Abstract
The support used in a composite catalyst has an important influence on the catalytic performance of the catalyst. Amorphous metals have good electron-transfer properties and the presence of defect structures on the surface will introduce additional active sites and should be excellent catalyst supports. In this study, an Ag@AgCl composite catalyst with amorphous Cu (a-Cu) as the support is prepared by a two-step precipitation method at room temperature and a light irradiation reduction method. Compared to the Ag@AgCl and a-Cu, the catalytic rate of the Ag@AgCl/a-Cu composite catalytic rate was 2.04 times and 6.69 times faster during the reduction of 4-NP in NaBH4 aqueous solution. The high-performance catalytic efficiency and reusability of Ag@AgCl/a-Cu may be attributed to the synergistic effect between Ag@AgC and amorphous metal elements. This work may provide an effective reference for the synthesis of high activity catalysts using amorphous metals as supports.
Introduction
4-Aminophenol (4-AP) is a valuable intermediate used to synthesize various drugs and plastic products, and can be obtained by reducing 4-nitrophenol (4-NP).1–7 Furthermore, the conversion of 4-NP to 4-AP is a common probe reaction to test the catalytic activity of some supported metal catalysts. Based on a previous report, 8 Ag@AgCl has bifunctional catalytic properties, which not only promotes the photo degradation of 4-NP under visible light but also reduced 4-NP to 4-AP in the presence of NaBH4 in aqueous solution. It was found that the catalytic performance of Ag@AgCl in the reduction of 4-NP by NaBH4 was assisted by the catalyst through the transfer of electrons and hydrogen atoms on BH4− to organic compounds, as reported previously.9,10 In order to further improve the Ag@AgCl catalytic performance, a standard method is to combine Ag@ AgCl with a support material to form a composite catalyst.11–15
As an amorphous material is in a metastable state, there are numerous active sites formed by defects on its surface, and many “dangling bonds” are present to increase the adsorption with the target, hence such materials are more efficient than the crystalline catalysts. Amorphous metal nanoparticles such as Au and Cu have been used in the reduction of N2 16 and CO2 17 and show excellent catalytic ability. Due to its important physical and chemical properties, non-noble amorphous Cu was the selected support for the composite catalysts, which should improve the catalytic performance of the composite material. However, as far as we know, there are very few reports in this research field.
In this study, an Ag@AgCl composite catalyst with amorphous Cu as the support is prepared by a two-step precipitation method at room temperature and a light irradiation reduction method. The structure, morphology, and chemical element states of the composite catalyst were characterized by X-ray diffraction, transmission electron microscopy, and X-ray photoelectron spectroscopy, which confirmed the successful synthesis of the composite catalyst. The catalytic reduction performance of the catalyst was studied with 4-NP as the reduction target, and the results showed that Ag@AgCl/a-Cu demonstrated high-performance catalytic efficiency and reusability.
Results and discussion
Characterization of the composite catalyst
The phase structure and diffraction pattern of a-Cu and Ag@AgCl/a-Cu were studied by powder X-ray diffraction (XRD). Figure 1 shows the typical powder XRD pattern of a-Cu and Ag@AgCl/a-Cu. CuCl2 can be reduced to a-Cu with the assistance of tannic acid (TA), a mild reducing agent, and a-Cu displayed the typical amorphous state with no diffraction peaks. The diffraction peaks at 27.8°, 32.3°, 46.2°, 54.8°, 57.5°, 67.5°, 74.5°, and 76.7° corresponded to the (1 1 1), (2 0 0), (2 2 0), (3 1 1), (2 2 2), and (4 0 0) planes of crystalline AgCl (JCPDS:31-1238), and the weak diffraction peak at 38.1° corresponded to the (1 1 1) plane of Ag0 crystal (JCPDS: 04-0783) in the Ag@AgCl/a-Cu composite.18,19 It indicated that a small amount of AgCl was reduced to Ag0, and the addition of a-Cu during the preparation process had no significant effect on the formation of AgCl crystals and Ag0. Except for the crystalline peaks of AgCl and Ag, no other diffraction peak existed, which also revealed that the elemental copper still was present in the amorphous state in the Ag@AgCl/a-Cu composite.
