of the SP system. A nonnegligible quantity of powders, deposited in the furnace tube, could not be recovered.
5.4 Characterization
FTIR spectrum of BZ was recorded from a Nicolet Magna 550 Fourier transform-infrared spectrometer by introducing the liquid molecule between two KBr windows. 1H NMR spectrum were obtained using a Bruker AM 300 spectrometer in CDCl3 operating at 300 MHz. Tetramethylsilane (TMS) was used as a reference for the NMR data. The XPS measurement of the final product was performed on a Perkin-Elmer PHI Quantera SXM multifunctional X-ray photoelectron spectroscope (Physical Electronics, USA), using Al-Kα radiation (photon energy 1476.6 eV) as the excitation source and the binding energy of Au (Au 4f7/2: 84.00 eV) as the reference. BN NPs and NPHs were characterized and analyzed by a transmission electron microscopy (TEM) with a TOPCON 002B working at 200 kV. Samples were characterized using a Philips PW 3040/60 X'Pert PRO X-ray diffraction system. Powder samples were prepared by placing ∼100 mg on the XRD sample holder (PVC). Cu Kα (λ = 1.54 Å) radiation with an Ni filter was used with a working voltage and a current of 40 kV and 30 mA, respectively. Scans were continuous from 10–85° 2θ for powders with a time per step of 0.85 s in increments of 0.017° 2θ. Peak positions and relative intensities were characterized by comparison with JCPDS files of the standard material (JCPDS card No 34–0421). The Specific Surface Area (SSA) of BN NPs was measured on a Micromeritics-ASAP 2010, BET 8 pts. Samples (450 mg) were degassed at 250°C for 12 h. Analysis was run at 77 K with N2. The BET specific surface area was calculated from the nitrogen adsorption data in the relative pressure range from 0.05 to 0.3. BN NPs were studied by high-temperature thermogravimetric analysis (HT-TGA, Setaram Setsys evolution TGA equipment) in argon and nitrogen atmospheres (50–1800°C; heating rates: 5°C/min) using tungsten crucibles.
6. Conclusions
This article reviews our recent advancements in the synthesis of 0D nanoBN, including nanoparticles (NPs) and nanopolyhedrons (NPHs) by spray-pyrolysis of borazine leading to BN NPs followed by the annealing of the latter to form NPHs at high temperature. In this review, we discussed the synthesis and characterization of borazine. Borazine offers the advantage of being a colorless liquid with an adequate vapor pressure, which is used as a spray-pyrolysis precursor for the synthesis of BN NPs. The extremely fast heating rate involved by this SP process (∼102 °C/s), and the short residence time of borazine at 1400°C (3 s) led to BN NPs with a low crystallization degree, a size ranging from 55 to 120 nm and a low specific surface area (SSA). Through annealing at high temperature under nitrogen, BN NPs underwent two weight losses. The first weight loss up to 1100°C was associated with evolution of hydrogen, whereas the second weight loss above 1100°C was associated with a gradual change in the morphology of NPs from BN NPs to BN NPHs. The production of BN NPs by spray-pyrolysis of borazine is a necessary step in forming at 1400°C under nitrogen disordered BN NPs from which the BN NPHs may grow via a solid-state process in the temperature range 1400–2000°C. It is envisaged that these structures might open new opportunities in exploring chemical and physical properties. We consider that these BN architectures could find applications in many areas of modern science and technology including adsorbents, optoelectronic devices, energy storage media as we recently demonstrated [34, 91], and catalyst support.
Footnotes
7. Acknowledgements
The authors acknowledge Prof. Sylvie Foucaud for the spray-pyrolysis investigation and Prof. Arnaud Brioude for the TEM observation of nanoparticles and samples annealed in the temperature range of 1450–2000°C. Dr. Samuel Bernard thanks University of Montpellier for providing financial support through the project entitled “Lenitrure de bore pour des applications « energie »” (2011, PP Energie type, Pole Chimie).