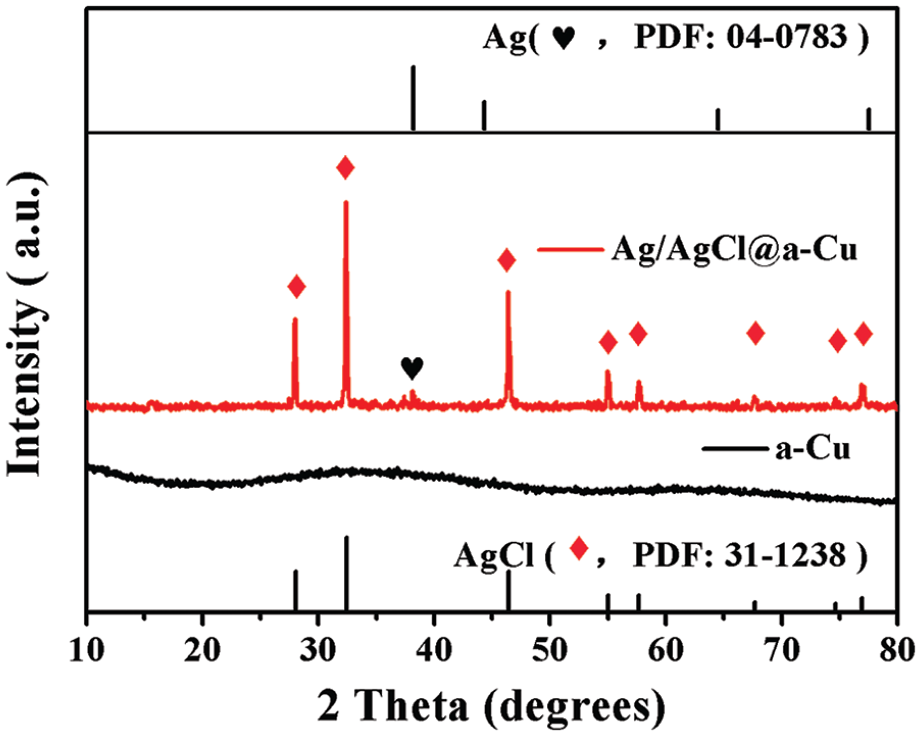
X-ray diffraction patterns of the different catalysts.
The morphology of Ag@AgCl/a-Cu was investigated with TEM. The TEM image of a-Cu is shown in Figure 2(a). The amorphous copper was mainly composed of irregular particles or fragments. Among them, the difference in brightness evident in Figure 2(a) may be caused by copper particles with different stacking thickness. In order to confirm the structure of amorphous copper, a high-resolution transmission electron microscope (HRTEM) and selective area electron diffraction (SAED) studies were applied (Figure 2(b) and (c)). No obvious lattice fringe was displayed in the HRTEM, and no diffraction ring or diffraction spots were observed in the SAED, which confirmed that the metallic copper was obtained as an amorphous structure. This result was also consistent with the XRD results. In the TEM of the Ag@AgCl/a-Cu composite, uniformly sized spherical dark areas and sharp borders were evident in Figure 2(d), which indicated that the particles may contain different substances and should have an Ag@AgCl structure. These black spheres were about 20 nm in diameter and were more evenly distributed in lighter shades (a-Cu) with traces of encapsulation. This indicated that the Ag@AgCl structure was not simply loaded in the aggregates of a-Cu, but may contain some Ag@AgCl covered by a-Cu, which is beneficial to improve the stability of Ag@AgCl/a-Cu composites. To further determine the composition, HRTEM and SAED were also applied and the results are shown in Figure 2(e)–(g). In the HRTEM image (Figure 2(g)), the lattice fringe spacings of 0.23 and 0.28 nm were consistent with cubic Ag (1 1 1) lattice plane spacing and cubic AgCl (200) lattice plane spacing, respectively. 20 In addition, the image also showed the tight interface between Ag and AgCl in the prepared sample and the formation of a heterojunction. This was also consistent with the results of the XRD pattern, which further confirmed that Ag0 was formed on the surface of the AgCl particle.

TEM, HRTEM, and SAED of a-Cu and Ag@AgCl/a-Cu. (a)–(c) are the TEM, HRTEM, and SAED of a-Cu support, (c) and (d) are the TEM images of Ag@AgCl/a-Cu, the SAED pattern is shown in (e) as an illustration, and (f) and (g) are HRTEM images at different magnifications.
X-ray photoelectron spectroscopy was applied to analyze the chemical states of the elements contained in the Ag@AgCl/a-Cu composite, and the energy spectrum was calibrated by the C1s peak (284.8 eV). From the analysis of the XPS spectrum (Figure 3(a)), the Ag@AgCl/a-Cu composites were mainly composed of three elements: Ag, Cl, and Cu. The C1s and O1s peaks may originate from ethanol or TA molecules adsorbed on the surface of the sample. From the high-resolution core-level XPS spectrum for Cl2p (Figure 3(b)), the Cl2p peak was composed of two peaks, 197.8 eV (Cl2p3/2) and 199.3 eV (Cl2p1/2), respectively, which indicated that the chlorine is in the Cl−1 state. In Figure 3(c), two double isolated peaks at ~368 and ~374 eV for Ag3d with a ca. 6 eV interval distance in the spectrum can be assigned to each Ag3d5/2 and Ag3d3/2 binding energy.21,22 The peaks of Ag3d5/2 and Ag3d3/2 can be further divided into different peaks at 367.45 and 368.25 eV, 373.45 and 374.15 eV. The peaks of 367.45 and 373.45 eV can be attributed to the Ag3d peaks of AgCl, and the peaks at 368.25 and 374.15 eV can be attributed to the Ag3d peaks of metallic silver. 8 However, in the case of Cu, it was found mostly in the metallic state Cu0 (Figure 3(d)). Two major peaks located at binding energies of 932.3 and 952.1 eV corresponded to Cu 2p3/2 and Cu 2p1/2, respectively, typically due to zero-valent Cu.17,23 In addition, the shake-up satellite peaks located at 941.2 and 944.1 eV were ascribed to the open 3d 9 shell of Cu2+, which meant that a small amount of unreduced Cu2+ was co-present in the amorphous copper. 24 Overall, XPS analysis confirmed the presence of metallic Ag, AgCl, and amorphous metallic Cu in the Ag@AgCl/a-Cu composite.
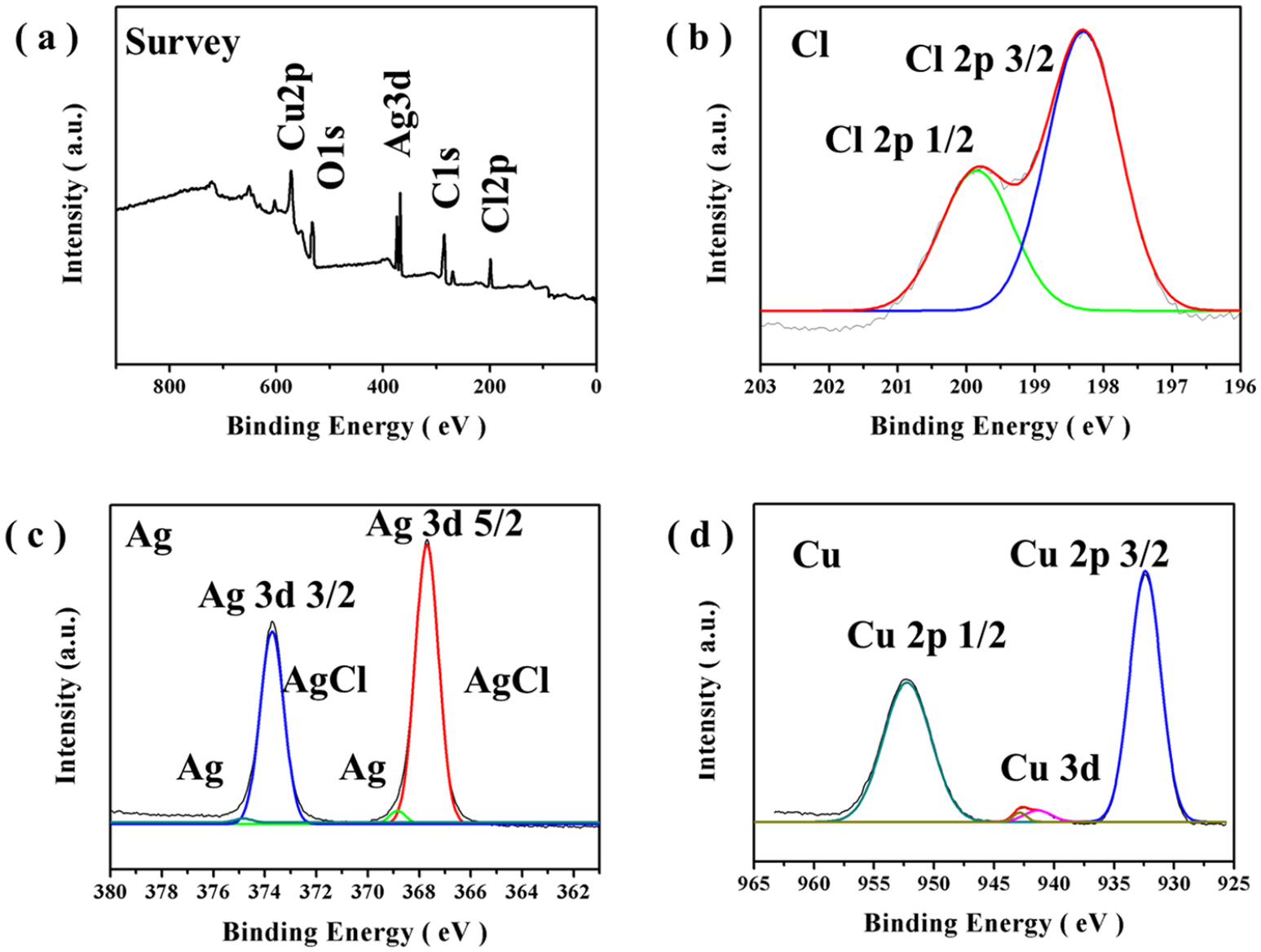
(a) The X-ray full-scan spectrum of Ag@AgCl/a-Cu. (b–d) The narrow scan spectra of the Cl 2p, Ag 3d, and Cu 2p peaks.
Catalytic studies
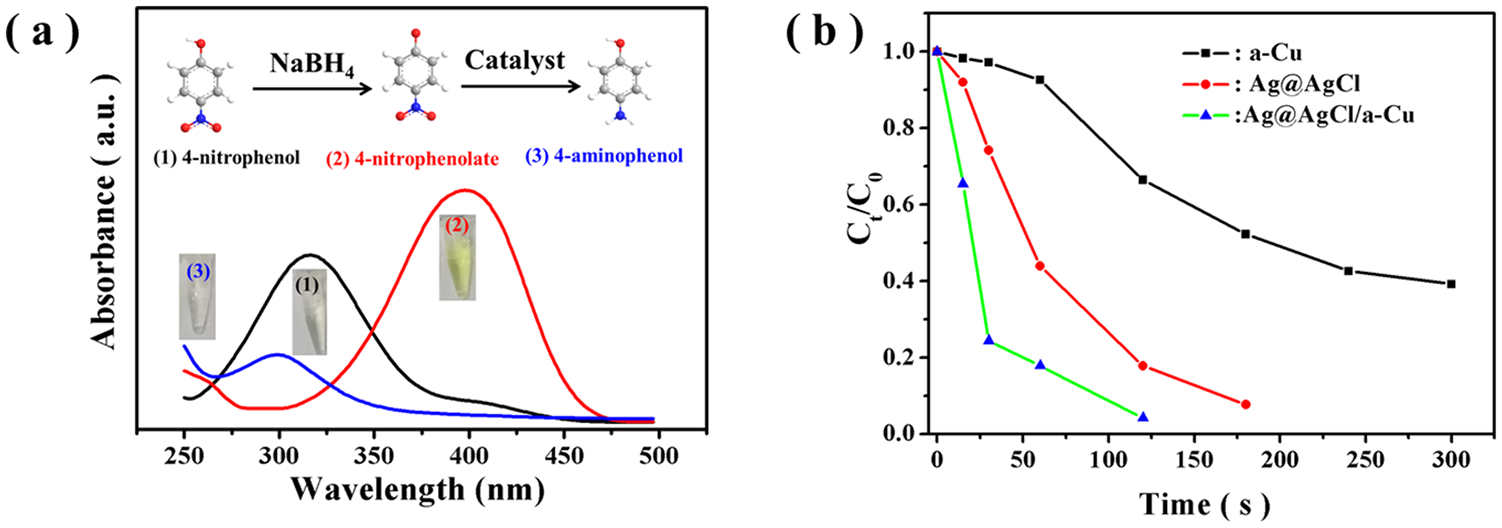
The catalytic activity of the Ag@AgCl/a-Cu composite was evaluated by reducing 4-NP using NaBH4 as the reducing agent. After NaBH4 had been added, the maximum absorption peak of 4-NP shifted from 317 to 400 nm, and the color of the solution changed from colorless to pale yellow due to the formation of 4-nitrophenolate ions under alkaline conditions. With the assistance of the Ag@AgCl/a-Cu composite catalyst, the solution became colorless within 120 s, and the peak at 400 nm disappeared, accompanied by a new peak at 300 nm, indicating that 4-NP had been reduced to 4-AP (Figure 4).

UV-Vis absorption spectra of 4-nitrophenol, 4-nitrophenolate ion, and 4-aminophenol.
The changes in UV-Vis absorption values in relation to time during the reduction of 4-NP to 4-AP with the assistance of a-Cu, Ag@AgCl, and Ag@AgCl/a-Cu composites are displayed in Figure 5, where Ct and C0 are the concentrations of 4-nitrophenol at time t and 0, respectively. Compared with a-Cu and Ag@AgCl, the Ag@AgCl/a-Cu composite exhibited the best catalytic activity.

Time-dependent UV-Vis absorption spectra for the reduction of 4-NP to 4-AP with (a) a-Cu, (b) Ag@AgCl, and (c) Ag@AgCl/a-Cu as the catalyst. The plot of (d) Ct/C0 and (e) –ln(Ct/C0) versus the reaction time for the different catalysts. (f) The recyclability of the Ag@AgCl/a-Cu composite catalyst for the reduction of 4-NP in NaBH4 solution.
NaBH4 was present in a large excess compared to 4-NP, so the NaBH4 concentration can be assumed as constant, and the reduction of 4-NP favors pseudo-first-order kinetics. The reduction rates of 4-NP by the as-prepared catalysts were calculated using the following pseudo-first-order kinetic equation

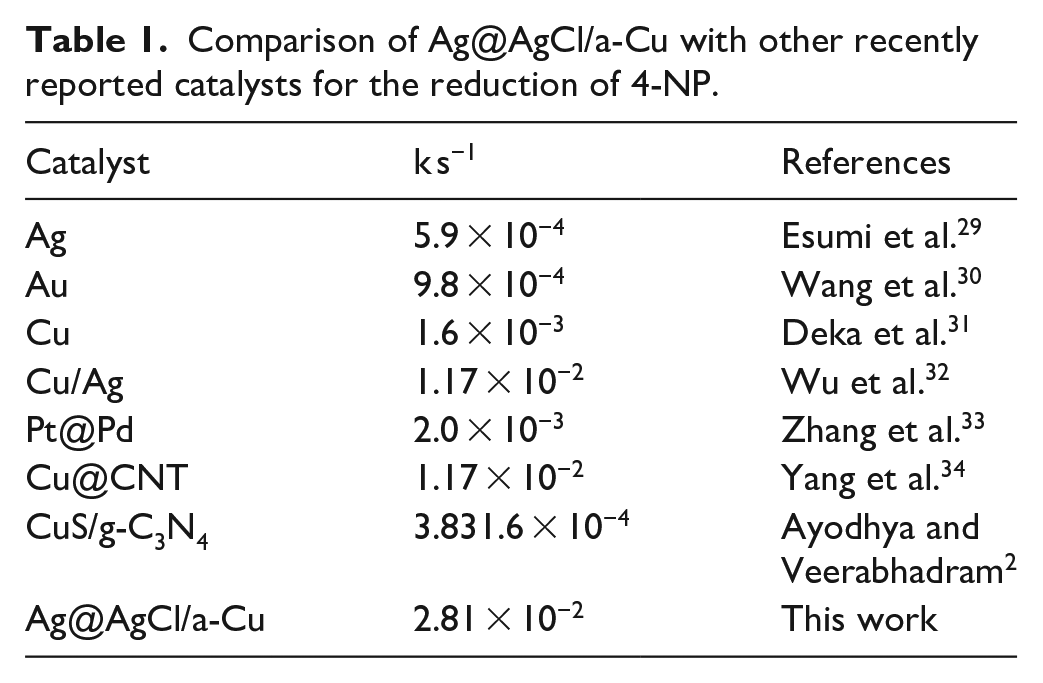
where k represents the rate constant (s−1). Figure 5(d) shows the 4-NP reduction rate constants for α-Cu, Ag@AgCl, and the Ag@AgCl/a-Cu composite. The results indicated that 4-NP reduction follows the pseudo-first-order model, which is consistent with previously reported works.25–28 The Ag@AgCl/a-Cu composite sample has the highest reduction rate constant of 2.81 × 10−2 s−1, which is 2.04 times that of Ag @ AgCl (1.38 × 10−2 s−1) and 6.69 times that of a-Cu (3.31 × 10−3 s−1). As previously reported by Wang et al., 8 the catalytic property of the AgCl@Ag composite for the reduction of 4-NP by NaBH4 is due to the donation of an electron or hydrogen atom from BH4-to organic compounds. As an amorphous Cu, it not only retains the good electron conductivity of the metal but also has many structural defects and dangling bonds distributed on the surface, which results in it having a good catalytic ability.8,16 In this study, the prepared Ag@AgCl/a-Cu composite exhibited excellent catalytic reduction properties for 4-NP, which may be due to the good synergistic effect between the two catalytic materials. In addition, Table 1 gives a detailed comparison of the analytical parameters between the proposed system and other recently reported catalysts. Compared with other catalysts, Ag@AgCl/a-Cu has higher catalytic activity.
Comparison of Ag@AgCl/a-Cu with other recently reported catalysts for the reduction of 4-NP.
For practical catalysts, especially those containing noble metal elements, recyclability is a very important issue. The catalyst was tested for its stability and reusability by recycling experiments. The results of five cycles are displayed in Figure 5(f), indicating that the catalytic efficiency decreases slightly based on the number of cycles. On the fifth reuse, the catalytic reduction efficiency was 84.7%.
Conclusion
In conclusion, an Ag@AgCl/a-Cu composite catalyst has been prepared by a two-step precipitation method at room temperature followed by a light irradiation reduction method. The reduction-catalytic activity of the Ag@AgCl/a-Cu composites was studied in the reduction of 4-NP in aqueous NaBH4 solution, and 95.1% of 4-NP was reduced to 4-AP in 120 s by fresh Ag@AgCl/a-Cu composite catalyst. The high-performance catalytic efficiency and reusability of Ag@AgCl/a-Cu can be attributed to the synergistic effect between Ag@AgC and amorphous metal elements. This research may provide an effective reference for the preparation of high-performance composite catalysts using amorphous metals as supports.
Experimental section
Materials and reagents
Copper chloride dehydrate (CuCl2·2H2O, AR, 99%), tannic acid (TA, 99%), sodium hydroxide (NaOH, AR, 96%), sodium borohydride (NaBH4, AR, 99%), silver nitrate (AgNO3, AR, 99%), and 4-nitrophenol (4-NP, AR, 99%) were purchased from Sinopharm Chemical Reagent Co., LTD. All reagents were used without further purification and distilled water with a resistance of 18.2 MΩ was used throughout the experiments.
Synthesis of amorphous Cu (a-Cu) support
CuCl2·2H2O (0.2 g) and TA (0.08 g) were added to deionized water (38 mL) and were completely dissolved by ultrasonic treatment and stirring. Next of NaOH (2.0 mL, 1M) was added dropwise to the above solution during stirring for 10 min, and the solution was then stirred for a further 15 min again. The product was collected by centrifugation, washed with water and ethanol several times, and then dried under vacuum overnight. All the operations described above were performed at room temperature.
Synthesis of the composite catalyst
CuCl2·2H2O (0.2 g) and TA (0.08 g) were added to deionized water (38 mL), and were completely dissolved by sonication and stirring. Next, during stirring, NaOH (2.0 mL, 1M) was added dropwise to the above solution for 10 min, and after stirring the solution for 15 min, AgNO3 (0.2 g) was added, and stirring was continued for 30 min. The product was collected by centrifugation, washed several times with water and ethanol, and then dried under vacuum overnight. The resulting powder was irradiated under an ultraviolet lamp for 60 min to ensure that Ag+ was partly reduced to Ag0 to give Ag@AgCl/a-Cu composite catalyst as a gray powder. The Ag@AgCl composite catalyst was synthesized by a similar method.
Characterization
The XRD patterns were recorded on the D8 advance Diffractometer (Bruker) equipped with CuKα (λ = 1.54056 Å) radiation (40 kV, 40 mA). TEM analysis was carried out by transmission electron microscopy (2010-Plus, JEOL) at an accelerating voltage of 200 kV. X-ray photoelectron spectroscopy (XPS) was carried out using a ThermoScientific ESCALAB 250 spectrometer with monochromatic Al Kα as the excitation source. The UV-Vis spectra were recorded on a UH4150 (Hitachi) UV-Vis spectrophotometer.
Reduction of 4-NP
The reduction of 4-NP was performed in a glass flask in the presence of the different catalysts (a-Cu, Ag@AgCl, and Ag@AgCl/a-Cu) and aqueous NaBH4 solution under constant stirring. In a typical procedure, first, 4-NP (20 mL, 0.10 mM) aqueous solution was purged with N2 to remove dissolved oxygen in the aqueous solution, and then freshly prepared NaBH4 (5 mL, 20 mM) were added. At this time, the colorless and transparent aqueous solution turned pale yellow. So, 4-NP showed a characteristic absorption peak at 317 nm in a neutral or acidic medium but after adding NaBH4, the 4-nitrophenolate ion was formed, and the absorption peak shifted to 400 nm. After the addition of the catalyst, the characteristic peak at 400 nm decreased with time and a new peak appeared at 300 nm. The solution was stirred during the reaction, and the supernatant was transferred to a quartz cuvette for UV-Vis spectrum measurements. The decrease in the 400 nm characteristic absorption peak indicated a decrease in the amount of 4-NP.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by West Anhui University (WGKQ201702022, WXBSH2019004, 201910376003), Anhui Provincial Education Department (gxgnfx2019029), and Anhui Provincial Natural Science Foundation (1808085QH233, 1808085QB33).
